Abstract
Skin injuries are a major healthcare problem that are not well understood or prevented in the critically ill, suggesting that underappreciated variables are contributing. This pilot study tested the hypothesis that perfusion-related factors contribute to skin injuries diagnosed as hospital-acquired pressure injuries (HAPIs). A total of 533 adult patients were followed over 2574 critical care days (mean age 62.4, standard deviation (SD) 14.3 years, mean body mass index 30.4 (SD 7.4) kg/m2, 36.4% female). This was a secondary analysis of prospective, non-randomised clinical data from an intensive care unit at a large urban teaching hospital. Factors related to perfusion, specifically two or more infusions of vasopressors/inotropes, temporary mechanical circulatory support (MCS), extracorporeal membrane oxygenation, and durable MCS, were analysed to determine whether they were more strongly associated with HAPIs than immobility due to prolonged mechanical ventilation (>72 h) or operating room time (>6 h). Patients diagnosed with a HAPI had a statistically significant higher risk of being exposed to variables related to perfusion and immobility (P < 0.05 for each variable). Perfusion-related variables, except durable MCS, had a larger effect on skin breakdown (number needed to harm (NNH) 4–10) than immobility-associated variables (NNH 12–17). The finding that perfusion-related variables predicted HAPIs may warrant consideration of alternative diagnoses, such as skin failure due to impaired perfusion as a pathophysiological process that occurs concurrently with multisystem organ failure. Differentiation of skin injuries primarily from circulatory malfunction, rather than external pressure, may guide the development of more effective treatment and prevention protocols. This pilot study suggests that the contribution of perfusion to skin injuries should be explored further.
Introduction
Skin breakdown in the critically ill has important implications for patient prognostication, recovery, morbidity, mortality, and family perception of care. The differential diagnosis for skin breakdown is broad, but pressure is a prevalent aetiology and major problem in Australia 1 and the United States 2 with more than 350,000 and 2.5 million pressure injury cases per year respectively, contributing to over 60,000 deaths in the latter. 3 The mortality risk associated with pressure injuries increases with the severity of pressure injury stage. 4 Pressure injuries are also associated with increased pain scores, rates of infection, and length of stay (LOS).5,6 Adult intensive care unit (ICU) patients are almost four times more likely to develop pressure injuries than their non-ICU acute-care patient counterparts, 7 with an incidence estimated at 10%–25%.8 –10
Hospital-acquired pressure injury (HAPI) cases, which are a designated nursing-sensitive quality indicator, 11 carry a huge economic toll for critical care health systems and patients. The annual total cost of chronic wounds in Australia was estimated at A$2.85 billion in 2012, which included pressure injury costs of A$1.64 billion. 1 This is comparable with other populations such as the United States where the total cost for HAPI cases was approximately US$10 billion, with a single HAPI incident costing hospitals up to approximately US$70,000, not counting the impact of malpractice.3,5,12 As such, there have been various quality improvement initiatives and systematic reviews for HAPI prevention.13 –15 However, despite such initiatives, pressure injuries have persisted, which has shifted the focus to finding significant risk factors and predictive models for injuries.16,17
Historically, the predictors and aetiologies of pressure injuries focused on sensory perception, moisture, mobility, nutrition, and shear.18,19 These factors unfortunately have significant heterogeneity and subjectivity alongside poor specificity and predictive value.18,19 This is probably because other factors make larger contributions to tissue survival, such as perfusion and oxygen delivery.9,20,21
The objective of the current study was to evaluate the association between perfusion with skin injuries classified as a HAPI. This hypothesis was tested in patients admitted to a cardiovascular intensive care unit (CVICU) by determining the correlation of factors related to perfusion/low flow states, such as infusions of two of more vasopressors/inotropes, temporary mechanical circulatory support (temp MCS), extracorporeal membrane oxygenation (ECMO), and durable mechanical circulatory support (i.e. left ventricular assist device (LVAD)). These perfusion variables were analysed to determine whether they were more strongly associated with skin injuries versus immobility due to prolonged mechanical ventilation or operating room (OR) time.
Methods
This cohort study was a secondary analysis of de-identified, prospective non-randomised, before and after clinical data. Adult patients were admitted to the CVICU at a large urban teaching hospital between 4 January 2018 and 30 June 2018 for the baseline group, and 16 September 2019 and 15 December 2019 during the quality improvement initiative. The primary study, during which data collection occurred, was a non-randomised, prospectively collected, before and after quality improvement project. The before group was a pressure mapping device study, which measured posterior pressure to identify patients at risk of skin injury, but had no intervention/pressure-relieving features. The after group study implemented an alternating pressure overlay device with the goal of decreasing the incidence, progression or severity of skin injuries due to pressure. The alternating pressure overlay device did not have an effect on skin injury outcomes (unpublished data). Institutional review board (IRB) approval was waived due to the study’s designation as a quality improvement initiative by the IRB/Human Research Protection Programs (PRO31758 and PRO35880 approved on 14 March 2018 and 16 September 2019, respectively). This quality improvement data were de-identified after collection, as recommended by the data analysts and compliance division. All aspects of this study were performed in accordance with institutional ethical standards of the responsible conduct of human research and the 1975 Declaration of Helsinki.
Data were extracted from the electronic medical record or acquired during daily chart audits for types of pressure-relieving surfaces and/or devices being used. Importantly, all the immobility and perfusion variables tested, as well as the comorbid conditions in the ICU, were present prior to and/or at the time the first skin injury developed. For example, the duration of mechanical ventilation might have been for 4 days or 2 weeks, but in either case there was greater than 72-h period prior to the first skin injury. All standard pressure injury risk assessments, skin assessments, and implementations were conducted in accordance with the hospital’s best practice bundle, including the Braden score, which consists of six subscales (sensory perception, moisture, activity, mobility, nutrition, friction/shear) and total scores that range from 6 to 23 with lower scores being worse. 19 If a HAPI was suspected, the hospital-approved procedure was followed by the bedside nurse, including documentation, notification, request for a wound, ostomy, continence nurse consult, and Safety Event reporting. Patients with device-related skin injuries were not excluded from this study because no patient had only a device-related skin injury and no other unrelated skin injury.
