Abstract
The purpose of this study was to identify haemodynamic factors that are associated with tissue hypoperfusion in flap/graft surgical patients that might be modified to reduce perioperative morbidity. We conducted a single-centre, retrospective, observational study of 1355 patients undergoing head and neck flap reconstructions. Logistic regression and chi-square analyses were employed to identify factors which signal perioperative complications. Study endpoints included postoperative lactic acidosis, acute kidney injury (AKI) and early surgical flap revision surgery. Intraoperative data were collected as time-weighted averages of the haemodynamic variables, including pulse pressure variation (PPV), mean arterial pressure, and vasopressor doses. Cumulative volume was used for intravenous (IV) fluids. Relevant patient comorbidities were also included in the analysis. The most common complication was hyperlactataemia (22.9%), followed by AKI (14.1%) and take-back surgery (3.3%). No patient factors were significantly correlated with flap complications. Elevated max PPV was significantly associated with elevated lactate and AKI in univariate regression, but only AKI in the multivariate analysis (P = 0.003). Case duration was the only variable associated with take-back surgery in the multivariate regression (P = 0.007); it was also associated with lactic acidosis (P = 0.003). Neither IV fluid administration nor the use of vasopressors appeared to be associated with study outcomes in the multivariate analysis.
Keywords
Introduction
Autologous tissue transfers are surgical techniques in which a portion of tissue is transferred from one location to another, most commonly to reconstruct a regional defect after tumour excision or trauma. Flap surgeries involve transfer of tissue with intact blood supply, whereas grafts have no blood supply and rely on neovascularisation. 1 The free flap with anastomosis has become a popular approach because of improvements in microvascular surgical techniques. 2 Otolaryngological reconstructions are generally long and complicated surgeries, often lasting several hours and requiring multiple surgical specialties.
Complications of this surgery include arterial thrombosis, delayed/failed healing, fistula formation, dehiscence, haematoma and infection; in severe cases, such complications can progress to tissue ischaemia/necrosis and outright flap failure.3,4 The major culprit of flap failure is believed to be vascular compromise, either from intravascular thrombosis or mechanical obstruction of the vessels at the site of microanastomosis.3,5,6 Vascular compromise, whether from pedicle malfunction or failure of neovascularisation, will rapidly abolish perfusion of the transferred/rotated tissue, which lacks collateral sources of blood flow. Thus, routine postoperative management involves close monitoring to promptly identify any change in blood flow.7 –10
Other potential contributors to flap/graft hypoperfusion and dysfunction include patient risk factors and intraoperative haemodynamic variables. Sustained decreases in mean arterial pressure (MAP) reduce perfusion systemically and to the autologous tissue. Historically, the use of vasoconstrictors was avoided for fear of curtailing blood flow due to unopposed arterial vasoconstriction of the denervated graft, a concern extrapolated from animal studies. 11 However, the vasodilating effects of anaesthesia, as well as increased preload from vasopressor-mediated venoconstriction, likely offsets the effects on the arterial vascular bed. Indeed, accumulating evidence now indicates that vasopressor use does not increase the rate of flap compromise.12,13 Moreover, avoiding vasopressors often necessitates a liberal fluid strategy, which has its own undesirable effects on flap perfusion, such as oedema.14,15 While the debate is yet unsettled, 16 there is a growing emphasis on goal-directed perfusion to guide haemodynamic decision-making in regard to fluid or vasopressor selection.17,18 There is an ongoing need for reliable predictors of poor perfusion to improve and individualise resuscitation therapy. Notably, although pulse pressure variation (PPV) is a common tool for guiding management of hypotension (either with fluids or vasopressors), it has not been evaluated in the setting of poor perfusion in surgical flap populations.
The purpose of this study was to identify modifiable factors of haemodynamic management that are associated with outcomes related to tissue hypoperfusion. We hypothesise that elevated PPV predisposes patients to postoperative complications from impaired global and local tissue perfusion. The decision to study this subject in flap/graft surgical patients was deliberate, as transferred tissues are exquisitely vulnerable to hypoperfusion.3,4 Thus, flap compromise represents a sensitive and meaningful endpoint to study tissue malperfusion and enhances the clinical relevance of this analysis for otolaryngological procedures compared with assessing non-specific indicators alone (e.g. lactic acidosis).
