Abstract
Patients with severe clinical manifestations of coronavirus disease 2019 (COVID-19) present particular diagnostic and management challenges to critical care physicians, including identifying and responding to concurrent bacterial and fungal coinfections. This study evaluates risk factors for in-hospital mortality in patients admitted to the intensive care unit with severe COVID-19 during circulation of the B.1.617.2 (Delta) variant, including the impact of immunomodulators and bacterial and/or fungal coinfection. This retrospective cohort study enrolled patients with severe COVID-19. A Cox proportional hazard ratio analysis identified risk factors for in-hospital mortality. Outcomes were also compared between patients receiving and not receiving immunomodulatory therapy alongside standard care. Ninety patients admitted to the intensive care unit were enrolled. On multivariate analysis, the greatest risk factors for in-hospital mortality were invasive mechanical ventilation (hazard ratio (HR) = 15.27; 95% confidence interval (CI) 3.29–71.0; P < 0.001), elevated body mass index (HR = 1.07 per unit; 95% CI 1.02–1.13; P = 0.007) and older age (HR = 1.53 per decade; 95% CI 1.05–2.24; P = 0.028). Bacterial and/or fungal coinfection occurred at equal frequency in patients receiving and not receiving immunomodulatory therapy. However, in patients receiving immunomodulators, coinfection carried a significantly higher mortality risk (63.0%) compared with those without coinfection (15.4%; P = 0.038). Mortality from severe COVID-19 is significantly higher in older patients and those with elevated body mass index and requiring mechanical ventilation. Immunomodulatory therapy necessitates vigilance towards evolving coinfection in the intensive care setting.
Introduction
Coronavirus disease 2019 (COVID-19) was declared a global pandemic by the World Health Organization on 11 March 2020, and the spread of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has brought unparalleled global health, social and economic disruption. 1 Although the spectrum of disease is broad, patients with severe disease present particular diagnostic and management challenges to clinicians, including identifying complications of the disease and initiating appropriate therapy in a timely manner. 2
Prognostic information is important to guide management decisions and to inform expectations. Immunomodulatory agents—in particular tocilizumab, sarilumab and baricitinib—form part of the backbone of therapy for severe COVID-19 and are known to have wide-reaching effects on the immune cascade, including the potential for development of opportunistic infections or dissemination of latent infections. 2 Meta-analysis 3 and randomised controlled trial data 4 have demonstrated overall reduced mortality in patients with COVID-19 receiving tocilizumab compared with placebo (odds ratio (OR) = 0.86; 95% confidence interval (CI) 0.79–0.95; P = 0.003 3 ; OR = 0.85; 95% CI 0·76–0·94; P = 0.0028 4 ), although these data did not specifically examine intensive care unit (ICU) cohorts nor tease apart the impact of coinfections in these patients, which may enable more targeted diagnostic algorithms. Detailed data are urgently needed to elucidate the impact of bacterial and fungal coinfection on associated mortality risk in patients receiving immunomodulatory therapy for COVID-19. Some studies to date suggest increased rates of bacterial infection (54% vs. 26%; P < 0.001) but no increased mortality associated with such infections (19% vs. 23%; P = 0.445),5,6 and others have demonstrated an increased risk of fungal infection, in particular with tocilizumab use. 7 Contributing factors may include an impaired ability of the dampened immune system to control infection even in the presence of appropriate antibiotic therapy.
This retrospective cohort study aims to analyse risk factors for mortality in an ICU cohort of patients with severe COVID-19 during the SARS-CoV-2 B.1.617.2 (Delta) wave in 2021. In addition, the interplay between immunomodulatory therapy and coinfection in SARS-CoV-2 infection is examined with a view to informing treatment algorithms for COVID-19, particularly for patients with severe disease.
Methods
Study design
This is a retrospective cohort study of patients with SARS-CoV-2 infection admitted to the intensive care unit (ICU) of an Australian tertiary hospital between 1 July 2021 and 30 November 2021. The study was conducted in a 520-bed hospital situated in a metropolitan area of Sydney, Australia. The study is reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) principles. 8
Patients were identified retrospectively from the hospital patient information management system using ICD-10 codes U07.1 ‘COVID-19’ or U07.2 ‘COVID-19, virus not identified’ with a filter for those patients managed in the ICU. A manual crosscheck was performed on all patients admitted to the ICU over the study period to identify any patients not captured by ICD coding.
COVID-19 was diagnosed by detection of SARS-CoV-2 RNA in a respiratory specimen by reverse transcriptase polymerase chain reaction assay (BD-Max, Becton, Dickinson and Company, Franklin Lakes, NJ; or GeneXpert, Cepheid, Sunnyvale, CA). Sample pooling (4:1) was adopted during the study period, and detailed analysis of the assay performance using this strategy is presented elsewhere. 9 Although an increase in cycle threshold of one to three cycles was observed, the diagnostic performance in a real-world setting demonstrated no significant difference in overall sensitivity of the assay.
Severe COVID-19 infection was defined as a requirement for admission to the ICU for respiratory support. Patients were excluded if they were previously enrolled in this study or if their admission to the ICU was for a non-COVID-19 indication, for example due to an alternative disease process with a coincidental finding of SARS-CoV-2 infection.
