Abstract

Postoperative complications are increasing in both frequency and impact on patients, health service capacity, and sustainability. 1 Under-detected and under-managed postoperative complications on wards, due to factors such as low nursing ratios and sparse junior medical cover, are increasingly recognised as a major issue affecting care. 2 Approaches such as ward rescue to what is often late stages of deterioration through rapid response teams, and traditional mixed use high dependency units (HDUs) and intensive care units (ICUs), have often struggled to demonstrate improvements in outcome. It has been suggested that early specialised perioperative input from anaesthetists, coordinated with surgical and other perioperative specialists, may be an effective alternative. 3
These have evolved in some places in the UK, and units have appeared out of necessity during the COVID pandemic.
Two hospitals, one in the Netherlands and one in Australia, have independently evolved to models which pivot away from mixed use units and into specialised extensions of recovery room (post-anaesthesia care unit) care for medium risk non-cardiac surgical patients, and which sit alongside more traditional HDUs and ICUs. In light of some scientific evidence of benefit in terms of patient and hospital outcomes from this approach,4,5 it is interesting to compare and share experiences and approaches.
Some of the issues and thinking which underpin these models include:
A perioperative service which includes preoperative, and intraoperative, assessment and planning; Continuation of the anaesthesia–surgeon working relationship and culture, into the early postoperative period; Structure and process to underpin high quality standards and consistency, supported by quality measurement and improvement; A focus on very proactive multimodal management to hasten recovery and minimise the time in high acuity care, and in hospital overall.
Table 1 provides a comparison of the elements taken in each hospital which appear important to the benefits achieved. There is increasing interest in this type of approach to alternative models of recovery room utilisation and care in Australia, New Zealand, the Netherlands and other countries. 6 We sense a number of challenges and opportunities, including:
A comparison of two anaesthesia-led facilities which evolved independently and for which a level of evidence of benefit exists.
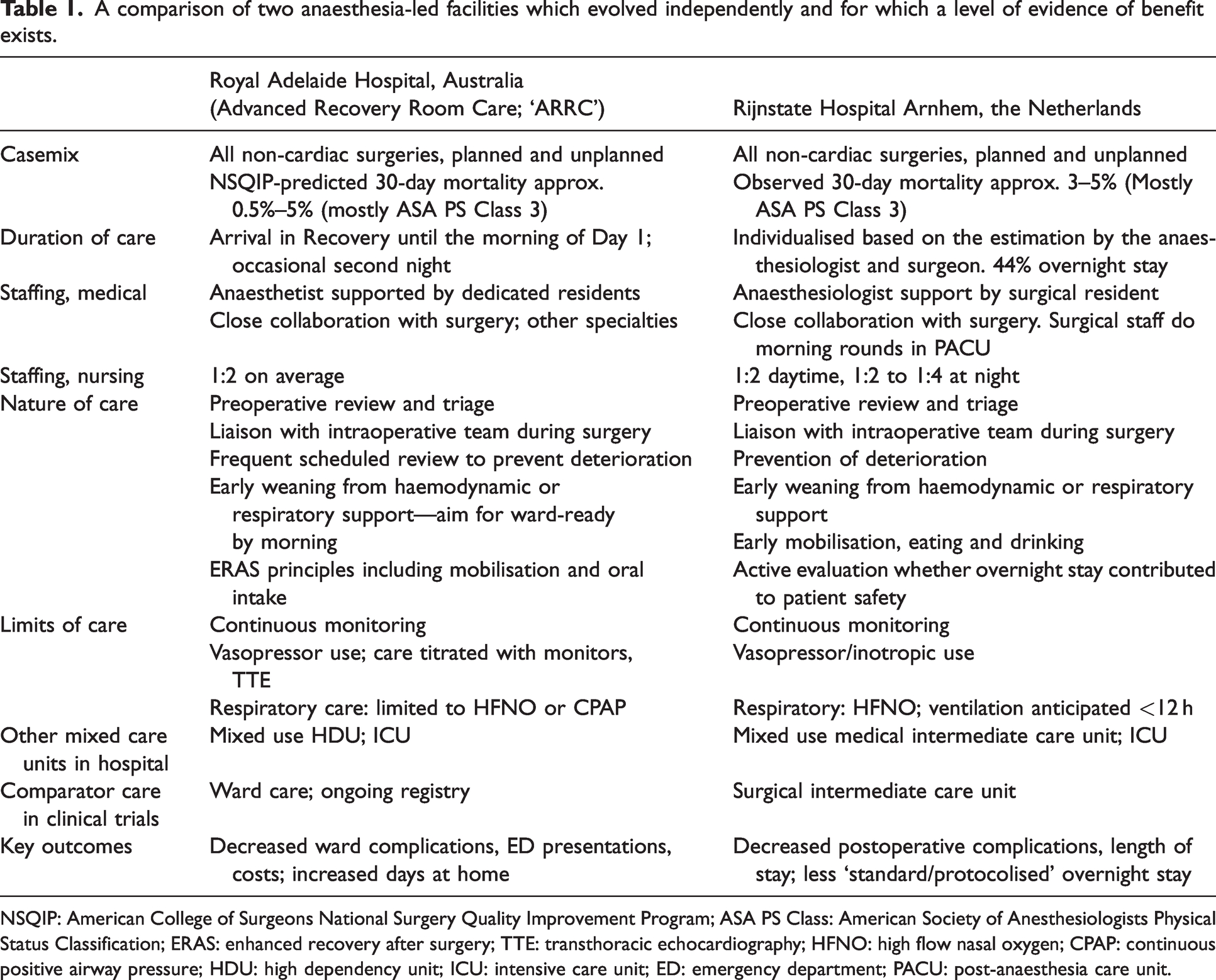
NSQIP: American College of Surgeons National Surgery Quality Improvement Program; ASA PS Class: American Society of Anesthesiologists Physical Status Classification; ERAS: enhanced recovery after surgery; TTE: transthoracic echocardiography; HFNO: high flow nasal oxygen; CPAP: continuous positive airway pressure; HDU: high dependency unit; ICU: intensive care unit; ED: emergency department; PACU: post-anaesthesia care unit.
Making sense of the nomenclature and models—several different names and models exist for such facilities, yet not all may provide benefit, or the same benefits;
Knowing what interventions make a difference, and why—low value elements are unhelpful in a resource-constrained future; there may be new technology or care modalities which further improve outcomes;
Which case-mix to select, and how—the ‘medium risk’ population to date appears a useful overall target based on published data, but benefits may vary with different surgeries and patient comorbidities, and higher- and/or lower-risk patients; use of conventional risk tools which predict 30-day mortality are at least available, but may not ultimately be the best to predict, for example, patients at risk of early deterioration;
Knowing the optimal endpoints to measure—what to measure and for how long to determine performance and value, and from what perspective.
There appears merit in a collaborative approach across and within countries to consider some of the issues raised, especially in an environment of increased interest in perioperative medicine and in a climate of increasingly scarce resources. High quality data, large patient numbers and careful analysis with relevant and validated outcome and cost endpoints will be necessary. A multicentre randomised trial seems unlikely, but pooled observational data can be very informative. Some of this work has commenced, and has started to identify key benefits and measurement endpoints of relevance to patients, providers and payers.
We recommend that, given the emerging evidence of benefit of this approach, and demonstration of feasibility in a range of settings, these models should be considered when looking to improve early postoperative care.
Footnotes
Author Contribution(s)
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