Methods
This retrospective observational study was conducted at a single academic tertiary care centre. It was approved by the Institutional Review Board (IRB 12-1087) on 30 May 2012, which was renewed annually throughout the study duration.
Patient population
The study population included all adult patients who underwent otolaryngological flap or graft reconstructions under general anaesthesia between 1 April 2014 and 30 June 2022. Procedures with Current Procedural Terminology (CPT) codes 15732, 15734, 15756, 15757, 15758, 20955, 20956, 20962 and 20969 were included. In total, 1355 patients satisfied the above criteria.
Data collection and processing
Patient electronic health record data, including high resolution intraoperative data, was obtained using BusinessObjects™ (SAP, Paris, France) software or manual extraction. Sensitive data were stored using REDCap® (Nashville, TN, USA) database software. Second- and third-party data collection teams performed numerous audits for internal quality control to minimise bias and human error. Among the data collected were baseline patient demographics, past medical history, intraoperative data including vital signs, vascular access, pertinent labs and medications administered (particularly vasopressor agents and dosage), as well as postoperative outcomes.
Independent variables and outcome measures
Patient comorbidities were collected using the following International Classification of Disease (ICD) diagnostic codes: types 1 (ICD-E10) and 2 diabetes mellitus (ICD-E11), hypertension (ICD I10), atrial fibrillation (ICD-I48), heart failure (ICD-I50), stroke (ICD-I63), chronic obstructive pulmonary disease (ICD-J44) and chronic kidney disease (ICD-N18).
Intraoperative MAP and PPV variables were collected at 1-min intervals. Time-averaging was performed as a moving mean over a sliding window of 30-min intervals, producing variables such as Time-averaged MAP and Time-averaged PPV. The maximum (Max) of these time-averaged vectors was used as input variables (Max MAP and Max PPV). We defined significant persistent hypotension as a time-averaged 30-min window with MAP <65 mmHg, yielding the binary variable MAP < 65 mmHg.
Vasopressor administration was recorded as both time-stamped bolus doses and as continuous data (if given by infusion). Continuous data were integrated over each administration period and then subsequently summed with the bolus doses to produce the Cumulative dose of each vasopressor. The time series vector for each medication was also iterated in 30-min windows, integrating and summing the total medication dose for that window, yielding the input variable Max dose for each vasopressor.
Intravenous (IV) fluid volume data were included simply as case totals. Data for both crystalloid and colloid solutions were collected. The crystalloids included were normal saline, Ringer’s lactate, and multiple electrolytes injection, type 1 (brand name Plasma-Lyte, Baxter International, Deerfield, IL, USA). Human albumin 5% solution was the only colloidal solution assessed. The variable All IV fluids represents the sum of both colloid and crystalloid fluids.
In 2010, Gaies et al first described the development and application of the Vasopressor Inotrope Score (VIS), a novel haemodynamic predictor for objectively quantifying the cumulative degree of vasoactive support. 19 Subsequent work further refined this tool.20,21 In simple terms, the VIS is the potency-weighted sum of all administered vasoconstrictors and inotropes. Though initially utilised in the paediatric intensive care setting, it has more recently been adapted for use in adult surgical patients.22,23
We made several modifications to the VIS for the purposes of this study. Previous iterations of the VIS employed weight- and time-based dosing (µg/kg per min); however, in the operating room setting, boluses of vasopressors/inotropes are frequently used alongside infusions, which complicates the simple use of time-based measures. Thus, as with the individual vasopressors, time-stamped bolus doses and continuous (infusion) data were integrated over each period and summed together. The maximum 30-min time-averaged VIS was used in the regression analysis. In addition, because adult patients are more appropriately managed with non-weight based (or ideal body weight) dosing regimens for hydrophilic drugs like vasopressors, we used absolute rather than weight-based values in our calculation. Last, we removed milrinone and dobutamine from the equation and added ephedrine, which was weighted according to estimated potency ratios as previously described.
