Abstract
Background: Headache has been reported to be the first clinical presentation in several patients with cerebral arteriovenous malformations (AVMs). Headache associated with AVMs often shows characteristics of migraine with and without aura. Angiographic characteristics of AVMs, such as their location, could determine the ‘migraine-like’ features of attacks.
Methods: We performed an observational study of the clinical and angiographic characteristics of a cohort of 40 consecutive patients with AVMs who had been admitted to our institute for endovascular embolization over a 4-year period. Headache was characterized according to ICHD-II criteria. The relationship between headaches and the angioarchitectural features of AVMs was also analysed.
Results: Migraine-like headache was the first clinical manifestation in 22.5% of patients. The location of the malformation was significantly associated with migraine-like presentation (p = 0.03) and the occipital lobe was the predominant site.
Conclusions: An occipital location may be linked with spreading depression, a pathogenic mechanism of migraine. Headache associated with arteriovenous malformations in the occipital lobe, although secondary in nature, could have clinical features similar to migraine.
Introduction
Headache has been reported to be the first clinical presentation in about 14% of patients with arteriovenous malformations (AVMs) (1). It has been suggested that there is a relationship between AVMs and headache, as also defined by the International Classification of Headache Disorders 2nd Edition (ICHD-II) (2). ICHD-II identifies diagnostic criteria of headache attributed to AVMs, but it gives no specific indication about the clinical pattern of attacks, except for their duration. Nevertheless, headache associated to AVMs often shows characteristics of migraine with (MwA) and without aura (MwoA).
The occipital lobe is a frequent location of AVMs (3), and headache has been described as the first symptom of presentation in several patients with occipital AVMs (4,5). Interestingly, ‘migraine-like’ headaches have been observed in patients with occipital AVMs, leading to the hypothesis that haemodynamic changes caused by AVMs might trigger spreading depression (SD), which is a key pathogenic mechanism of migraine (6–8). Thus, it could also be hypothesized that the angiographic characteristics of AVMs, such as their locations, could determine migraine-like features of attacks. However, most of the previous studies on AVMs have not classified headaches according to the ICHD-II criteria (1).
The aim of our study was to evaluate headache, according to ICHD-II criteria, in a group of patients with AVMs, and the relationship of headache with angioarchitectural AVM features.
Methods
This study has been approved by an internal review board. We performed an observational study of a cohort of 40 patients with AVMs who had been consecutively admitted to the Neurological Clinic of Perugia University for endovascular embolization in a 4-year period and included in the registry of the Interventional Neuroradiology Unit (1 January 2005 to 1 August 2009).
For each patient we evaluated sex, mean age at the time of diagnosis and initial clinical manifestation. Headache was characterized according to ICHD-II criteria (2).
For patients with migraine-like headache reported as the first symptom of AVMs, we evaluated the family history and the age of onset of headache.
In all patients diagnostic cerebral angiography was performed after written consent.
AVMs were analysed according to their angioarchitectural characteristics: location, size, type of nidus, the presence of feeding from the external carotid artery and characteristics of venous drainage. Locations were classified in: frontal, temporal, parietal, occipital, multilobar, callosal, cerebellar and basal ganglia AVMs. AVMs were distinguished according to size in small (less than 3 cm), medium (3 to 5 cm) and large (more than 5 cm). Type of nidus was classified as plexiform, fistulous and mixed. Venous drainage was classified as deep, superficial or mixed; moreover, we also considered the number of draining veins, single or multiple.
Fisher’s exact test was used to analyse whether there was a possible association between migraine-like presentation and some AVM angioarchitectural features (location, the presence of feeders from the external carotid artery and the type of venous drainage).
Results
Patient population
Demographic, clinical and angiographic features

Initial clinical manifestations were epilepsy in 15 patients (37.5%), headache in 10 (25%) and intracranial bleeding in 11 (27.5%). Three patients (7.5%) were asymptomatic at the time of the diagnosis. In one case (2.5%) the diagnosis was made because of a focal neurological deficit (Table 1).
Angiographic characteristics
AVMs were frontal in 27.5% (n = 11), temporal in 12.5% (n = 5), parietal in 10% (n = 4) and occipital in 17.5% (n = 7) patients. A multilobar AVM was observed in 10% (n = 4) of patients, and 5% (n = 2) had an AVM located in the corpus callosum. Finally, 7.5% (n = 3) of the AVMs were in the basal ganglia and 10% (n = 4) in the cerebellum.
