Abstract
The practice of anaesthetists relating to the administration of intraoperative oxygen has not been previously quantified in Australia and New Zealand. The optimal regimen of intraoperative oxygen administration to patients undergoing surgery under general anaesthesia is not known, and international recommendations for oxygen therapy are contradictory; the World Health Organization (WHO) recommend administering an intraoperative fraction of inspired oxygen of at least 0.8, while the World Federation of Societies of Anaesthesiologists, British Thoracic Society, and Thoracic Society of Australia and New Zealand recommend a more restrictive approach. We conducted a prospective observational study to describe the pattern of intraoperative oxygen administration among anaesthetists in Australia and New Zealand and, second, to determine the proportion of anaesthetists who administer intraoperative inspired oxygen in accordance with the WHO recommendations. We identified 150 anaesthetists from ten metropolitan hospitals in Australia and New Zealand and observed the patterns of intraoperative oxygen administration to American Society of Anesthesiologists physical status classification (ASA) 3 or 4 patients undergoing prolonged surgery under general anaesthesia. The median (interquartile range) intraoperative time-weighted mean fraction of inspired oxygen (FiO2) for all participants in the study was 0.47 (0.40–0.55). Three out of 150 anaesthetists (2%, 95% confidence interval 0.4 to 5.7) administered an average intraoperative FiO2 of at least 0.8. These findings indicate that most anaesthetists routinely administer an intermediate level of oxygen for ASA 3 or 4 adult patients undergoing prolonged surgery in Australia and New Zealand, rather than down-titrating inspired oxygen to a target pulse oximetry reading (SpO2) or administering liberal perioperative oxygen therapy in line with the current WHO recommendation.
Introduction
Patients undergoing anaesthesia for surgery receive oxygen at levels higher than that found in room air in order to maintain arterial oxygen saturations as measured by a pulse oximeter (SpO2) that are determined to be safe by the anaesthetist. As a result, a degree of supplemental oxygen is routinely administered to patients under general anaesthesia. However, it is not known whether the fraction of inspired oxygen (FiO2) administered under anaesthesia impacts recovery from surgery, and if so, what FiO2 is optimal. In 2016, the World Health Organization (WHO) published the first recommendations that included guidance on perioperative oxygen therapy as one of 16 intraoperative and postoperative measures for surgical site infection prevention, four of which pertained directly to anaesthesia care. This recommendation was that an FiO2 of at least 0.8 should be administered during anaesthesia and for two to six hours postoperatively for all patients having surgery under general anaesthesia with tracheal intubation, in order to reduce the risk of surgical site infection.1–3 This was one of only two ‘strong recommendations’ put forward in the publication. However, this WHO recommendation has been openly challenged by the World Federation of Societies of Anaesthesiologists and contradicts related guidelines for acute oxygen administration from the British Thoracic Society and from the Thoracic Society of Australia and New Zealand, who advocate for a more restrictive approach to oxygen therapy, aiming to give the minimum amount required to maintain SpO2 above 93%, in order to prevent potential respiratory, cardiovascular, cerebrovascular and reperfusion-related adverse effects associated with prolonged administration of higher fractions of inspired oxygen.4–7 Therefore, widespread agreement on perioperative oxygen administration best practice is lacking and the WHO recommendation continues to be questioned.8–13
Additionally, despite the widespread administration of oxygen under anaesthesia, the patterns of oxygen administration during surgery under anaesthesia have not been quantified in Australia and New Zealand. Accordingly, we have conducted a prospective observational study to outline patterns of intraoperative oxygen administration, and to determine the proportion of anaesthetists in Australia and New Zealand who administer intraoperative FiO2 ≥0.8 in accordance with the WHO recommendation.
Materials and methods
Study design
We performed a prospective multicentre observational study using data collected from intraoperative anaesthesia records of patients undergoing anaesthesia care provided by anaesthetists across ten large metropolitan hospitals with established links to the Australian and New Zealand College of Anaesthetists Clinical Trials Network.
To ensure approximately proportionate representation from anaesthetists at each site, the number of anaesthetists recruited at each site was determined on the basis of the respective number of operating theatres relative to the total number of operating theatres across all participating sites. Specifically, this resulted in a planned enrolment rate of 0.8 anaesthetists per theatre per site. Informed consent was obtained from anaesthetists at participating sites.
