Abstract
A 32-year-old multigravida woman, with known familial hypokalaemic periodic paralysis, underwent spinal anaesthesia for an elective lower segment caesarean section. There are several case reports in the literature discussing the optimal anaesthetic technique. In the past there has not been an emphasis on aggressive and early potassium replacement. A target level to commence replacement of potassium at 4.0 mmol/L or less is proposed. Careful preoperative preparation, frequent perioperative monitoring and early potassium replacement resulted in no perioperative episodes of weakness in this case, in contrast with other case reports where potassium was either not monitored or not replaced early enough, resulting in postoperative attacks. Another factor to consider in hypokalaemic periodic paralysis is the avoidance of triggers, including certain medications. Misoprostol was used in this instance to avoid potential electrolyte derangements from other uterotonics.
Keywords
Case history
The patient was a 32-year-old multigravida woman, admitted for an elective lower segment caesarean section (LSCS). The patient gave consent for publication. She had no other comorbidities except a known diagnosis of familial hypokalaemic periodic paralysis (hypoKPP), diagnosed at approximately 8 years old. She had undergone two previous surgeries, a septoplasty under general anaesthesia and a previous LSCS under spinal anaesthesia. She identified several triggers for her episodes of weakness, including high carbohydrate intake and rest after prolonged exercise. She had no history of palpitations or cardiac arrhythmias, or any respiratory or oropharyngeal muscle involvement. She was on preventive medication consisting of acetazolamide, slow-release oral potassium (K+) (Slow K, Novartis Pharmaceuticals Australia Pty Ltd.) and an additional faster acting oral potassium replacement (Chlorvescent, Aspen Pharmacare Australia Pty Ltd.) as required for symptom control.
She had been reviewed in the anaesthetic antenatal assessment clinic during her pregnancy to formulate a plan for her surgery. Preparations consisted of continuing her regular acetazolamide and oral potassium the morning of surgery, active warming in the anaesthetic bay with a convection blanket to minimise the risk of hypothermia as a trigger and being first on the list to minimise anxiety. A radial artery cannula was inserted in the anaesthetic bay for regular collection of arterial blood gases for K+ measurements perioperatively (Figure 1). A spinal anaesthetic was given using 2.2 ml heavy bupivacaine, 25 μg fentanyl and 100 μg morphine, the standard practice in the author’s institution. Phenylephrine was chosen as the planned vasopressor to avoid any beta-adrenergic effects; however, she did not require any throughout the case. Glucose-containing fluids were avoided. Misoprostol (given sublingually immediately after delivery of the baby) was chosen as the drug for primary prevention of postpartum haemorrhage, to avoid any possible fluctuations in electrolytes. She had an estimated blood loss of 300 ml intraoperatively with a well contracted uterus post-delivery of the placenta. During the surgery the K+ was closely monitored every 15–20 minutes using a blood gas analyser situated in the theatre complex. The treatment aim was to maintain K+ greater than 3.5 mmol/L as weakness is associated with levels less than the normal range. Once K+ fell to 3.5 mmol/L an intravenous (IV) potassium chloride infusion was commenced at 30 mmol/hour. This was continued during the case and ceased in the postoperative care unit (PACU). She complained of some discomfort at the IV cannula site but was able to tolerate it during surgery. She had no arrhythmias.