Patients who received immunomodulatory therapy (i.e. baricitinib or tocilizumab) with or without other medical therapies were compared with patients who did not receive immunomodulatory therapy. Of note, sarilumab and other novel immunomodulators were not available in our setting at the time of this study. Use of immunomodulatory therapy for COVID-19 was guided by recommendations of the Australian COVID-19 taskforce at the time of the study, which suggested consideration of tocilizumab, sarilumab or baricitinib with commencement of oxygen supplementation (but not mechanical ventilation), particularly if there was concurrent biochemical evidence of inflammation (for tocilizumab). 10 Additional pharmaceutical treatments for COVID-19 administered at the time included a monoclonal antibody formulation (sotrovimab), systemic corticosteroids (typically dexamethasone orally or intravenously at a dose of 6 mg/day), antivirals (remdesivir) and/or antimicrobials (if indicated). All treatment decisions were at the discretion of the treating physician. Potential confounders, including diabetes mellitus, number of medical comorbidities, initial severity of illness (as measured using Acute Physiology and Chronic Health Evaluation-III (APACHE-III) score, quick Sepsis-related Organ Failure Assessment (qSOFA) score and PaO2/FiO2 ratio) 11 and extracorporeal membrane oxygenation (ECMO) use were considered in the statistical analysis. Participants were considered fully immunised if symptom onset occurred 14 days or more after the second dose of SARS-CoV-2 vaccine. Obesity was defined as a body mass index (BMI) of 30 kg/m2 or more at the time of admission. 12
The term ‘coinfection’ is used in this article to refer to the finding of a non-SARS-CoV-2 pathogen, specifically a bacterial or fungal pathogen, during the participant’s hospital admission. The term is used regardless of the timing of infection onset relative to SARS-CoV-2 infection, and data on acquisition timing are presented and analysed in detail. Coinfections have been divided into two subgroups: hospital-acquired infections, defined as infections for which the date of sample collection was less than 48 hours following admission or within 48 hours of a prior discharge, or in association with an indwelling device such as a central venous line; and community-acquired infections, defined as infections not fulfilling the aforementioned criteria. 13 In assessing the presence of bacterial infection, organisms typically associated with colonisation or contamination (including single blood cultures with growth of coagulase negative Staphylococci or Gram-positive bacilli, bacteriuria without pyuria or Candida spp. grown from urine without disseminated disease) were regarded as negative findings. For the purposes of this analysis and considering this patient cohort was a group with severe respiratory disease in the context of SARS-CoV-2 pneumonitis, all significant organisms isolated from lower airway specimens were deemed to represent pathogens, with the exception of Candida spp. from a respiratory sample in the absence of disseminated Candidal disease.
This study was approved by the Nepean Blue Mountains Local Health District Human Research and Ethics Committee (2021/ETH11932). A waiver of consent was granted due to the negligible risk to individual participants.
Data collection
For all participants who met inclusion criteria, detailed demographic, clinical and laboratory data were collected from the electronic medical record. All data were deidentified prior to analysis. Of note, there is some overlap in patient recruitment between this study dataset and that reported by Gerhardy et al. 14 However, ethics constraints prevented crosslinking of data across these two studies.
Statistical analysis
The effects of immunomodulator administration and bacterial and/or fungal coinfection on mortality were analysed using univariate survival analysis. Survival curves between the groups were compared using the log rank (non-parametric) test without weighting. Cox proportional hazard model was used to investigate the association between hospital survival time and a set of predictors determined a priori. These predictors were age, BMI, immunomodulator use, coinfection, number of comorbidities and invasive mechanical ventilation. The full model incorporated all of the selected predictors, and the final model contained only those which remained statistically significant (P < 0.05) after stepwise backward selection procedures. Multicollinearity was checked using variance inflation factor. The Akaike information criterion (AIC) was used to select the final model with the best fit (with the least parameters) in nested models. Proportional hazards assumptions were tested using Schoenfeld test. For patients transferred to other facilities (e.g. for ECMO), total length of stay and mortality data were accessible from internal databases and incorporated into the current analysis, but other clinical data were not accessible due to ethics constraints. Outcomes data were reanalysed with exclusion of these transferred patients to determine the impact on study findings. All follow-ups were censored on 31 December 2021.
Categorical variables are presented as count (percentage), and continuous variables are presented either as mean (standard deviation) or median (interquartile range). Student’s independent t-test and chi-square analysis were used to compare differences between patient groups. Log transformations were performed for skewed data (e.g. length of stay) before group comparison. Ninety-five percent confidence intervals (CI) were reported for all continuous variables and for hazard ratios (HRs) associated with the multivariable analysis. A P-value of <0.05 was regarded as statistically significant. Statistical analysis was performed using Jamovi v2.2.3 15 and R v4.1.0 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Patient demographics
Over the study period, 95 patients were admitted to the ICU with a diagnosis of COVID-19, as confirmed by positive polymerase chain reaction result for SARS-CoV-2 on an upper or lower respiratory tract specimen. During the period of this study, 100% of circulating SARS-CoV-2 strains in the local community were the B.1.617.2 (Delta) variant. 16 Five patients were excluded as their requirement for ICU-level support was due to a non-COVID-19-related indication (trauma, diabetic ketoacidosis or infection control purposes). One participant was excluded from analyses of timing of COVID-19 onset and length of stay due to in-hospital acquisition of COVID-19 on day 274 of an admission for another indication, but their data were incorporated into other aspects of the analysis. No patients were lost to follow-up. Of the 90 patients included in this study, 66 (73.3%) received immunomodulatory therapy, and 24 (26.7%) did not. Of the patients receiving immunomodulatory therapy, most patients received baricitinib (73%) compared with tocilizumab (27%); no patient received sarilumab.
Table 1 summarises the demographic features of the patients, and there were no significant discernible differences in demographic features between the immunomodulator and non-immunomodulator groups. In line with obesity as a known risk factor for severe COVID-19, there was a high proportion of obese patients in both groups, with mean BMIs of 36.9 ± 11.7 kg/m2 and 37.8 ± 12.0 kg/m2 in the immunomodulator and non-immunomodulator groups, respectively (P = 0.763). Only one patient across both groups was fully immunised against SARS-CoV-2. APACHE-III scores were similar in the immunomodulator compared with non-immunomodulator groups (50.8 ± 18.8 vs. 57.3 ± 30.4; P = 0.230), as were qSOFA scores on admission (2.0 ± 0.7 vs. 2.3 ± 0.8; P = 0.536) and PaO2/FiO2 ratios on admission (119.9 ± 61.4 vs. 110.4 ± 41.2; P = 0.085), suggesting the groups were matched in disease severity at the time of admission to the ICU.
Baseline characteristics of patients receiving immunomodulatory therapy plus standard care versus those receiving standard care only for severe COVID-19 infection.