Anaesthesia staff who did not practise clinical anaesthesia nor care for any patients that met the eligibility criteria were excluded. Eligible and consented anaesthetists were alphabetically ordered by name, assigned a corresponding number and randomly selected for study participation. Anaesthetists’ lists were prospectively screened for eligible cases and the first case that met eligibility criteria was included for each participating anaesthetist. To reduce the potential for observer bias, anaesthetists were not informed of whether they had been randomly selected for study inclusion, or which anaesthesia event would be included for data collection.
A case was considered eligible if the patient was 18 years or older, judged by the attending anaesthetist to be American Society of Anesthesiologists physical status classification (ASA) score 3 or 4, and was having non-cardiothoracic surgery under general anaesthesia with an expected surgical duration of at least 120 min and anticipated hospital stay of at least one postoperative night. A case was excluded if the site primary investigator determined that there was a specific indication for either liberal or restrictive perioperative oxygen therapy, or the patient was pregnant.
Data collection
Exposure data including FiO2, SpO2 and positive end-expiratory pressure (PEEP) were obtained by research staff from the intraoperative anaesthesia record after completion of surgery. Data collection commenced at the time of skin incision and continued until the time of wound closure. FiO2 and SpO2 data were collected at ten min intervals for the first 120 min, and 30 min intervals for the remaining intraoperative data collection period. PEEP data were recorded at 30 min intervals.
Baseline variables were obtained from the anaesthesia preoperative assessment form and relevant patient medical records by research staff at each site.
Outcome variables
Key outcome variables were the time-weighted average intraoperative FiO2, and the proportion of cases where the time-weighted average intraoperative FiO2 was ≥0.8. Other outcomes were the median highest and lowest intraoperative FiO2, median highest, lowest, and time-weighted average intraoperative SpO2, median time-weighted average PEEP, and duration of surgery.
Baseline variables of interest
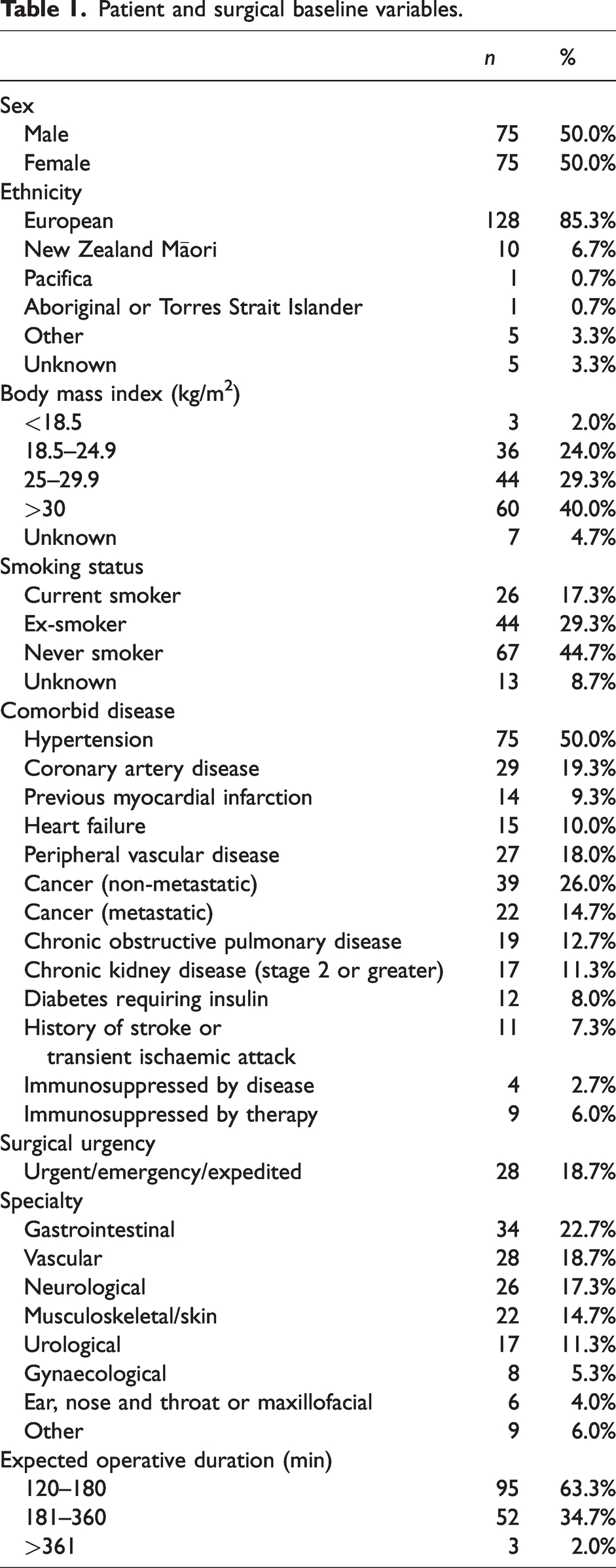
Baseline variables were collected to describe the study patient population and included patient and surgical data, including age, sex, ethnicity, body mass index, chronic comorbidities (hypertension, coronary artery disease, ischaemic heart disease, previous myocardial infarction, heart failure, peripheral vascular disease, cerebrovascular disease, previous transient ischaemic attack or stroke, cancer, chronic obstructive pulmonary disease, moderate or severe renal disease (estimated glomerular filtration rate ≤44 ml/min per 1.73 m2, diabetes requiring insulin, immunosuppressed state), smoking status, category of surgery, urgency of surgery, and immediate preoperative SpO2 and respiratory rate.
Statistical analyses
