Abstract

Captain Meredith of the Royal Artillery ‘came under Dr Rollo’s care on the 16th of October 1796’. 1 It was clear from his symptoms—excessive production of sweet-tasting urine, extreme thirst, weight loss and exhaustion—that he was suffering from diabetes mellitus, a disease which had ‘in general resisted every remedy hitherto recommended for its removal’. The treatment Rollo prescribed was drastic. It included a diet of animal protein, principally ‘game, and old meats which have been long kept … fat and rancid old meats’, regular application of hog’s fat to the skin and a nightly draught of wine and opium. Fortunately for Captain Meredith, he recovered completely under this regimen and was soon able to resume a relatively normal diet. Rollo drew some insightful conclusions, including that the disease was related to saccharine digestion and that many of the symptoms, such as excessive urine production and dry skin, were secondary features. The cause, he acknowledged, would remain obscure until ‘the physiology of healthful digestion be properly explained and established’.
It would be many decades before ‘healthful digestion’ was properly understood. Over those years, while many diabetic adults were successfully treated with dietary restrictions, opium and bloodletting remained the mainstays of medical therapy, along with a wide range of quack remedies marketed as digestive elixirs. Meanwhile, childhood diabetes remained a separate, perplexing and universally fatal disease.
In the second half of the nineteenth century, scientists gradually began to unravel the mysteries of the pancreas and its role in digestion and fat metabolism. In 1869, Paul Langerhans described distinct, highly innervated ‘heaps’ of clear cells in the pancreas but wrongly assumed they had a lymphatic function.2,3 Gustave-Édouard Laguesse in Lille conducted extensive histological studies on pancreatic tissue from 1889 onwards, eventually concluding that the cells that Langerhans had described had a role in the regulation of glucose. He developed the concept of a substance which altered the ‘milieu interieur’, coining the term ‘endocrine’.4,5
In 1889, a chance meeting between Oskar Minkowski, a young German physician with a passion and talent for vivisection, and Josef von Mering, a German doctor researching diabetes, led to a major breakthrough. 6 After a heated debate about fat metabolism in a library in Strasbourg, von Mering sent Minkowski a dog from their laboratories so that he could attempt a total pancreatectomy. The dog survived but became incontinent of urine and was unexpectedly found to have diabetes.7,8 In further experiments, they established that diabetes following pancreatectomy could be prevented by leaving the pancreatic blood supply intact, while removing all connections to the gastrointestinal tract. Minkowski recognised that this was likely ‘a functional disturbance of the pancreas’, which would one day lead to ‘successful therapy, just as happened with myxedema, once it was established that the disorder is associated with the loss of thyroid function’. 8
