Abstract
In Australia, neuromuscular blocking agents are the leading cause of perioperative anaphylaxis. Current investigation of suspected anaphylaxis includes tryptase levels, serum immunoglobulin E (IgE) levels, and skin testing, including intradermal testing and skin prick testing. The gold standard for the diagnosis of a hypersensitivity reaction is a challenge test, but this poses a risk to patient safety. An alternative test, known as the basophil activation test (BAT) is a form of cellular in vitro testing using flow cytometry to measure the degree of basophil degranulation within a sample of blood following exposure to an allergen. This acts as a surrogate marker for mast cell and basophil activation, thereby identifying IgE-mediated allergy. It is most commonly used to supplement equivocal findings from initial in vitro testing to assist in confirming the diagnosis of a hypersensitivity reaction and identify the causative agent. We present a case series in which five patients with suspected anaphylaxis underwent a BAT, demonstrating its role and limitations in allergy testing within Australia.
Introduction
Current investigations for identifying the causative agents of perioperative anaphylactic reactions include skin testing and specific immunoglobulin E (sIgE) levels. The gold standard for drug allergy testing is an in vivo challenge test,1 –3 but this may pose a risk to patient safety. An alternative test, known as the basophil activation test (BAT), utilises flow cytometry to measure the degree of basophil degranulation in a peripheral blood sample after stimulation with an allergen. 4 A positive BAT result has the potential to identify the medication incriminated in causing anaphylaxis without exposing a patient to the significant risk of a drug challenge. Studies have shown the BAT to be beneficial as a supplemental test to assist in identifying the causative allergen in cases of suspected perioperative anaphylaxis when routine investigations are equivocal or negative.3,4 Despite this, the BAT is not yet commercially available in Australia and is predominantly used only as a research tool due to various limitations, which include accessibility, cost, lack of technical expertise, lack of standardised testing3,4 and limited clinical validation. 5 We present a case series in which the BAT was successfully used to identify the medication implicated in perioperative anaphylaxis and discuss its role in perioperative allergy testing in an Australian context.