The sample size was determined on the basis of an expected maximum proportion of 6% of anaesthetists administering FiO2 of ≥0.8 to their patients. A desired confidence of 95% with a confidence interval upper limit of less than 10% resulted in a total required sample size of 150 anaesthetists. A conservative value of 6% was chosen as a recent survey among Australian and New Zealand anaesthetists reported that 2.9% supported the routine use of FiO2 ≥0.8. 14 We considered that a finding of fewer than 10% of overall cases having oxygen administered at an FiO2 ≥0.8 would indicate a lack of general support for the current WHO recommendation.
The time-weighted average intraoperative FiO2 was calculated for each anaesthetist and reported as an overall study median (interquartile range (IQR)). Values for intraoperative oxygen administration were reported for all participants combined, and by site. Adherence to the WHO recommendation was reported as the proportion of cases with a mean time-weighted intraoperative FiO2 ≥0.8. The Clopper–Pearson exact method was used to calculate 95% confidence intervals. Continuous secondary outcome data including SpO2, PEEP and duration of surgery were reported as median (IQR) values.
Ethics approval was given by the New Zealand Health and Disability Ethics Committee (NZ/1/5252114), and local approvals were completed at all participating sites. Prospective trial registration was obtained prior to enrolment of the first participant (ACTRN12620001013954). Analyses are reported in accordance with the Strengthening the Reporting of Observational studies in Epidemiology checklist. 15
Results
One hundred and fifty anaesthetists were recruited into the study across the ten study sites, with six sites from Australia and four from New Zealand. The total number of anaesthetists at each site ranged from 13 at the smallest to 19 participants at the largest site. Data collection was completed over a 12-month period from 11 December 2020 to 6 December 2021. Patient and surgical baseline variables for patients under the care of participating anaesthetists are presented in Table 1. The median (IQR) actual operative duration was 170 (130–231 min) min.
Patient and surgical baseline variables.
Oxygen administration
The overall median (IQR) of intraoperative time-weighted mean FiO2 for all participants in the study was 0.47 (0.40–0.55). The median highest FiO2 across all cases was 0.82 (0.48–0.98) and median lowest FiO2 was 0.40 (0.34–0.45). Intraoperative FiO2 recordings for all study participants at all intraoperative timepoints are shown in Figure 1. Mean intraoperative FiO2 recordings at all timepoints for study participants ordered by individual study sites are shown in Figure 2. The range of median time-weighted mean FiO2 for participants at individual sites (Figure 3) was 0.39 at the site with the most restrictive use of oxygen to 0.60 at the site with the most liberal use of oxygen. Out of a total of 150 anaesthesia events, three anaesthetists (2%, 95% confidence intervals 0.4% to 5.7%) administered a time-weighted average intraoperative FiO2 of at least 0.8.
