Abstract
Patients who exhibit high systemic inflammation after cardiac surgery may benefit most from pre-emptive anti-inflammatory treatments. In this secondary analysis (n = 813) of the randomised, double-blind Intraoperative High-Dose Dexamethasone for Cardiac Surgery trial, we set out to develop an inflammation risk prediction model and assess whether patients at higher risk benefit from a single intraoperative dose of dexamethasone (1 mg/kg). Inflammation risk before surgery was quantified from a linear regression model developed in the placebo arm, relating preoperatively available covariates to peak postoperative C-reactive protein. The primary endpoint was the interaction between inflammation risk and the peak postoperative C-reactive protein reduction associated with dexamethasone treatment. The impact of dexamethasone on the main clinical outcome (a composite of death, myocardial infarction, stroke, renal failure, or respiratory failure within 30 days) was also explored in relation to inflammation risk. Preoperatively available covariates explained a minority of peak postoperative C-reactive protein variation and were not suitable for clinical application (R2 = 0.058, P = 0.012); C-reactive protein before surgery (excluded above 10 mg/L) was the most predictive covariate (P < 0.001). The anti-inflammatory effect of dexamethasone increased as the inflammation risk increased (–0.689 mg/L per unit predicted peak C-reactive protein, P = 0.002 for interaction). No treatment-effect heterogeneity was detected for the main clinical outcome (P = 0.167 for interaction). Overall, risk predictions from a model of inflammation after cardiac surgery were associated with the degree of peak postoperative C-reactive protein reduction derived from dexamethasone treatment. Future work should explore the impact of this phenomenon on clinical outcomes in larger surgical populations.
Introduction
Systemic inflammation is central to the development of chronic disease.1,2 Lifestyle factors that associate with low-grade chronic inflammation are linked to poor health outcomes. 3 Several anti-inflammatory treatments are established to slow the progression of heart disease and cancer.4 –6 In the acute setting, systemic inflammation is associated with higher mortality in severe sepsis 7 and trauma. 8 Given its importance in these varied conditions, it is plausible that systemic inflammation is causally related to morbidity after surgery.
Cardiac surgery with cardiopulmonary bypass (CPB) is a particularly potent inflammatory stimulus. 9 Exposure of tissues to high levels of circulating cytokines after surgery has been posited as a cause of organ dysfunction and poor outcomes. 10 Consequently, a number of large multicentre trials have evaluated the anti-inflammatory effects of perioperative glucocorticoid administration on mortality and major morbidity.11 –14 Two of these trials focus specifically on cardiac surgery populations,13,14 including the Intraoperative High-Dose Dexamethasone for Cardiac Surgery (DECS) trial. 13 Although neutral results have been reported in all these trials, the point estimates for the primary outcome effect have consistently favoured glucocorticoid administration, raising the possibility that there are subpopulations that derive benefit, and subpopulations that are unaffected or even harmed. Indeed, in a recent study of patients experiencing systemic inflammation and sepsis, four distinct phenotypes were identified that were likely to respond differently to various treatments including anti-inflammatory agents. 15
The surgical patients who are most likely to derive net benefit from anti-inflammatory treatments are those experiencing the most profound inflammatory responses. This was recently described as ‘postoperative systemic inflammatory dysregulation’, defined on the basis of exaggerated increases in circulating concentrations of C-reactive protein (CRP). 16 CRP is an acute phase protein that is primarily synthesised in the liver following exposure to the dominant postoperative inflammatory cytokine, interleukin-6 (IL-6). 17 Increases in IL-6 in turn are proportional to the degree of tissue injury during surgery and the subsequent burden of damage and pathogen-associated molecular patterns. 18 Routine serial CRP measurement during surgical admission has become common in some institutions to indicate recovery from postoperative inflammation or to facilitate early detection of secondary infections. 19 At present, alternative measures of inflammation are not available in large surgical populations due to technical or cost-based restrictions.
Tools to accurately distinguish patients who are vulnerable to high levels of postoperative inflammation as measured by CRP are lacking. In other settings, similar risk-prediction tools have successfully identified heterogeneity of treatment effects and now underpin precision-medicine approaches. 20 We therefore set out to develop an inflammation risk model based on CRP responses in patients enrolled in the DECS trial, 13 with a view to exploring glucocorticoid treatment-effect heterogeneity according to inflammation risk.
Materials and methods
Data from patients in the DECS trial 13 enrolled at the Isala Clinics (Zwolle, The Netherlands) were analysed, as frequent perioperative CRP measurements with a standardised normal-sensitivity assay were routine at this site. The DECS trial methods (NCT00293592) and results have been published elsewhere, including details of research ethics committee approval. 13 Briefly, adult patients undergoing any type of elective or urgent cardiac surgery with CPB at eight centres in The Netherlands (n = 4494, 2006 to 2011) were randomly assigned to receive a single intraoperative dose of 1 mg/kg dexamethasone intravenously after induction of anaesthesia but before initiation of CPB. No additional perioperative glucocorticoid treatments were permitted. Exclusion criteria included an emergent or off-pump procedure and a life expectancy of less than 6 months. The primary outcome was a composite of death, myocardial infarction, stroke, renal failure, or respiratory failure within 30 days of randomisation. Data collection encompassed the following demographic and clinical covariates in the preoperative phase: age, sex, hypertension, diabetes mellitus, pulmonary disease, cerebrovascular accident, peripheral vascular disease, creatinine, recent myocardial infarction, left ventricular function, medication with beta blocker or statin, type of surgery (isolated coronary bypass graft or more complex procedures), and repeat surgery.