Scientists now knew they were searching for a particular glucose-lowering substance, which probably originated in the pancreas, most likely in the islets of Langerhans. But the dual functions of the pancreas were complicating the search, with digestive enzymes contaminating and confusing all available extraction techniques. 9 In Berlin, Georg Ludwig Zülzer had some success in treating diabetic dogs using alcoholic extracts from calf pancreases and, in 1906, performed probably the first human experiment, injecting a patient in a diabetic coma. The patient showed some improvement before deteriorating rapidly and dying. Several further brief experiments on patients resulted in fevers, tremors and sweats which were attributed to contamination, but in retrospect, they were also possibly symptoms of hypoglycaemia. World War I then intervened, disrupting his research and that of many other scientists around the world who were equally close to a solution. 10
In the immediate aftermath of the war, Frederick Banting, who had served as a medical officer in the Canadian Army, was teaching medical students in Toronto to supplement his struggling medical practice. While contemplating the mechanisms of carbohydrate metabolism in preparation for a lecture, he realised that tying off the pancreatic duct might allow the exocrine pancreas to atrophy and remove the digestive enzymes naturally. To obtain research facilities to test this hypothesis, he approached physiologist and diabetes expert, Professor John Macleod at the University of Toronto. Macleod was sceptical but assigned a final-year physiology student, Charles Best, to work with him over the summer of 1921. 10
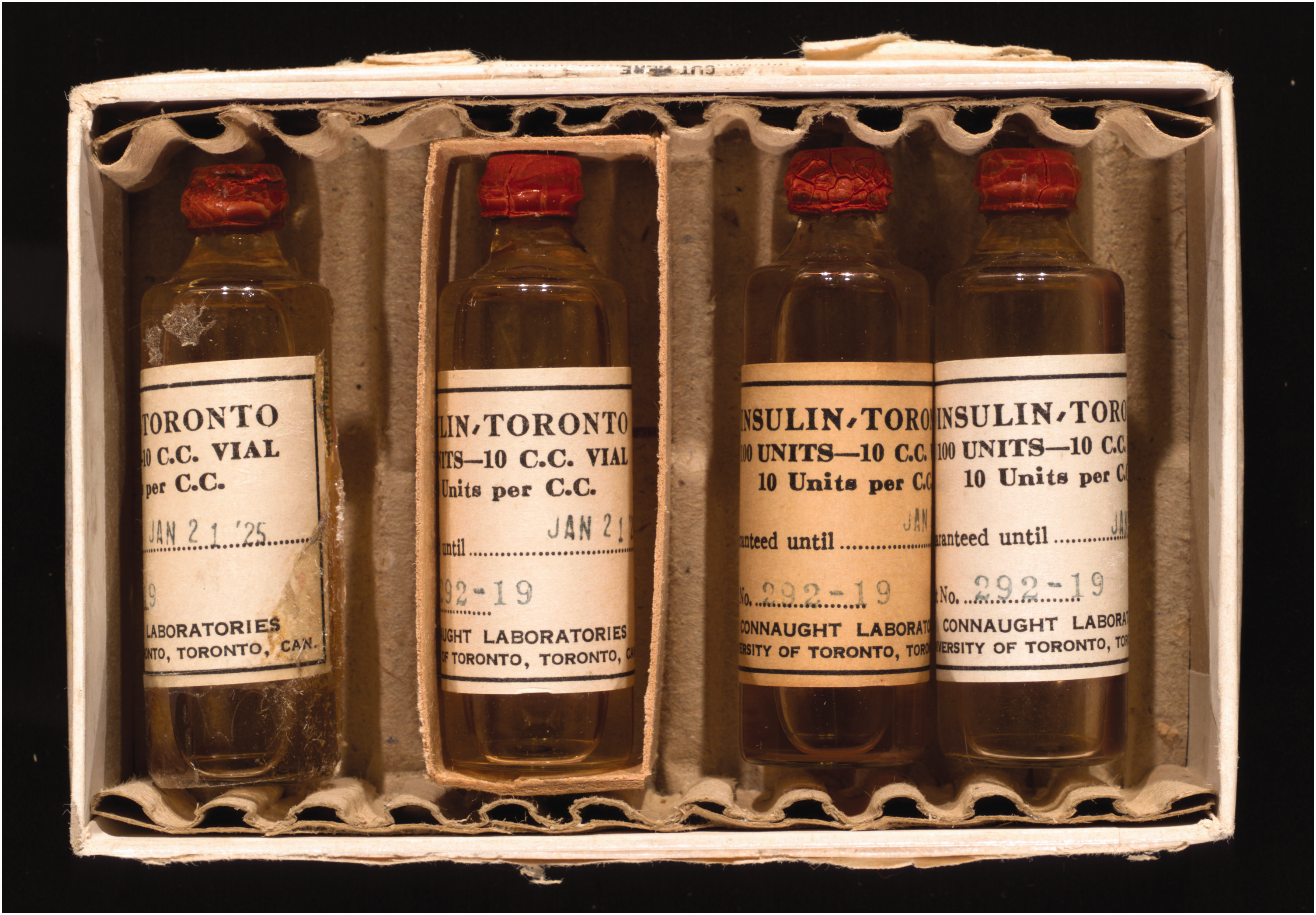
There were many failed experiments over the next few months, interspersed with some encouraging results. When Macleod returned from a summer trip to Scotland, he was sufficiently convinced to increase the laboratory resources for the project, adding James Collip to the team. Collip, a visiting biochemistry professor from Edmonton on sabbatical in Toronto, was quickly able to improve the extraction technique. Banting’s original theory about ligating the pancreatic duct had proved less satisfactory and reliable than they had hoped, and few dogs survived the process for long. Eventually, Banting realised that fetal calf pancreases would be a good source of material, as the islet cells were present in high numbers but the gastrointestinal functions had not matured. 11 This proved to be a far more promising source of insulin.