Comparisons were made for demographics, comorbidities that were present on admission, conditions developed in the ICU, as well as the immobility and perfusion variables to be tested, including mechanical ventilation greater than 72 h (Vent >72 h), cumulative OR time greater than 6 h (OR >6 h), two or more inotropes, temp MCS (intra-aortic balloon pump, Impella® Pertcutaneous Assist Device, (Abiomed Inc., Danvers, MA, USA), or Protek Duo (CardiacAssist Inc, Pittsburgh, PA, USA)), ECMO, and LVAD. Two or more inotropes was selected because prior literature has shown that this variable is associated with skin injuries and might be more specific than a single inotrope. 22
Descriptive statistics are presented as mean with standard deviation (SD), median with range for continuous variables, and counts with percentage for categorical ones. Between-group comparisons were performed using t-tests for continuous variables, Wilcoxon rank sum tests for ordinal measures, and chi-square tests for categorical outcomes; exact versions of the tests were used when the expected counts were under five for at least one cell. Number needed to harm (NNH) was calculated for each of the perfusion and immobility variables as the inverse of the risk difference between patients with and without the risk factor. Confidence intervals are presented using the method of Altman et al. 23
Results
The final study cohort included 533 patients admitted to the CVICU and followed over approximately 2574 critical care days. The patients had a mean age of 62.4 (SD 14.3) years, a mean body mass index of 30.4 (SD 7.4) kg/m2, and 36.4% were female (Table 1); these characteristics were similar between the groups that were and were not diagnosed with a new or worsened HAPI. The primary service for these patients was primarily 103 (39.2%) cardiothoracic surgery, 82 (31.2%) cardiology, 35 (13.3%) critical care anaesthesia, and 30 (11.4%) critical care medicine. Comorbidities and characteristics of pressure injuries present on admission were similar between the groups that were and were not diagnosed with a new or worsened HAPI while in the ICU (Table 2). A total of 17 unique patients in this cohort were diagnosed with new or worsened HAPIs while in the ICU (Table 3), which represents a 3.2% incidence per patient admission, or 6.6 patients diagnosed with HAPIs per 1000 critical care days.
Demographics and characteristics. Patients admitted that did or did not develop a new or worsened skin injury diagnosed as a hospital-acquired pressure injury while in the intensive care unit.
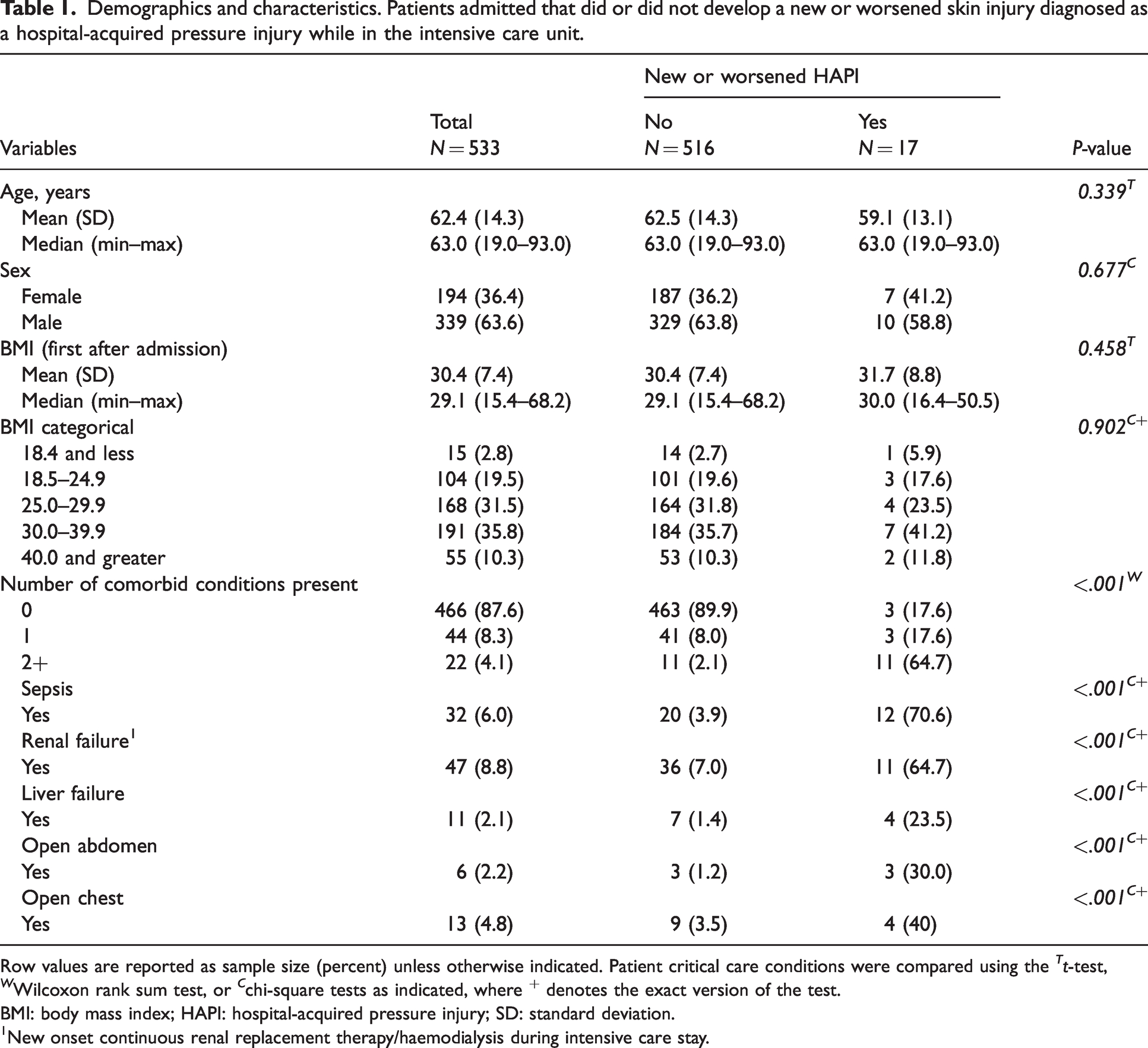
Row values are reported as sample size (percent) unless otherwise indicated. Patient critical care conditions were compared using the Tt-test, W Wilcoxon rank sum test, or C chi-square tests as indicated, where + denotes the exact version of the test.