Case series
In our perioperative allergy testing centre, 242 patients were investigated for suspected perioperative anaphylaxis based on clinical history between 2015 and 2021. Of those patients, five were identified as needing further investigations and underwent a BAT and provided consent for publication in this case series. In each case, there was a strong clinical suspicion of perioperative anaphylaxis, but initial testing (sIgE levels, dynamic tryptase and skin testing) demonstrated ambiguous results. The perioperative anaphylaxis workup results are summarised in Table 1.
Summary of drugs administered prior to suspected anaphylaxis and the results of tryptase, skin and basophil activation tests.
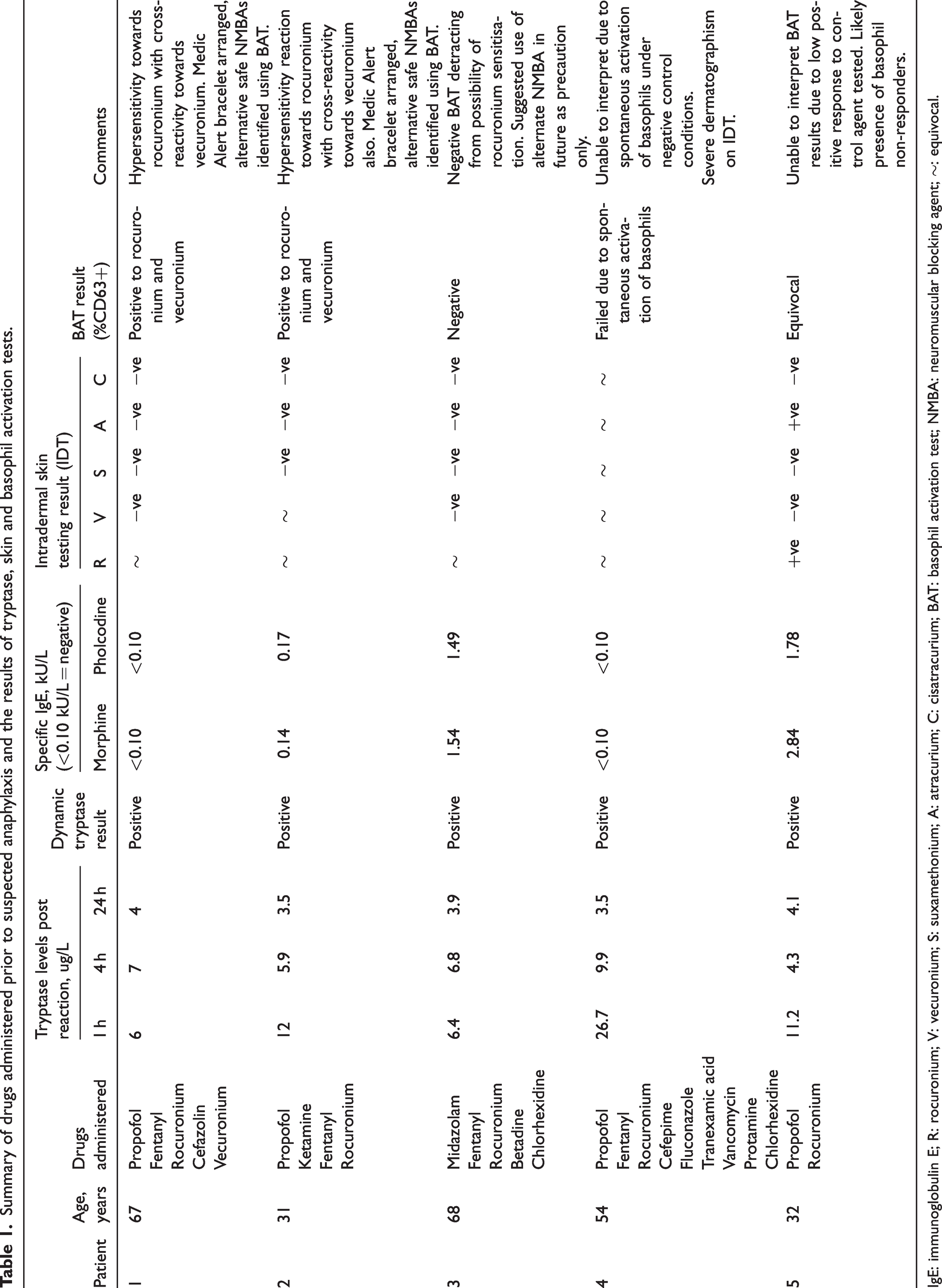
IgE: immunoglobulin E; R: rocuronium; V: vecuronium; S: suxamethonium; A: atracurium; C: cisatracurium; BAT: basophil activation test; NMBA: neuromuscular blocking agent; ∼: equivocal.
Case 1
A 67-year-old female developed features suspected to be anaphylaxis during induction of anaesthesia and the procedure was abandoned. She had no significant past medical history, or history of previous anaesthetic complications. Dynamic tryptase level (increase of peak tryptase by 120% of baseline plus 2 μg/L) 3 was elevated at this time. Intradermal testing (IDT) revealed an equivocal response towards rocuronium and negative cross-reactivity testing of other neuromuscular blocking agents (NMBAs). Skin prick testing (SPT) was performed, which was positive for rocuronium only. The surgery was rescheduled with vecuronium as the NMBA of choice instead. However, following induction, clinical features suggestive of a Grade 1 anaphylaxis became evident and the surgery was completed with symptomatic management. On repeat skin testing, all NMBAs tested negative on both IDT and SPT. Considering these ambiguous results, a BAT was arranged to safely assist in identifying the causative agent, as well as suitable alternative NMBAs for future use. The BAT showed a positive reaction to rocuronium, a low positive reaction to vecuronium suggestive of sensitisation (Figure 1), and a negative reaction to atracurium. This patient later went on to have an uneventful general anaesthetic, with atracurium as the NMBA of choice.
