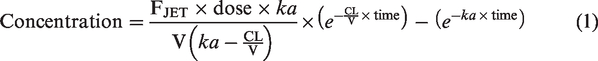Abstract
Jet injection is a drug delivery system without a needle. A compressed liquid drug formulation pierces the skin, depositing the drug into the subcutaneous or intramuscular tissues. We investigated the pharmacokinetics and patient experience of dexmedetomidine administered using jet injection in six healthy adult study participants. This needleless jet injection device was used to administer dexmedetomidine 0.5 μg/kg to the subcutaneous tissues overlying the deltoid muscle. Serum concentrations of dexmedetomidine were assayed at approximately 5 minutes, 15 minutes, 30 minutes, 1 hour and 4 hours after administration. Pharmacokinetic interrogation of concentration time profiles estimated an absorption half time for jet-injected dexmedetomidine of 21 minutes (coefficient of variation 69.4%) with a relative bioavailability assumed unity. In our samples the measured median peak (range) concentration was 0.164 μg/l (0.011–0.325 μg/l), observed in the sample taken at a median (range) of 13.5 minutes (11–30 minutes). The Richmond agitation sedation scale was used to assess the sedative effect, and scored 0 (alert and calm) or –1 (drowsy) in all participants. Five of the six participants stated they would prefer jet injection to needle injection in the future and one had no preference. The findings suggest that the use of a larger dose (>2 μg/kg) would be required to achieve the clinically relevant target concentration of 1 μg/l necessary to achieve deeper sedation (Richmond agitation sedation scale ≤3).
Introduction
Dexmedetomidine is a highly selective alpha 2-adrenergic receptor agonist that is often used as a premedication to decrease the stress and anxiety involved with induction of anaesthesia. It is usually given using intravenous or intranasal routes. 1 , 2 There is considerable between-subject variability associated with intranasal bioavailability (FIN 40.7%, coefficients of variation (CV) 31.8%) and absorption half time (TABS 58 minutes, CV 29%).1 –4 The intravenous route is commonly used intraoperatively, but bradycardia and bimodal blood pressure changes are associated with the rapid changes in plasma concentration. 5 , 6
A potential alternative method of administration is jet injection whereby liquid drug is compressed to a high pressure (>10 MPa) and formed into a jet approximately the width of a human hair. 7 This hair-thin jet allows the fluid itself to penetrate the skin and deposit in the subcutaneous or intramuscular tissues. 8 , 9 By performing delivery without a needle, jet injection avoids the associated issue of needle phobia. 7 Jet injection is a rapid procedure with delivery of drug taking less than 1 second. These benefits could allow jet injection to overcome the difficulties (e.g. patient anxiety, pain) associated with intravenous cannula placement for administration of dexmedetomidine while also improving the accuracy of dose administration compared with intranasal delivery. We investigated dexmedetomidine pharmacokinetics when given using a jet injection device and the experience of the delivery method by the participants. Time concentration profiles from the limited number of participants in this current study were pooled with time concentration profiles determined after intravenous administration in order to estimate absorption half time (TABS) of the drug delivered by jet injection, a clinically useful parameter to compare with other methods of administration.
Materials and methods
This was a healthy adult pharmacokinetic study of dexmedetomidine administered using a jet injection device. Physiological observations and sedation scores were recorded and participants were asked about pain on injection of this delivery method.
The study protocol was approved by the New Zealand Health and Disability Ethics Committee (reference 21/STH/212). Inclusion criteria were age 18 years or older, capacity to understand the purpose and requirements of the study, adequate written and verbal English language skills for all communication to be conducted in English and to provide written consent. Exclusion criteria were pregnancy or plans of pregnancy in next 6 months, a known medical condition, resting heart rate lower than 45 beats/minute (bpm), conduction issues on ECG, known allergy or sensitivity to dexmedetomidine and any other medication taken within the previous 24 hours.
Ten fasted (6 hours for food and 2 hours for clear fluids) participants were admitted to the study site on a single day. On arrival, baseline observations and ECG were performed and written consent was obtained. Three participants were excluded after minor conduction variants on their ECG were noted. A further participant was excluded because medication had been taken the previous evening. A 20 g intravenous (IV) cannula was inserted in the hand or forearm for blood sampling and administration of fluid or medications in case of unexpected adverse reaction. Dexmedetomidine 0.5 µg/kg was prepared from a 200 µg/ml ampoule (Teva Pharma, Australia), and checked in duplicate; and then administered to each participant using the Comfort-in jet injection device (Mika Medical Co., Busan, Korea) into the subcutaneous tissue over the deltoid muscle.