BMI: body mass index; HAPI: hospital-acquired pressure injury; SD: standard deviation.
New onset continuous renal replacement therapy/haemodialysis during intensive care stay.
Comorbidities present on admission. Patients that did or did not develop a new or worsened skin injury diagnosed as a hospital-acquired pressure injury while in the intensive care unit.
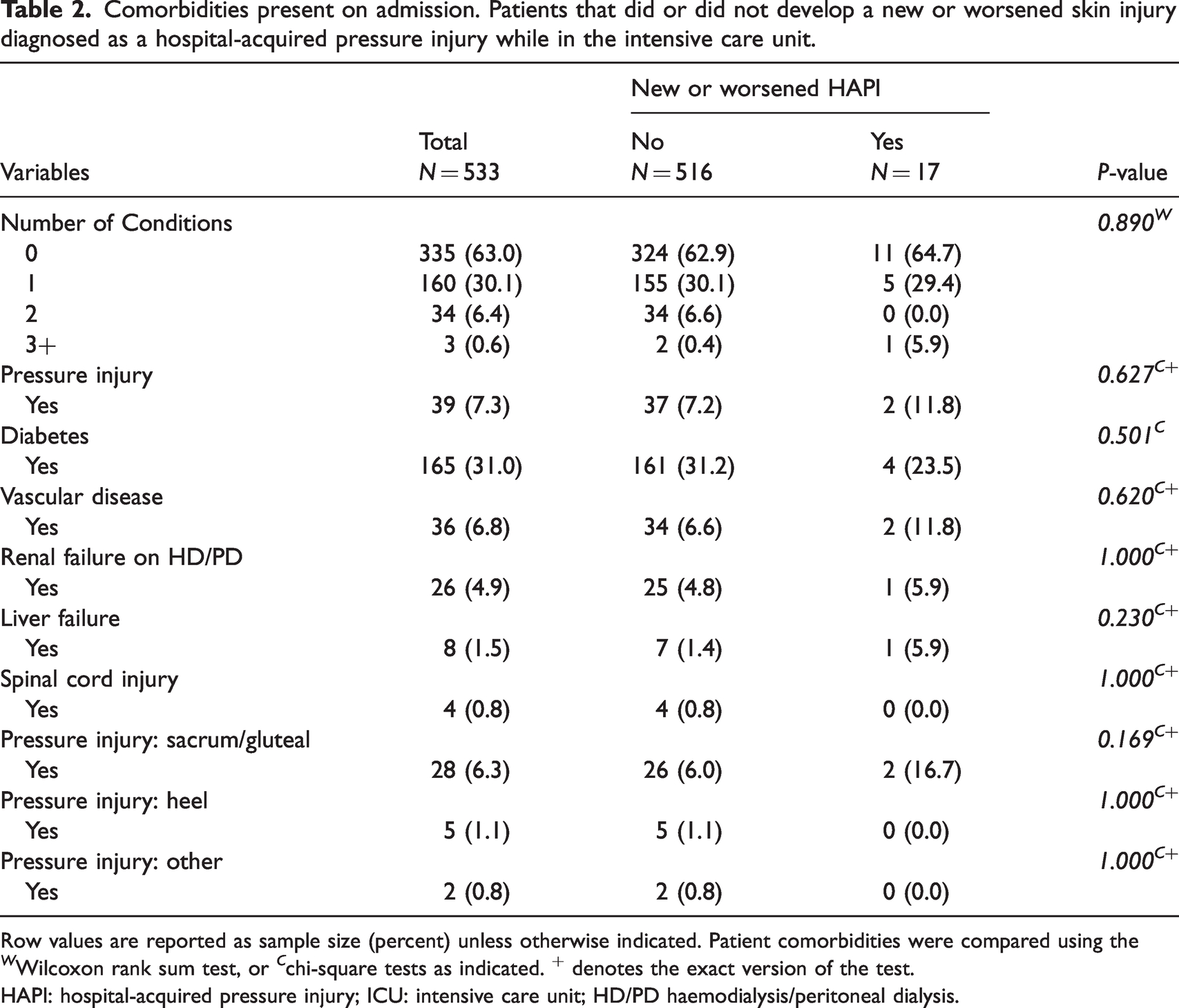
Row values are reported as sample size (percent) unless otherwise indicated. Patient comorbidities were compared using the W Wilcoxon rank sum test, or C chi-square tests as indicated. + denotes the exact version of the test.
HAPI: hospital-acquired pressure injury; ICU: intensive care unit; HD/PD haemodialysis/peritoneal dialysis.
Perfusion and immobility variables. Patients that did or did not develop a new or worsened skin injury diagnosed as a hospital-acquired pressure injury while in intensive care were compared for clinical interventions that treat poor perfusion or are associated with prolonged immobility.