24
Our final VIS was calculated as follows:
For our outcome measures, lactic acidosis was defined as an intraoperative plasma lactate greater than 2 mmol/l. 25 A diagnosis of acute kidney injury (AKI) was made if there was an increase in plasma creatinine by 26.5 µmol/l or greater within 48 h of the surgery, or an increase of 1.5× the baseline within seven days as per 2012 criteria defined by KDIGO (Kidney Disease: Improving Global Outcomes). 26 Surgical complications were defined as any case requiring take-back surgery within seven days of the primary operation. Specifics regarding the indication for surgical re-exploration (thrombosis, infection, fistula formation, haematoma, etc) were not retrievable because a generic code (CPT 35800) was used for all take-back case postings. Flap complications not requiring take-back surgery were not reliably documented with ICD codes and so were not captured in the outcome measure.
Logistic regression and statistical analysis
Statistical analysis was performed using MATLAB (MathWorks, Natick, MA, USA). All three outcomes were binary (discrete) variables (lactate greater than 2 mmol/l, flap take-back and AKI). Independent variables included both continuous and discrete variables. For the continuous variables (age, body mass index, duration of anaesthesia, minimum haemoglobin, vasopressor doses, VIS, IV fluid volumes and PPV), a logistic regression was employed. The odds ratio for these variables indicates the percent increase of the response variable per unit increase. For the discrete variables (i.e. patient comorbidities), Pearson’s chi-square test was used. The odds ratio for discrete binary variables represents the fold change in probability of having a present condition (1) relative to absent condition (0). For our multivariate binomial logistic regression model, we included all pertinent variables previously used in the univariate analysis. In total, the multivariate linear model included 21 terms with 20 variables. For all analyses, a two-sided t-test with a P < 0.05 was initially considered significant. Given that our statistical plan has three primary endpoints, we have implemented multiple test corrections in the form of a Bonferroni correction. Considering this correction, a more stringent threshold of P < 0.05 = 0.017 was required for statistical significance.
Results
Baseline characteristics of study population and perioperative data