Discomfort during jet injection was assessed using a visual analogue pain scale (0, no pain; 10, worst pain imaginable) during and after dexmedetomidine administration. Heart rate and oxygen saturations were monitored continuously while participants were observed in the recovery unit of our operating theatres by anaesthetic staff and nurses trained and experienced in sedation care.
Clinical observations (heart rate, blood pressure, respiratory rate, blood oxygen saturation) were measured every minute for the first 10 minutes, every 5 minutes for the following hour, and every 30 minutes for next 6 hours. Sedation scores using both the Richmond agitation sedation scale (RASS), 10 which ranges from unrousable (−4) to combative (+4), and a subjective sedation scale were also recorded at these times by nursing staff.
Five venous blood samples (5 mL each sample) were obtained from the IV cannula at time intervals of 3–5 minutes, 7–15 minutes, 20–40 minutes, 1–1.2 hours and 4–6 hours. IV cannulae were flushed with 2 mL normal saline 30 seconds prior to tourniquet placement for sampling. The initial draw (5 mL) was discarded prior to each sample to avoid cross-contamination between samples or with saline flush. The intravenous cannulae were flushed with 5 mL of saline after each sample was taken. Plasma from the samples was separated using a centrifuge at 500 g for 15 minutes and stored at −80°C before drug assay.
The jet injector
Jet injection achieves needle-free drug delivery by forming the drug into a high-speed jet that can break through the skin. These jets are typically around 200 µm in diameter, and travel at a speed between 100 m/s and 200 m/s. 7 Intradermal, subcutaneous, and/or intramuscular delivery has been demonstrated using needle-free jet injection; typically limited to a volume much less than 1 mL (Figure 1). The device used in this study uses a spring to pressurise the drug and form the high-speed jet. This device was designed for subcutaneous delivery of insulin and the speed of the jet is consistent with other studies in which subcutaneous delivery was observed. 9 However, a small amount of injectate reaching the muscle cannot be excluded in subjects where the subcutaneous fat layer is thin.
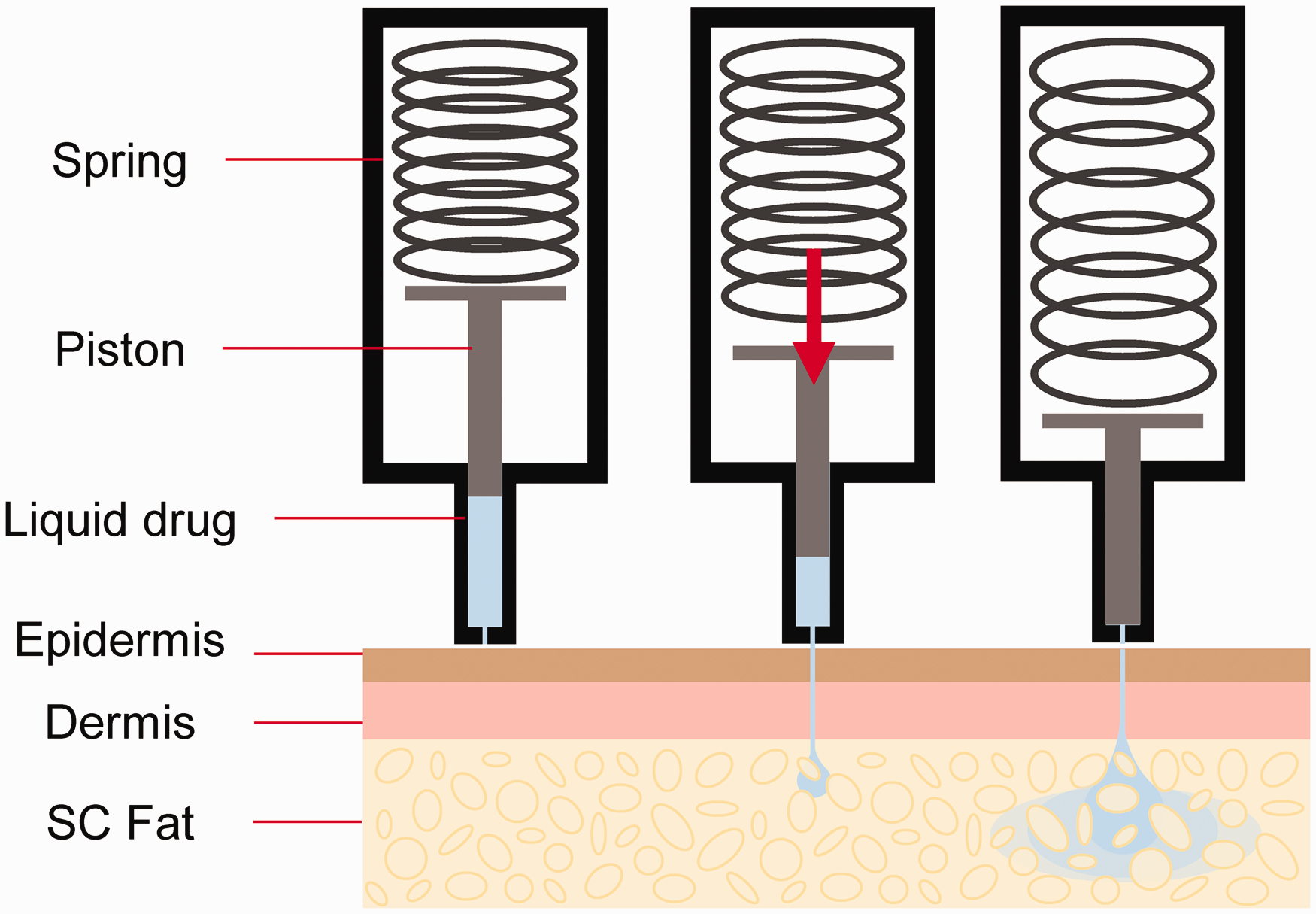
Spring driven jet injection. SC: subcutaneous.