Inflammation risk model eligibility
Patients in the placebo arm of the DECS trial 13 enrolled at the Isala Clinics were used to develop an inflammation risk model linking the preoperatively available covariates to postoperative CRP responses. Patients were only eligible if they had no missing preoperative data, a CRP value before surgery and at least one further CRP measurement on postoperative day 2, 3 or 4. Individuals with a preoperative CRP value greater than 10 mg/L were excluded as they were considered already to have an active inflammatory or infective condition before surgery. 21
Statistical methods
Data were summarised with mean (standard deviation (SD)), median (interquartile range (IQR)) (range), or number (percentage) as appropriate. In patients with at least one CRP measurement in the first 5 days, the time-course of the rise in CRP values in the placebo arm was described with a linear mixed regression model that compensated for missing data. Consequently, the peak postoperative CRP value for each individual was assigned from the highest value recorded across postoperative days 2, 3 and 4.
Inflammation risk model
A linear regression model using preoperatively available covariates to predict peak postoperative CRP was constructed in the placebo arm. No model selection was applied. This model was then used to predict peak postoperative CRP for all patients, under the assumption of no treatment with intraoperative dexamethasone. This prediction defined an individual’s preoperative inflammation risk score.
Heterogeneity of anti-inflammatory treatment effect (the primary endpoint)
Patients across both arms of the trial with a preoperative inflammation risk score and a peak postoperative CRP value were used to evaluate any heterogeneity in the anti-inflammatory treatment effect of dexamethasone. This was assessed with linear regression, in which the effect of interest was the interaction between the covariates of treatment and risk score, in determining the dependent variable of peak postoperative CRP.
Heterogeneity of clinical treatment effect
Patients across both arms of the trial with a preoperative inflammation risk score and a DECS trial 13 primary outcome were used to evaluate any heterogeneity in the clinical treatment effect of dexamethasone. This was assessed with the use of binomial regression with an identity-link function, in which the effect of interest was the interaction between the covariates of treatment and risk score, in determining the dependent variable of the DECS trial primary outcome (a composite of death, myocardial infarction, stroke, renal failure, or respiratory failure within 30 days of randomisation).