Demographic information and pertinent patient comorbidities of the study population were collected, as well as relevant perioperative data (Table 1). Invasive blood pressure (BP) monitoring with PPV data were available for 1081 of the 1355 surgeries, with the remaining cases using non-invasive BP cuff measurements. Patients lacking PPV data were excluded from the analysis.
Patient demographics, comorbidities and procedural details.
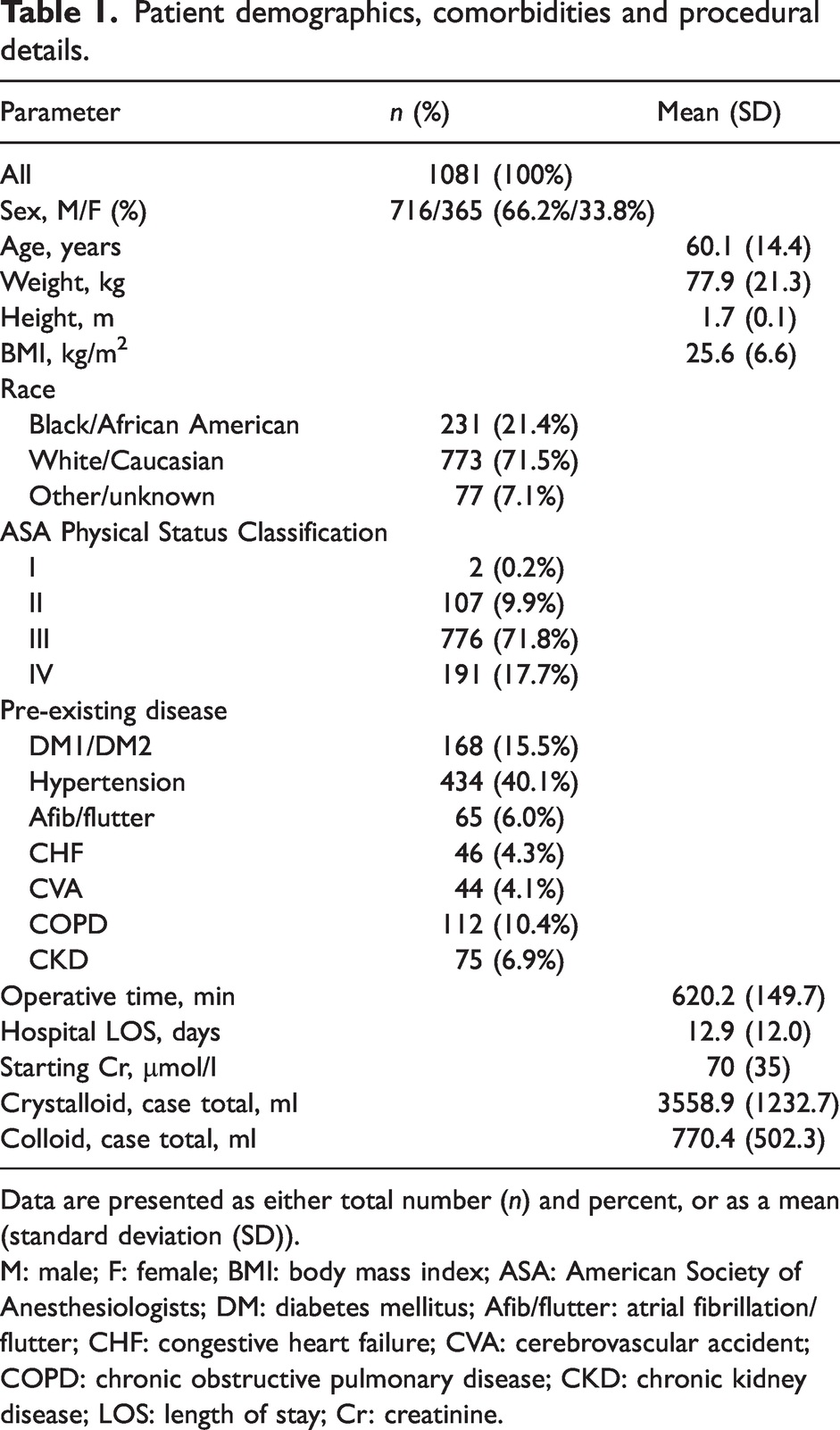
Data are presented as either total number (n) and percent, or as a mean (standard deviation (SD)).
M: male; F: female; BMI: body mass index; ASA: American Society of Anesthesiologists; DM: diabetes mellitus; Afib/flutter: atrial fibrillation/flutter; CHF: congestive heart failure; CVA: cerebrovascular accident; COPD: chronic obstructive pulmonary disease; CKD: chronic kidney disease; LOS: length of stay; Cr: creatinine.
Incidence of study endpoints
The incidence of the three outcomes was determined based on the predetermined diagnostic criteria described above. The most frequent endpoint was elevated lactate, which occurred in 265 cases (22.9%). There were 182 instances of postoperative AKI (14.1%). The rate of flap/graft dysfunction was extremely low; only 3.3% of cases were complicated by a surgical site complication requiring revision surgery within the seven-day time frame. This number is somewhat lower compared with complication rates previously reported in the literature.3,27 –29 Of note, although we report the rate of take-back surgery in our study, the actual rate of true flap failure is likely below the calculated 3.3%, presuming that some fraction of the salvage surgeries was successful.
Patient variables associated with study outcomes
Nine patient risk factors were included in our analysis: patient age, body mass index (BMI), diabetes (both insulin and non–insulin dependent), hypertension, atrial fibrillation/flutter, congestive heart failure, cerebrovascular accident, chronic obstructive pulmonary disease (COPD) and chronic kidney disease (CKD; all stages). Selection was based on a synthesis of several previous large scale observational studies of flap surgery patients.30,31 In our preliminary univariate analysis, heart failure, atrial arrhythmias, hypertension, CKD and age all bore an association with AKI, while BMI and COPD were associated with lactic acidosis (Table 2). Diabetes correlated with both AKI and lactic acidosis. No patient risk factors were associated with surgical site complications leading to re-exploration.
Univariate regression analysis of patient comorbidities, perioperative variables and study outcomes.
