Abstract
This case report describes the management of a patient with a lumbar arachnoid cyst requesting epidural labour analgesia. The patient was otherwise healthy but had suffered from multiple dural punctures during attempted epidural placement in a previous pregnancy. Magnetic resonance imaging following the dural punctures revealed the cyst, which was at the level of the L3–4 interspace, effacing the posterior epidural space at that level. Multidisciplinary management in this subsequent pregnancy, involving radiology and anaesthesia specialists, allowed ultrasound-guided identification of a higher epidural space, and successful and uneventful epidural placement during labour. Multiple dural punctures with attempted epidural placement should prompt anaesthetists to consider spinal magnetic resonance imaging to identify anatomical abnormalities that may be present, which may have implications for future management.
Keywords
Introduction
Spinal arachnoid cysts are typically benign collections of fluid in the spinal canal or epidural space.1,2 Most occur in the thoracic spine,3–5 with a mean span of 3.7 vertebral bodies. 6 They are also often positioned posteriorly, 1 which has implications for translaminar neuraxial access. Histologically, these cysts consist of a fibrous connective tissue wall, and may be lined with arachnoid cells. 7 Some communicate with the existing subarachnoid space, filling with cerebrospinal fluid (CSF), while others are separate, secreting their own fluid from cells within the cyst.7,8 Their exact pathogenesis remains unclear. Proposed causes include primary cysts, due to a probable congenital defect in the dura, and secondary cysts, due to infection, inflammation, or trauma, which may be iatrogenic. 9 Cysts are usually asymptomatic and are often found incidentally. 4 They may become symptomatic if compression of the associated spinal cord or nerve roots occurs, resulting in motor weakness, back pain and paraesthesia.5,7 Various classifications of spinal cysts have been proposed, 10 the most relevant for anaesthetists is likely that proposed by Nabors et al., in which cysts are classified based on their association with nerve root fibres. 11
Consent was obtained from the patient for presentation of the following case report. This report adheres to the case report (CARE) guidelines.
Case report
A healthy primiparous woman presented for induction of labour for a small-for-gestational age foetus and requested epidural analgesia. Her height was 161 cm and body mass index at antenatal booking was 23 kg/m2. She was healthy apart from a history of anxiety treated with desvenlafaxine.
Initial epidural placement was attempted by a trainee anaesthetist with the patient in the sitting position using a 16 gauge Tuohy needle with a loss of resistance to saline technique. At the L3–4 level, CSF was identified at a depth of 4 cm. A consultant anaesthetist was called to assist and similarly identified CSF at a depth of 5 cm at the L4–5 level. During this attempt, 2 ml of ropivacaine 0.2% with fentanyl 5 µg/mL was administered to establish analgesia. A third attempt, also at the L3–4 level, resulted in a ‘subtle’ loss of resistance. However, when a 5-ml test dose of ropivacaine 0.2% with fentanyl 5 µg/mL was injected, the inserted catheter behaved like an intrathecal catheter, with the patient experiencing a block extending up to C5, a systolic blood pressure in the 90s, nausea, but no respiratory compromise, and therefore was managed as such. A few hours after a ventouse delivery, the patient developed a headache with verbal numerical rating scale intensity of six to seven out of ten. This was managed initially with oral analgesia; however, the next day the patient developed an acute neurological syndrome with hallucinations, hyperreflexia and clonus that was diagnosed as serotonin syndrome after she had received tramadol in addition to her usual desvenlafaxine. She was transferred to the intensive care unit for observation and symptomatic treatment, and this syndrome and her headache resolved over the course of her admission without specific treatment.
Approximately 2 months after discharge when the patient was well and asymptomatic, she was followed up with investigations that included lumbar spine magnetic resonance imaging (MRI). This scan identified an oblong-shaped intraspinal extradural collection at the level of the L3–4 intervertebral disc with approximate dimensions of 4 × 40 × 6 mm, with a signal consistent with CSF (Figure 1). There was no compression of the thecal sac. The conclusion from the reporting radiologist was that this would ‘likely represent an area of controlled small CSF leak’. A provisional plan to repeat the scan to assess the collection’s progress was made, but no other specific intervention was undertaken.