Statistical analysis was performed using Stata v.15 (StataCorp LLC, College Station, TX, USA) and statistical significance was set at a P value less than 0.05.
Results
From the DECS trial 13 participants enrolled at the Isala Clinics, 283 patients were used to develop the inflammation risk model, 552 patients were used to test heterogeneity of the anti-inflammatory treatment effect, and 813 patients were used to test heterogeneity of the clinical treatment effect (Figure 1).
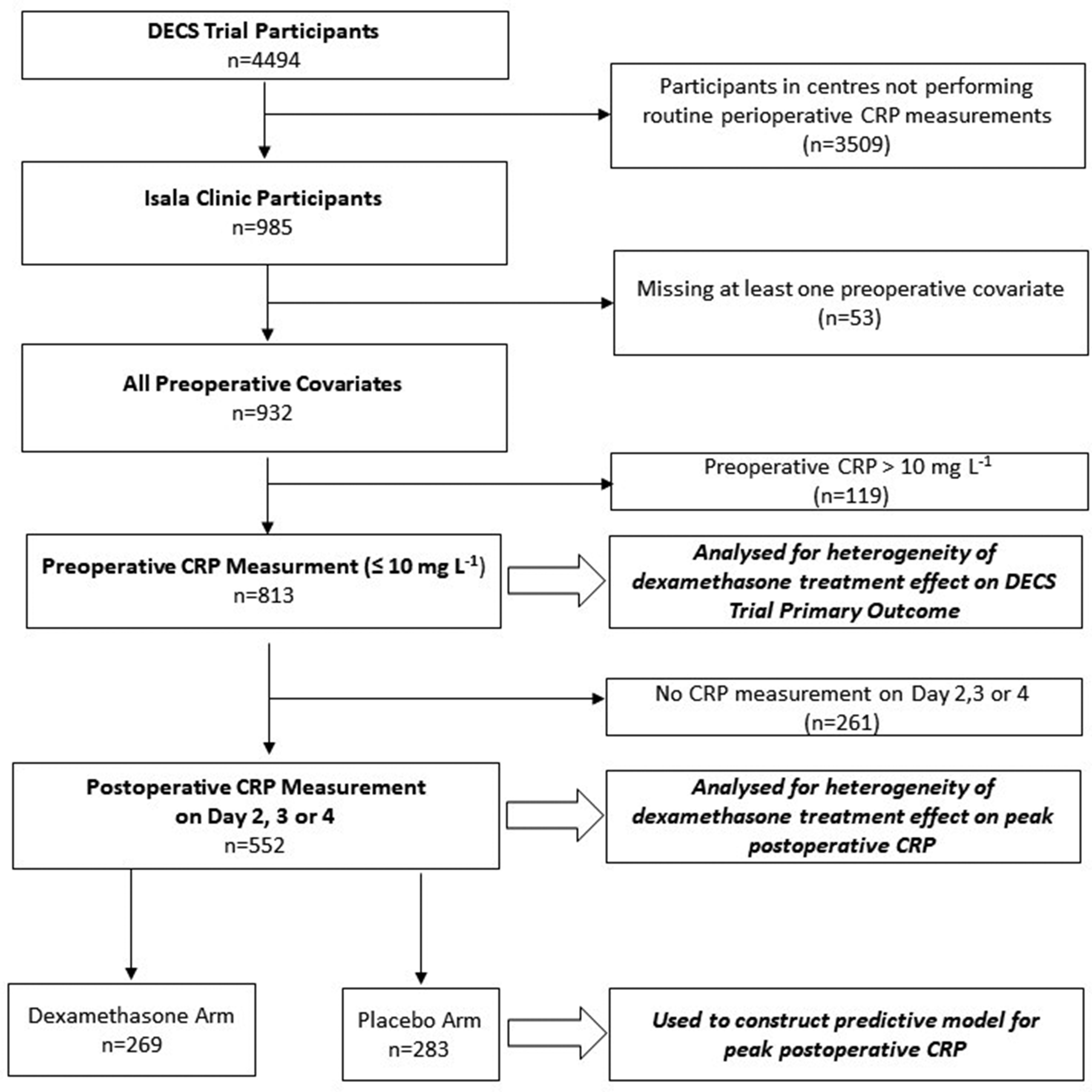
Flow diagram showing cohorts drawn from Intraoperative High-Dose Dexamethasone for Cardiac Surgery (DECS) trial participants for the purposes of inflammation risk model development (n = 283), testing heterogeneity of anti-inflammatory treatment effect (n = 552), testing heterogeneity of clinical treatment effect (n = 813).
CRP measurements after surgery were highest on postoperative day 3 (Figure 2). The peak postoperative CRP for each individual, derived from the highest measurement between day two, three or four was normally distributed with a mean (95% CI) of 202 (193 to 211) mg/L. The mean (SD) peak postoperative CRP in patients receiving placebo and dexamethasone was 202 (80) mg/L and 117 (75) mg/L respectively, equivalent to a 42% reduction with treatment.
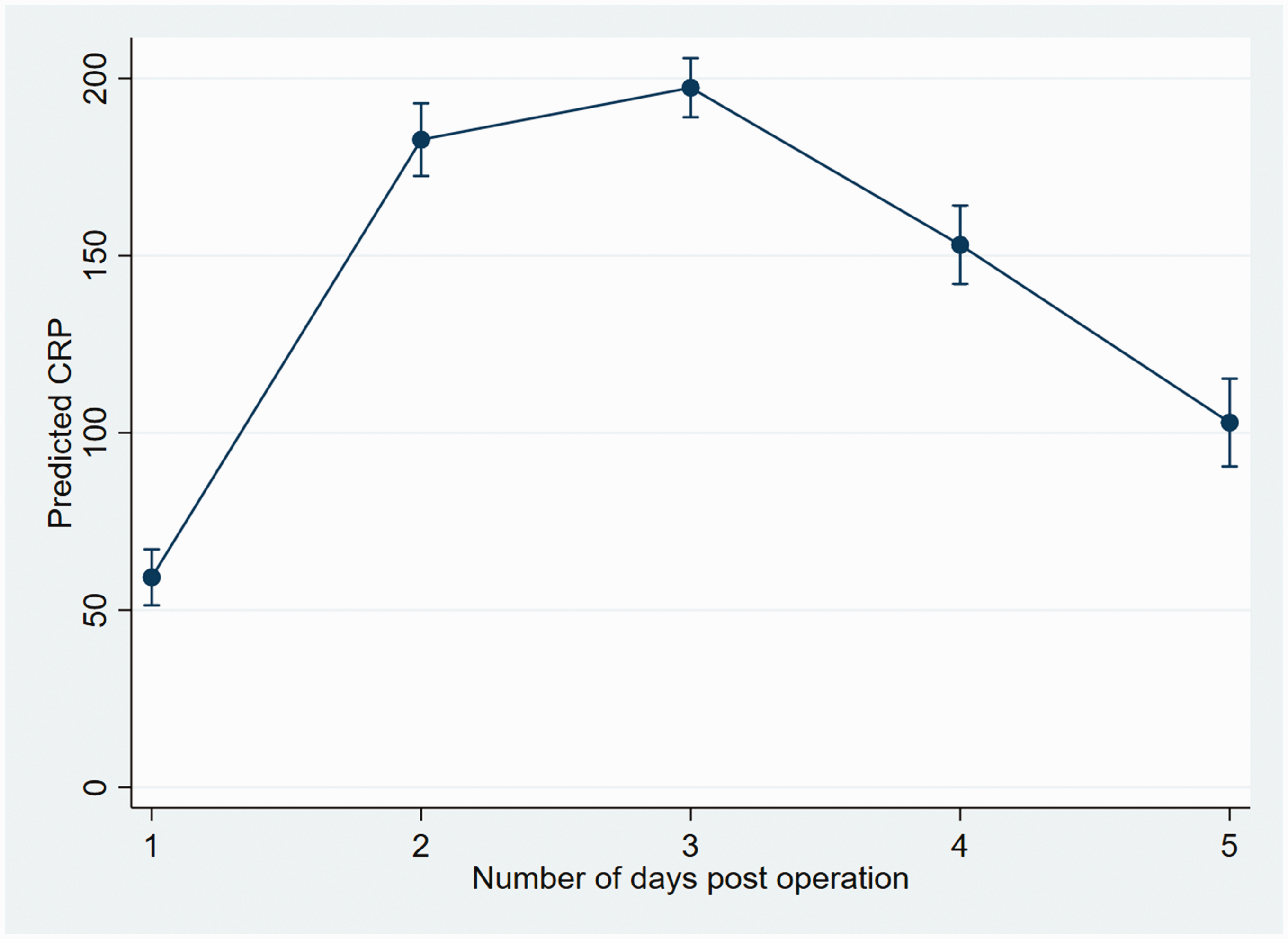