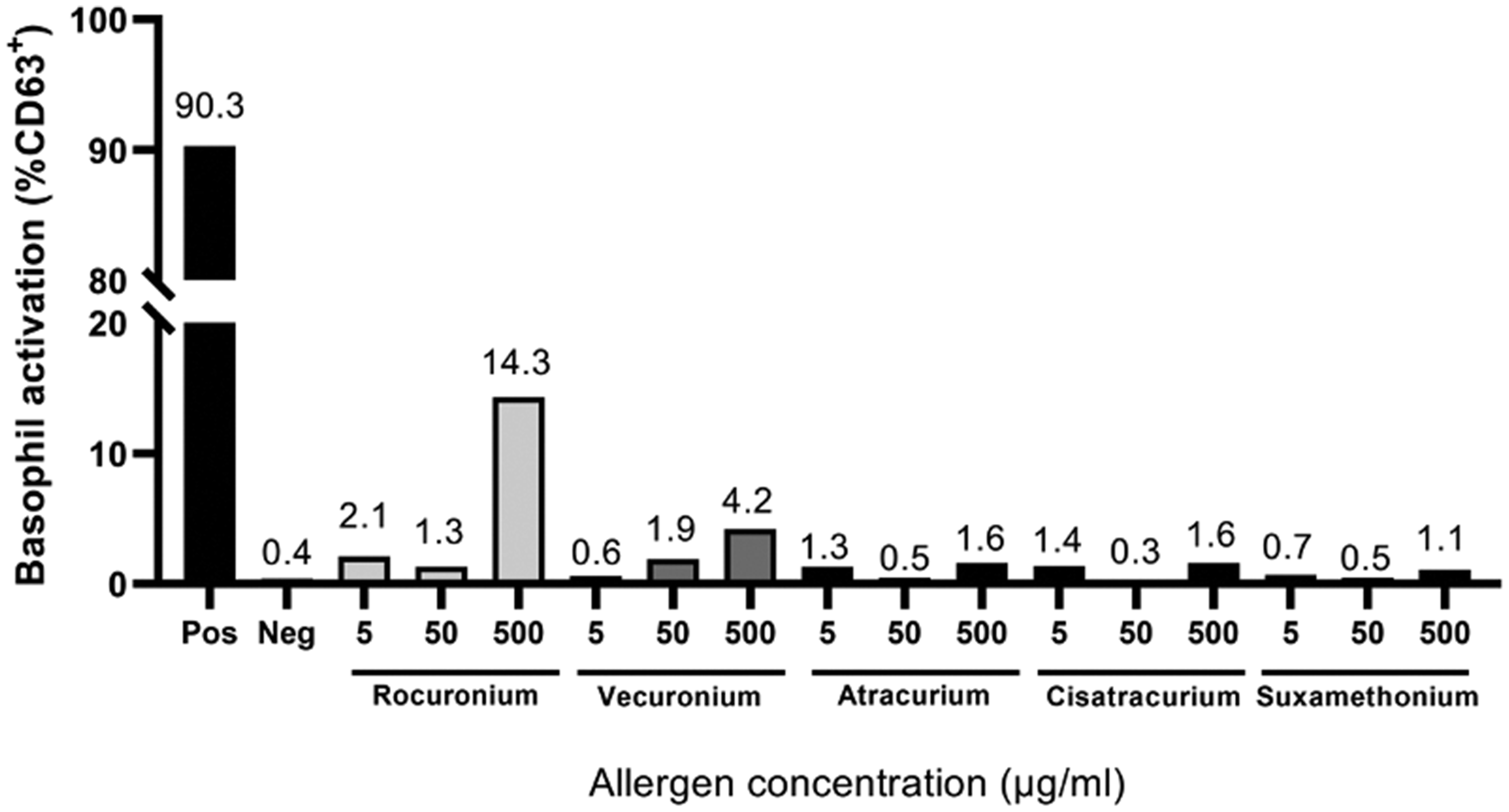
Basophil activation test results showing positive reactions to rocuronium and vecuronium.
Case 2
A 31-year-old female underwent an exploratory laparotomy during which she developed clinical features consistent with severe anaphylaxis shortly after induction. Serum mast cell tryptases performed following the episode were supportive of anaphylaxis and she was subsequently referred to our centre for perioperative allergy testing. One year prior, she had had an episode of cardiac arrest secondary to suspected perioperative anaphylaxis. At that time, dynamic tryptase levels and skin testing were negative. Due to a high clinical suspicion of rocuronium anaphylaxis, a challenge test was performed, but was declared negative after 4.6 mg of rocuronium was uneventfully administered. A presumptive diagnosis of non-IgE-mediated anaphylaxis was made. Of note, she was prescribed several antipsychotic medications, which had not been withheld during her previous skin testing. Antipsychotic medications are known to interfere with skin testing results due to their antihistamine side-effects. 6
In our centre, after discussion with her psychiatrist, we temporarily withheld one of her antipsychotic medications for 7 days and skin testing was repeated with the same drugs used on induction. Only rocuronium returned an equivocal result, raising suspicions for possible sensitisation. Skin testing of additional NMBAs to assess for cross-reactivity returned equivocal results for vecuronium and suxamethonium. Due to the severity of the patient’s previous reactions and her comorbidities, an alternative test such as a BAT was preferred. It showed a clear positive reaction to both rocuronium and vecuronium, thereby suggesting a cross-reactivity towards vecuronium (Figure 2).