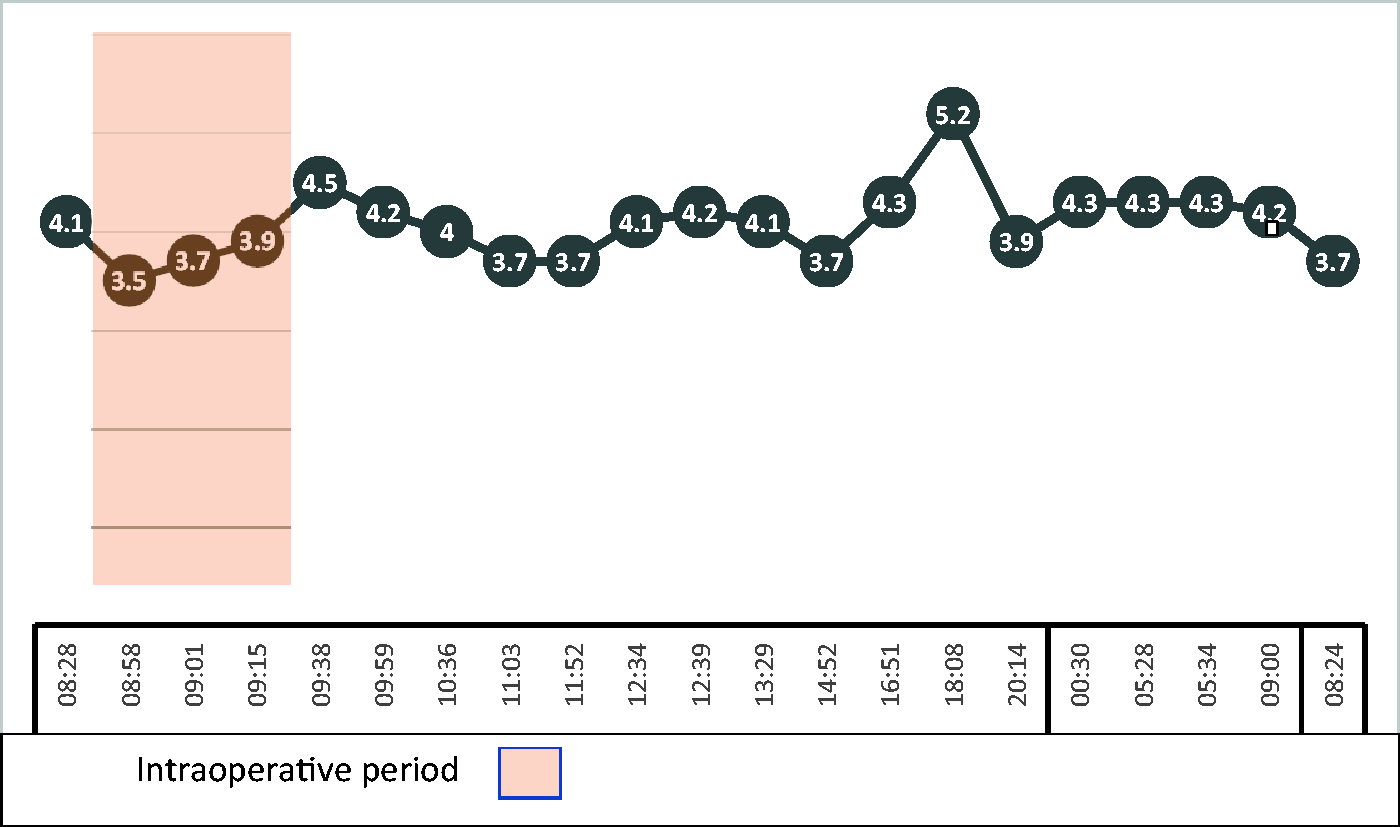
Arterial blood gas potassium levels (mmol/L). Timing of potassium (K+) administration: 08:50 potassium chloride (KCl) infusion commenced at 30 mmol/hour; 09:40 KCl infusion ceased in postoperative care unit (PACU); 11:03 oral K+ replacement given in the PACU.
In the PACU her K+ fell to 3.7 mmol/L, and she was given two tablets of rapid-acting oral effervescent potassium replacement, each containing 14 mmol of K+. Her K+ levels continued to be monitored closely every 30 minutes in the PACU until transfer to the high dependency unit (HDU) for postoperative care. Once her spinal block regressed, she had full power and no episodes of weakness.
Immediately postoperatively she recommenced her usual K+ replacement and preventive therapy. Her K+ levels were closely monitored overnight in the HDU without any requirement for IV replacement. She experienced no arrhythmias or episodes of weakness and was discharged from the HDU to the maternity ward early on postoperative day 1. On review by the pain team that day she was comfortable using oral analgesia, had mobilised and showered independently with no episodes of weakness and was able to nurse her newborn independently. She was discharged home on day 3 and followed up with home midwife visits and a phone call by the author at 3 weeks. She had not had any episodes of weakness at any time following discharge.