Linear mixed model of postoperative C-reactive protein (CRP) values in the Intraoperative High-Dose Dexamethasone for Cardiac Surgery (DECS) trial (all patients with at least one postoperative CRP included). Error bars represent the 95% confidence interval. Number (%) of measurements at each time point: day 1 = 258 (91%), day 2 = 141 (50%), day 3 = 227 (80%), day 4 = 118 (42%), day 5 = 92 (33%).
The linear regression model predicting peak postoperative CRP was limited and accounted for only a minority of the inter-individual variation observed (adjusted R2 = 0.058, P = 0.012, n = 283). Preoperative inflammation risk scores generated by the model were normally distributed. Preoperative CRP (≤10 mg/L) was the most significant predictive covariate (P = 0.001) – see Table 1.
Preoperative covariates and their relationship in a linear regression model to peak postoperative C-reactive protein.
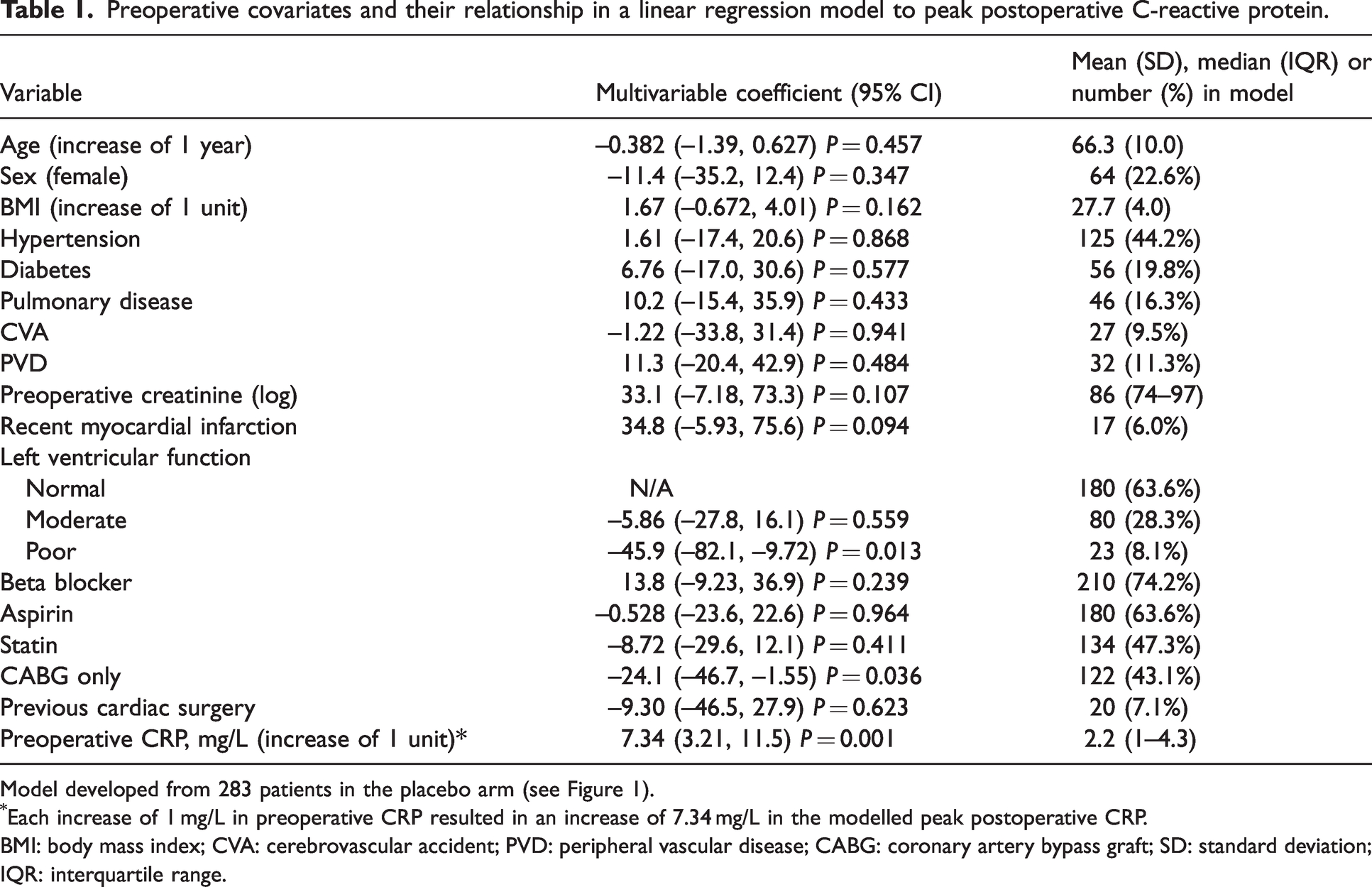
Model developed from 283 patients in the placebo arm (see Figure 1).
Each increase of 1 mg/L in preoperative CRP resulted in an increase of 7.34 mg/L in the modelled peak postoperative CRP.
BMI: body mass index; CVA: cerebrovascular accident; PVD: peripheral vascular disease; CABG: coronary artery bypass graft; SD: standard deviation; IQR: interquartile range.
The anti-inflammatory effect of dexamethasone on peak postoperative CRP was substantial across all patients (peak CRP –84.1 mg/L, P < 0.001, n = 552) and increased as the inflammation risk increased (–0.689 mg/L per unit predicted peak CRP, P = 0.002 for interaction) – see Figure 3.
