Abstract
A bag-valve-mask (BVM) is a portable handheld medical device commonly used in airway management and manual ventilation. Outside of the operating theatre, BVM devices are often used to pre-oxygenate spontaneously breathing patients before intubation to reduce the risk of hypoxaemia. Pre-oxygenation is considered adequate when the end-tidal expiratory fraction of oxygen is greater than 0.85. There are reports that some BVM devices fail to deliver a satisfactory inspired oxygen (FiO2) in spontaneously breathing patients due to variability in design. The primary aim of this study was to evaluate the efficacy of oxygen delivery of a broad range of adult and paediatric BVM devices at increasing tidal volumes using a mechanical lung to simulate spontaneous ventilation. The secondary aim was to evaluate the effect of BVM design on performance.
Forty BVM devices were evaluated in a laboratory setting as part of a safety assessment requested by HealthShare New South Wales. The oxygen inlet of each BVM device was primed with 100% oxygen (15 l/min) for two min. The BVM device was then attached to the mechanical lung and commenced spontaneous breathing at a fixed respiratory rate of 12 breaths/min with an inspiratory: expiratory ratio of 1:2. For each device FiO2 was measured after two min of spontaneous breathing. This process was repeated with small (250 ml), medium (500 ml) and large (750 ml) tidal volumes simulating adult breathing in adult BVM devices, and small (150 ml), medium (300 ml) and large (450 ml) tidal volumes simulating paediatric breathing in paediatric BVM devices. The test was repeated using up to five BVM devices of the same model (where supplied) at each tidal volume as a manufacturing quality control measure.
Eight of the 40 devices tested failed to deliver a FiO2 above 0.85 for at least one tidal volume, and five models failed to achieve this at any measured tidal volume. Concerningly, three of these devices delivered a FiO2 below 0.55. Six of the eight poorly performing devices delivered reducing concentrations of inspired oxygen with increasing tidal volumes. Devices which performed the worst were those with a duckbill non-rebreather valve and without a dedicated expiratory valve.
Several BVM devices available for clinical use in Australia did not deliver sufficient oxygen for reliable pre-oxygenation in a spontaneously breathing in vitro model. Devices with a duckbill non-rebreather valve and without a dedicated expiratory valve performed the worst. It is imperative that clinicians using BVM devices to deliver oxygen to spontaneously breathing patients are aware of the characteristics and limitations of the BVM devices, and that the standards for manufacture are updated to require safe performance in all clinical circumstances.
Keywords
Introduction
A bag-valve-mask (BVM) is a portable handheld medical device commonly used in airway management and manual ventilation. BVMs are sometimes used to provide positive pressure ventilation, and to pre-oxygenate spontaneously breathing patients before intubation to extend the safe apnoea time. There are reports1– 5 that some of the current BVM devices do not deliver a satisfactory fraction of inspired oxygen (FiO2) in spontaneously breathing patients, despite complying with the current International Organization for Standardization (ISO) standards of design.
Pre-oxygenation is necessary for the optimal preparation of patients for intubation to reduce the risk of hypoxaemia. In spontaneously breathing patients pre-oxygenation is performed by delivering a high FiO2 to a patient to facilitate nitrogen washout from the functional residual capacity of the lungs. This provides a reservoir of oxygen for the body to extend the safe apnoeic period following the onset of neuromuscular blockade. Pre-oxygenation is considered adequate when the end-tidal fraction of oxygen (ETO2) is greater than 0.85.6– 8 In the operating theatre, a closed circuit connected to a patient with a well-sealed facemask can deliver a FiO2 close to 1. Outside the operating theatre, self-inflating BVM devices are commonly used for pre-oxygenation, and are superior to alternative methods such as the non-rebreather mask. 9 In addition, BVM devices enable manual positive pressure ventilation whereas alternatives like high-flow nasal oxygen and non-rebreather masks do not.