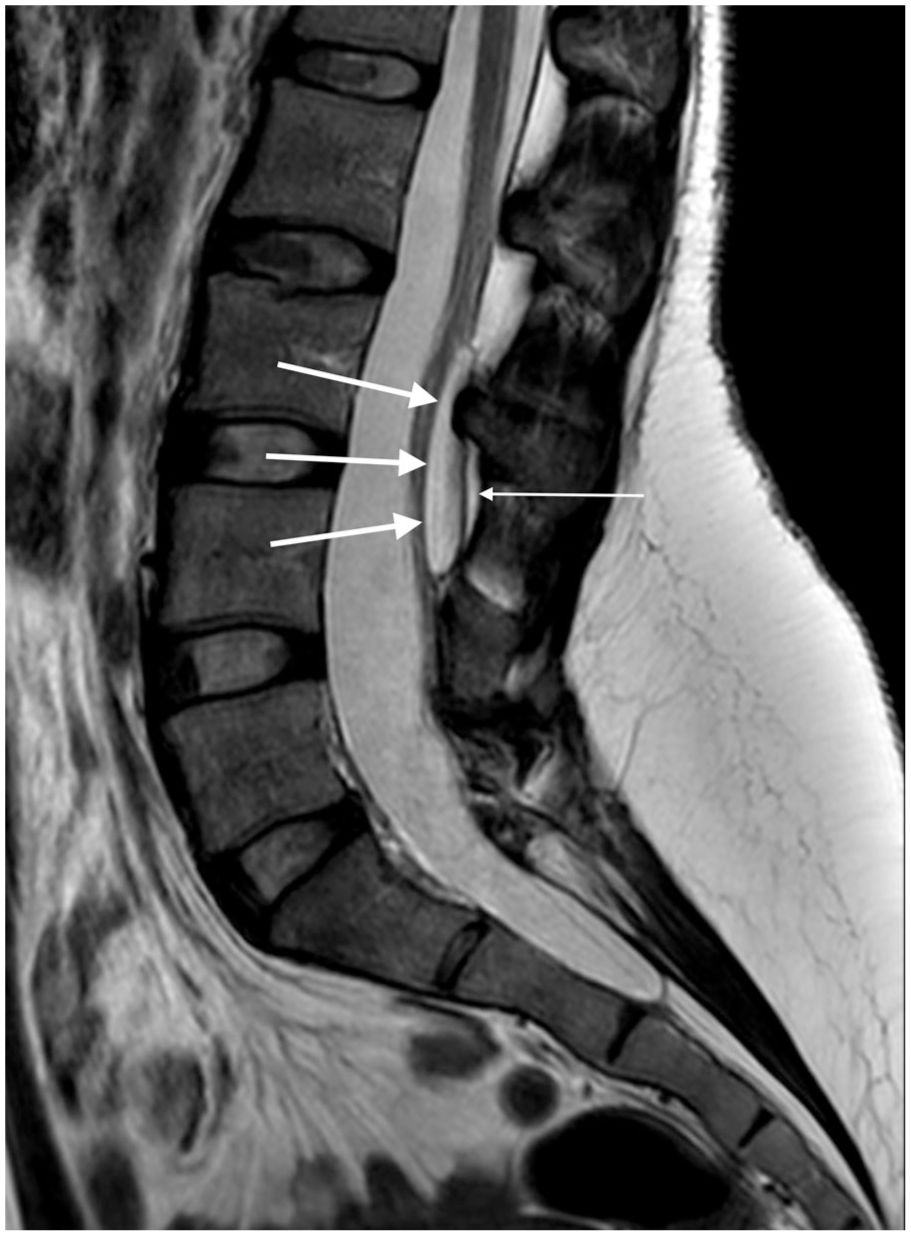
Magnetic resonance imaging 2 months postpartum. Thick arrows point to the cystic lesion, thin arrow points to the effaced epidural space.
One year later, a further lumbar spine MRI was performed (Figure 2), and the collection present on the previous scan was again noted, with a minimal increase in dimensions. The patient was similarly asymptomatic. Subsequent to this scan it was felt that the cystic lesion most likely represented a pre-existing arachnoid cyst and that the original dural punctures were related to effacement of the posterior epidural space12,13 caused by the presence of the cyst.