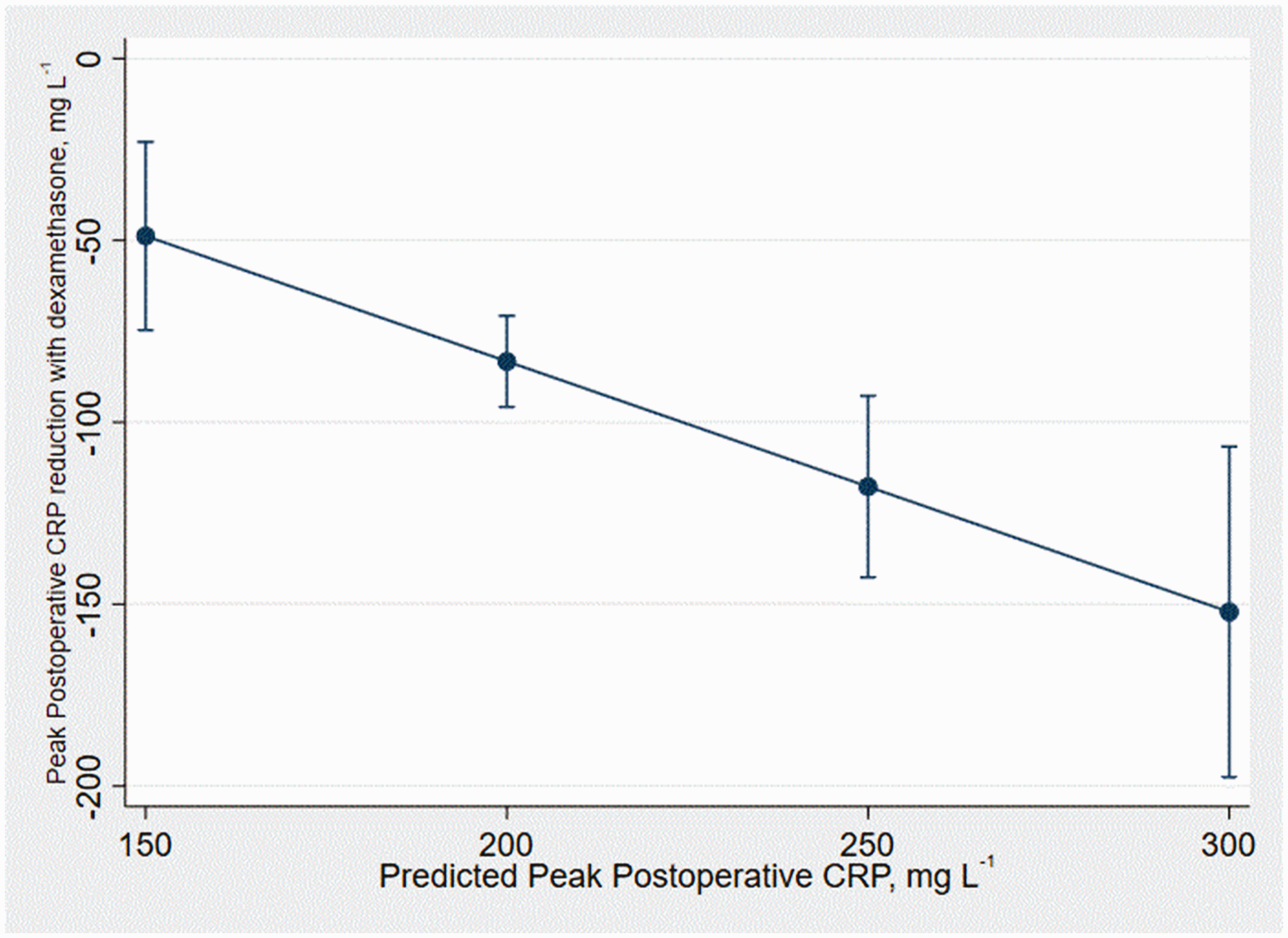
The reduction of peak postoperative C-reactive protein (CRP) by dexamethasone treatment according to the preoperative inflammation risk score; a graphical representation of the interaction term (P = 0.002) between these two covariates. Error bars signify 95% confidence interval.
The main clinical outcome occurred in 29 out of 400 patients receiving placebo (7.25%) and 24 out of 413 patients receiving dexamethasone (5.81%). No dexamethasone treatment-effect heterogeneity according to inflammation risk was detected for the main clinical outcome (P = 0.167 for interaction, n = 813, Figure 4). Patient and operative characteristics in low (below median risk) and high (above median risk) inflammation risk groups are summarised in Table 2.
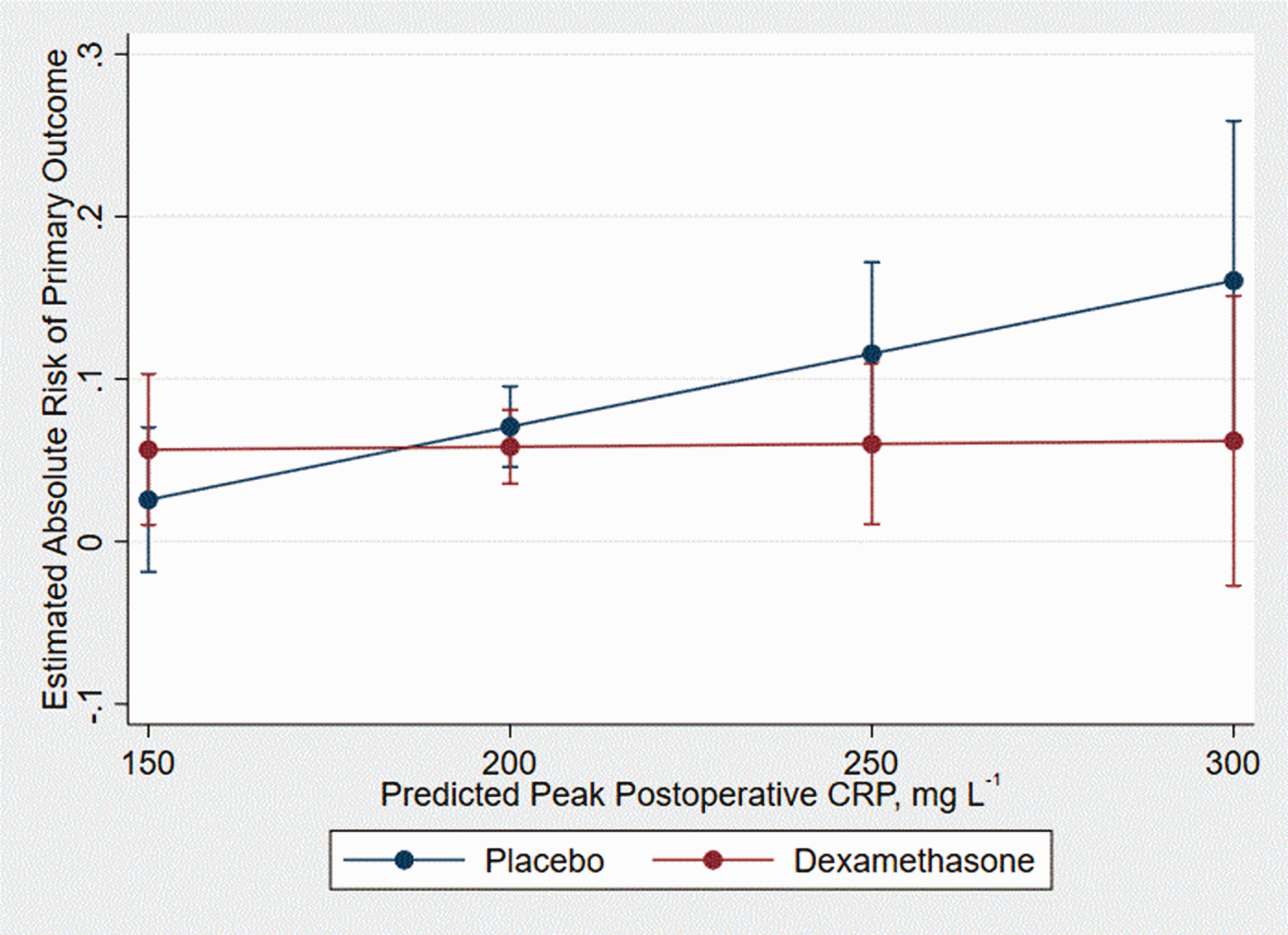
The Intraoperative High-Dose Dexamethasone for Cardiac Surgery (DECS) trial primary outcome according to the preoperative inflammation risk score in the placebo and dexamethasone treatment arms (P = 0.167 for interaction). Straight lines indicate the modelled regression coefficients for the preoperative inflammation risk score in placebo (blue) and dexamethasone (red) arms. Error bars signify 95% confidence interval.