Pre-oxygenation via a BVM device is performed by administering supplemental oxygen (100%) to a patient with a well-sealed facemask, with the aim of delivering a FiO2 close to 1.10,11 There is limited evidence to suggest that measurement of FiO2 and ETO2 via a gas analyser may predict the partial pressure of oxygen in plasma, and thus may be an indicator of pre-oxygenation efficacy. 12 In emergency situations where ETO2 monitoring is not available, pre-oxygenation with a FiO2 close to 1 for three min provides adequate pre-oxygenation. 13 However, a recent study highlighted that inadequate pre-oxygenation may still occur in up to 74% of cases where BVM devices are used for pre-oxygenation without ETO2 monitoring. 14 There are few data available from manufacturers about the performance of their devices for pre-oxygenation in spontaneous ventilation despite the routine use of BVM devices for this purpose. Furthermore, the components of BVM devices and differences between models are poorly understood by clinicians. 1
The primary aim of this in vitro study was to evaluate the efficacy of oxygen delivery of a broad range of commercially available adult and paediatric BVM devices at increasing tidal volumes using a mechanical spontaneously breathing lung. The secondary aim was to evaluate the effects of some design elements of these BVM devices on their performance.
Materials and methods
Forty BVM devices available from HealthShare New South Wales (NSW) were evaluated in a laboratory setting using a simulation lung following a request for an assessment by HealthShare NSW due to safety concerns raised. Each device was tested for the FiO2 delivered after two min of spontaneous breathing at various tidal volumes. An ASL 5000 mechanical lung simulator (IngMar Medical, Pittsburgh, PA, USA) was used to generate small (250 ml), medium (500 ml) and large (750 ml) spontaneous tidal volumes simulating adult breathing for adult BVM devices, and small (150 ml), medium (300 ml) and large (450 ml) tidal volumes simulating paediatric breathing for paediatric BVM devices. The software employed in this study (LLEAP®, Laerdel, Stavanger, Norway) requires a programmed inspiratory pressure to generate a tidal volume. Therefore, this required titration of the inspiratory pressure against the measured tidal volume until the experimental tidal volume was achieved. To calibrate the lung simulator, this titration was performed prior to attaching each BVM device to the lung simulator.
Each BVM device was connected to the lung simulator via its patient port and the 22 mm tapered port on the circuit tubing and, therefore, a facemask seal was not required for this study. The oxygen inlet of each BVM device was connected to 15 l/min of 100% oxygen. Each device was primed for two min to fill the oxygen reservoir bag prior to commencement of ventilation. The lung simulator then commenced spontaneous breathing for two min at a fixed respiratory rate of 12 breaths/min with an inspiratory: expiratory ratio of 1:2. An in-line oxygen sampling device (Datex Ohmeda, Instrumentarium Corp., Helsinki, Finland) attached between the patient port of each BVM device and the circuit tubing was used to measure inspiratory FiO2 after two min. This protocol was repeated on up to five BVM devices of the same type (where supplied) at each tidal volume as a manufacturing quality control measure. The oxygen analyser was calibrated using 100% oxygen.
Results
The characteristics of the BVMs evaluated are presented in Table 1. The results of testing for the BVMs that achieved an adequate FiO2 with spontaneous ventilation are presented in Table 2.
Characteristics of tested bag-valve-mask devices.