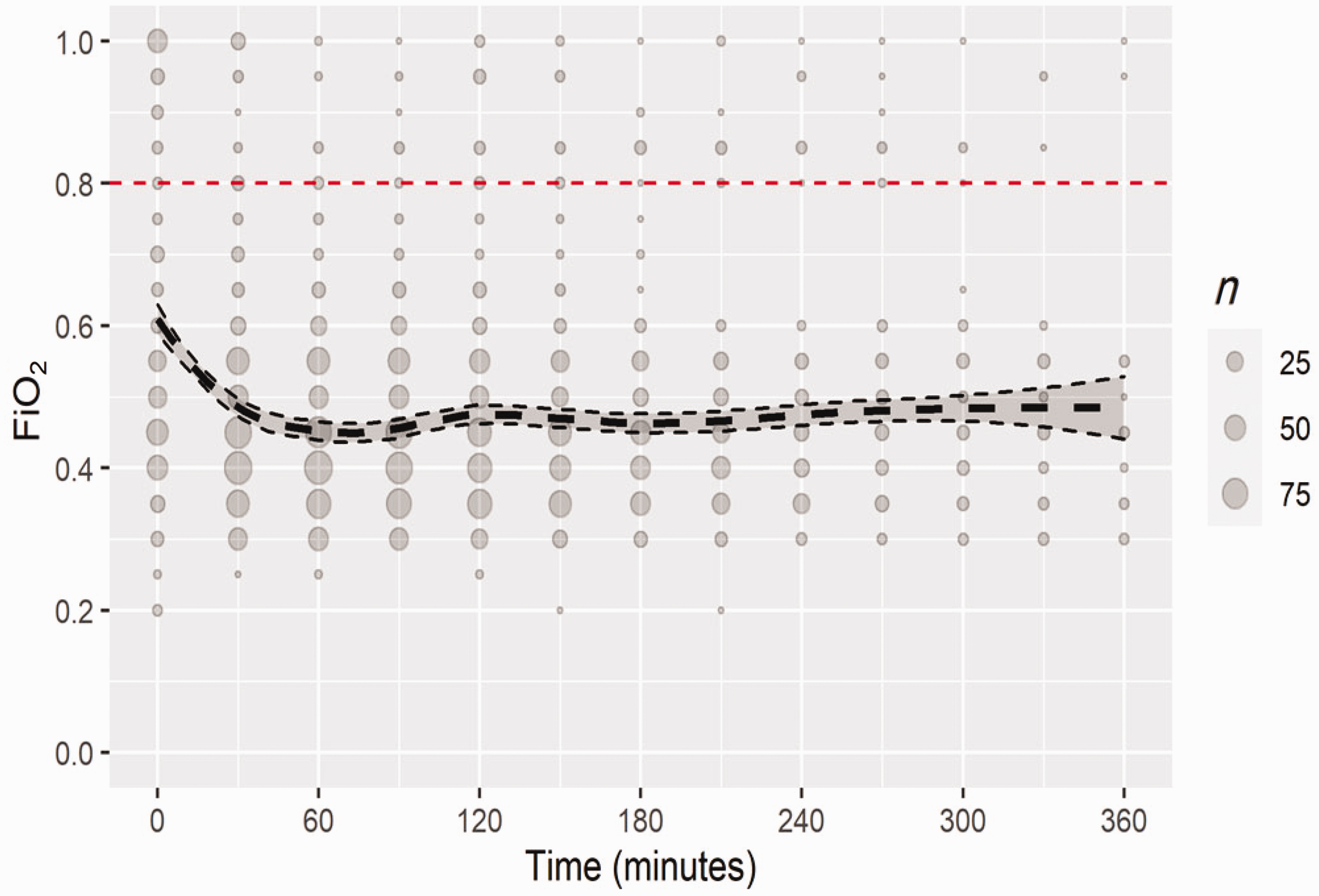
Overall intraoperative FiO2 for all timepoints using locally estimated scatterplot smoothing, with a window of 180 min. The smaller dashed lines are the 95% confidence bounds of the estimate of the regression model.