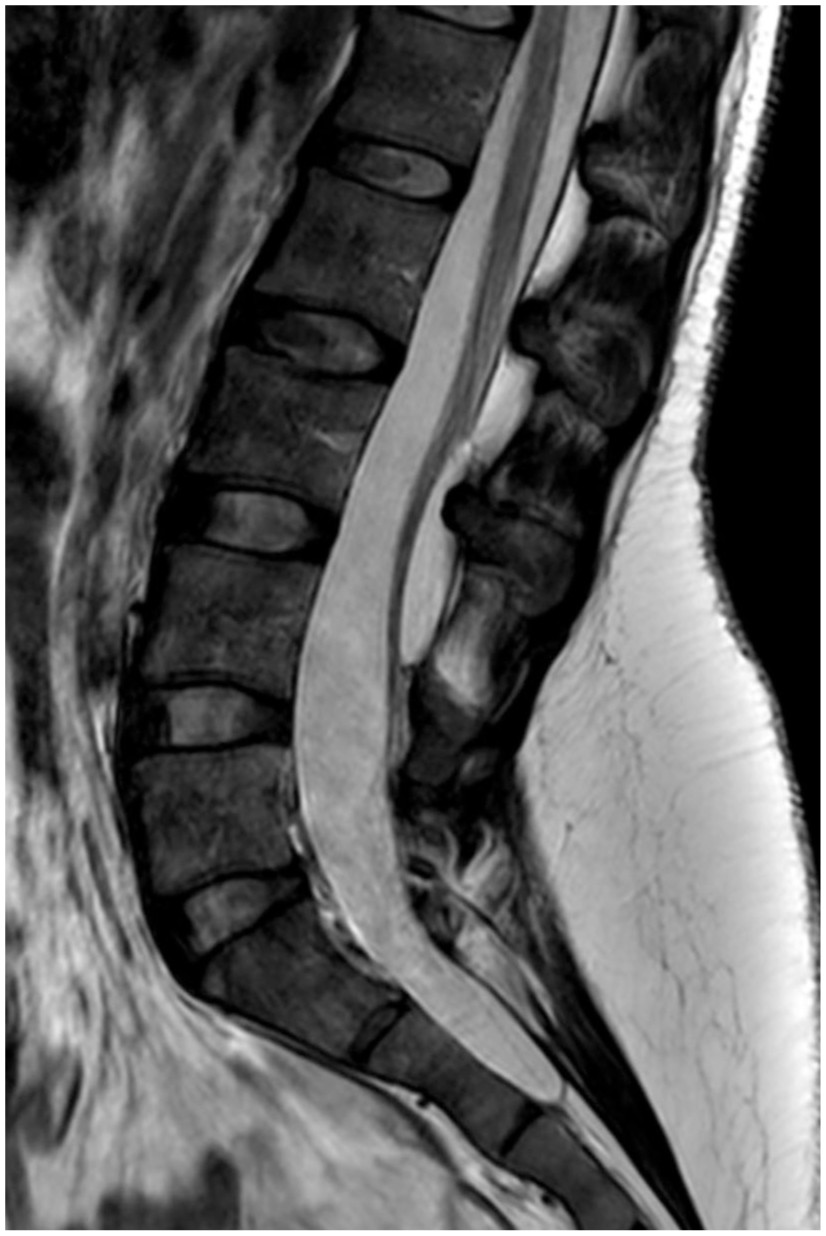
Magnetic resonance imaging 14 months postpartum showing persistence of the lumbar cystic lesion.
Approximately one year later, the patient was pregnant with her second child. A vaginal delivery was planned, and she was seen in the antenatal anaesthetic assessment clinic to discuss planning for labour analgesia. Given that the cyst was unchanged on two prior MRI scans, we elected not to repeat the scan, and to assume that the cyst was still present at the L3–4 level. The patient’s health was otherwise unchanged, although she was anxious about labour and felt that, despite her previous experience, epidural analgesia was required to allow successful vaginal delivery. Her case was discussed at a departmental meeting, and it was felt that if delivery by Caesarean section was required, an attempt at placement of a single-shot subarachnoid anaesthetic would be reasonable (given that the previous presumed intrathecal catheter had been effective) but that treating anaesthetists should have a low threshold for general anaesthesia.