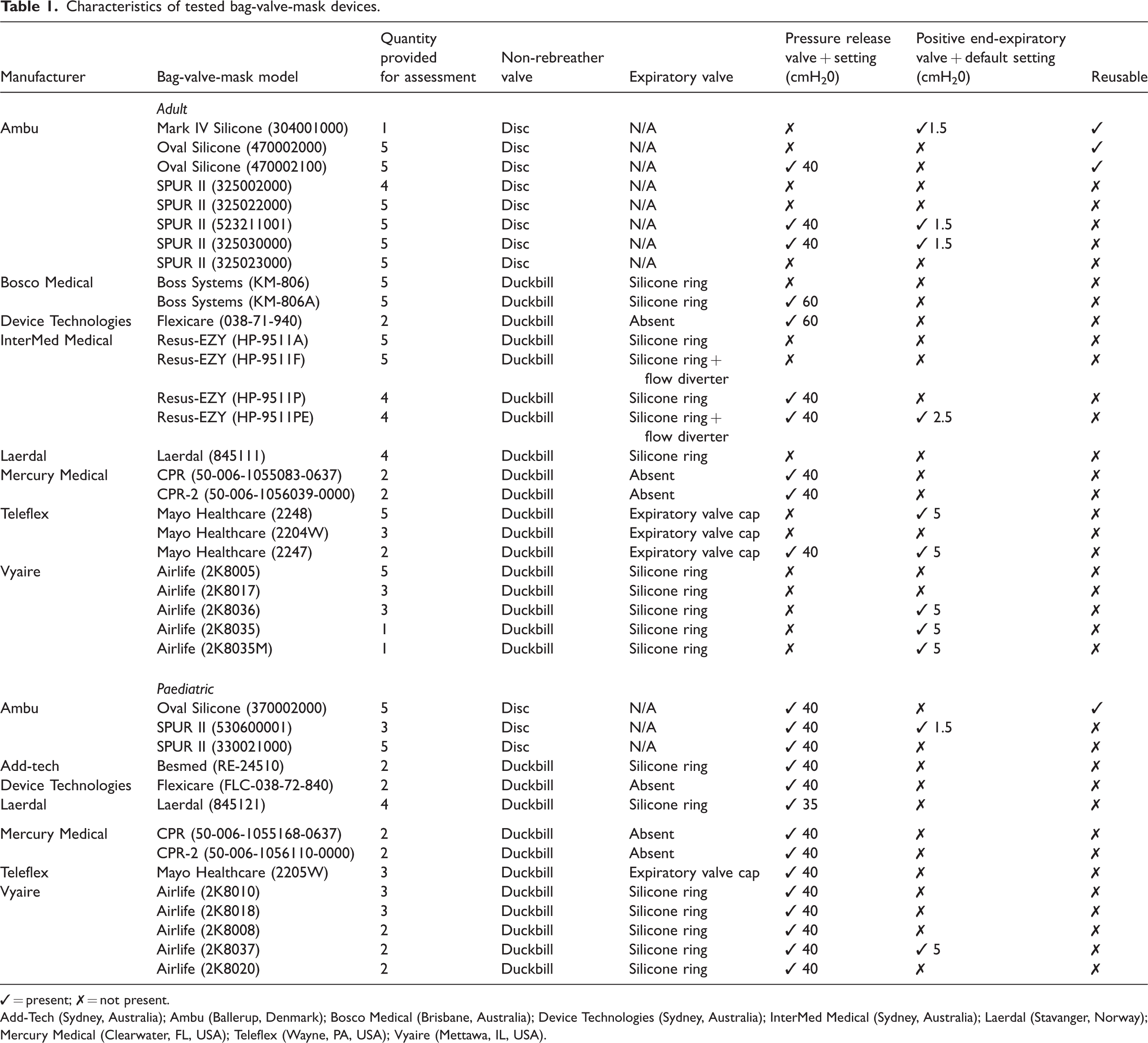
✓ = present; ✗ = not present.
Add-Tech (Sydney, Australia); Ambu (Ballerup, Denmark); Bosco Medical (Brisbane, Australia); Device Technologies (Sydney, Australia); InterMed Medical (Sydney, Australia); Laerdal (Stavanger, Norway); Mercury Medical (Clearwater, FL, USA); Teleflex (Wayne, PA, USA); Vyaire (Mettawa, IL, USA).
Adequately performing bag-valve-mask devices (fraction of inspired oxygen >0.85): average fraction of inspired oxygen following 2 min of simulated spontaneous ventilation.