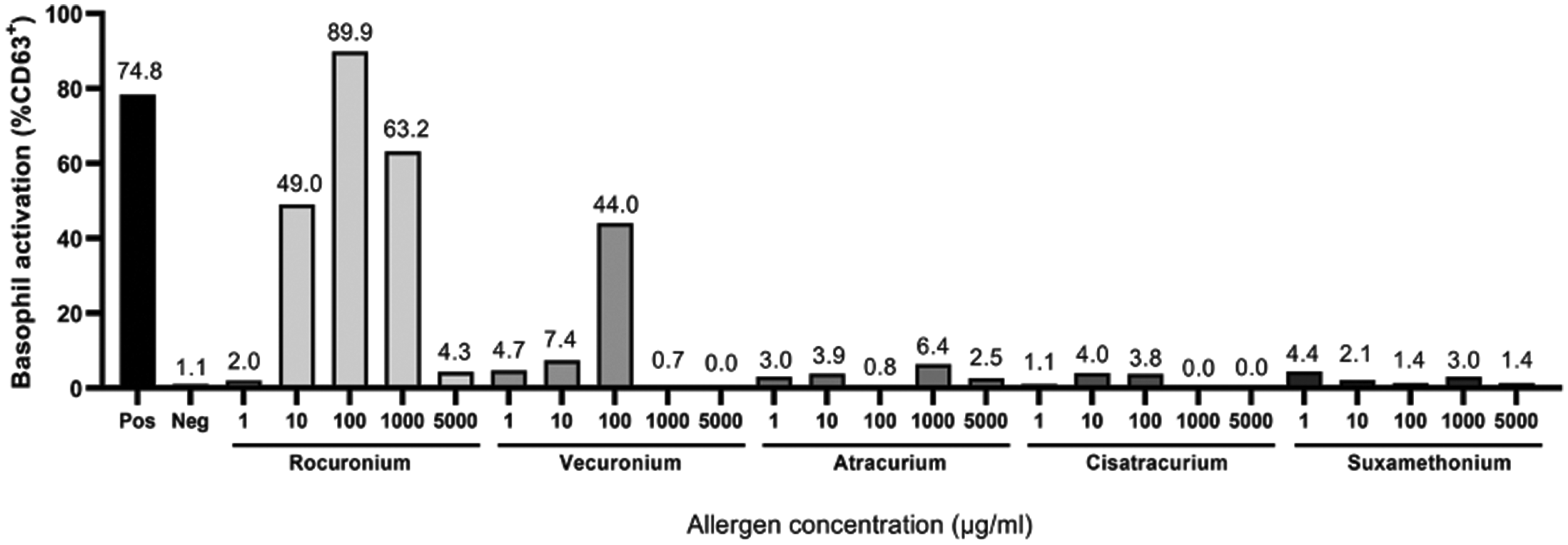
Basophil activation test results showing positive reactions to rocuronium and vecuronium.
Case 3
A 58-year-old female underwent open aortic valve replacement for severe aortic stenosis. Following anaesthesia induction with midazolam and fentanyl, and the administration of rocuronium, features of anaphylaxis were noted. Dynamic tryptase results were negative and initial skin testing showed negative results to all drugs tested. Specific IgE levels to morphine and pholcodine were positive at 1.54 and 1.49 kU/L, suggesting sensitisation to NMBAs. Due to a strong clinical suspicion of anaphylaxis, skin testing was repeated at a later date. On the second occasion, she demonstrated an equivocal response to rocuronium. Given the conflicting results, a BAT was performed, which showed no evidence of reactivity towards rocuronium or vecuronium, overall suggesting no anaphylaxis towards these agents (Figure 3). In light of this, a direct provocation test was offered to the patient, but was declined due to personal stressors. Ultimately, anaphylaxis was considered unlikely, and her profound hypotension was attributed to her comorbidities.