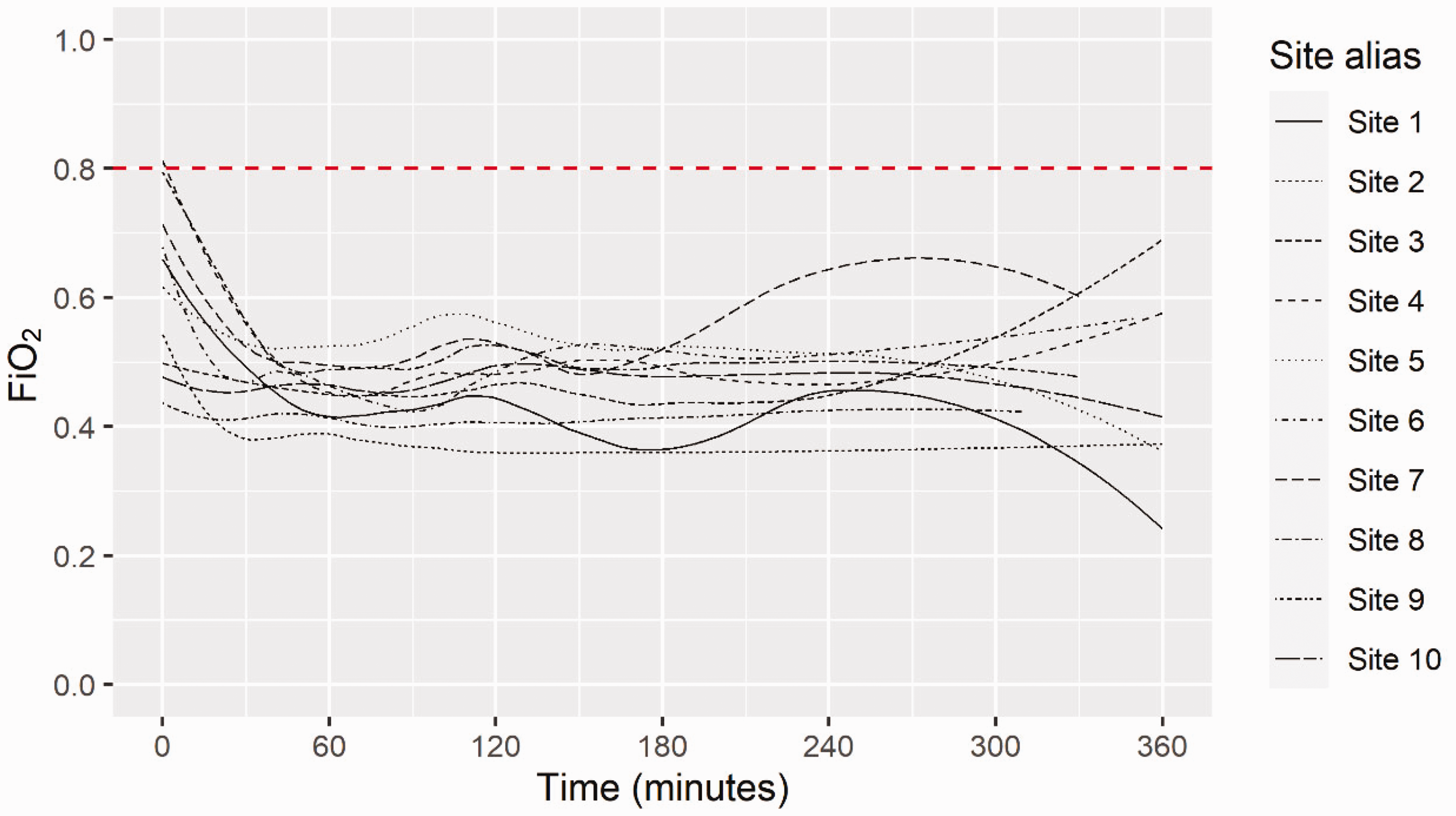
Mean FiO2 for all timepoints, displayed by site.