Preoperative covariates in low (below median risk) and high (above median risk) inflammation risk groups tested for heterogeneity of clinical treatment effect.
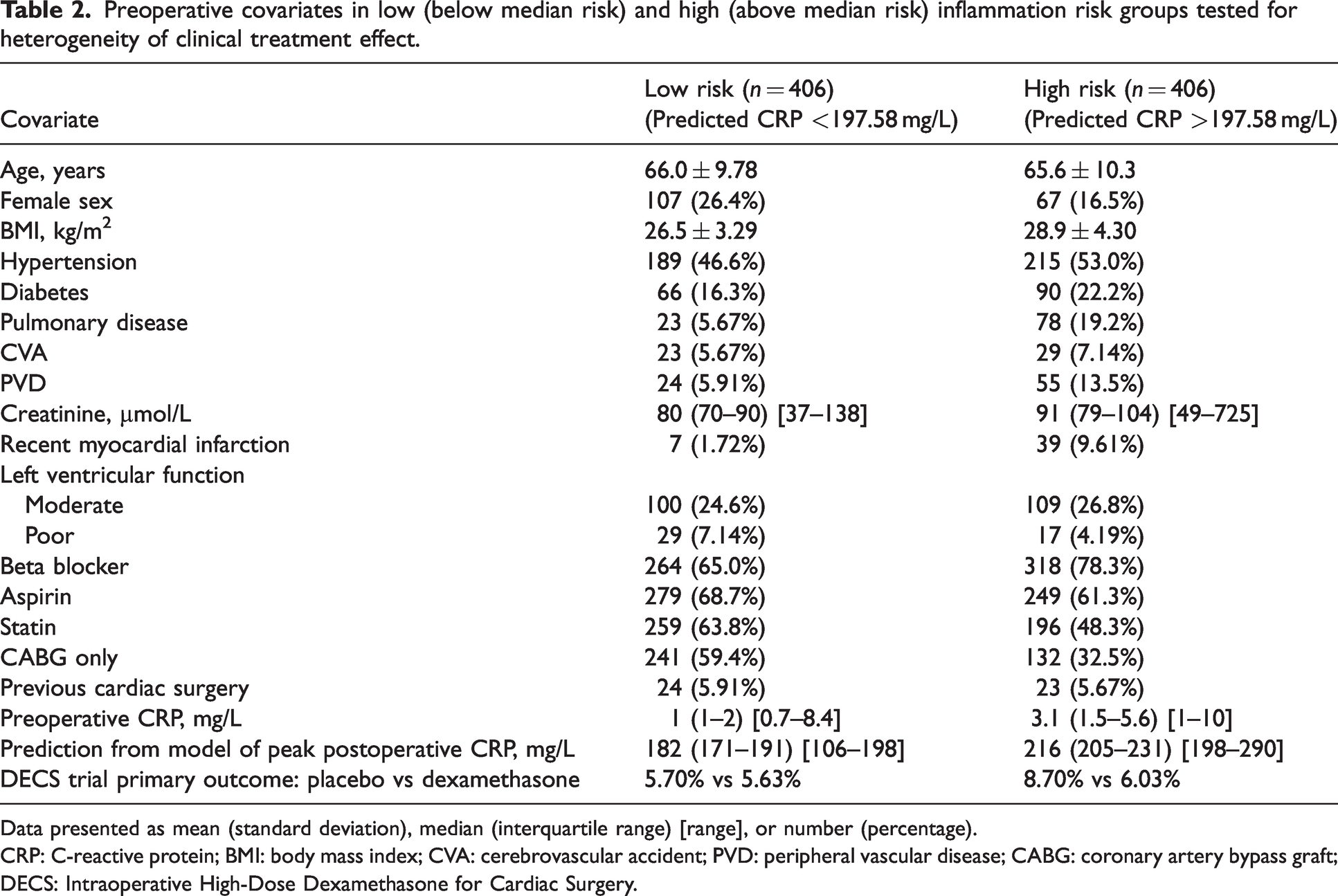
Data presented as mean (standard deviation), median (interquartile range) [range], or number (percentage).