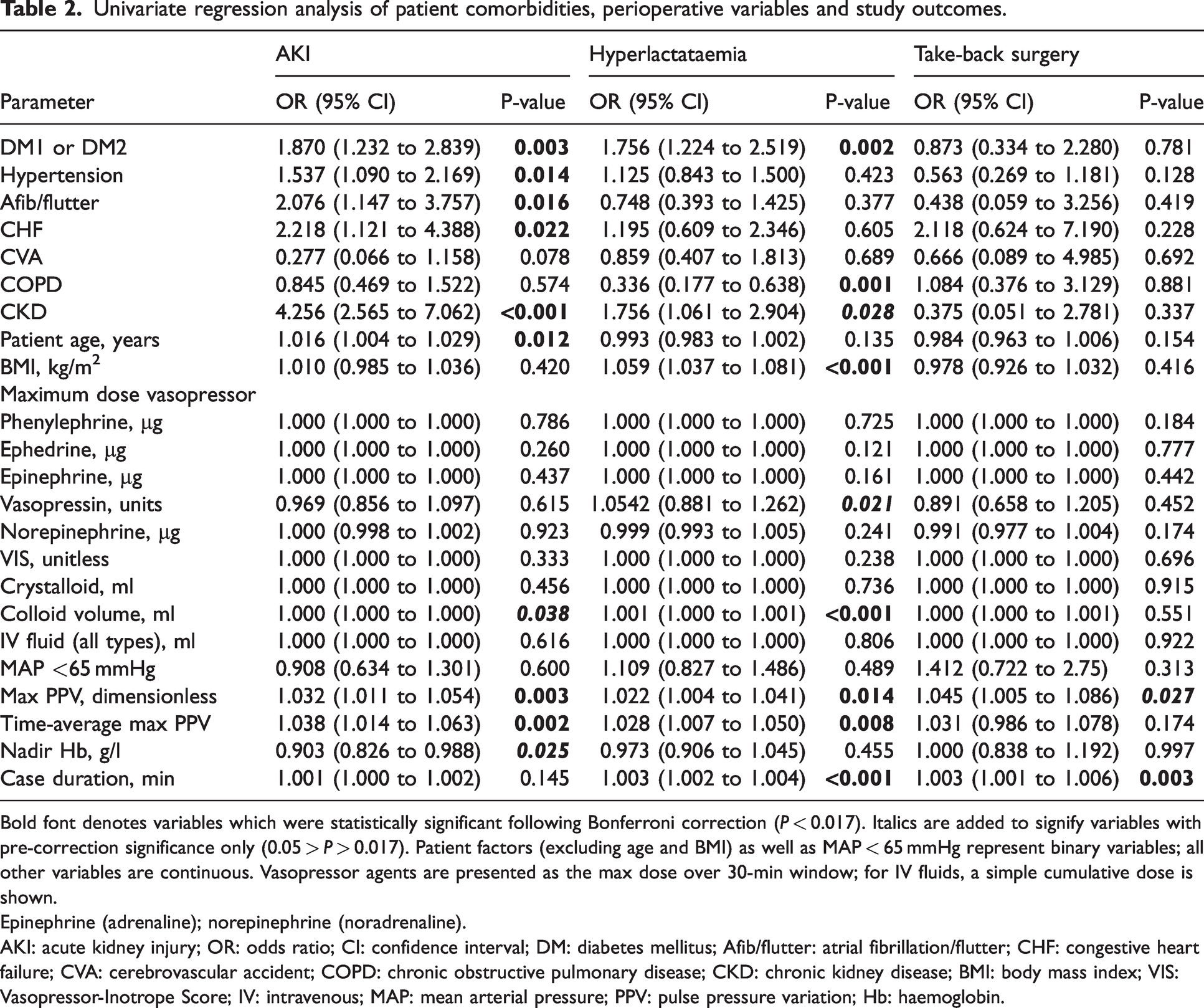
Bold font denotes variables which were statistically significant following Bonferroni correction (P < 0.017). Italics are added to signify variables with pre-correction significance only (0.05 > P > 0.017). Patient factors (excluding age and BMI) as well as MAP < 65 mmHg represent binary variables; all other variables are continuous. Vasopressor agents are presented as the max dose over 30-min window; for IV fluids, a simple cumulative dose is shown.
Epinephrine (adrenaline); norepinephrine (noradrenaline).