Dexmedetomidine assay
Measurements of dexmedetomidine in plasma were performed using an in-house developed and validated liquid chromatography with tandem mass spectrometry (LC-MS/MS) assay. Briefly, a simple one-step protein precipitation with acetonitrile was used for plasma sample preparation. After precipitation, the clear supernatant was directly injected into the LC-MS/MS system. Dexmedetomidine and its corresponding isotopically labelled internal standard were resolved on a C18(2) column and detected using electrospray ionization LC-MS/MS in the positive mode. Dexmedetomidine standard curve was adequately fitted by 1/x weighted quadratic regressions (r > 0.999) over the concentration range of 0.001–0.1 µg/L (r > 0.999) and the lower limit of the quantification was 0.001 µg/L. The accuracy and precision were assessed at the low, medium and high-level quality controls. There was no constant direction to the bias quality controls and the mean values were within 10% of the spiked values. The intra and inter-day CV over the analysed concentration ranges were less than 10%. The recoveries of dexmedetomidine from plasma at concentrations of quality controls were similar and consistent, with mean values greater than 95%. No significant matrix effects were observed. Two assays were performed for each sample and the average is reported.
Population modelling
Data pooling
This was a small preliminary study with six participants and a limited number of samples per participant. Therefore, estimation of pharmacokinetic parameters (e.g. clearance and volume) using the study data alone was imprecise. However, the clearance (CL) and volume (V) of dexmedetomidine have been described and this prior knowledge can be used to facilitate estimation of absorption parameters (absorption half time TABS, relative bioavailability FJET).
11
When using a one-compartment mammillary model:
Pharmacokinetic analysis
A two-compartment pharmacokinetic model with first order elimination was used to describe dexmedetomidine pharmacokinetics. The model was parameterized in terms of clearance (CL), volumes of distribution (V1, V2) and intercompartmental clearance (Q2). Allometric theory was used to quantify size-related changes in pharmacokinetic parameters. A depot compartment was used to model the absorption rate constant (ka) that was expressed as an absorption half-time (TABS):
Pharmacokinetic parameters (e.g. CL, Q2, V1, V2) were standardized to an adult measure of body size with a standard weight of 70 kg using allometric scaling.20 –22 Population parameter variability (PPV) was accounted for using an exponential model for the random effect variables (η). This assumes a log-normal distribution and avoids parameter estimates falling below biologically plausible values.
Residual unidentified variability (RUV) was modelled using both proportional and additive residual errors. The between-subject variability (ηRUV,i) of the RUV was also estimated for data.
Population parameter estimates were obtained using non-linear mixed effects models (NONMEMs; 7.5 ICON Development Solutions, USA) with first-order conditional estimation and a convergence criterion set to three significant digits. The population mean parameters, between-subject variance and residual variance were estimated using the first order conditional interaction estimate method using ADVAN13 TOL = 9 of NONMEMs.
Model selection was also based on parameter plausibility and prediction-corrected visual predictive checks (PC-VPC) plots. 23 Shrinkage considers the quality of the observed data. The term η-shrinkage refers to the between-subject variability. When the observed data are informative η-shrinkage approaches zero and when the data are less informative it approaches 1. Bootstrap methods provided a means to evaluate parameter uncertainty. 24 A total of 100 bootstrap replications were used to estimate parameter means and confidence intervals. Results from the population models are presented as parameter estimates, together with their 95% confidence intervals. Between-subject parameter variability is expressed as an apparent coefficient of variation obtained from the square root of the variance estimate.