This contrasts with her previous two anaesthetics, both of which resulted in some episodes of weakness postoperatively. The author was unable to obtain details of the anaesthetic for the septoplasty or if there had been any intraoperative K+ supplementation as the patient could not recall where she had the surgery. Her previous LSCS was under a spinal anaesthetic also but with no intraoperative K+ replacement. She was transferred to the HDU for postoperative care on that occasion where a K+ infusion was commenced. She describes two periods overnight of being ‘30–40% paralysed, able to move but weak, like after a run’. However, the next morning she was walking and showering with help and the episode resolved after a few hours.
Discussion
HypoKPP is a rare genetic disorder with a prevalence of approximately 0.13 per 100,000,1,2 typically affecting the Caucasian population. 3 Two thirds of cases are genetically determined by autosomal dominant inheritance and the rest are sporadic. There is variable penetrance, it being decreased in women, leading to men being more clinically affected.1,3–5 This skeletal muscle channelopathy is caused by point mutations in voltage-gated calcium (Ca2+) and sodium (Na+) channels. Aberrant depolarisation results in muscle membrane inexcitability and paralysis, triggered by hypokalaemia. There are two types of familial hypoKPP. Type 1 is the most common, with the mutation on chromosome 1, involving the muscle CACNA1S Ca2+ channel on the dihydropyridine receptor. Type 2 accounts for approximately 10% of cases, with the mutation on chromosome 17, involving the muscle SCN4A Na+ channel. 1
Features of hypoKPP
Symptoms
Symptoms usually appear in the second decade of life, but this can be variable. After the age of 25 years, thyrotoxic periodic paralysis becomes the more likely diagnosis, as it is the most common acquired form of periodic paralysis. 3
There are key features of the attacks, which help to diagnose the condition. The attacks are reversible and treatable and have clearly identifiable triggers (Table 1).6,7 The attacks are episodic, being as frequent as daily to once every few years and lasting from hours to days in duration. The weakness typically affects proximal more than distal limb muscles and very rarely affects ocular, bulbar or respiratory muscles. On examination during an attack, flaccid paralysis with loss of muscle stretch reflexes, but importantly, no myotonia, is seen and consciousness is maintained throughout the attack. The weakness fully resolves between attacks but later in the disease patients may develop severe, fixed, proximal lower limb weakness. Life-threatening cardiac arrhythmias may occur during an attack. The attacks coincide with a drop in serum K+, usually less than 3 mmol/L.1 –3,6
Triggers in hypoKPP.

Diagnosis
Diagnosis is usually made clinically during an attack, demonstrating the listed clinical features in association with a measured low serum K+. Provocative testing with glucose and insulin may be used to precipitate an attack.1,3 Nerve conduction studies can demonstrate decreased amplitude in motor nerves and electromyography can demonstrate electrical silence in the affected muscles during an attack, with complete recovery of normal function in between.4,8 Genetic testing is used to identify point mutations in about 60–70% of cases, with the remainder having no identifiable mutation. 1 Secondary causes of hypokalaemia must be excluded for the diagnosis to be made.