AKI: acute kidney injury; OR: odds ratio; CI: confidence interval; DM: diabetes mellitus; Afib/flutter: atrial fibrillation/flutter; CHF: congestive heart failure; CVA: cerebrovascular accident; COPD: chronic obstructive pulmonary disease; CKD: chronic kidney disease; BMI: body mass index; VIS: Vasopressor-Inotrope Score; IV: intravenous; MAP: mean arterial pressure; PPV: pulse pressure variation; Hb: haemoglobin.
Association of intraoperative haemodynamic variables with study outcomes
Both case duration and maximum PPV were associated with all three study endpoints in univariate regression at a P-value of 0.05 (Table 2). The odds of needing take-back surgery increased by 18% for each additional hour of operative time (or 0.3% per minute). For each percentage point increase in maximum PPV, the odds of take-back surgery increased by 4.5%. However, PPV lost significance when the more stringent Bonferroni correction was applied. Time-averaged maximum PPV is also associated with hyperlactataemia (P = 0.008) and AKI (P = 0.002), but not surgical take-back (P = 0.174). Time-averaged MAPs <65 mmHg (binary variable) showed no significant correlation with outcomes.
Univariate regression analysis of maximum vasopressor doses was performed for each agent individually. The use of vasopressin was associated only with elevated lactate (P = 0.021), but only prior to Bonferroni correction (Table 2). For each additional unit increase in the maximum dose of vasopressin, the odds of having lactate over 2 mmol/l increased by 5.4%. No other vasopressor was found to be associated with any study outcomes. To more uniformly capture the true amount of vasoactive support being administered over a given time frame, we calculated a VIS as described above. Similar to individual vasopressor calculations, the time-averaged maximum VIS did not show any association with study outcomes (Table 2).
We next considered how IV fluid administration might impact postoperative outcomes. The mean (SD) volume of crystalloid used in our population was 3558.9 (1232.7) ml (Table 1); for albumin, the mean (SD) was 770.4 (502.3) ml. Larger volumes of crystalloids did not appear to lead to any complications in the univariate regression. Albumin had a P-value <0.05 for both elevated lactate (P < 0.001) and AKI (P = 0.038); however, there was very low magnitude of effect, with an odds ratio of essentially 1 in both cases (Table 2). When all fluid types were considered in aggregate (total crystalloid + total colloid), the results were again non-significant. The seemingly identical confidence intervals (e.g. 1.000, 1.000) seen throughout Table 2 are secondary to rounding artefacts.
To further scrutinise these results, a linear multivariate logistic regression was performed, and utilised the same set of covariates included in the univariate analysis. Of the patient factors included, the multivariate regression showed BMI, COPD and CKD all associated with hyperlactataemia (Table 3). As expected, CKD history was also correlated with AKI (P < 0.001). Case duration once again was statistically significant for flap complications (P = 0.007) and hyperlactataemia (P = 0.003) although it lost significance for AKI (Table 3). Maximum PPV preserved its association with postoperative AKI (P = 0.003), but not either of the other two outcomes. Last, while vasopressin and albumin were both associated with complications in the univariate model, neither was so within the multivariate context.
Multivariate regression analysis of selected predictor variables and study outcomes.