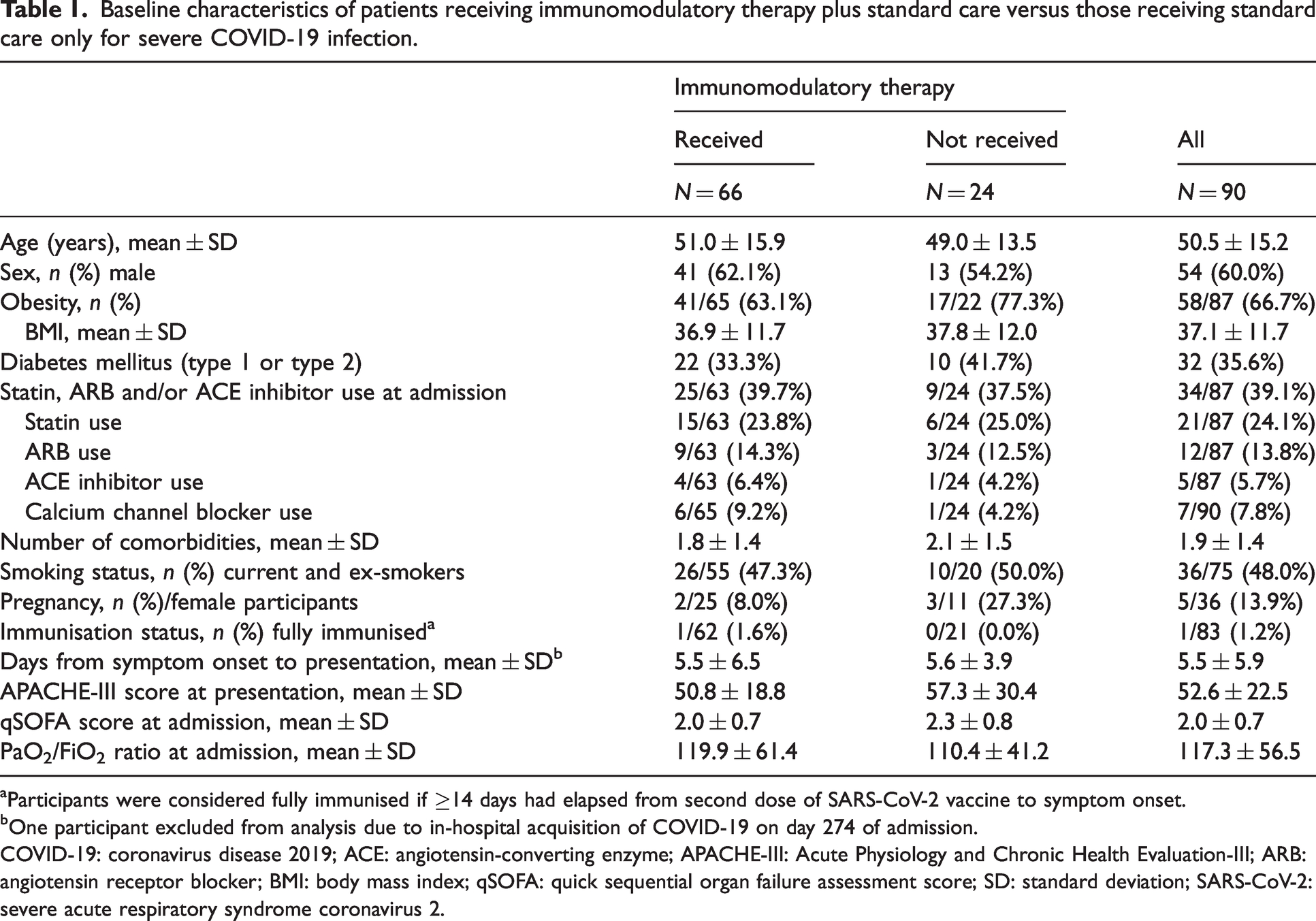
Participants were considered fully immunised if ≥14 days had elapsed from second dose of SARS-CoV-2 vaccine to symptom onset.
One participant excluded from analysis due to in-hospital acquisition of COVID-19 on day 274 of admission.
COVID-19: coronavirus disease 2019; ACE: angiotensin-converting enzyme; APACHE-III: Acute Physiology and Chronic Health Evaluation-III; ARB: angiotensin receptor blocker; BMI: body mass index; qSOFA: quick sequential organ failure assessment score; SD: standard deviation; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2.
Infections
Amongst the 90 patients of the study, 38 (42.2%) experienced coinfections, comprising 62 bacterial and fungal isolates (Table 2). These included 10 bloodstream isolates, 37 respiratory tract isolates and 15 isolates from other sites. The proportion of patients with coinfections (40.9% vs. 45.8%; P = 0.680) and the number of infections per patient (mean 0.71 ± 1.1 vs. 0.71 ± 1.0; P = 0.989) were not significantly different between patients who received immunomodulators and those who did not. There were three episodes of polymicrobial infection, all in patients from the immunomodulatory therapy group (Table 2). The majority of coinfections were hospital acquired, and this finding was most notable in the immunomodulatory therapy group (89% of coinfected patients) compared with the non-immunotherapy group (64%). The most common pathogens associated with bacteraemia included Staphylococcus aureus and Streptococcus pneumoniae, as well as Enterobacterales and Gram-negative non-fermenters, as expected in the context of marked respiratory inflammation. There was one episode of fulminant candidaemia. Onset of coinfection relative to hospital admission and relative to initiation of immunomodulatory therapy (in the relevant patient group) was highly variable (mean 6.7 ± 6.7 days in the latter case; Table 3).
Microbiological characteristics of 62 isolates from patients with severe COVID-19 infection.