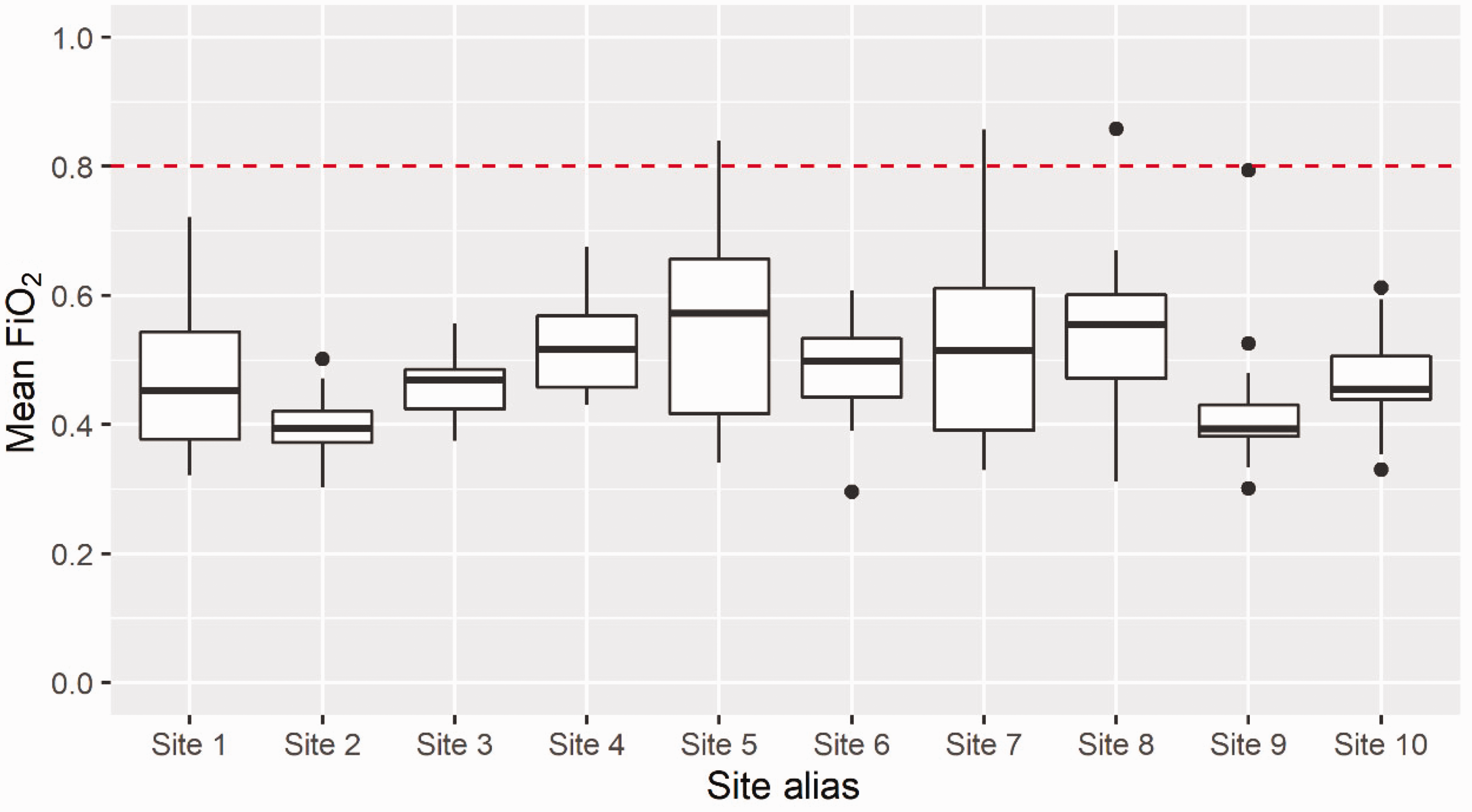
Distribution of overall time-weighted average FiO2 for patients, by site.
Oxygenation and positive end-expiratory pressure
The overall study median (IQR) of individual case time-weighted average SpO2 for all patients was 98.3% (97.5%–99.2%). The median (IQR) highest SpO2 in all cases was 100% (99%–100%) and lowest 97% (IQR 95%–98%, range 88%–99%). The median (IQR) time-weighted average PEEP was 5.1 mmHg (4.96–7.23 mmHg).