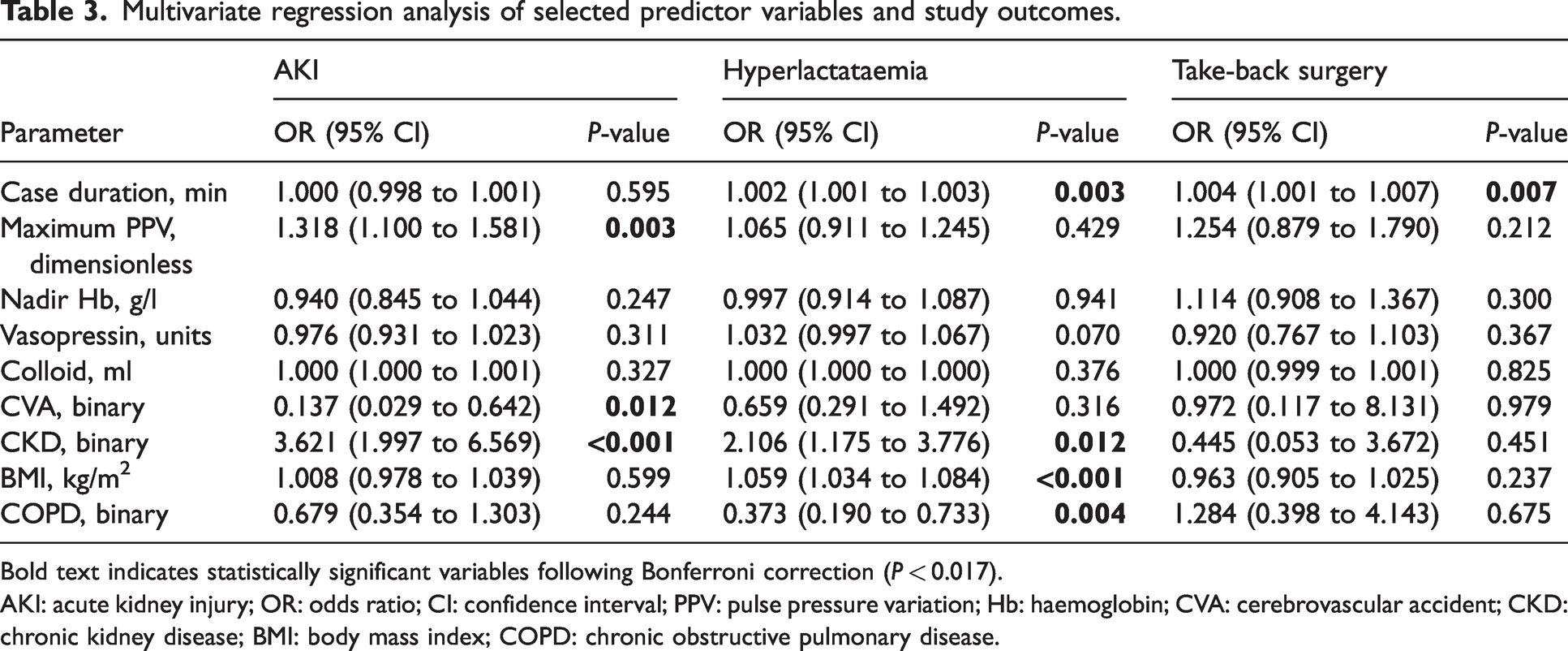
Bold text indicates statistically significant variables following Bonferroni correction (P < 0.017).
AKI: acute kidney injury; OR: odds ratio; CI: confidence interval; PPV: pulse pressure variation; Hb: haemoglobin; CVA: cerebrovascular accident; CKD: chronic kidney disease; BMI: body mass index; COPD: chronic obstructive pulmonary disease.
Discussion
Goal-directed perfusion has become a topic of considerable interest in perioperative medicine.32,33 Among the most useful and well-validated tools for guiding fluid and vasopressor administration is PPV. In ventilated patients, it functions as a dynamic indicator of a patient’s location on the Frank–Starling curve, in essence, a marker of preload responsiveness. 34 Recognition of elevated maximum PPV as a potential predictor of perioperative complications is noteworthy because, unlike other variables (such as case duration), maximum PPV is potentially modifiable by the anaesthesiologist, as the use of lower thresholds to give fluids might prevent further elevation. Of note, the great majority of patients in this study had a maximum PPV above 13% (online Supplementary material Figure 1), which is the commonly referenced threshold for preload dependence. 35 It is worth noting that the fluid totals in our study cohort were relatively low compared with older trials where goal-directed, fluid-sparing Enhanced Recovery after Surgery protocols had not been implemented worldwide. For example, a landmark 2001 study linking IV fluids with flap dysfunction did not observe an increase in complication rates until fluid administration exceeded 7 l. 36
When patients are relatively normotensive, the appropriate response to elevated PPV is more ambiguous. However, our results show that maximum PPV is an independent risk factor for AKI in flap surgery. Thus, one possible implication of this study is the potential for PPV monitoring to reduce the risk of postoperative complications. Unfortunately, we are not able to comment on our main aim, which was flap surgical outcomes. Although our data suggest that allowing extremes in maximum PPV can be harmful, it is not clear what range of PPV values should be targeted. Theoretically, it may be possible to mitigate postoperative morbidity by actively keeping PPV below a certain critical threshold, even in the setting of normotension. A future cohort study would be necessary to confirm these as predictors. Additional investigation is also needed to identify if and what PPV ceiling would be optimal.