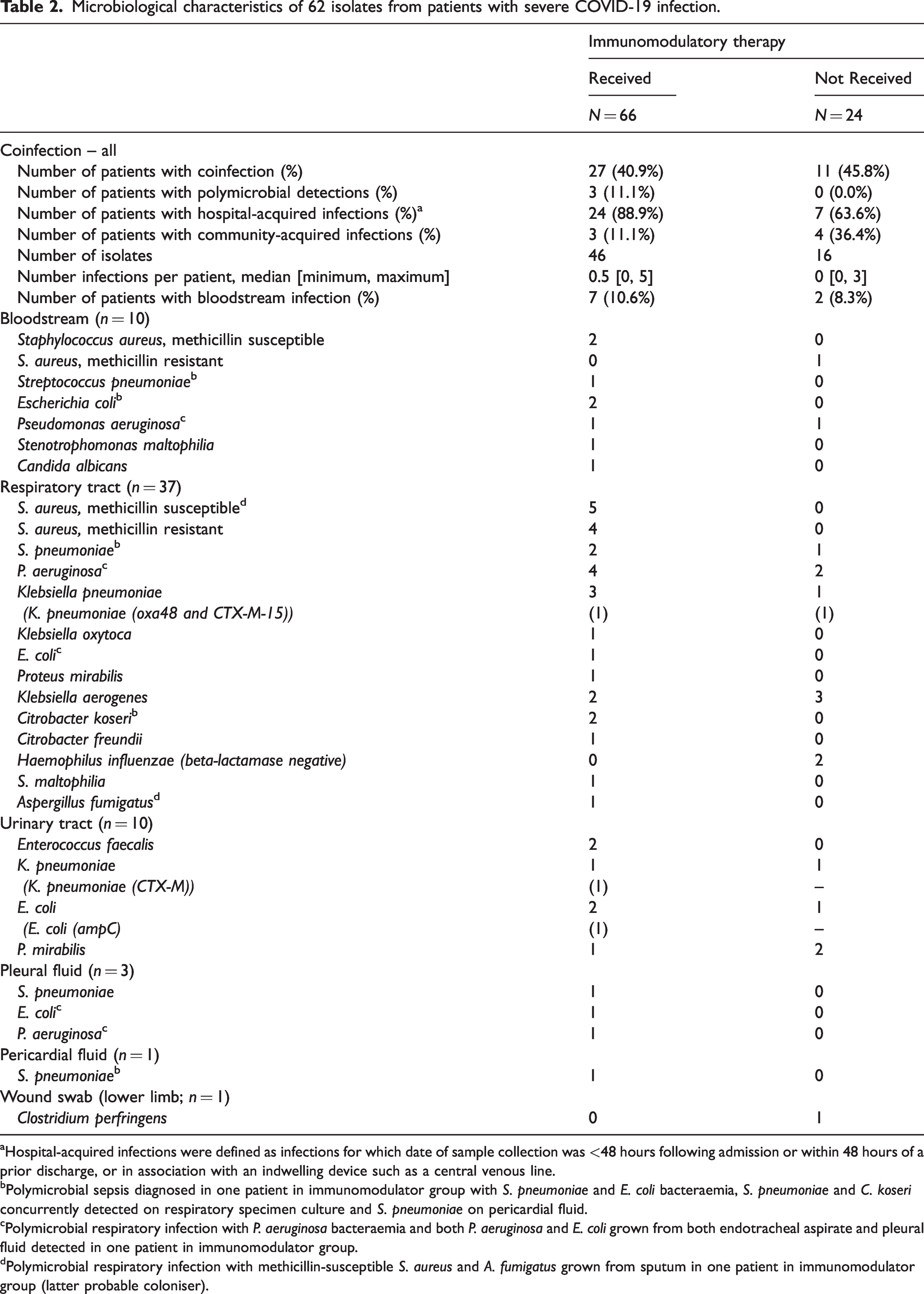
Hospital-acquired infections were defined as infections for which date of sample collection was <48 hours following admission or within 48 hours of a prior discharge, or in association with an indwelling device such as a central venous line.
Polymicrobial sepsis diagnosed in one patient in immunomodulator group with S. pneumoniae and E. coli bacteraemia, S. pneumoniae and C. koseri concurrently detected on respiratory specimen culture and S. pneumoniae on pericardial fluid.
Polymicrobial respiratory infection with P. aeruginosa bacteraemia and both P. aeruginosa and E. coli grown from both endotracheal aspirate and pleural fluid detected in one patient in immunomodulator group.
Polymicrobial respiratory infection with methicillin-susceptible S. aureus and A. fumigatus grown from sputum in one patient in immunomodulator group (latter probable coloniser).
Clinical progress and outcomes for patients receiving immunomodulatory therapy plus standard care compared with those receiving standard care only.
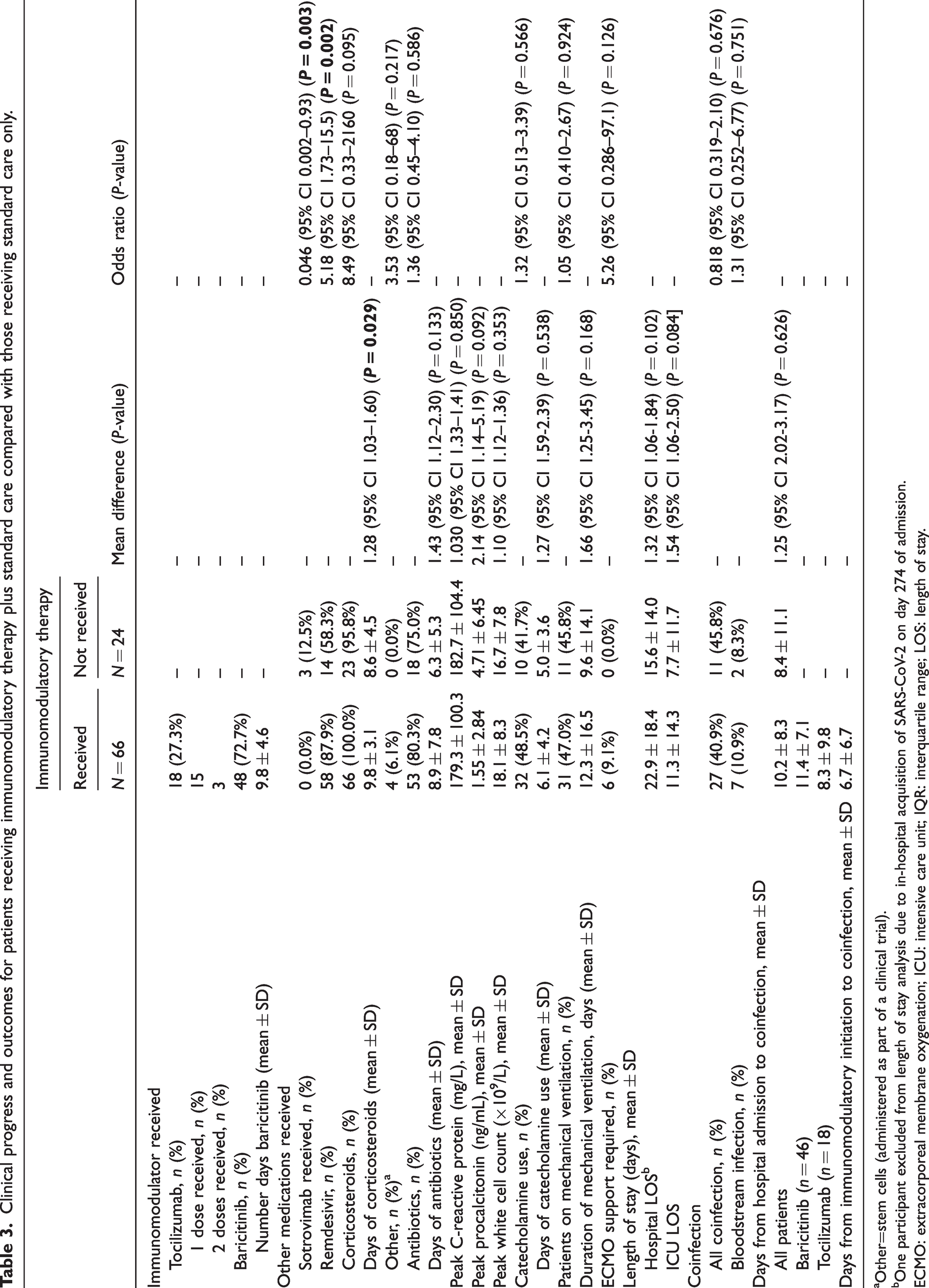
Other=stem cells (administered as part of a clinical trial).