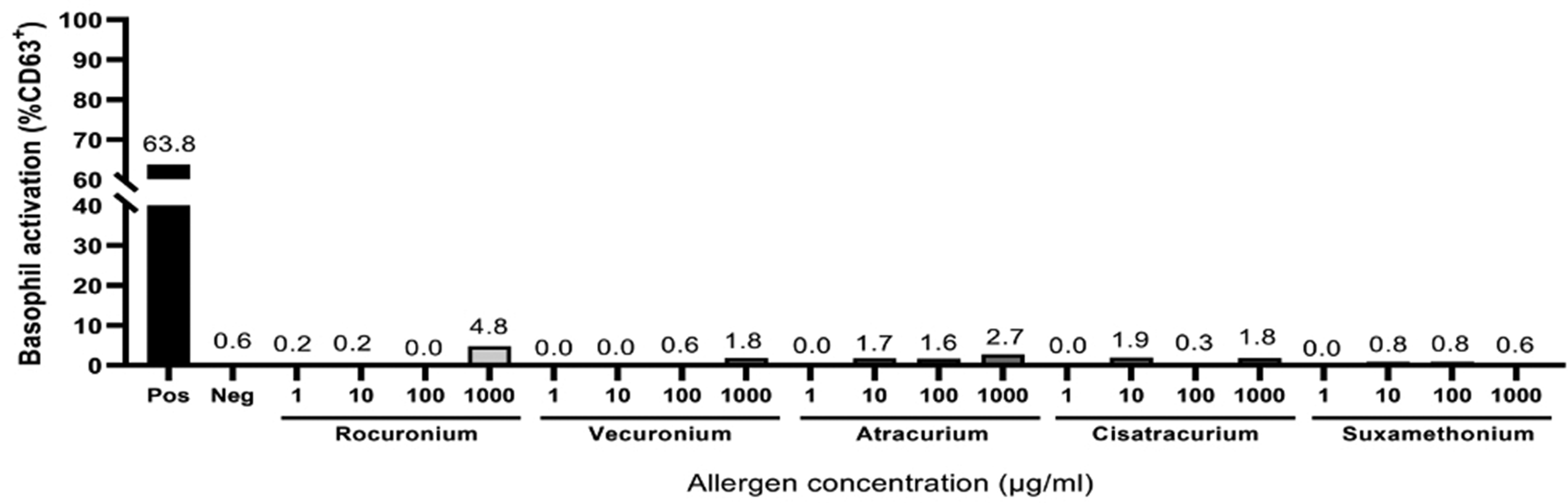
Basophil activation test results showing no evidence of sensitisation towards various neuromuscular blocking drugs.
Case 4
A 55-year-old male underwent left ventricular-assist device insertion during which he developed suspected severe anaphylaxis manifesting as significant hypotension. Dynamic tryptase was positive, but skin testing was complicated by severe dermatographism, whereby the patient had a wheal and flare reaction to all drugs tested including the negative control, rendering it uninterpretable. Repeat testing revealed similar results. Due to severe cardiac comorbidities, the patient was not suitable for a challenge test. A BAT was arranged instead. The BAT demonstrated spontaneous activation of basophils to all samples tested, including the negative control (Figure 4). Hence, the BAT was considered inconclusive, without a clear causative agent for his clinical presentation. He is now awaiting surgery with an anaesthetic plan that avoids any drugs with a high probability of causing anaphylaxis.
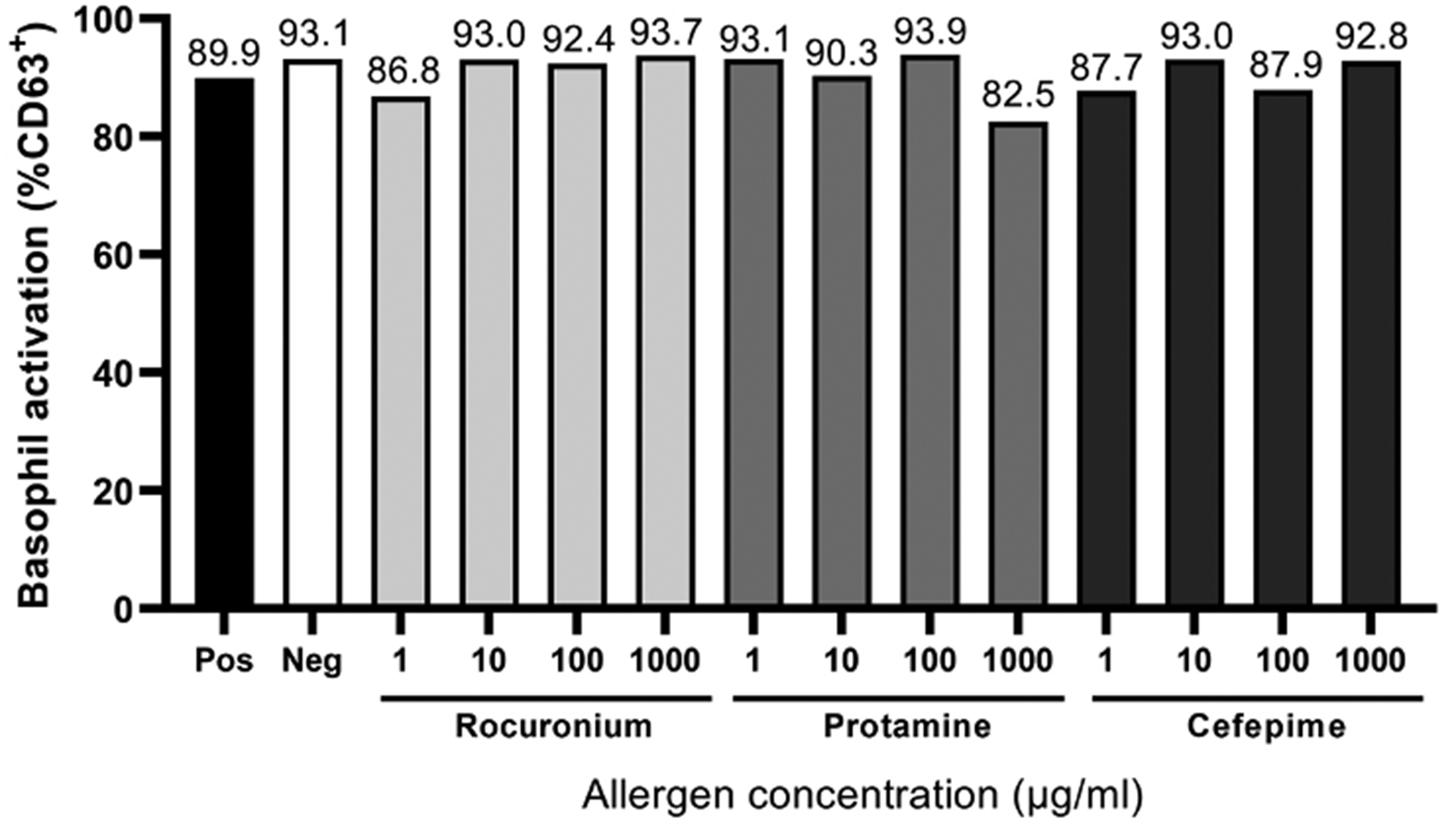
Basophil activation test results showing spontaneous activation of basophils under negative control conditions.
Case 5