Simulation using parameter estimates
To see how our results might translate into clinical practice, a simulation study was performed using a differential equation solver (Berkeley Madonna, Robert Macey and George Oster of the University of California at Berkeley, USA) to investigate the likely plasma concentrations in adults when given a sedative dose of dexmedetomidine (0.5 mg) using the jet injector; and compared with intranasal dexmedetomidine 3 µg/kg. Pharmacokinetic parameter estimates and their variability from this current analysis were used to predict individual time–concentration profiles after jet injection. An absorption half-time for nasal administration of 58 minutes (CV 29%) with a relative bioavailability of 40% (CV 31.9%) and an effect compartment equilibration half-time of 3.3 minutes was assumed for intranasal administration. 2
Results
There were six participants aged 19–30 years (four women and two men), weighing 58–95 kg available for data collection for the study.
Pharmacokinetics
Descriptive non-compartmental analysis
The observed median peak concentration (CMAX) was 0.164 µg/l (range 0.011–0.325 µg/l) and was observed (TMAX) at a median of 13.5 minutes (range 11–30 minutes) after jet injection. There was one participant with very low plasma concentrations (Figure 2), which was associated with incomplete delivery of the dexmedetomidine dose to that subject, evident by injectate present on the skin surface after jet injection.
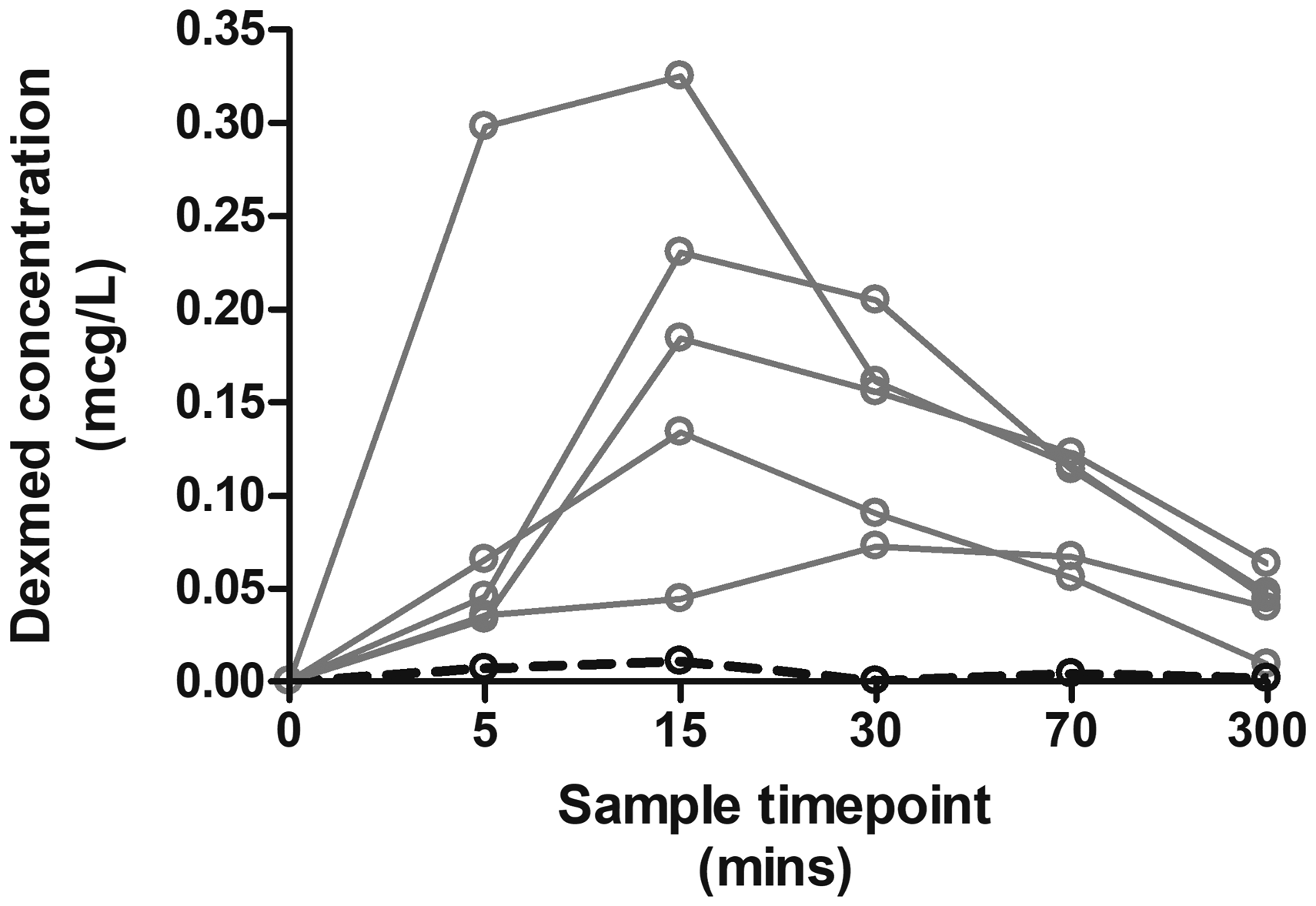
Dexmedetomidine serum concentration profiles for each subject. There was one participant with a very low serum concentration (dashed line), which was associated with incomplete delivery of the dexmedetomidine dose to that participant. Dexmed: dexmedetomidine.