One participant excluded from length of stay analysis due to in-hospital acquisition of SARS-CoV-2 on day 274 of admission.
ECMO: extracorporeal membrane oxygenation; ICU: intensive care unit; IQR: interquartile range; LOS: length of stay.
Medications
The proportion of patients who received antibiotics was not significantly different between the patient groups (80.3% vs. 75.0%; P = 0.586), and although the duration of antibiotic therapy was longer in the immunomodulator group, this did not reach statistical significance (8.9 ± 7.8 vs. 6.3 ± 5.3 days; P = 0.133). More patients in the immunomodulator group received remdesivir compared with the non-immunomodulator group (87.9% vs. 58.3%; P = 0.002), and the only patients to receive sotrovimab prior to ICU admission were in the non-immunomodulator group (three patients, 12.5%; P = 0.003). Duration of corticosteroid use was significantly longer in the immunomodulatory therapy group (9.8 ± 3.1 vs. 8.6 ± 4.5 days; P = 0.029). Otherwise, there was no statistically significant difference in the administration of additional therapies between the two patient groups (Table 3). The proportion of patients requiring mechanical ventilation (P = 0.924) and duration of mechanical ventilation (P = 0.168) were also comparable between the immunomodulatory and non-immunomodulatory therapy groups. Similarly, no significant differences in rates (P = 0.566) or duration of catecholamine use (P = 0.538) were observed between the groups.
Outcomes
Patients who received immunomodulatory agents had a numerically longer median hospital stay (22.9 vs. 15.6 days; mean difference 1.32 (1.06, 1.84); P = 0.102) and ICU length of stay (11.3 vs. 7.7 days; mean difference 1.54 (1.06, 2.50); P = 0.084), but these did not reach statistical significance. Similarly, a higher proportion of patients required ECMO in the immunomodulator group (9.1%; 6/66 participants) compared with the non-immunomodulatory group (0.0%; 0/24 participants), although this difference did not reach statistical significance (P = 0.126). ECMO support required transfer from our centre to a quaternary facility. Given the limited clinical data available for patients transferred externally for ECMO (n = 6), a post-hoc sensitivity analysis of overall outcomes data was performed with exclusion of these patients from the statistical analysis. The overall findings and statistical significance of the key study outcomes were unchanged (data not shown).
Overall mortality was not significantly affected by coinfection (55.3% vs. 15.4%; P = 0.051) or immunomodulator use in isolation (34.8% vs. 25.0%; P = 0.930; Figure 1(a) and (b)). However, in the group of patients on immunomodulatory therapy, mortality was significantly worse in patients who developed a bacterial and/or fungal coinfection compared with those with no coinfection (63.0% vs. 15.4%; P = 0.038; Figure 1(d)). This effect was not seen in patients who did not receive immunomodulators (36.4% vs. 15.4%; P = 0.890).
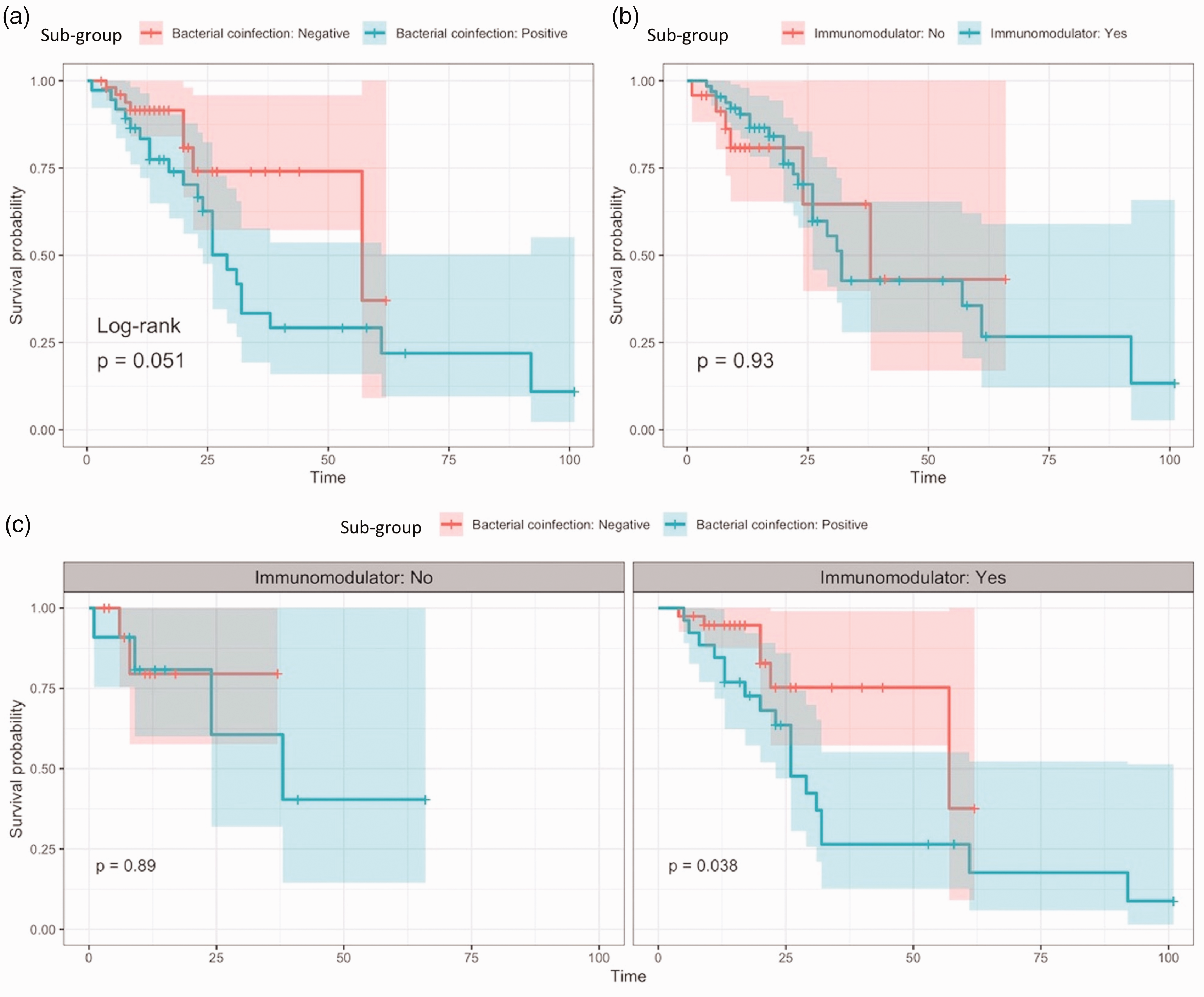
Overall survival curves for patient groups with (a) coinfection versus no coinfection, (b) immunomodulator use versus no immunomodulator use and (c) coinfection versus no coinfection, separated by those who did not receive immunomodulators (left-hand panel) and those who did receive immunomodulators (right-hand panel).