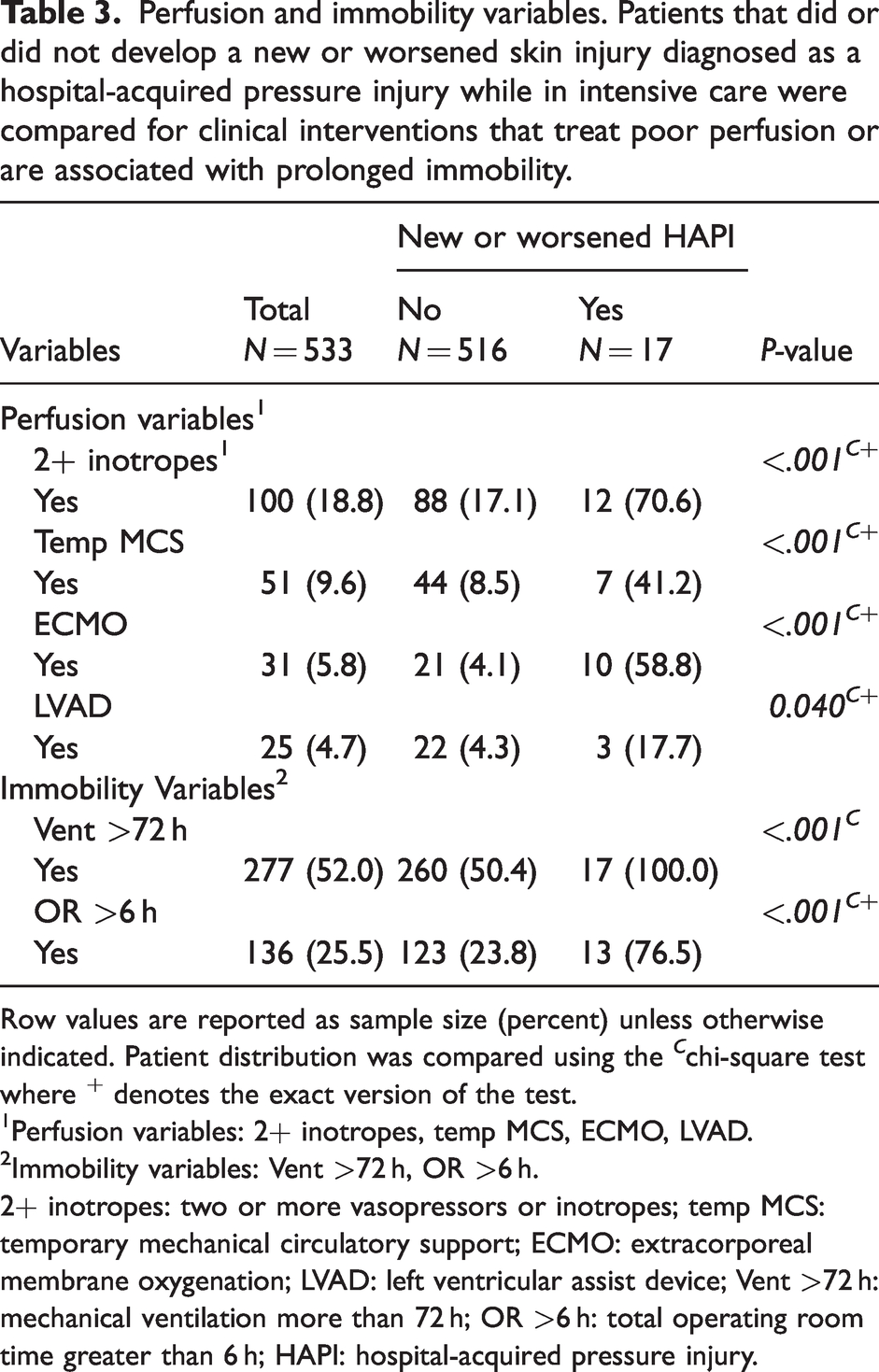
Row values are reported as sample size (percent) unless otherwise indicated. Patient distribution was compared using the C chi-square test where + denotes the exact version of the test.
Perfusion variables: 2+ inotropes, temp MCS, ECMO, LVAD.
Immobility variables: Vent >72 h, OR >6 h.
2+ inotropes: two or more vasopressors or inotropes; temp MCS: temporary mechanical circulatory support; ECMO: extracorporeal membrane oxygenation; LVAD: left ventricular assist device; Vent >72 h: mechanical ventilation more than 72 h; OR >6 h: total operating room time greater than 6 h; HAPI: hospital-acquired pressure injury.
Critical care conditions were significantly different between the groups that did or did not develop a new or worsened HAPI while in the ICU. Patients with HAPIs had significantly more hours of two or more inotropes, more hours of mechanical ventilation, more comorbid conditions present in the ICU (sepsis, renal failure, liver failure; Table 1), longer ICU and hospital lengths of stay (eTable 1), more hours dependent for mobility, higher number of times repositioned while dependent, lower Braden score on transfer, and greater hours with Braden mobility subscore <3 (eTable 2). Interventions to prevent HAPI were utilised more often in the group that developed injuries (eTable 2).
The distribution of patients that were managed with interventions to treat poor perfusion/low flow states or had clinical care associated with prolonged immobility was significantly different between patients developing a new or worse HAPI (Table 3). All perfusion and immobility variables tested, including two or more inotropes, temp MCS, ECMO, LVAD, Vent >72 h, and total OR >6 h, demonstrated significantly higher prevalence in the patients diagnosed with a new or worsened HAPI while in the ICU compared to those that did not (Table 3). The distribution of patients diagnosed with a new or worsened HAPI compared to those patients that were not, was significantly different in terms of the number that met perfusion or immobility criteria (Figure 1).