The application of the VIS as an experimental tool to investigate flap reconstructions is another novel aspect of this work. Many previous studies have demonstrated no apparent increase in the incidence of flap/graft dysfunction in conjunction with vasopressor infusions when evaluated as a nominal variable.37 –40 However, the use of a composite variable reflective of total vasoactive dose burden in this study population has not been described. While it is difficult to assess the net effect of vasoactive support in retrospective analyses, especially when multiple agents are co-administered, the VIS score provides one means to simplify this process. In our multivariate analysis, neither the VIS nor any individual vasopressors were associated with perioperative complications.
Previous work has implicated excessive IV fluid use as a cause of flap dysfunction, presumably by increasing venous congestion and tissue oedema.15,36,41 However, our univariate logistic regression failed to demonstrate a link between IV fluids and flap complications requiring take-back surgery (or any other study outcome). The statistical significance seen with albumin use and AKI/hyperlactataemia in the univariate analysis illustrates the limitations of the P-value, as the odds ratio is of essentially no clinical significance. Furthermore, statistical significance was lost in the multivariate analysis.
Although preliminary univariate regression suggested a possible association of vasopressin with elevated lactate, we suspect this finding is confounded by the complicated clinical situations in which vasopressin is typically required. As vasopressin is not a first-line agent for hypotension, the need to resort to it (as either a bolus or infusion) likely signals the intraoperative course was characterised by significant vasoplegia or hypovolaemia. Thus, it more likely indicates an effect rather than a cause of an underlying process associated with poor perfusion. Moreover, these results were not recapitulated in the multivariate analysis.
Limitations of this study include the retrospective design, which did not allow for direct comparison between groups, for example, patients with either high or low PPV. In addition, the take-back rate for surgery was very low relative to the sample size of our study, which was unexpected. Our initial designs had hoped to look at flap failures as an indicator of perfusion. However, this objective was limited by study power, and thus precluded our ability to assess this variable more reliably. Consequently, these data must be interpreted with caution. We primarily view these findings as pilot data for a future project, and may serve a purpose in hypothesis-building. Subsequent work aiming to investigate factors associated with malperfusion and surgical flap complications will need to be considerably larger in scale. The inclusion of a modified VIS allowed for semiquantitative consideration of the net vasopressor burden used in these cases, an important factor not previously studied due to its complexity. However, this method has not been rigorously validated and warrants further investigation, with a promising role in studies of goal-directed perfusion.
Overall, our findings are in alignment with the prevailing view that a fluid-sparing approach, when mediated by indicators of poor perfusion such as high PPV, is safe and appropriate in tissue transfer surgeries, even when it necessitates early use of vasopressors. Rather than restricting fluids or avoiding vasopressors, our data suggest that perhaps a more important objective might be to avoid extremes in maximum PPV and, to the extent possible, limit the time spent under anaesthesia. Although case duration was significantly associated with take-back surgery, this relationship was likely confounded by surgical difficulty. We did not quantify or correlate surgical complexity, and it is difficult for the anaesthesiologist to modify or control surgical difficulty intraoperatively. On the contrary, PPV is well-understood, readily modifiable and also significantly associated with complications. Further investigation is warranted to explore novel ways to incorporate PPV targeting into goal-directed perfusion strategies.
Supplemental Material
sj-pdf-1-aic-10.1177_0310057X241275112 - Supplemental material for Factors associated with poor intraoperative perfusion and postoperative complications in otolaryngological autologous tissue transfers: A single-centre retrospective observational study
Supplemental material, sj-pdf-1-aic-10.1177_0310057X241275112 for Factors associated with poor intraoperative perfusion and postoperative complications in otolaryngological autologous tissue transfers: A single-centre retrospective observational study by Steven C Eastlack, Adriano A Bellotti, Wesley H Stepp, Joshua B Cadwell and Alan M Smeltz in Anaesthesia and Intensive Care
Footnotes
Author Contribution(s)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