Pooled compartment pharmacokinetics
Parameter estimates for the pooled data determining the dexmedetomidine model are shown in Table 1. There were 2204 dexmedetomidine concentrations that were amenable for modelling in the pooled dexmedetomidine pharmacokinetic analysis. This pooled data analysis included 30 drug concentrations from the six participants administered dexmedetomidine using the jet injector. A two-compartment pharmacokinetic model was adequate to describe concentration profiles. The final model included allometric scaling of pharmacokinetic parameters using total body weight. The PC-VPC plots are shown in Figure 3. The absorption half time was 21 minutes (CV 69.4%) with a relative bioavailability of the jet (FJET) assumed unity. The relative bioavailability in the one participant who had a poorly performed injection, and with low plasma concentrations, was 16%.
Dexmedetomidine population pharmacokinetic parameter estimates.
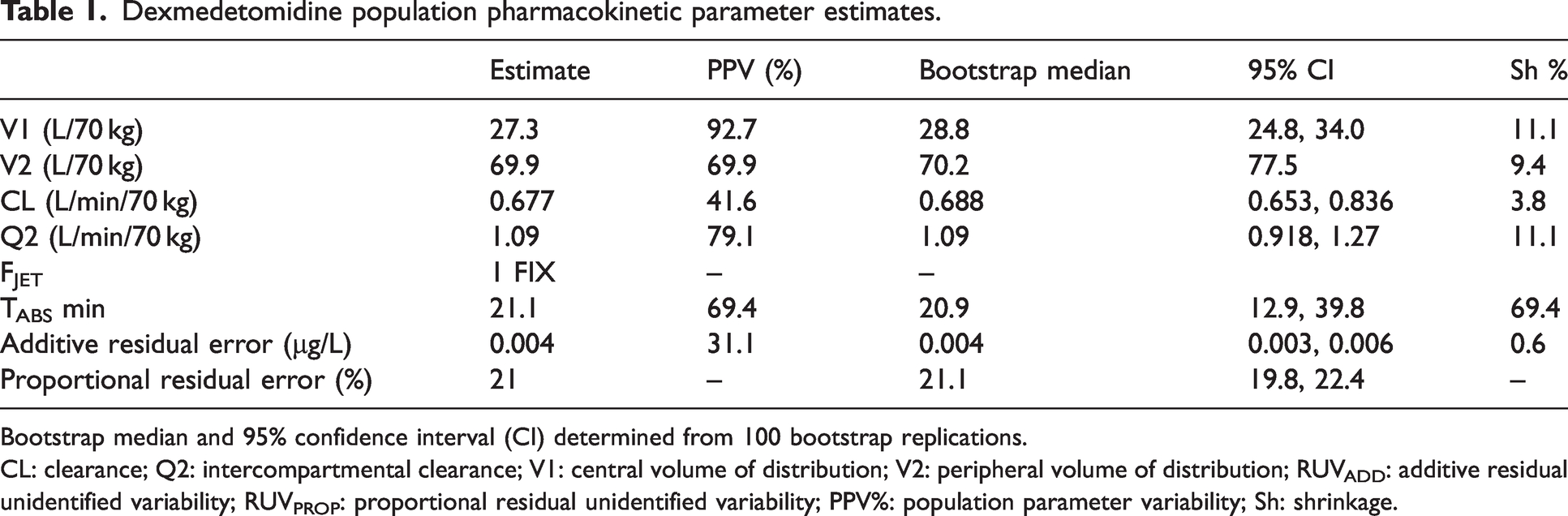
Bootstrap median and 95% confidence interval (CI) determined from 100 bootstrap replications.
CL: clearance; Q2: intercompartmental clearance; V1: central volume of distribution; V2: peripheral volume of distribution; RUVADD: additive residual unidentified variability; RUVPROP: proportional residual unidentified variability; PPV%: population parameter variability; Sh: shrinkage.