Invasive mechanical ventilation was strongly associated with mortality on multivariate Cox analysis (Figure 2), with a 15-fold increase in hazard of death for patients who were on invasive mechanical ventilation. Every decade increase in age resulted in a 53% increase in hazard, and every unit increase in BMI was associated with a 7% increase in hazard (Figure 2). When adjusted for invasive mechanical ventilation and other predictors, there was no evidence that immunomodulator administration (HR = 0.84; 95% CI 0.33–2.11) and presence of coinfection (HR = 0.98; 95% CI 0.36–2.68) were associated with survival. The number of comorbidities was also not associated with survival (HR = 1.02; 95% CI 0.78–1.33).
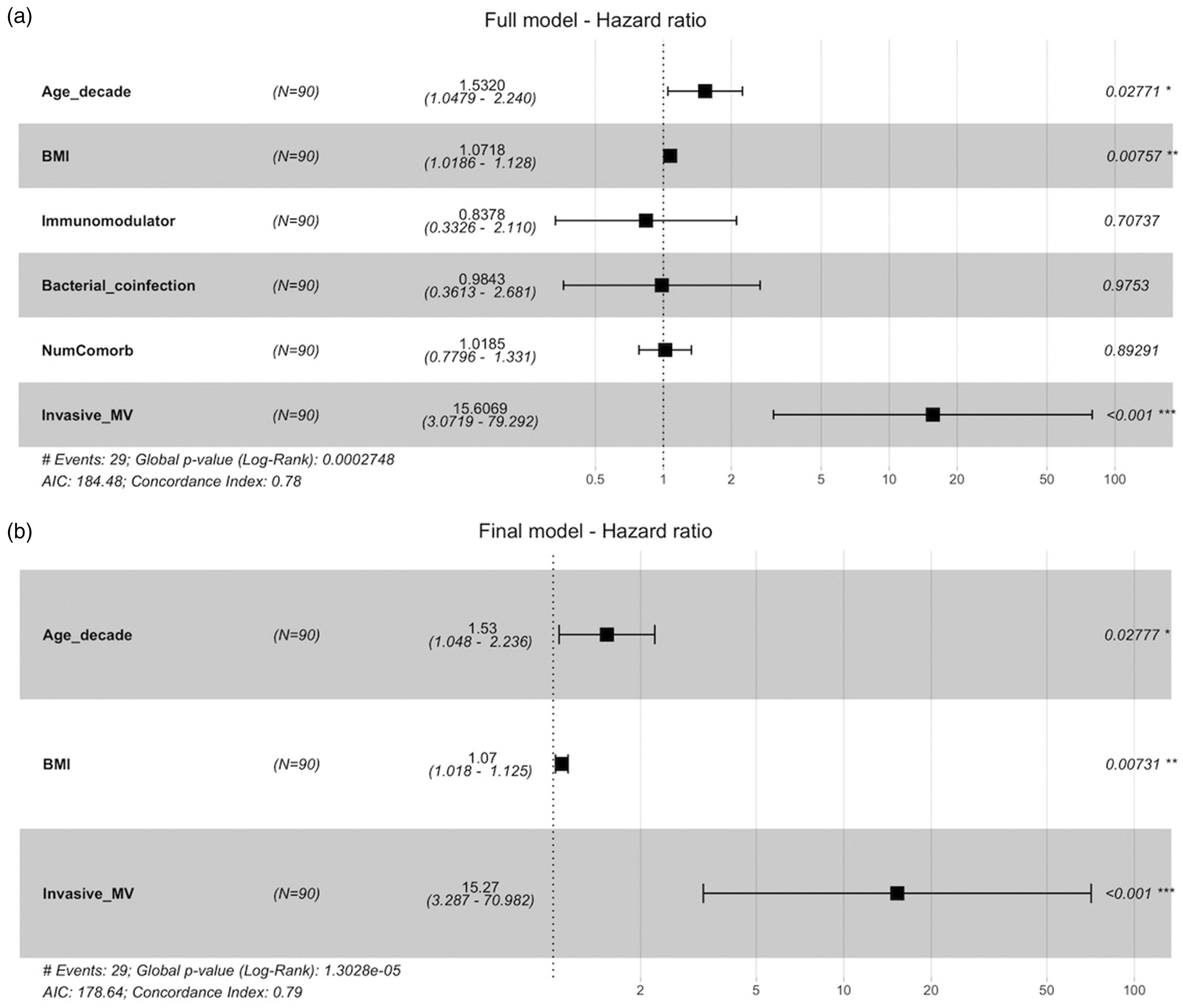
Forest plot of multivariable analysis showing contributors to mortality in patients admitted to the intensive care unit with COVID-19. Panel (a) incorporates a range of possible contributors and (b) the three individual variables reaching statistical significance: age, BMI and invasive MV. BMI: body mass index; Invasive_MV: invasive mechanical ventilation; NumComorbid: number of comorbidities.
Figure 3 shows the predicted survivor curves for ‘model patients’ receiving invasive mechanical ventilation versus those who did not, derived from mathematical modelling with different combinations of age and BMI based on the final model. Figure 3(c) depicts the curve for a model patient with the mean age (50 years) and mean BMI (37 kg/m2) of the cohort. Increased hazard of death with mechanical ventilation was seen across all age groups and all BMIs (Figure 3). Increasing age (Figure 3(b)–(d)) and increasing BMI (Figure 3(a), (c), and (e)) had a detrimental effect on survival, both with and without mechanical ventilation.