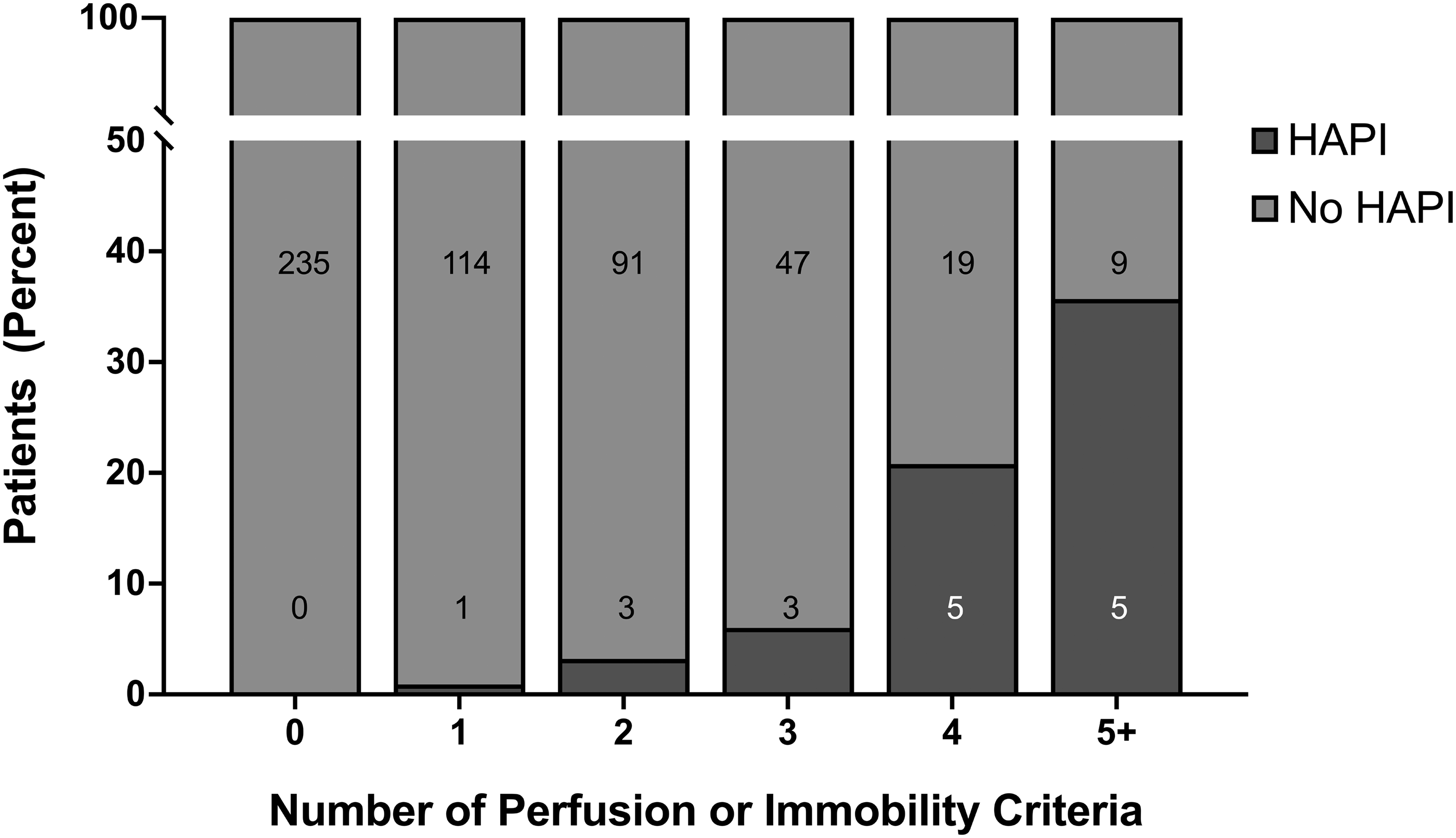
Number of perfusion or immobility criteria in patients with and without a HAPI diagnosis. The distribution of patients in the HAPI and no HAPI groups are significantly different based on the number of perfusion or immobility criteria met (P < 0.001) according to the Wilcoxon rank sum test. The number of patients in each group is identified within or immediately adjacent to the representative subcolumn. HAPI: hospital-acquired pressure injury.
Therefore, an NNH analysis for exposure to individual perfusion and immobility criteria was performed, which demonstrated 95% confidence intervals that did not include infinity, except for durable LVAD support (Table 4), consistent with their association to skin injury. The variables associated with perfusion, except LVAD, had a larger effect size on skin breakdown (NNH 4–10) than variables associated with immobility (NNH 12–17). Additionally, there was a significant increase in the risk of developing a skin injury when a patient was exposed to more than three of the perfusion or immobility criteria (Table 5).
Number needed to harm or benefit for exposure to individual criteria to contribute to skin injury.