Discussion
In this prospective observational study including 150 anaesthetists providing care to ASA 3 or 4 patients undergoing prolonged surgery under general anaesthesia across ten large metropolitan hospitals in Australia and New Zealand, we observed that standard care intraoperative oxygen administration is an intermediate level of oxygen rather than titrated inspired oxygen to SpO2, or liberal oxygen of FiO2 >0.8 during surgery. A minority of anaesthetists administered a time-weighted average FiO2 of at least 0.8, suggesting a general lack of compliance with the WHO recommendation pertaining to perioperative oxygen therapy for the prevention of surgical site infection, and indicating that the current WHO recommendation is a departure from standard care.
Several limitations were identified in the present study. First, the number of participants was relatively small, reflecting approximately 2.3% of the total number of anaesthetists in Australia and New Zealand. Additionally, although a broad geographical spread of hospitals was achieved in New Zealand, the method of hospital selection on the basis of ANZCA Clinical Trials Network affiliation in combination with not all regions from Australia being represented (no hospitals included from the states of South Australia and Tasmania, or the Australian Capital Territory or Northern Territory) represents a degree of hospital selection bias and may limit the generalisability of study findings. While it is not certain whether the findings reflect practice of anaesthetists in smaller hospitals or regional/rural hospitals, given the consistency of findings between the six large Australian hospital sites in Victoria, New South Wales, Western Australia and Queensland, as well as all New Zealand sites, we submit that the findings are likely to be reflective of practice at other similar sized hospitals across Australia and New Zealand. Only ASA 3 or 4 patients were included in this study and therefore the findings may not be generalisable to ASA 1 or 2 patients. ASA 3 or 4 status was chosen as the indicator of more severe patient comorbidity; these patients may be more likely to receive ‘best practice’ care from anaesthetists due to their higher rate of postoperative complications, and furthermore are likely to be the population of interest for future trials designed to investigate the impact of perioperative oxygen administration on postoperative outcomes, due to the higher occurrence of postoperative complications in this group. We allowed for cases to be excluded if the site coordinating investigator felt that there was a specific oxygen requirement for that case. However, we did not collect information on the number of patients who were excluded or why they were excluded. Accordingly, we cannot be certain that such exclusions affect the generalisability of our findings. That said, we consider that this is unlikely as clear specific indications for particular oxygen regimens in patients undergoing anaesthesia are rare.
Because informed consent was obtained from all participating anaesthetists, it is possible that observer bias may have altered participants’ practice with regard to oxygen administration during the study period. However, anaesthetists were not explicitly told whether they were selected for study participation, or which of their cases would be chosen for study inclusion over the 12-month recruitment period, and data were obtained from anaesthesia records by independent research staff. Informed consent was obtained from all participating anaesthetists; however, not all anaesthetists at every included site gave consent to participate. This may have resulted in some selection bias, with anaesthetists who routinely use a particular regimen of oxygen therapy potentially not giving consent to participate. However, the study investigators did not assert that any particular strategy of oxygen therapy would be preferable so anaesthetists should not have felt compelled to provide any specific oxygen regimen.
The study recruitment period spanned approximately 12 months. This was longer than anticipated; however, several sites experienced delayed or interrupted recruitment due to COVID-19 related disruptions. While it is possible that oxygen administration patterns may have changed throughout the COVID pandemic, to our knowledge there were no changes to major perioperative oxygen administration recommendations during this period of time, nor any related publications that would have been expected to alter clinician behaviour. Similarly, no study sites were affected by actual or threatened oxygen supply shortages. In any case, the practice observed reflects current day practice against a backdrop of the COVID-19 pandemic. Finally, it is known that higher levels of oxygen are frequently used during both the induction and emergence phases of anaesthesia, whereby creating an oxygen store with pre-oxygenation provides a prolonged apnoeic desaturation time and a margin of safety in the event of airway difficulty at intubation, extubation or during patient transfer. 16 This practice is likely to be responsible for the higher FiO2 observed in the early post-induction phase in our study, as well as the higher overall average FiO2 than that typically selected by anaesthetists during the maintenance phase of anaesthesia, due to the gradual washout of the higher FiO2 used at induction. However, such washout reflects the oxygen exposure that occurs in usual practice.