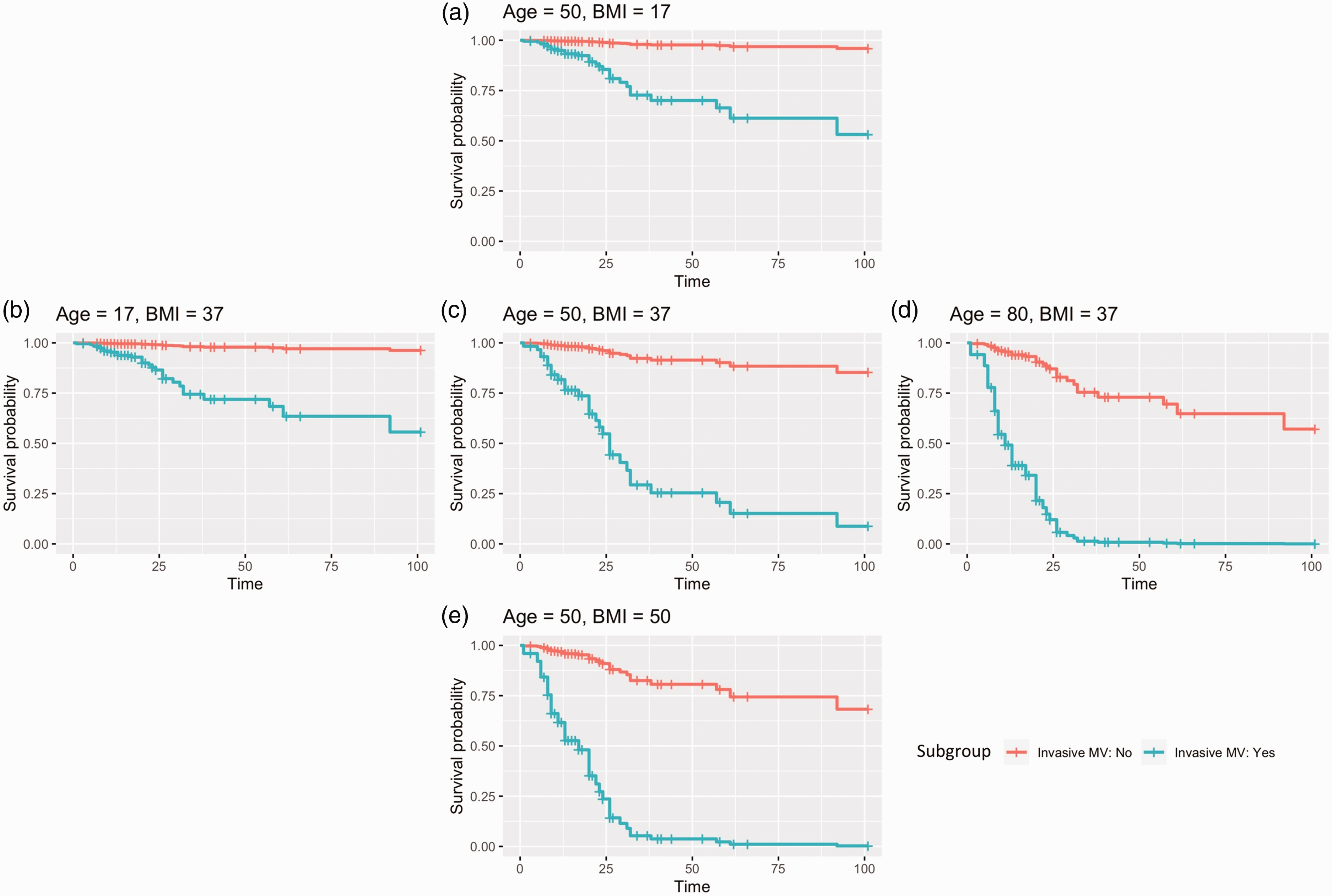
Mathematically predicted survival curves for ‘model patients’ of various ages and BMIs, comparing those who received invasive mechanical ventilation (MV) (blue) with those who did not (red). Panel (c) represents a patient with age and BMI at the average of the patient cohort (age 50 years, BMI 37 kg/m2). Other panels depict representative patients with ages and BMIs above or below this mean.
Discussion
Decision-making in severe COVID-19 is complex, as patients may present with a multiplicity of clinical phenotypes due to escalating viraemia, marked inflammatory dysregulation, bacterial or fungal coinfection or additional complications, and the management approach to each of these can be quite disparate. Care of this cohort, representing the most unwell patients during the COVID-19 pandemic, requires thorough and frequent consideration of multiple clinical factors and often necessitates treading a tenuous path between anti-infective and anti-inflammatory therapy with constant vigilance toward a plethora of potential disease complications.
This study demonstrates that coinfection is common in patients admitted to the ICU with severe COVID-19, occurring in 42% of such patients. This figure is on par with published meta-analysis data from critical care cohorts 17 and contrasts with lower rates of infection on the general wards (4%). 18 Furthermore, and consistent with previous meta-analyses, 3 the number of infections per patient and the proportion of patients with coinfections were comparable between patients receiving and not receiving immunomodulatory therapy. In line with other studies, the most common bacterial pathogens identified in our cohort were S. aureus, Pseudomonas aeruginosa, S. pneumoniae, and the Enterobacterales, particularly Escherichia coli, Klebsiella pneumoniae and Klebsiella aerogenes. We hypothesise that the impaired barrier function of the respiratory mucosa due to SARS-CoV-2-induced inflammation, with or without associated endotheliitis, predisposes to bacterial translocation and possible bacteraemia by pathogens commonly resident in the respiratory tract. The onset of bacterial or fungal coinfection was quite variable relative to immunomodulatory therapy initiation (Table 3), likely representing a range of pathophysiological and immune states at infection onset. Hence, a causative link between immunomodulators and coinfection has not been established in this study. Furthermore, invasive mechanical ventilation is itself an established risk factor for hospital-acquired infection. 5
Mortality in patients with severe COVID-19 requiring ICU-level support is high, reaching 32% in our study. This is in line with earlier systematic review data showing a mortality of 42% in ICU cohorts internationally during circulation of the B.1.617.2 (Delta) variant of SARS-CoV-2. 19 This study has demonstrated that mechanical ventilation, BMI and older age were significantly associated with mortality on multivariable regression. The fact that mechanical ventilation carried a 15-fold increased risk for death is likely reflective not only of the severity of the underlying COVID-19-related disease in patients requiring mechanical ventilation but also of the complications of multiple interventions and prolonged hospitalisation experienced by ventilated patients in the ICU. This increased mortality risk with mechanical ventilation was observed across the age and BMI spectra (Figure 3). Both obesity and older age are known to be associated with severity of disease in COVID-19. 20 Thus, the strong correlation of both these factors with risk of death in our ICU cohort was not surprising.