Multidisciplinary discussion took place to allow coordination of a delivery plan (induction on a given date), radiological consultation to allow ultrasound identification of the L3–4 level (to avoid) and a higher level (to place an epidural), and senior anaesthesia support to place an early epidural. Despite these plans, the patient presented early with ruptured membranes and contractions. Fortunately, a radiologist familiar with the patient was on duty and performed an ultrasound scan that identified the L3–4 level, and the anaesthetist who had seen her twice in the antenatal anaesthetic clinic was also on duty and placed the epidural at the L1–2 level. Initial attempts at the L2–3 level had led to cannulation of an epidural vein, but no CSF was encountered. The patient had successful labour analgesia, as well as a top-up for removal of retained placenta. This top-up consisted of divided doses of 15 ml of lidocaine 2% with adrenaline and achieved a block to T4–T6 and patient comfort throughout the procedure. She was well when discharged from hospital, and when followed up by phone several weeks postpartum.
Discussion
A key consideration in this case is whether the arachnoid cyst was a consequence of repeated dural puncture, or whether the failed epidural in her first labour was due to an existing arachnoid cyst.
Several case reports describe the formation of, or development of symptoms related to, a spinal arachnoid cyst following subarachnoid and epidural anaesthesia (Table 1).4,14–20 A common theme in these cases is a slow, progressive development of symptoms, often presenting many months after the procedure. Accordingly, it is challenging to determine whether the neuraxial anaesthetic procedure was the cause of the cyst, or whether it simply uncovered a pre-existing problem. Furthermore, arachnoiditis is an important cause of cyst formation, 12 and most of the reported cases occurred in the late 20th century when anaesthetic formulations and techniques were different to those at present. Additionally, ultrasound and/or MRI in these cases broadly demonstrates cysts located at spinal levels markedly higher than where neuraxial puncture was performed.
Case reports of arachnoid cyst following anaesthetic procedures.
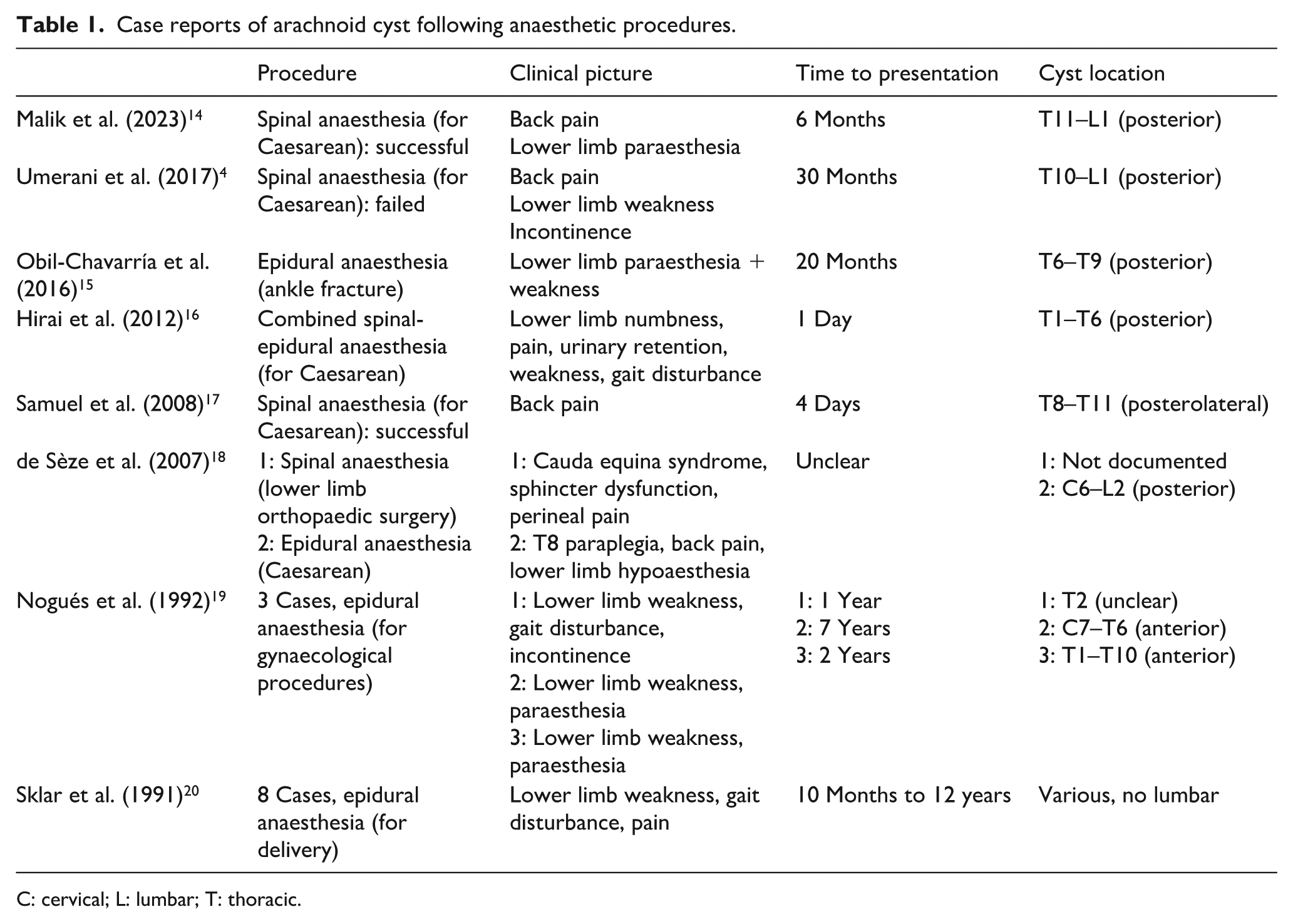
C: cervical; L: lumbar; T: thoracic.
Supporting the hypothesis that the cyst was pre-existing are case reports describing the distribution of CSF after dural puncture.21–24 MRI from these reports shows extensive spread of CSF into the epidural space following both deliberate puncture for diagnostic purposes, and inadvertent puncture during epidural anaesthesia. Subsequent MRI in the following days and months then demonstrates resolution of this CSF spread. Given this, it is the authors’ hypothesis that the difficult epidural anaesthetic in our patient in her first labour was a consequence of an existing arachnoid cyst, which is likely to be congenital in origin.