A 32-year-old female developed suspected anaphylaxis during laparoscopic surgery resulting in procedural abandonment. Dynamic tryptase levels were positive. The patient had a history of severe needle phobia, and skin testing was a prolonged process, which was unfortunately poorly tolerated by the patient, despite the use of pharmacological anxiolysis. Initial skin testing revealed an equivocal response to rocuronium and atracurium. Specific IgE levels to both morphine and pholcodine were also positive. Repeat testing later revealed a positive response to both rocuronium and atracurium but a negative response for other NMBAs tested. Due to these results, a BAT was arranged. The BAT demonstrated the ‘non-responder’ phenomenon, seen in a minority of patients 2 (Figure 5). This was evidenced by the lack of response to the positive control utilised in the BAT. As such, the remainder of the BAT results could not be accurately interpreted. Subsequently she tolerated a direct provocation with cisatracurium and an alert was created to avoid both rocuronium and atracurium. She is awaiting further direct provocation testing with vecuronium and suxamethonium.
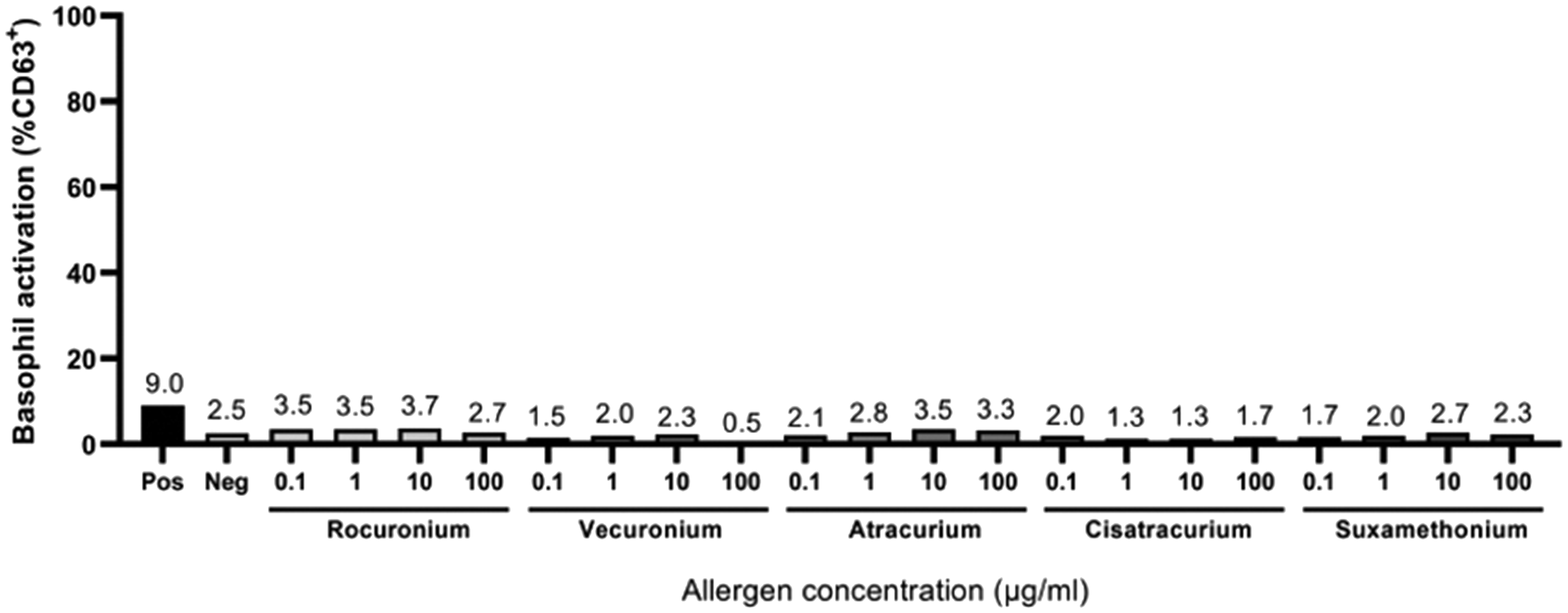
Basophil activation test results showing evidence of non-responder basophils, demonstrating no reaction to all drugs tested, including the positive control (consistent with equivocal results).