CRP: C-reactive protein; BMI: body mass index; CVA: cerebrovascular accident; PVD: peripheral vascular disease; CABG: coronary artery bypass graft; DECS: Intraoperative High-Dose Dexamethasone for Cardiac Surgery.
Discussion
In this study we have shown that predictive modelling using commonly available preoperative covariates only explains a small proportion of the interindividual variation in peak postoperative CRP values after cardiac surgery. This model is therefore not currently appropriate for individual risk prediction. Nevertheless, patients with higher preoperative inflammation risk scores generated from the model demonstrated greater reductions in peak postoperative CRP after treatment with dexamethasone. The impact of this on major postoperative morbidity and death appears promising but was inconclusive in this relatively small sample. Larger definitive studies are now warranted.
The preliminary data presented here remain consistent with the hypothesis that inflammation is causally related to poor postoperative outcomes. However, given that most anti-inflammatory drugs have a side-effect profile, it is plausible that the risk-benefit balance of dexamethasone only tips towards net benefit in patients with higher inflammation risk before surgery (above the point of intersection on Figure 4). Such patients may also have a greater comorbidity burden at baseline (Table 2) and therefore a greater vulnerability to inflammation-driven organ damage.
The study findings are also consistent with the conventional subgroup analyses reported in the original DECS trial, 13 suggesting that younger patients (higher inflamers) may benefit most from dexamethasone treatment (P = 0.08 for interaction). The analysis approach described herein is a type of subgroup analysis, in which all the relevant preoperative covariates are combined to create a single inflammation risk score. As per expert guidance, 22 the impact of this risk score on the dexamethasone treatment effect was explored on the continuous scale rather than within arbitrary categories. However, the gain in power from combining covariates was offset by a smaller sample size, and the results with respect to clinical outcomes were similarly non-significant. Overall, definitive trials specifically targeting high inflammatory-risk patients appear well justified, with some underway including DECS-II (age less than 75 years). 23 These will be enhanced by the development and validation of inflammation-risk prediction tools. Although we have taken a first step towards such a tool, models with better predictive performance are required that encompass novel covariates linked to inflammation control including autonomic 24 and genomic parameters. 25
The finding that preoperative CRP demonstrated the most significant association with postoperative CRP warrants further discussion. There is a wide range of literature on how CRP is a good marker of chronic low-grade inflammation and associates with all-cause mortality over long time-periods. 26 Our study supports the concept that a chronic inflammatory phenotype that is harmful over decades also results in exaggerated acute inflammation that may be harmful over days and weeks. Indeed, as many baseline characteristics linked to inflammation also associate with CRP, 3 this parameter may serve as a summarising metric of an individual’s inflammatory phenotype.
This study has a number of limitations. First, the analysis was exploratory in relation to the main clinical endpoint (power calculated post hoc as 24%). Second, a relatively large number of covariates were modelled relative to the sample size and any interpretation of the impact of individual covariates would be mostly speculative. Future studies should look to apply non-linear and machine learning modelling approaches 27 to larger datasets and publish a statistical analysis plan in advance. Third, CRP is well recognised to be a non-specific measure of inflammation. However, it is the only measure currently recorded on scale in clinical trials that is suitable for the analyses described herein. An acute inflammatory condition cannot be excluded in our patients with an elevated preoperative CRP, and future studies should consider examining a strictly elective cohort with a high-sensitivity assay. Missing CRP data may also have been non-random and a source of bias. Fourth, multiple imputation was not used to compensate for missing data. Finally, patients undergoing cardiac surgery with CPB will all have relatively large postoperative inflammatory responses. Greater distinction between patients with low and high inflammation risk scores may be more achievable in trials targeting non-cardiac surgery populations, although glucocorticoid doses in these settings are substantially lower. Indeed, we plan to address this and other limitations in an analysis of the recently completed Perioperative Administration of Dexamethasone and Infection (PADDI) trial, which randomly assigned 8880 patients undergoing non-cardiac surgery to receive 8 mg dexamethasone or placebo. 12
In conclusion, readily available preoperative covariates exhibited limited performance within a linear regression model predicting peak postoperative CRP after cardiac surgery. Nevertheless, risk predictions from this model were associated with the degree of peak postoperative reduction in CRP derived from dexamethasone treatment. Future studies should address whether this translates into improved clinical outcomes in high-risk patients, using larger datasets and the development of better inflammation models, inclusive of preoperative CRP and novel covariates.
Footnotes
Data sharing statement
Author Contribution(s)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Anaesthesia and Pain Medicine, Royal Perth Hospital, Perth, Australia.