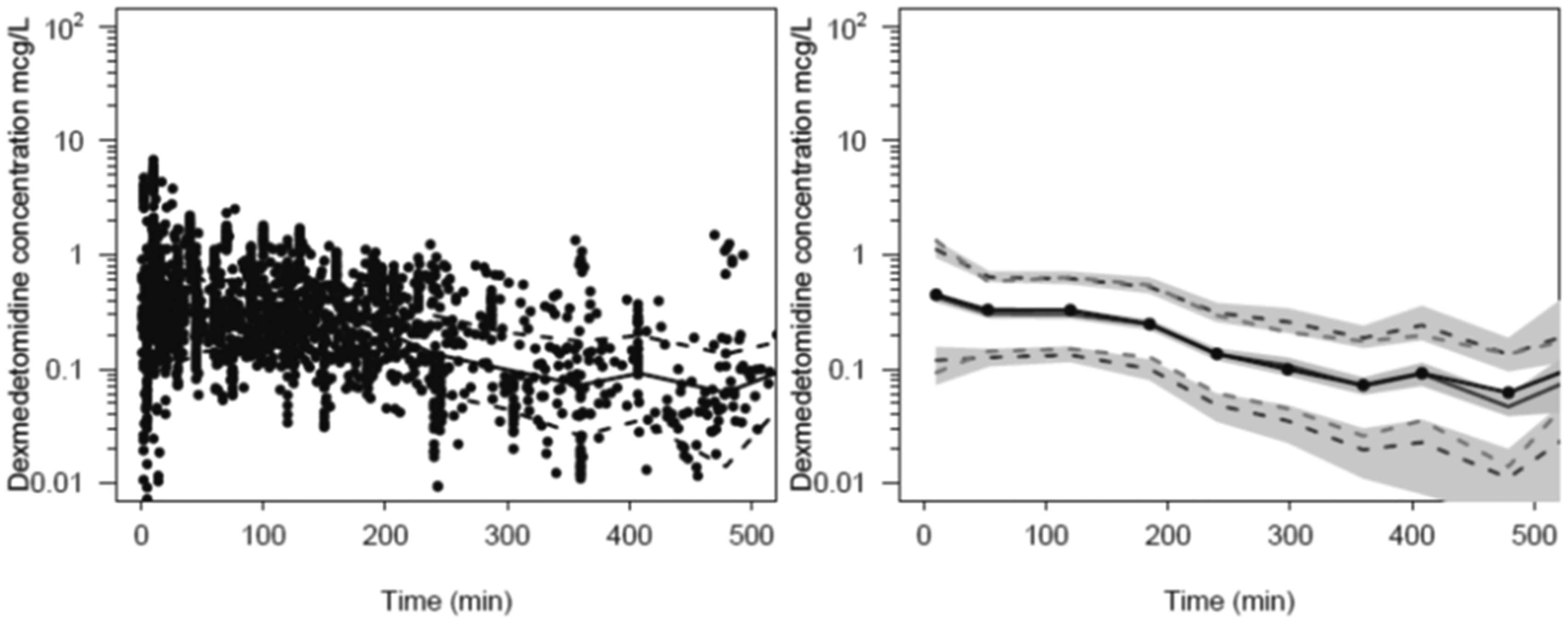
Prediction-corrected visual predictive check (PC-VPC) for the universal dexmedetomidine pharmacokinetic model. Plots show median (solid) and 90% intervals (dashed lines). The left-hand plot shows all prediction corrected observed dexmedetomidine concentrations. The right-hand plot shows prediction corrected percentiles (10%, 50% and 90%) for observations and predictions (dashed lines) with 95% confidence intervals for prediction percentiles (median, 5th and 95th) with shading.
Simulation
Due to the nature of our study, serum samples were only available for analysis at discrete time points. The limited number of these, especially just after injection meant the true plasma peak may have occurred just before or after the observed highest measured serum concentration. There is consequently considerable imprecision with observed CMAX and TMAX estimates. Simulation concentration profiles using estimated pharmacokinetic parameters and their associated between-subject variability allows better estimation of these confounded parameters (CMAX and TMAX).
Time–concentration profiles (1000 simulations) after a modelled jet injection of dexmedetomidine 0.5 mg/kg, and after intranasal delivery of dexmedetomidine 3 mg/kg are shown in Figure 4. The CMAX of 0.41 µg/l was achieved at a TMAX of 25 minutes after jet injection. The CMAX 0.5 µg/l was achieved at a TMAX of 40 minutes after intranasal administration.