Treatment
Treatment consists of a three-pronged approach: avoidance of known triggers (Table 1), prophylactic medication and acute K+ replacement during an attack.1,4 Attacks have been successfully prevented using carbonic anhydrase inhibitors (acetazolamide and dichlorphenamide), to induce a metabolic acidosis and maintain K+ levels; K+-sparing diuretics (spironolactone and triamterene); and regular oral K+ replacement (10–20 mmol up to three times daily).1,9 It should be noted that about 50% of genotyped patients respond to acetazolamide, most with the Ca2+ channel mutation, exacerbation of the condition being reported with its use in patients with the Na+ channel mutation.1,10 An attack can be staved off with commencement of gentle exercise at its onset, but the mainstay of treatment acutely is K+ replacement, either orally with 0.2–0.4 mmol/kg every 30 minutes (maximum 250 mmol per day) or intravenously with 40 mmol/L in 5% mannitol at 20 mmol/hour (maximum 250 mmol per day). 1
HypoKPP and pregnancy
Labour and parturition are both significant triggers for an attack in hypoKPP, due to the pain involved and subsequent sympathetic nervous system activation and the intermittent intense exercise involved in the second stage of labour. Considering this, epidural analgesia has been successfully used for many years to manage patients during labour.11 –13 Epidural and spinal anaesthesia have been used for many years for LSCS and other surgeries to avoid general anaesthesia because of concern regarding hypoKPP’s association with malignant hyperthermia (MH), 11 to avoid a drop in K+ associated with muscle relaxants, 14 and to attenuate the stress response, minimising the sympathetic stimulation associated with labour and surgery.12 –14 General anaesthesia using both inhalational15 –17 and total intravenous anaesthesia (TIVA)18,19 techniques has been reported in pregnancy. All these techniques have been used successfully, with no precipitation of MH. There seems therefore to be no advantage of one over the other from the perspective of the hypoKPP patient during pregnancy.
Issues for consideration in hypoKPP
There are some debated issues surrounding the anaesthetic management of familial hypoKPP. These include the potential association of familial hypoKPP with MH, the optimal anaesthetic technique to employ (general anaesthesia vs. regional techniques), other anaesthetic considerations, and when and how rapidly to administer K+ replacement during an anaesthetic. However, there is little in the literature regarding a definitive target K+ at which to commence replacement.
MH association
There are case reports in the literature in which inhalational anaesthetics, which are known triggers for MH, have been used without triggering an MH episode in patients with known or undiagnosed familial hypoKPP.15,16,17 It is noteworthy to mention that in the cases of Marchant et al. the muscle rigidity was interpreted as suspected MH, but on testing only one of the patients was positive and had a recognised mutation for MH. 20 This muscle rigidity observed may well be the slowed twitch relaxation that was noted in three of the 13 patients with familial hypoKPP in the study of Lehmann-Horn and Iaizzo, 21 when muscle fibres from these patients were exposed to very high concentrations of caffeine and halothane. This abnormally slowed relaxation, causing a type of mild contracture, which did not seem related to the pathologically increased reticular release of Ca2+ as are the contractures seen in MH. 21 The case report of Lambert et al. 22 of a patient with hypoKPP and MH did not actually demonstrate any conclusive diagnosis of MH following the patient’s intraoperative incident. MH testing results classified the patient as MH-equivocal and may well demonstrate this slowed relaxation. Furthermore, in the study of Lehmann-Horn and Iaizzo 21 and the study of Rayan and Hanna, 23 suxamethonium, another known trigger for MH, did not cause contracture in any of the patients with hypoKPP. The findings in these case reports suggest a lack of association between MH and familial hypoKPP.
Neuromuscular blockers
Depolarising muscle relaxants should be avoided due to the potential changes they produce in the membrane potential and serum electrolytes and the possibility of precipitating adverse events.14,15,18 However, non-depolarising muscle relaxants (NDMRs), have been used without adverse events,15,18 and it appears that there is no contraindication to their use. It is advised to use short-acting NDMRs in preference to longer-acting agents. 18 The author suggests that another option is to avoid the use of muscle relaxation altogether, using an alternative technique involving TIVA with remifentanil and propofol to achieve ablation of airway reflexes and a degree of muscle relaxation, along with topicalisation of the larynx and airway with 2% lidocaine by way of an atomiser.