Discussion
In this case series, we explored the utility of the BAT in situations with a strong clinical suspicion of anaphylaxis with ambiguous test results. Of the five cases assessed with a BAT, two had clear identification of the causative agents, and one had a definite exclusion of anaphylaxis. The remaining two were inconclusive due to patient factors attributed respectively as the non-responder phenomenon and spontaneous basophil activation.
Routine investigations of suspected perioperative anaphylaxis include serum tryptase (dynamic as well as peak) levels. Specific IgE levels and skin testing (SPT or IDT) are then recommended to complete the workup to identify the culprit drug causing the allergic reaction. Evaluation of sIgE antibodies is available for a select group of perioperative drugs and their utility is limited by their poor predictive value 7 and hence, the results should always be interpreted in conjunction with the clinical manifestation and skin testing results. 6 Measuring sIgEs to NMBAs are limited to suxamethonium and rocuronium in some centres.
In our centre, skin testing is performed as IDT based on standardised concentrations as recommended by the Australian and New Zealand Anaesthetic Allergy Group (ANZAAG) guidelines. Intradermal testing involves assessing all drugs administered during the suspected perioperative anaphylaxis incident, but may also include additional drugs where clinically relevant, such as in the case of NMBA or antibiotic cross-reactivity screening. Relevant sIgE levels, where available, are measured to corroborate the diagnosis.
These routine investigations, along with the clinical history, may be sufficient to support the diagnosis of anaphylaxis. However, cases exist in which there is a strong clinical suspicion of perioperative anaphylaxis, with negative or equivocal serum tryptase and skin testing results. An anaphylactic reaction cannot safely be excluded based on these results alone, and suitable alternative drugs should be identified for future anaesthesia. A provocation test is the gold standard,1,2 but this is not without significant risk and impracticality, such as in cases of testing numerous NMBAs. 8 The BAT presents a minimally invasive alternative option for patients. 4
The BAT is a form of cellular in vitro testing that utilises flow cytometry to measure the degree of degranulation following exposure to an allergen, acting as a surrogate marker for mast cell and basophil activation.1,4,5 The BAT measures the expression of basophil activation markers, which are upregulated following IgE cross-linking and basophil degranulation, indicating the proportion of basophils that respond to a given stimulus.3,4 A positive BAT result is suggested to equate to an immediate IgE-mediated allergic reaction. 5 The most commonly used marker in the BAT is CD63 followed closely by CD203c. 5 CD63 is expressed on the inner leaflet of preformed cytoplasmic granules and is detectable on the basophil surface upon fusion of granules to the cytoplasmic membrane in the process of degranulation. The results are usually expressed as a percentage of upregulation above the negative control, and the stimulation index, which is the ratio of the fluorescence index of the stimulated sample and the negative control. 3 The testing centre in our state considers a BAT to be positive when the percentage of basophils that are CD63 positive is greater than 5% that of the positive control. A test is considered equivocal when the negative control meets this definition of positive, or when the positive control (non-specific stimulation with polyclonal anti-human IgE antibodies) yields less than 15% CD63 positivity (non-responder). It is worth noting that the optimum concentration of the test drugs (NMBAs) for basophil activation appears to be between 100 and 500 μg/mL. This is much higher than for protein allergens, for which optimal is usually around 1 μg/mL. Interestingly, we see a similar dose effect as with protein allergens, wherein basophil activation begins to decrease at allergen concentrations past the optimum.