Concerning size, AVMs were small in 17.5% (n = 7), medium in 62.5% (n = 25) and large in 20% (n = 8) of the patients. Type of nidus was plexiform in 20% (n = 8), fistulous in 17.5% (n = 7) and mixed in 62.5% (n = 25) of the patients. AVMs had some arterial feeders from the external carotid artery in 30% (n = 12) of the patients.
Moreover, 50% (n = 20) of AVMs had a superficial venous drainage, 15% (n = 6) deep and 35% (n = 14) mixed. Concerning the number of draining veins, 85% (n = 34) of AVMs had multiple venous drainage.
Migraine as initial manifestation of AVMs
According to ICHD-II criteria (2), migraine-like headache was the first clinical manifestation of AVM in 22.5% (9 out of 40) of the patients. In this group, ‘MwoA-like’ headache was observed in 55.6% (n = 5) of the patients and 22.2% (n = 2) had a ‘MwA-like’ headache. The remaining two patients presented with attacks both with and without aura.
All patients showing migraine-like headache were female and the mean age of onset of headache was 21.1 ± 3.3 years. In 22.2% (2 out of 9) of patients there was a family history of migraine.
As regards AVM angioarchitectural features of patients with migraine-like headache, all AVMs were of medium size, and 44.4% had some arterial feeders from the external carotid artery. Moreover, 44.4% (n = 4) of the AVMs had a superficial venous drainage, in 11.2% (n = 1) of the patients it was deep and in 44.4% (n = 4) it was mixed.
The AVM location in patients with migraine-like headache was the occipital lobe in 55.6% (n = 5), frontal lobe in 22.2% (n = 2), corpus callosum in 11.1% (n = 1) and cerebellum in 11.1% (n = 1) of cases.
Distribution of AVM location in relation with migraine-like presentation
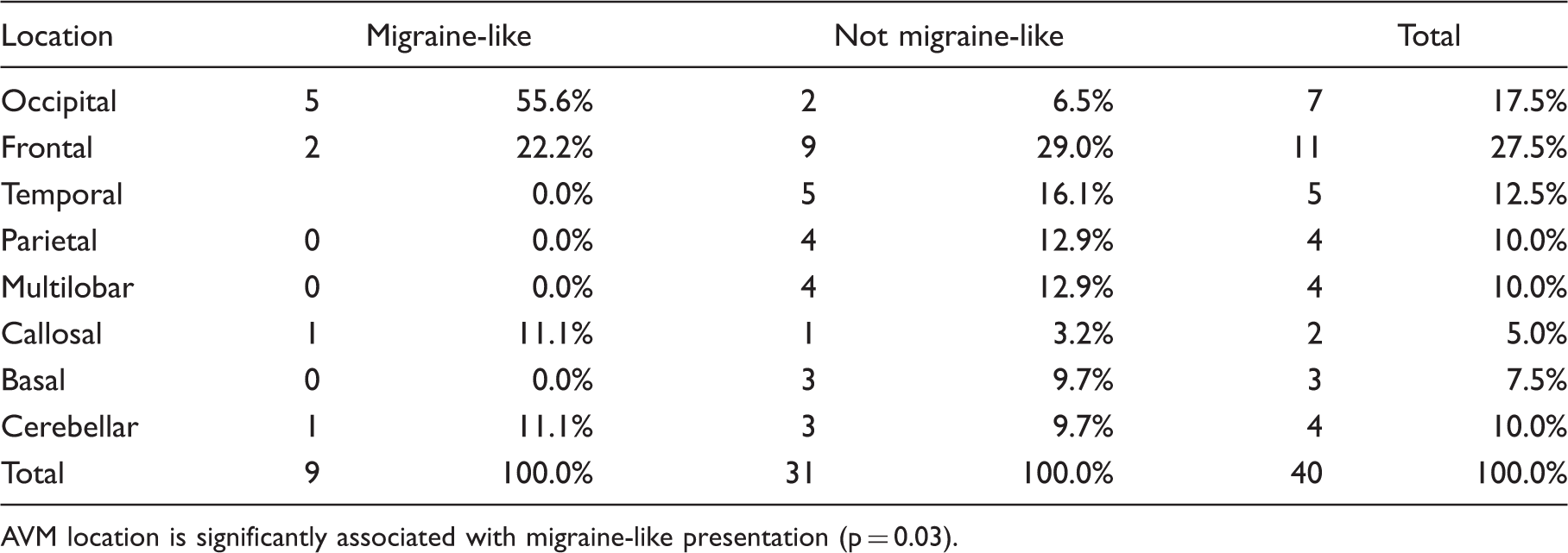
AVM location is significantly associated with migraine-like presentation (p = 0.03).