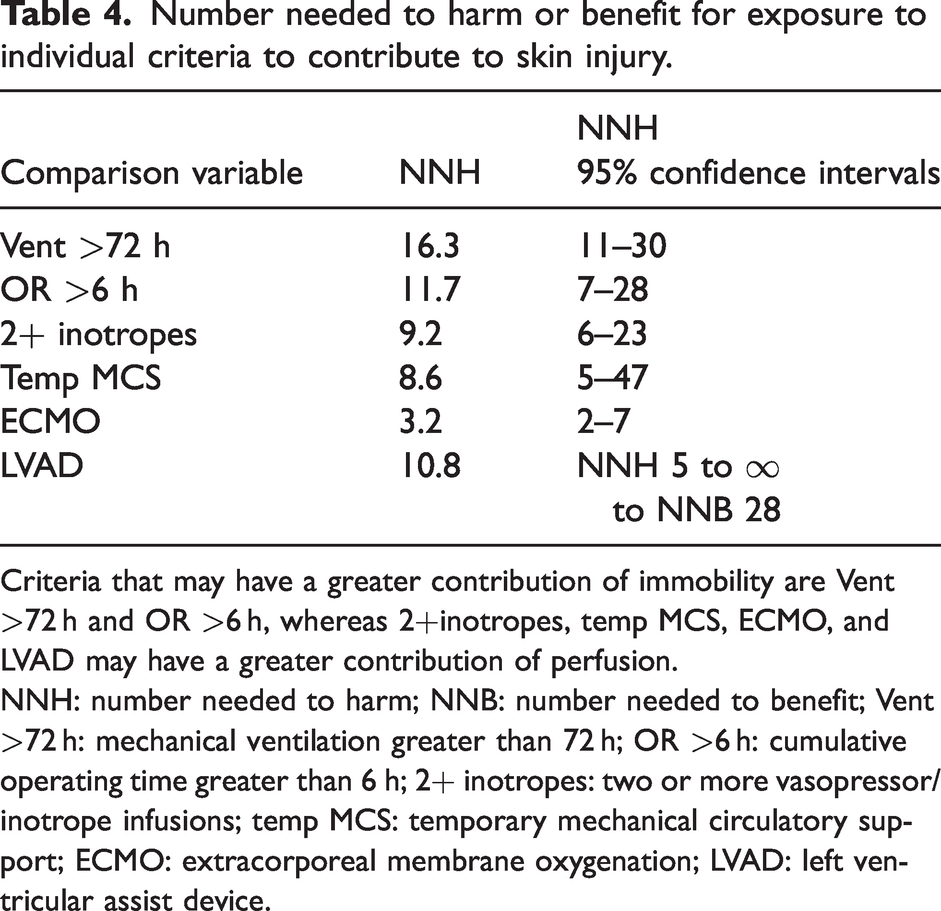
Criteria that may have a greater contribution of immobility are Vent >72 h and OR >6 h, whereas 2+inotropes, temp MCS, ECMO, and LVAD may have a greater contribution of perfusion.
NNH: number needed to harm; NNB: number needed to benefit; Vent >72 h: mechanical ventilation greater than 72 h; OR >6 h: cumulative operating time greater than 6 h; 2+ inotropes: two or more vasopressor/inotrope infusions; temp MCS: temporary mechanical circulatory support; ECMO: extracorporeal membrane oxygenation; LVAD: left ventricular assist device.
Number needed to harm or benefit for exposure to multiple criteria to contribute to skin injury.
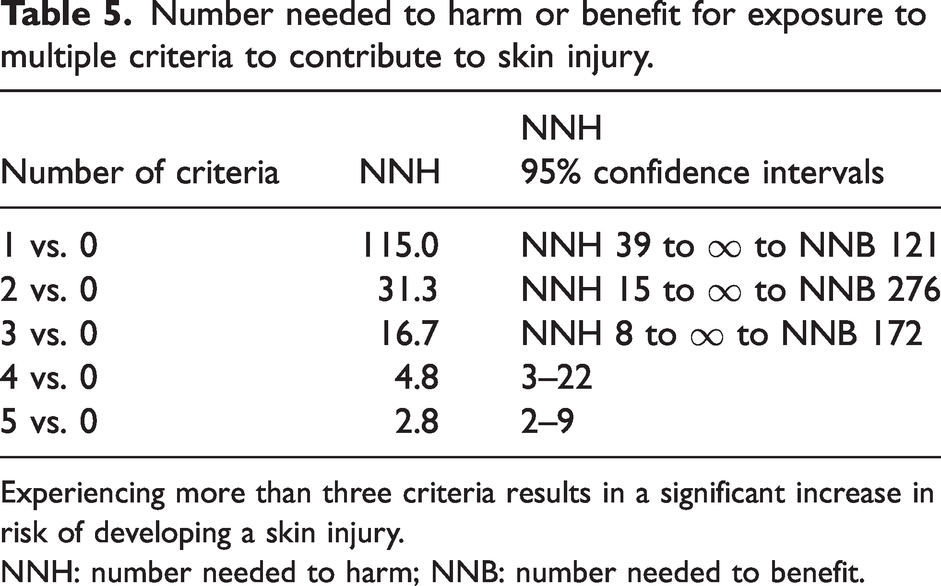
Experiencing more than three criteria results in a significant increase in risk of developing a skin injury.
NNH: number needed to harm; NNB: number needed to benefit.
Discussion
This data were consistent with the hypothesis that malperfusion due to cardiogenic shock is associated with an increased risk of skin failure, defined here as a pathophysiological process that occurs concurrently with multisystem organ failure due to circulatory malfunction.24 –27 The presumption being that these criteria reflect a treatment for the underlying pathophysiological process, namely cardiogenic shock, rather than increasing the risk of skin injury themselves. Criteria that may have had a greater perfusion contribution were two or more inotropes, temp MCS, ECMO, and LVAD, whereas mechanical ventilation >72 h and cumulative OR time >6 h may have had a greater contribution of immobility as a surrogate for prolonged exposure to pressure. Based on the NNH analysis, the perfusion-related variables had a larger effect size than variables associated with immobility. The exception to that pattern was the incidence of HAPI in patients with a durable LVAD, which was highly variable. This is probably because the trajectory of patients with a durable LVAD was bimodal, such that some had no complications and thus were protected from skin injury, while others were plagued by coagulopathy, fluid shifts, and right ventricle failure that contributed to prolonged low flow states and decreased perfusion of all organs, including the skin. The NNH analysis also demonstrated that temp MCS and ECMO had a lower NNH (suggesting a larger risk of skin injury) compared to two or more inotropes, which could be interpreted as reflective of disease severity given that inotropes/vasopressors are often first-line agents compared to MCS.