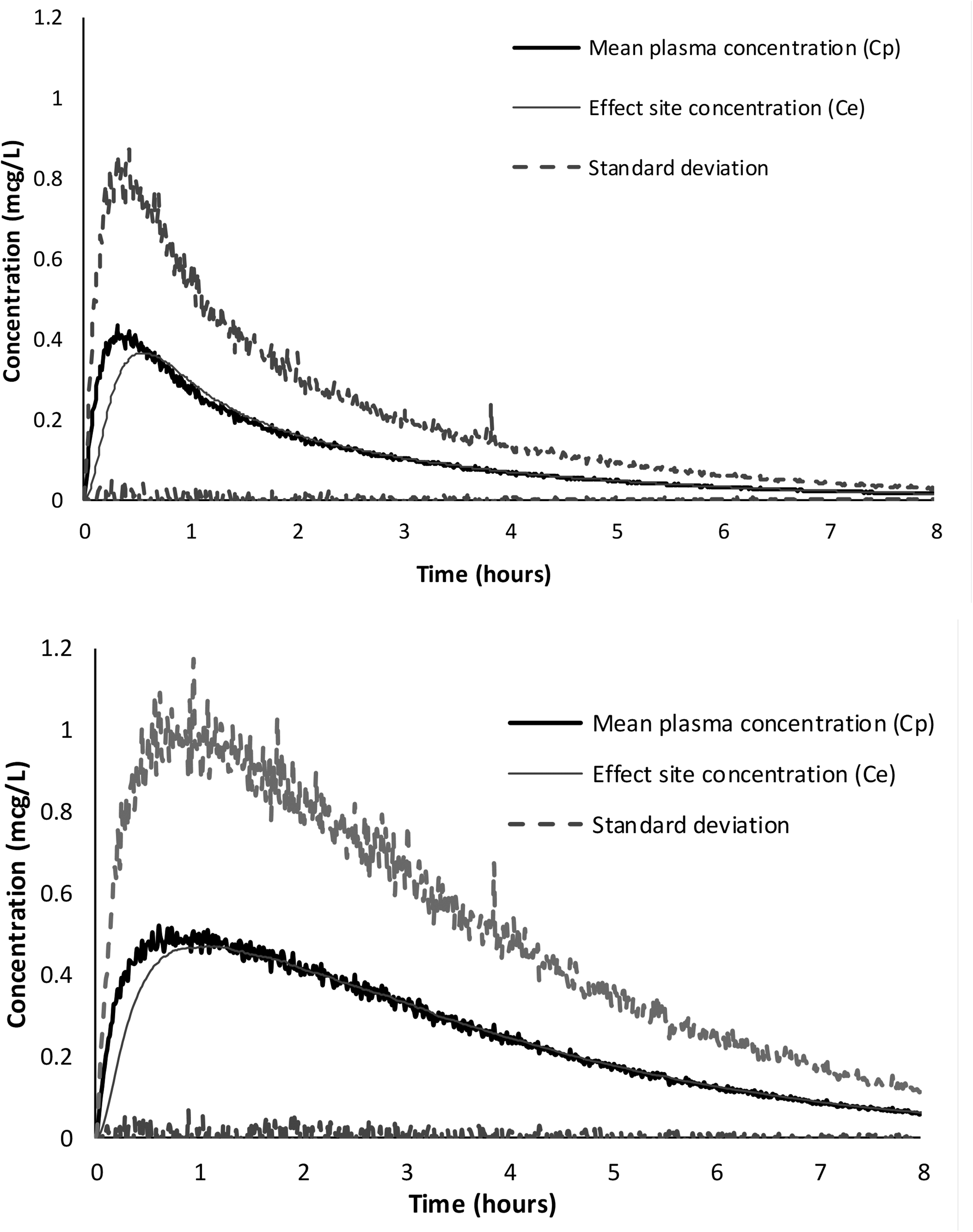
Simulated time–concentration profiles comparing a jet injection of dexmedetomidine 0.5 µg/kg (upper panel) and intranasal delivery of dexmedetomidine 3 µg/kg (lower panel). An absorption half-time for nasal administration of 58 minutes (coefficients of variation (CV) 29%) with a relative bioavailability of 40% (CV 31.9%) and an effect compartment equilibration half-time of 3.3 minutes was assumed for intranasal administration.
Pharmacodynamics
Sedation
In this study the objective sedation scores (RASS) were all 0 (alert and calm) or −1 (drowsy). These sedation scores reflect minimal sedation. Some subjective sedation was self-reported by five participants, and no subjective sedation was reported by one participant. This participant was not the one with the lowest observed concentration profile and their observed peak plasma concentration (CMAX) was below 0.2 µg/l. A relaxed feeling was reported by the participant with the lowest measured plasma concentrations.