In all the patients with occipital AVM cephalic pain was ipsilateral to the vascular lesion. In two cases typical ‘MwA-like’ headache was the clinical presentation of AVM, and one patient presented a ‘basilar-type-like’ headache (see case reports in Online-Only Material). For example, Figure 1 shows an intra-arterial angiogram from a woman with a typical ‘visual aura’ as a manifestation of a AVM in the medial part of the left occipital lobe before (Figure 1A) and after (Figure 1B) treatment by embolization (case 1 in Online-Only Material).
Intra-arterial angiogram revealing occipital arteriovenous malformation in the medial part of the left occipital lobe before (A) and after (B) embolization (see case 1 in Online-Only Material).
Discussion
AVMs could be considered to be among the causes of symptomatic headache, as stated in the ICHD-II (2). Headache has been reported to be the first clinical presentation in about 14% of AVM patients (1). Conversely, our study has shown that in about 25% of patients headache led to an AVM diagnosis.
Although most of previous studies did not use current headache criteria to characterize clinical features of attacks, some authors have hypothesized that headache related to AVMs could be migraine-like (9,10). Interestingly, we found that in large majority of patients with headache as AVM presentation, attacks showed clinical features of migraine, according to ICHD-II criteria (2). Moreover, prevalence of migraine-like headache as AVM initial symptom was higher than the prevalence of this kind of headache in the general population. We found a predominance in female patients and the same age of onset of migraine as reported in the general population (11).
AVM location was the angioarchitectural feature that was associated with migraine-like presentation. This type of presentation was not related to the presence of arterial feeders from the external carotid artery or to the characteristics of venous drainage. Interestingly, in patients with migraine-like headache, we found a clear predominance of the occipital lobe location.
A prevalence of migraine-like headache of 18.9%, using the previous International Headache Society criteria (12), has been previously found in occipital AVMs (9). Thus, in accordance with that study and with our findings, it could be hypothesized that haemodynamic and structural changes surrounding AVMs located in the occipital lobe may be involved in initiation and propagation of SD, a key pathogenic mechanism underlying migraine (6,7). As suggested by the clinical history of our three described cases (see Online-Only Material), patients with occipital AVMs show migraine-like headache that has clinical features very similar to the idiopathic form, showing characteristics resembling those of migraine with visual aura.
It could be postulated that AVM could lower the local threshold for attacks, possibly triggering SD, in patients having genetic predisposition for migraine (7). Vascular events, such as ischemic and haemorrhagic stroke, could create conditions that would induce SD (13,14).
Ischemic and haemorrhagic events could occur in the environment of an AVM, reflecting continuous haemodynamic modifications in the arteriovenous shunt (15). The rapid lowering of blood flow between feeding arteries and draining veins could create a sort of ‘steal’ phenomenon in the perilesional region, realizing a relative ischemia (15). Conversely, sudden pressure variations and vessel stenosis could predispose to haemorrhagic events (15). Thus, it might be supposed that AVM could contribute to triggering SD.
Interestingly, occipital location could be considered a necessary condition for this phenomenon, as for the pathogenesis of migraine (not only with aura). SD is considered, in fact, to be the crucial phenomenon in migraine pathogenesis when it occurs in specific cortical and subcortical structures (6,16). AVM, by favouring SD, could determine the lateralization of the visual aura and of cephalic pain (7). It has been reported that the side where the SD occurs is ipsilateral to the headache and controlateral to the visual symptoms (17). This clinical feature has usually represented a ‘red flag’ suggesting that a headache is secondary in nature.
Results obtained in the present study should be interpreted with caution because of some potential biases. Firstly, patients have been selected for endovascular therapy at a single centre. This might influence the clinical and angiographic characteristic of the population studied. Moreover, it should be considered possible that patients with haemorrhage, seizures and/or focal deficits could have a history of migraine before these events.
Finally, we cannot provide clear information on the effects of AVM embolization on migraine frequency because this aspect has not been properly addressed in our study. We are aware that a large multi-centre international trial (ARUBA) is ongoing to evaluate the benefit of AVM eradication not only for the prevention of stroke, but also for reducing the frequency of seizures and migraine. Results from this trial might provide more data on this interesting issue.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of interest
PC serves as an editorial board member of Lancet Neurology, the Journal of Neuroscience, and Synapse. He receives research support from Bayer Schering, Biogen, Boehringer Ingelheim, Eisai, Novartis, Lundbeck, Sanofi-Aventis, Sigma-Tau, and UCB Pharma, and from Ricerca Corrente IRCCS, Ricerca Finalizzata IRCCS (European Community Grants SYNSCAFF and REPLACES), the Italian Minister of Health and AIFA (Agenzia Italiana del Farmaco).