Preventing skin injuries has begun to evolve from pressure-relieving interventions to optimising skin perfusion and oxygenation, which has garnered increased attention and sophistication over the years. 28 For example, skin perfusion pressure (SPP), defined as mean arterial pressure (MAP) minus tissue pressure has been studied to determine the SPP threshold for the restoration of microcirculatory or capillary flow that would promote wound healing or avoid skin failure. 29 In a study of patients that underwent amputation, SPP ≥30 mmHg predicted complete healing in 90% of cases, while SPP <30 mmHg predicted the failure of healing in 75% of cases. 30 In another study of patients with foot ulcers, almost all healed cases had values of SPP >27 mmHg and those that did not heal were mainly associated with severe infections. 31 It would not be surprising if these thresholds were dependent on patient comorbidities. For example, the SPP threshold for detecting atherothrombosis complications in diabetic haemodialysis patients was found to be 10 mmHg lower than in non-diabetic haemodialysis patients. 32 Notably, that study found cut-off points at 53 and 62 mmHg, which is substantially higher than most studies that investigated 30–40 mmHg thresholds for skin healing. 29 While SPP values have been shown to be a good predictor of wound healing in patients with limb ischaemia it is unknown the extent to which the SPP threshold in a limb ischaemia patient population can be extrapolated to critically ill patients. For that matter, SPP might not be the ideal endpoint to target to protect the skin given that SPP would be a population-average target. Other non-invasive measures of skin blood flow using laser Doppler or tissue oxygenation are being investigated that could be implemented to individualise care.
The skin is particularly vulnerable during critical illness since decreased systemic perfusion results in adaptive mechanisms to protect visceral organs at the expense of the cutaneous circulation.24,28 Due to this, skin injuries are associated with multisystem organ failure and mortality, mostly as a marker of illness severity rather than as the cause of death.4,33 –35 Consistent with that notion, peripheral tissue perfusion is associated with organ failure and death in out-of-hospital cardiac arrest patients. 36 While there is extensive literature on the haemodynamic parameters that predict mortality,37,38 there is limited literature on specific perfusion variables and their thresholds for predicting skin failure or wound healing. Interestingly, the skin has been used as an endpoint to determine resuscitation efficacy based on increases to both skin blood flow and oxygen consumption for patients in shock, the majority of which were septic.39,40 Notably, skin perfusion is further diminished in the setting of tissue oedema due to fluid overload, which increases the risk of mortality 41 and probably increases the risk of skin injury, similar to oedema in other organs. 42 Also, it is not yet known whether different aetiologies of multisystem organ failure have unique thresholds for optimising tissue oxygenation to protect against skin injury. It would be reasonable to hypothesise that different diagnoses might have different thresholds because the underlying pathophysiology is distinct. This would be logical, but there are arguments supporting and refuting that hypothesis. Supporting the theory that thresholds will be different is that there are different underlying mechanisms. For example, even within distributive shock states, those induced by elevations in nitric oxide, such as sepsis, burns or pancreatitis, might respond differently than adrenal insufficiency or anaphylaxis as vasodilatory states. Even though all of the above distributive conditions are regularly temporised with similar vasopressors, they respond differently to treatments. For example, nitric oxide–mediated shock states respond well to anti-vasodilators, such as methylene blue and hydroxocobalamin (discussed further below). Also, increasing doses of norepinephrine (noradrenaline) to target higher MAPs can improve cutaneous microvascular flow and tissue oxygenation in sepsis, 43 but the same approach may be harmful in cardiogenic shock. Of note, vasopressors/inotropes do not necessarily restore normal flow to the microcirculation44,45 or autonomic control of specific vascular beds. 46 Likewise, the limited evidence in humans on whether current pulsatile flow technology improves patient-oriented outcomes primarily comes from cardiopulmonary bypass, which suggests that continuous non-pulsatile flow is mostly non-inferior to pulsatile perfusion, with the exception of marginal improvements on renal and pulmonary outcomes.47,48 These findings are, of course, in contrast to the durable LVAD literature that demonstrates that non-pulsatile flow is associated with compromised splanchnic blood flow, bowel ischaemia, feeding intolerance, and gastrointestinal bleeds.49 –51 The literature related to ECMO that supports that pulsatility improves microvascular perfusion due to endothelial/glycocalyx and inflammatory mechanisms is relatively limited to canine studies.52 –54 However, in support of the alternative hypothesis, that the risk of skin failure is not based on the indication for critical care, there is some data, albeit from a few relatively small studies. First, skin blood flow changes were independent of aetiology when comparing septic to cardiogenic/hypovolaemic shock.38,55,56 Furthermore, decreased sublingual microvasculature perfusion is associated with organ failure and death in patients following out-of-hospital cardiac arrest 36 and in septic shock. 57 These similarities between post–cardiac arrest syndrome and septic shock have been proposed to be due to similar autonomic nervous system changes, namely decreased sympathovagal tone. 58 Again, consistent with skin failure being a marker of disease severity across shock states of multiple aetiologies. Unfortunately, although sublingual measurements are convenient, minimally invasive, and common in temporary MCS studies, 59 skin perfusion does not correlate with sublingual microvascular flow index measurements, at least in septic patients. 60 This highlights again that all microvascular beds and shock states are not equivalent, which is especially troublesome for the study of the microcirculation.