Cardiovascular changes
The participants’ heart rate decreased between 20% and 30% following dexmedetomidine administration over a wide range of time. The lowest heart rates generally occurred between 2 minutes and 180 minutes, but there was one self-resolving symptomatic 46% reduction in heart rate to 42 bpm with systolic blood pressure of 81 mmHg, consistent with a vasovagal episode, associated with intravenous line removal 365 minutes after dexmedetomidine administration. Excluding this event, the systolic blood pressure systolic reduction range was 11–22% with nadirs occurring between 30 and 90 minutes. These modest effects are consistent with previously reported cardiovascular pharmacodynamics of dexmedetomidine.
Local injection site effects
A range of pain on injection was reported from 1/10 to 8/10 (visual analogue scale 0–10). Five participants stated a preference for jet injection over a needle in the future. The remaining participant felt they would be equivalent. The participant with the lowest pain score on injection had an incomplete injection with some injectate present on the skin after injection. There were no visible signs of irritation at any of the injection sites during the study and no pain, redness, irritation or any other issues were reported on phone follow-up the next day.
Discussion
This preliminary study demonstrates the potential of jet injection as a method of dexmedetomidine administration in awake participants. The device proved satisfactory for most participants, but is operator dependant; drug delivery was poor in one participant in whom injectate was evident on the skin surface after jet injection, and relative bioavailability was only 16%. The absorption half-time of 21 minutes (95% confidence interval 12.9 to 39.8 minutes) was associated with considerable between-subject variability (CV 69.8%).
Absorption of jet-injected dexmedetomidine from subcutaneous tissue was moderately slow (TABS 21 minutes), although more than twice as quick as that reported with intranasal delivery (TABS 58 minutes). 2 Rapid achievement of sedation using dexmedetomidine is undesirable because the drug causes adverse cardiovascular effects, worsened with the high concentrations associated with a rapid bolus dose. 25 It has been suggested that the administration of an intravenous loading dose of dexmedetomidine should be over 10–20 minutes, at a dose rate of 6 µg/kg/hour. 26 Thus the absorption half-time after jet injection (Figure 4) could be considered akin to slow intravenous infusion. The onset time after jet injection is shorter than after nasal administration, but concentrations are sustained above a target concentration for a longer duration than intravenous bolus.
Dexmedetomidine 0.5 µg/kg delivered by jet injection in this study achieved plasma concentrations below 0.4 µg/l. We report RASS scores of 0 (alert and calm) or –1 (drowsy) in all volunteers. These sedation scores are consistent with a sedation pharmacodynamic model that reported modified observer’s assessment of alertness/sedation (MOAA/S 0–5) score of greater than 4 with a corresponding bispectral index of 80–100 with plasma concentrations less than 0.4 µg/l. 1 In order to achieve some clinical sedation (i.e. bispectral index 80–60 and MOAA/S 3–4), it would be necessary to achieve an effect compartment concentration range of 0.6–1 µg/l. These concentrations would require a larger dose of dexmedetomidine of around 1.5 µg/kg. Consistent with our observations, previous studies have shown that subcutaneously delivered dexmedetomidine has attenuated cardiovascular effects compared with IV drug administration. 27 The slower absorption avoids the high peak plasma concentrations, and thus has fewer consequent cardiovascular changes, which would be an advantage over IV administration.
None of the participants would prefer a needle injection to this spring-loaded jet injection in the future. However, some reported pain scores on jet injection that might be unacceptable in some patient groups (e.g. children). The comfort and success of a jet injection depends on the jet characteristics, such as the speed and shape of the jet, 9 , 28 as well as how the device contacts the skin surface tissue. Electronically controllable jet injectors are a future option that could improve understanding of the relationship between jet injector characteristics and comfort. 29 Bennett et al. compared jet and needle injection of midazolam and saline. 30 The authors showed that although jet injection was less painful than needle injection in both groups, jet injection of midazolam was more painful than needle injection of saline. When administered to children jet-injected midazolam was found to have a lower parental satisfaction score than oral midazolam, 31 but jet-injected midazolam had higher parental satisfaction when compared with IV midazolam. 32 Greenburg et al. noted groups of children receiving larger volumes of jet-injected midazolam were associated with more discomfort and crying over a volume range of approximately 0.1–0.7 ml. 33 Despite the limitations of jet injection in some settings, avoidance of the needle alone may have advantages in agitated adult patients, to avoid the danger of sharps injuries, or for specific needle phobia.
The delivery of doses required to reach clinically useful levels of sedation in the adult population using our study device would require more than one jet injection. To achieve this in a single injection two options could be considered for future investigation; use of a larger injectate volume or use of a more concentrated formulation of dexmedetomidine. Concerns about pain on injection may limit the larger injectate volume.
Footnotes
Author Contribution(s)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Waikato Medical Research Foundation.