K+ target levels and replacement
K+ levels need to be maintained within a normal range to prevent attacks in hypoKPP. The decreases in K+ can occur quickly and precipitously. It is therefore very important to have a clear target range for replacement. Reviewing the case reports available, the lowest K+ measurement which did not result in paralysis postoperatively was 3.3 mmol/L but in both cases the K+ rose quickly, within 20 minutes, to levels greater than 3.5 mmol/L.15,19
A comparison of the case reports in which there was no postoperative paralysis compared to paralysis, often prolonged, has highlighted a very important aspect of the management of patients with hypoKPP. In the case reports in which there was preoperative K+ measurement and replacement if it was in the low normal range, in conjunction with frequent intraoperative measurement of K+ levels and intraoperative K+ replacement there were no reports of postoperative episodes of paralysis.13 –15,18,19 In contrast, in the case reports in which there was no preoperative replacement despite low K+ levels or no intraoperative (or infrequent) 16 measurement of K+ levels and no intraoperative replacement, in each case prolonged postoperative paralysis was reported.11,16,17,24 –26
In view of this, every patient with hypoKPP should receive preoperative K+ replacement if it is in the low to low normal range. Intraoperative monitoring of serum K+ levels should be frequent, at least every 20 minutes and K+ should be replaced aggressively (20–30 mmol/hour) and without delay once the K+ begins to drop.
If access to a blood gas analyser in the theatre complex is not possible, another option is a point-of-care electrolyte testing system, such as the Abbott i-STAT 1.
A K+ level of 3.5 mmol/L was chosen, in this case, to commence the intra-operative K+ infusion as the lowest reported K+ level found in the literature, not causing paralysis, was 3.3 mmol/L.15,19 It may be prudent to consider a target level for commencement at an even higher level, of 4.0 mmol/L, considering the rapidity with which the K+ levels fall in hypoKPP. The author suggests a target K+ for initiation of K+ replacement of 4.0 mmol/L or less. The K+ infusion needs to be ceased with a target in mind: the author suggests once a K+ level of 4.5–5.0 mmol/L is reached, the infusion can be ceased.
When running a peripheral IV K+ infusion, consideration should be given to the size and site of its insertion. A large-bore (16–18 G) IV cannula in the cubital fossa should reduce the discomfort associated with the K+ infusion.
The author also suggests that as there is an increased requirement for K+ in the postoperative period, due to its requirement in cellular repair in healing following surgery, that close monitoring should continue until the patient has resumed their usual prophylactic medications and can take oral supplements for treatment, as dictated by symptoms, in the event of an attack.
Vasopressors and uterotonics
The use of any drug with any beta-adrenergic activity should be avoided. In this case phenylephrine, a pure alpha agonist, was used in preference to metaraminol, which has very mild beta-adrenergic activity. The author suggests phenylephrine is the preferred vasopressor of choice for the parturient with hypoKPP.
One aspect which has not been discussed in the literature is the choice of agent for primary postpartum haemorrhage (PPH) prevention in parturition. The following medications are used in the primary prevention of PPH: oxytocics (carbetocin and oxytocin), ergot alkaloids (ergometrine), and prostaglandins (carboprost and misoprostol). All these medications have side effects, including bronchoconstriction, tachycardia, flushing, nausea, vomiting, diarrhoea, abdominal pain, fever and chills. The oxytocics, by virtue of their similar structure to vasopressin, can also precipitate hyponatraemia. These side effects can produce a degree of emotional and physiological stress that can potentially trigger an episode of hypoKPP. Misoprostol has the most favourable side effect profile of all these medications. It also avoids the risk of precipitating hyponatraemia and thus avoiding any potential electrolyte derangements intraoperatively and postoperatively which can trigger an episode. For these reasons, misoprostol might be considered the most favourable drug to use as a first-line uterotonic agent in the primary prevention of PPH in this condition.