Point-of-care ultrasound has become common practice in modern anaesthesia, 25 including ultrasound of the spine for neuraxial procedures. 26 Several systematic reviews have confirmed the benefits of neuraxial ultrasound in epidural and spinal procedures,27–30 including in obstetric patients. 31 Ultrasound imaging of the spine by both anaesthetists 32 and radiologists 33 has been reported for facilitating neuraxial anaesthesia, and has been shown to reduce the number of skin punctures and needle redirections.26,34 Although anaesthetists often have appropriate ultrasound skills and training, involving a radiologist familiar with this case was felt to offer the best chance of avoiding complications in our patient.
The implications of arachnoid cysts for obstetric anaesthesia have been described in various case reports. The successful use of epidural, subarachnoid and general anaesthesia has been described in obstetric patients with intracranial arachnoid cysts.8,35–37 Yamaki et al. reported a case of failed subarachnoid anaesthesia for Caesarean delivery, with postoperative MRI discovering a spinal arachnoid cyst from T11 to L3 that may have contributed. 38 Gaiser and Antonik reported successful epidural anaesthesia for delivery in a patient with a known spinal arachnoid cyst extending from T11 to L1. 5 The epidural was inserted at the L3–4 level without complication. Gaiser et al. have also described a successful epidural blood patch in a patient with a T10 level arachnoid cyst and postdural puncture headache after myelogram for investigation of the cyst. 2
Another consideration, particularly for subarachnoid anaesthesia, is whether the cyst communicates with the subarachnoid CSF. If subarachnoid anaesthesia were to be attempted and a cyst was unwittingly entered, fluid flashback would be observed. For non-communicating cysts, anaesthesia would then likely be ineffective. For communicating cysts, there may be unpredictable spread related to the degree of communication and any alteration of subarachnoid CSF movement caused by compression. In our case, subarachnoid analgesia was achieved during the patient’s first labour despite probable delivery of medication into the cyst, indicating that the cyst likely had communication with the subarachnoid CSF.
To the best of the authors’ knowledge, no reports have described successful epidural placement in a patient with a low lumbar arachnoid cyst. Importantly, in patients who suffer multiple accidental dural punctures, strong consideration should be given to performing MRI of the spine to identify a possible anatomical cause. Successful epidural insertion is possible in these patients, particularly when multidisciplinary input is sought.