We found no significant correlation between immunomodulator use and increased survival in our ICU cohort. Data encompassing all hospitalised COVID-19 patients from the RECOVERY (tocilizumab) 4 and COV-BARRIER studies (baricitinib) 21 suggested a significant net mortality benefit to the use of these agents in COVID-19, and the ACTT-2 trial showed a non-significant improvement in mortality with baricitinib versus placebo. 22 Of note, these trials did not specifically examine outcomes in ICU cohorts, and detailed Bayesian analysis has since suggested that the mortality benefit of tocilizumab may be less certain in patients who are mechanically ventilated. 23 The lack of significance for immunomodulator use in our dataset likely reflects a population more heavily skewed to the severe end of the disease spectrum with an associated high rate of bacterial coinfection (42% overall) as well as a small sample size. It is also important to note that these earlier trials largely encompassed infections with the ancestral SARS-CoV-2 variant in contrast to the circulating B.1.617.2 (Delta) variant at the time of our study. Correlation of existing data with emerging variants of concern of SARS-CoV-2 will be an ongoing research priority.
Within the subgroup of patients on immunomodulatory therapy, mortality was significantly higher in patients who developed a bacterial or fungal coinfection compared with those without (63% vs. 15%; P = 0.038; Figure 1(c)). While not unexpected, this finding is nonetheless intriguing in the context of the previously demonstrated net mortality benefit with tocilizumab amongst a broader COVID-19 cohort. 6
Our study has several limitations. As a single-centre retrospective cohort study with limited sample size, the current findings are subject to inherent biases which may have impacted on the reported outcomes. In particular, certain outcomes of importance such as ECMO requirement did not reach statistical significance between the two groups, and sample size was likely a contributing factor. Availability of immunomodulatory therapy from the Australian National Medical Stockpile at the time of the study was limited to baricitinib and (to a lesser extent) tocilizumab. Hence, there was greater use of baricitinib in this study. For pragmatic purposes, this study combined patients receiving baricitinib and tocilizumab into a single immunomodulator cohort, although the mechanisms of these drugs are different, and outcomes may not be directly comparable. Furthermore, this study is susceptible to confounding, most notably the impact of an increased proportion of patients in the non-immunomodulator group who received sotrovimab in the early phase of their illness and increased use of remdesivir in the immunomodulator group. This study has compared rates of bacterial and/or fungal infections between the two patient groups at a range of sites of disease. For respiratory and urinary tract isolates in particular, incorporation of these coinfection rates without specific interrogation of clinical details to delineate the significance of each as a pathogen versus a colonising organism may have influenced the findings of the study, given the frequency of bacterial colonisation at these sites in ICU patients. The high reported rates of hospital-acquired infections (36.4%, 24/66 patients in the immunomodulatory therapy group; 29.2%, 7/24 patients in the non-immunomodulatory therapy group) may in part be attributable to this approach, although a differential impact would not be expected between the two patient groups.
Additionally, it is important to acknowledge that the global SARS-CoV-2 virological context and also the immunity profile of the human population represent constantly shifting landscapes. The Delta (B.1.617.2) variant examined in this current study is no longer the predominant variant in Australasia or other regions globally, and the SARS-CoV-2 milieu comprises many cocirculating subvariants, largely of the Omicron and Omicron/Delta recombinant lineages at the time of publication. The Australasian population, like much of the world, is no longer immunologically naïve to the SARS-CoV-2 virus, with vaccine-derived, natural infection–derived and hybrid immunity all at play at a population level. Available treatment options, including novel antiviral medications such as nirmatrelvir/ritonavir (Paxlovid) and molnupiravir, and monoclonal formulations such as tixagevimab/cilgavimab (Evusheld), as well as improved treatment algorithms, have evolved considerably since the time of this study. As a result, while the current study provides useful data to understand the clinical context of severe COVID-19 during the Delta (B.1.617.2) wave of 2021, the generalisability of these findings to a different COVID-19 context warrants careful consideration. Overall, randomised data are welcomed on the use of immunomodulatory therapy specifically in ICU patients with severe COVID-19 and also in the context of new SARS-CoV-2 variants of concern.
Antimicrobial stewardship is well established as a key priority in COVID-19 patients due to the risks of evolving antimicrobial resistance and the possibility for transmission of multi-drug–resistant organisms. 24 Moreover, robust published data have demonstrated that low rates of empiric antibiotic use under close antimicrobial stewardship guidance are not associated with adverse outcomes in COVID-19 pneumonitis. 25 Hence, a careful balance must be struck to prioritise wise empiric prescribing of antimicrobials with thorough and frequent clinical review to identify evolving coinfection. In this context, early sampling of blood and other specimens for culture and prompt empiric antimicrobial therapy when indicated should form part of routine practice when caring for patients on immunomodulatory therapy for COVID-19.
Conclusions
Amongst patients admitted to the ICU with COVID-19, invasive mechanical ventilation, elevated BMI and older age were identified as key risk factors for in-hospital mortality. While use of immunomodulatory therapy in patients with severe COVID-19 was not itself associated with a significantly increased risk of coinfection, a higher fatality rate was observed in our cohort in those patients who acquired such an infection during immunomodulatory therapy. Recognising the limitations of the small sample size of this study, these results raise the possibility that the interplay of novel immunomodulatory therapy and coinfection may contribute to poor outcomes in ICU patients. Clinicians would be wise to consider bacterial and fungal coinfection thoroughly in patients on immunomodulatory therapy and think carefully about empiric antibiotic coverage while culture results are pending and clinical progress is unfolding.
Footnotes
Author Contribution(s)
Acknowledgements
The authors would like to thank the staff of the intensive care unit and the infectious diseases and microbiology departments for their contributions to the clinical management of the patients in this study. We also thank Michelle Thomson for assistance with APACHE-III scoring.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