Work escalated from that point, but so too did the tension in the team. Banting was passionate about his work but reportedly lacked public-speaking skills. 10 As a result, Macleod often intervened when the work was presented, but Banting felt that he was unfairly claiming credit for work that wasn’t his. And even though Collip was invited at Banting’s request, he too became a source of conflict, as Banting felt his own extraction technique was better.
There were many external stresses as well. Senior members of the university ‘who had not invested an hour’s work before the discovery’ were anxious to see clinical results. And as word spread beyond the university, patients began arriving in Toronto, desperate to be part of a clinical trial. ‘Patients with diabetic children found their way right into our laboratory and, in spite of anything we could do, came day after day until plans were made to care for them’. 12 These patients were desperate. Whereas previously children had died on presentation, they were now surviving up to a year on severe calorie-restricted diets. Introduced around 1915 by prominent diabetes specialists Frederick Allen and Elliott Joslin, these diets were a prolonged balancing act requiring close medical supervision. 13 They were difficult for patients to maintain, especially if they returned home. Even Joslin realised how fraught this treatment was: ‘The aspect of these children was truly dreadful. We literally starved the child and adult with the faint hope that something new in treatment would appear’. 14
One of these desperate patients was 14-year-old Leonard Thompson. 15 He had been living with diabetes for more than two years when he was carried into Toronto General Hospital, severely malnourished and unable to walk. As he was deteriorating rapidly, his family agreed to the trial of Banting’s original preparation on 11 January 1922. 10 Despite slight clinical improvement, there were serious local inflammatory reactions to the injections, and further treatment was delayed while Collip further refined his product. On 23 January, Thompson began a daily treatment regimen with dramatic improvement in his health. He was discharged in May, but problems with insulin supply saw him return to his starvation diet without insulin for a few more months before he, and so many other children, were able to obtain a consistent supply of insulin due to an agreement between the university and Eli Lilly and Company of Indianapolis. Thompson managed to live a relatively normal life until his death at the age of 27 from pneumonia and diabetes.
Despite their successes, the tension in the team continued to build, compounded by the awarding of the Nobel Prize in Physiology or Medicine to Banting and Macleod in 1923. 10 There were many other scientists around the world who felt cheated of recognition for their earlier work in the field, and there was considerable debate around the prize. Macleod’s nomination was particularly controversial, as many felt he had not contributed significantly to the discovery. Schack Krogh, a previous Nobel Prize winner who had nominated both Macleod and Banting separately, had personally visited the research facilities. He felt that the work would not have succeeded without Macleod’s resources and Banting's supervision. 4 Others have since written that Macleod made substantial original contributions to the research and that this information was presented to the Nobel Prize committee. 11 No one questioned Banting’s nomination, but he was so upset that Best was not nominated that he contemplated refusing the nomination. Ultimately, he opted to share his prize money with him. Macleod subsequently shared his prize money with Collip, who had also not been nominated.
It is unfortunate, but probably not surprising, that such an enormous breakthrough led to so much controversy and angst within the research team. But the awarding of the Nobel Prize 100 years ago, so soon after the first clinical trials of insulin, is a testament to the vital role of insulin in modern medicine.