Cardiogenic shock states treated with the criteria defined in this study as perfusion variables are, of course, disproportionately present in CVICUs, and that patient population may be at risk of skin injury that is refractory to pressure injury prevention bundle interventions. While the presenting problem in patients treated with MCS devices is often cardiogenic shock, vasoplegia is also a major problem. As such, vasoconstrictors can be a life-saving treatment, but they are associated with risks, 61 including increased rates of HAPI, most likely due to underlying disease severity and diverting blood away from the skin. 62 The usage of anti-vasodilators, such as methylene blue and hydroxocobalamin, has increased in vasoplegic patients63 –65 because they are able to reduce dependency on vasopressors while improving overall haemodynamics, microvascular perfusion, haemodynamic coherence, and perfusion pressure.66 –68 In fact, approximately 50% of patients on ECMO with vasoplegia can be expected to respond to methylene blue with a 10% increase in MAP, 69 and hydroxocobalamin can treat refractory vasoplegia after cardiopulmonary bypass.70 –72 However, hydroxocobalamin has also been reported to interfere with multiple laboratory tests. 73 Sepsis is the most common cause of distributive shock in an ICU setting, but this study could not control for sepsis as a covariate because two or more inotropes could be reflective of treatment of distributive shock and so would have resulted in duplication of a similar variable in the analysis. As such, this data cannot be used to speculate about the relative risk of skin injury due to cardiogenic shock compared to distributive shock. 44 Nevertheless, there is increasing evidence that nitric oxide pathways are disturbed in critically ill patients due to underlying pathophysiology and current care, which can contribute to microvascular vasoconstriction.74,75 As such, anti-vasodilators are promising treatments that may decrease the risk of skin injuries for CVICU patients, but larger randomised controlled trials are needed to confirm this.
There are several other limitations of this study given that it represents prospectively collected data from patients admitted to a single ICU that was retrospectively analysed to understand the effects of HAPI in this facility. Ideally, this study would have had a derivation and validation cohort, but this was not feasible given the rarity of skin injuries. Another limitation is the risk of confounding due to unmeasured variables, particularly due to allocation bias (i.e. the group of patients with cardiogenic shock that developed a new or worsened HAPI had a common cause besides the tested criteria that was linked to perfusion deficits or immobility). As such, the associations found here cannot prove causation. This study did not include LOS as a covariate in the model because both LOS and HAPI are outcomes that mutually affect each other. Also, a proper analysis to statistically control for LOS would require data on the timing of HAPI development, which was not available in the dataset. Of note, the analysis considered the incidence of each criterion separately, but most of the patients either met none of the criteria (Figure 1; n = 235) or met two or more criteria (n = 182) with 115 patients (22%) meeting a single criterion. Of the group meeting only a single perfusion or immobility criterion, one patient was diagnosed with a HAPI. Thus, the effect of having exactly one criterion had an NNH of 115, which was a weaker effect than any of the criteria by themselves (NNH 17 or lower), since many of those patients met two or more criteria. The NNH analysis should be interpreted with caution, in the setting of a univariate analysis, because there are confounds that are not accounted for and therefore should be taken as a quantification of the effect size and confidence intervals of a single variable. A multivariable analysis was not feasible due to the rare incidence of HAPI (n = 17) and the comparison with six perfusion/immobility criteria. The lack of a multivariate analysis greatly limits the confidence in these findings, which need to be replicated in well-designed studies. Although the care in this ICU was standardised based on bundles and protocols, the findings might not be generalisable to other ICU patient populations, even those that are predominantly treated with MCS. Several of these limitations are common for investigations into skin injuries due to the rarity of these events and heterogeneity between patients. Attempts were made to control for these by limiting the case to a single ICU that primarily treats patients with cardiogenic shock.
While the data presented here were consistent with our hypothesis, the findings from this pilot study should be considered preliminary due to the above limitations. This investigation might be useful as a guide for well-designed, appropriately powered trials that can implement multivariate models to test the contribution of perfusion to skin failure. The DecubICUs study did not find an association between vasopressor use and skin injuries, 4 but such a large-scale epidemiological study is not optimal for a targeted scientific question. Furthermore, the DecubICUs study tested for any vasopressor use rather than two or more vasopressors as done in this study, which might be more specific for predicting skin injuries.
The incidence of skin injuries is likely to continue to increase given that advancements in critical care are allowing for the survival of an ageing population with a higher severity of critical illness. 76 The findings presented here suggest that cardiogenic shock variables may have a greater correlation with skin injuries compared to immobility or skin pressure variables. As such, skin failure is likely to be underdiagnosed, but notably the definition still needs consensus from interprofessional and multidisciplinary stakeholders.24 –27 Further studies are necessary to refine the variables tested to control for disease severity to determine which clinical variables are most amenable to interventions to optimise skin perfusion. Studies should also test whether cardiogenic shock or distributive shock variables increase the risk of skin failure to a greater degree. Investigations into the pathophysiological conditions that contribute to skin failure in critically ill patients are essential so that personalised physiological endpoints can be targeted with individualised treatments.
Supplemental Material
sj-pdf-1-aic-10.1177_0310057X241264575 - Supplemental material for Skin injury: Associations with variables related to perfusion and pressure
Supplemental material, sj-pdf-1-aic-10.1177_0310057X241264575 for Skin injury: Associations with variables related to perfusion and pressure by Christopher J Roberts, Jennifer A Popies, Abrahim N Razzak, Xi Fang, Octavio A Falcucci and Paul J Pearson and Aniko Szabo in Anaesthesia and Intensive Care
Footnotes
Author contribution(s)
Acknowledgements
The authors would like to thank the entire interprofessional and multidisciplinary team that serves patients and their families in the CVICU for tirelessly striving to deliver exceptional care and for their support of the data collection used in this project as well as in clinically piloting the algorithm developed as a result.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the United States Department of Veterans Affairs Biomedical Laboratory Research and Development Service Career Development (award no. IK2 BX005600 (CJR)) and by the Medical College of Wisconsin Departments of Anesthesiology and Surgery, Division of Cardiothoracic Surgery.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