The practice we observed in Australia and New Zealand appears similar to that in other parts of the world. A cross-sectional observational study of 1498 patients undergoing surgery at 43 hospitals over a five-day period in Japan found that the median intraoperative FiO2 administered was 0.47 (IQR 0.4–0.6), with a mean intraoperative FiO2 of between 0.31 and 0.6 observed in 92% of patients. 17 The authors did not report the proportion exposed to mean FiO2 ≥0.8; however, fewer than 8% were exposed to a time-weighted average intraoperative FiO2 of greater than 0.6, suggesting a low rate of adherence to the WHO recommendation. The intraoperative oxygenation in adult patients undergoing surgery (iOPS) multicentre observational study in the United Kingdom reported that, among patients undergoing major surgery with placement of an intra-arterial catheter for intraoperative blood pressure and/or arterial blood gas monitoring, only 4% of measured intraoperative FiO2 data points were ≥0.80. 18 The overall mean intraoperative FiO2 was 0.49; however, the proportion of patients that were administered FiO2 ≥0.8 for the duration of surgery and up to two to six hours postoperatively in accordance with the current WHO recommendation was not reported. An American registry study of 73,922 patient records aiming to investigate a potential association between intraoperative FiO2 and the risk of major respiratory complications reported a median FiO2 of 0.52 (0.49–0.54). 19
A recent single-centre retrospective observational study from Australia sought to determine the frequency of arterial hyperoxaemia in 100 patients undergoing surgery where placement of an arterial cannula was determined to be indicated by treating anaesthetists. 20 The authors recorded patient partial pressure of oxygen in the arterial blood (PaO2) from arterial blood gas analysis at three intervals during surgery, as well as the highest and lowest median FiO2 at a single timepoint. This study found that 70% of PaO2 values in the study population showed arterial hyperoxaemia, defined as a PaO2 > 150 mmHg. The median (IQR) lowest FiO2 in the study population was 0.45 (0.41–0.50) and the median (IQR) highest FiO2 was 0.70 (0.50–0.96), which is comparable to the findings of our present study. Unfortunately the overall median FiO2 during surgery was not reported. Although the authors concluded that anaesthetists adopted a relatively liberal approach to oxygen therapy, it was not possible to determine the proportion of anaesthetists that adhered to WHO recommendations for perioperative oxygen therapy. It is possible that the higher ‘median lowest FiO2’ observed in this study may have reflected the selection of a higher risk patient population, as indicated by the need for invasive arterial monitoring. Although very few anaesthetists systematically used a FiO2 of 0.8 or more in our present study, the observed median highest SpO2 of 100% at the very least suggests that the practice of Australian and New Zealand anaesthetists results in potential exposure to hyperoxaemia.
The reasons for the fraction of inspired oxygen administered by anaesthetists cannot be determined from the present study. However, a previous survey of Australian and New Zealand anaesthetists where fewer than 5% of the survey respondents indicated they aimed to administer perioperative oxygen therapy in accordance with the WHO recommendation of ≥0.8 found that 2.9% of respondents believed liberal oxygen therapy could help reduce postoperative complications, and only 0.7% believed liberal perioperative oxygen reduced the risk of postoperative infection specifically. 14 It is not known whether the low observed rate of adherence to the WHO recommendation is due to a lack of awareness of the recommendation or the evidence upon which it is based, or due to clinician disagreement with the recommendation for liberal oxygen therapy due to a different interpretation of the available evidence, or to concerns about potential adverse effects resulting from liberal oxygen administration.
Conclusion
The practices observed in this study indicate that most anaesthetists routinely administer an intermediate level of oxygen rather than either a liberal or a restrictive-titrated regimen of inspired oxygen for ASA 3 or 4 adult patients undergoing prolonged surgery in Australia and New Zealand. The findings suggest a general lack of support for the WHO recommendation of administering FiO2 >0.8 during and for two to six hours after surgery. Future research investigating the impact of oxygen on postoperative outcomes should include an intermediate or standard care arm in addition to restrictive or liberal perioperative oxygen therapy interventions.
Footnotes
Author Contribution(s)
Acknowledgements
PROSOX study participating investigators: Ruby Han, Wendy Purcell, David Highton, Susanna van Haeringen, Allison Kearney, Stefan Dieleman, Tomas Corcoran, Yvonne Buller, Pauline Coutts, Davina McAllister, Kelly Byrne, Michael Nottingham, Jeremy Young, Ross Kennedy, Margie Mckellow.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a project grant from the Campbell Barrett Anaesthesia Research Trust.