Anaesthetic considerations
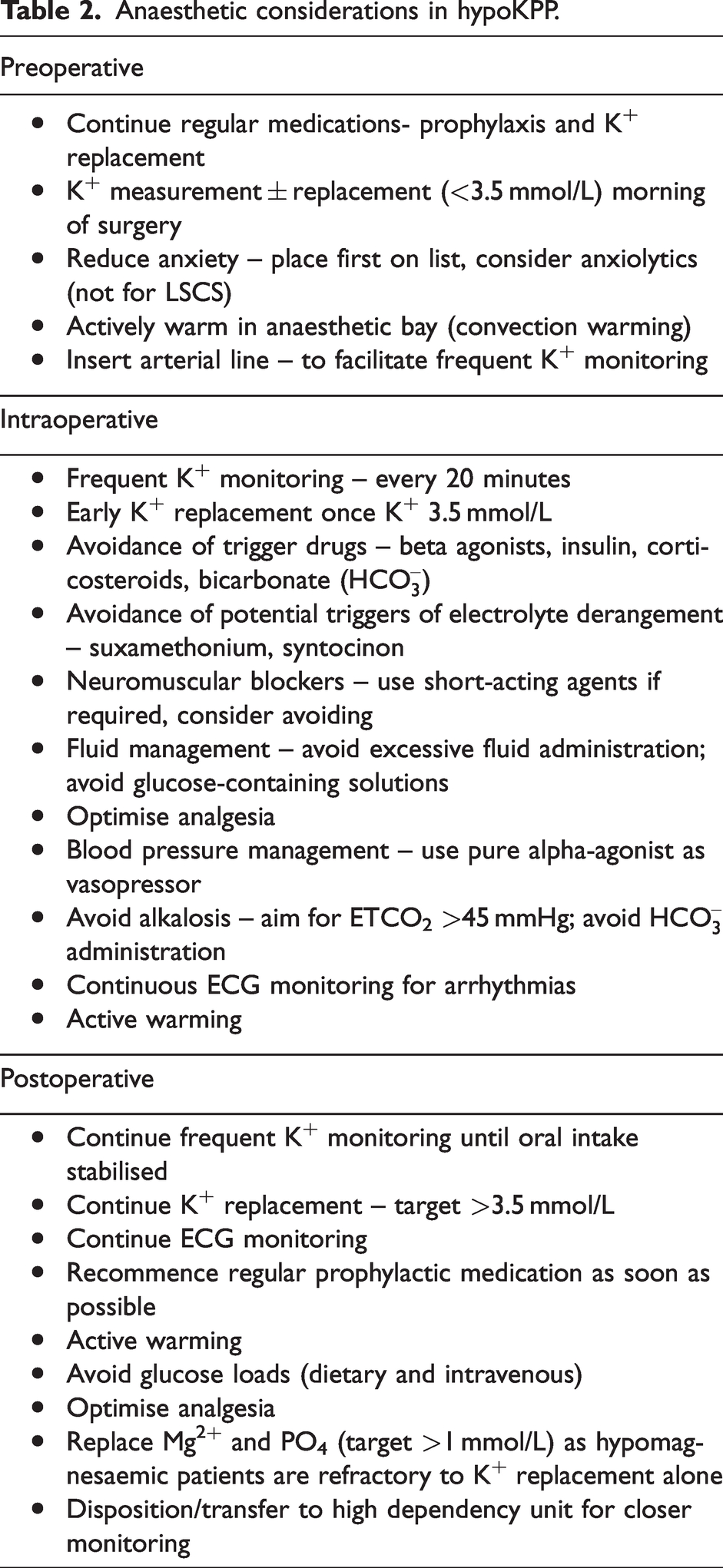
There are a number of anaesthetic considerations in treating patients with hypoKPP, as listed in Table 2.
Anaesthetic considerations in hypoKPP.
Conclusion
There have been reports of successful anaesthetics using both regional and general (inhalational and TIVA) techniques in patients with hypoKPP. There has been debate as to the association of hypoKPP with MH, but this has never been definitively proved. There are many well recognised triggers to an attack and a carefully planned and considered anaesthetic will avoid or prevent these. There is, however, a clear difference in how K+ monitoring and replacement has been approached in the past. Lacking in the literature is a clear guideline of the K+ level at which to initiate K+ replacement. The author suggests that K+ measurement and replacement should commence preoperatively and continue intra and post-operatively to maintain a K+ greater than 4.0 mmol/L. This requires frequent measurements intraoperatively, every 20 minutes, continuing into the postoperative period, until the patient can recommence oral prophylaxis and K+ replacement. Any drugs which can potentially cause electrolyte disturbances should also be avoided, including depolarising muscle relaxants and oxytocin.
Footnotes
Author Contribution(s)
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship and publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