The BAT has been found to be beneficial in supporting the diagnosis of IgE-mediated anaphylaxis where initial investigations have been shown to be equivocal.3,4,8 The BAT has repeatedly demonstrated high diagnostic accuracy in the perioperative setting.3,4,8 In general, perioperative hypersensitivities were identified with a sensitivity of 50% to 90% and specificity of more than 90% with the BAT. 8 In Australia, NMBAs and antibiotics are the leading cause of perioperative anaphylaxis.1,2 In a 2019 Australian study with 120 subjects, Li et al. demonstrated a sensitivity of 77% and specificity of 76% to NMBAs. 3 Santos et al. demonstrated that the BAT specifically for rocuronium has a sensitivity of 80% and a specificity of 96%. 4 They also demonstrated a sensitivity of 55% and a specificity of 80% for beta-lactam antibiotics. 4 Takazawa et al. showed variable sensitivity for NMBAs in general, ranging from 36% to 91% and a specificity between 81% and 100% for the BAT. 8 When compared with sIgE levels, the BAT demonstrated a higher sensitivity of approximately 50% and a specificity of 90% overall, making it a useful adjunct in the diagnosis of perioperative anaphylaxis. 8 For a detailed description of the BAT and its application, readers are directed to a recent publication. 3 The BAT has been shown to be beneficial across numerous studies, but the literature recommends its use only in specific scenarios, namely where tryptase levels, sIgE levels and skin testing is equivocal.3,4 ANZAAG recommends skin testing after 4 to 6 weeks post incident. The BAT may be useful where early testing is unviable – for instance, semi-urgent surgery after the initial event. 9
The BAT cannot currently be used routinely due to various limitations including limited accessibility by testing sites, technical requirements, cost, the presence of non-responders and a lack of standardised testing.3,4 Currently the BAT in South Australia is entirely funded by research grants and is not subject to Medicare rebate, and as such is limited to a few selected cases. It is accessed by directly liaising with immunologists with a special interest in this modality. Patients undergoing the BAT are required to present to one specific pathology clinic in which their test will be performed. This is largely due to the technical requirements of the test. The BAT should be performed on a fresh sample of blood, ideally as soon as possible, but realistically within 24 h of collection.3,4,10 Delays in testing the sample result in a reduced basophil reactivity over time, potentially leading to false negatives.4,5 Additional testing requirements also stipulate that the test must be performed within 1 year of exposure to the suspected allergen.5,10
A proportion of patients who undergo a BAT are deemed to have ‘non-responder’ basophils. 5 These basophils will elicit a response to a non-IgE-mediated positive control, but not to an IgE-mediated stimulus. 5 This results in an inconclusive test in this population as seen in Case 5. Furthermore, there is a lack of standardised testing for the BAT both locally and globally, and this is largely credited as a significant contributor to a lack of its availability as a clinical diagnostic test.4,5,11 Different testing sites utilise different methodologies in order to perform the test, all of which cause variability between BAT results. 11 This includes use of different anticoagulant agents in the testing sample, the use of different basophil activation markers (e.g. CD63 versus CD203c), and the use of different cut-off measurements and different drug dilutions to determine whether a BAT is positive.3 –5 This variability compromises the diagnostic accuracy and reproducibility of a result, which would hinder the validation of this test.3,5 A standardised protocol would need to be adopted in order to overcome this issue.4,8 Despite this, in our experience, NMBAs are peculiar amongst non-protein allergens in consistently eliciting a strong basophil activation. Few non-protein molecules efficiently cross-link surface IgE, 12 which has led to difficulty in defining cut-offs for positivity for other small-molecule drugs.
Due to these various limitations of the BAT and its restricted availability across the country, no current ANZAAG guidelines exist for its use, and it is predominantly used in research settings. The existing literature suggests it should be used in cases in which initial testing (e.g. dynamic tryptase levels, sIgE testing or skin testing) results are equivocal or negative, but there remains a strong clinical suspicion for anaphylaxis. Until these issues are addressed, the BAT will continue to exist as a research tool and will not be readily implemented as a diagnostic test despite promising results.3 –5
One of the limitations of our case series is the possibility of selection bias. Not all cases of suspected perioperative anaphylaxis were referred for a BAT, rather, only those in which the initial tests returned equivocal results, thereby limiting the validation of the test.
Conclusion
Our case series highlights the role of the BAT in the investigation of suspected cases of perioperative anaphylaxis. In most of the cases a BAT provided additional benefits by facilitating the use of safer alternative drugs in the place of the identified allergen. Strong clinical suspicion of anaphylaxis should always warrant further investigation, and in equivocal cases, the BAT assists in identifying the causative allergen without compromising patient safety.
Footnotes
Author Contribution(s)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