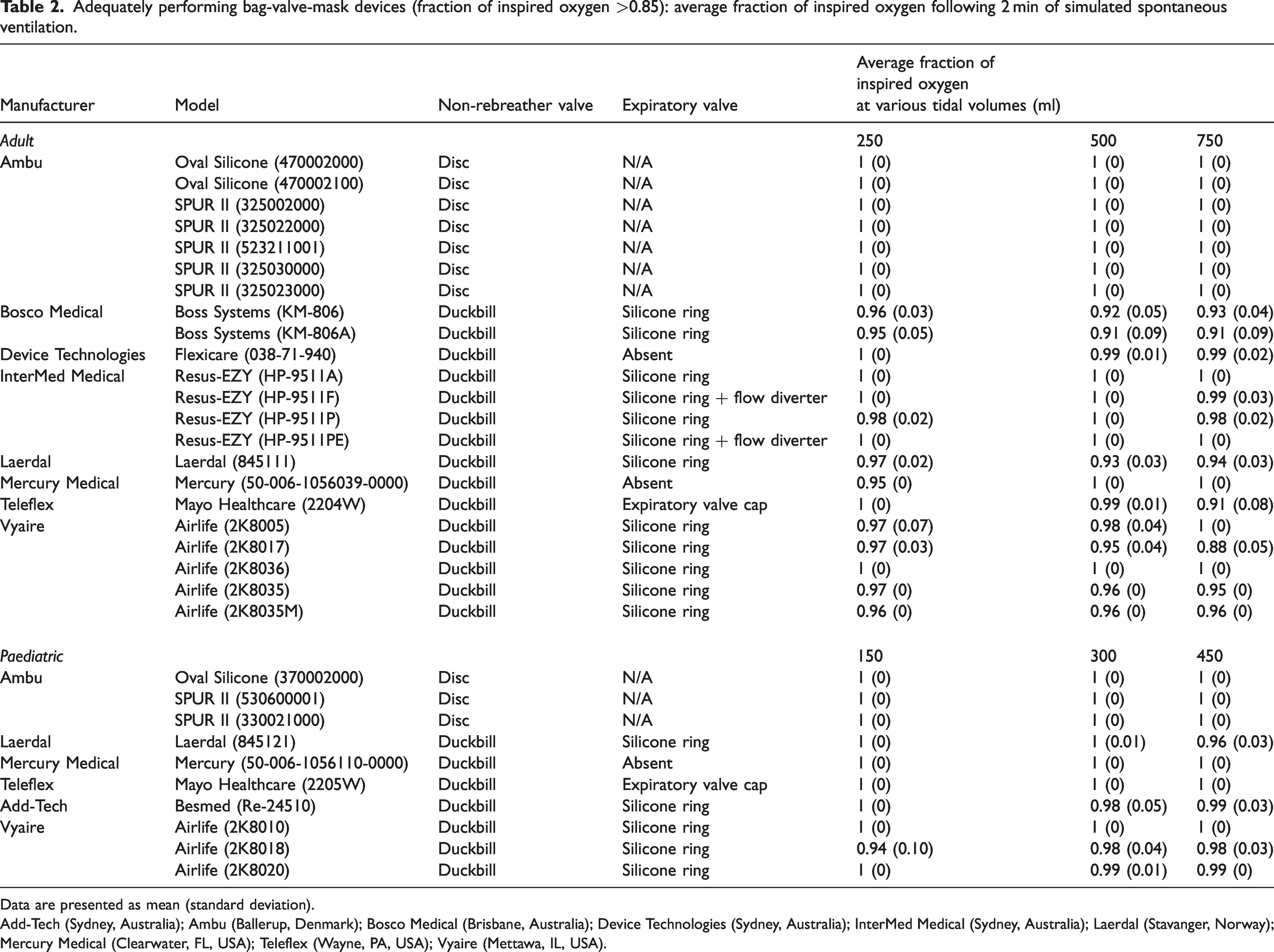
Data are presented as mean (standard deviation).
Add-Tech (Sydney, Australia); Ambu (Ballerup, Denmark); Bosco Medical (Brisbane, Australia); Device Technologies (Sydney, Australia); InterMed Medical (Sydney, Australia); Laerdal (Stavanger, Norway); Mercury Medical (Clearwater, FL, USA); Teleflex (Wayne, PA, USA); Vyaire (Mettawa, IL, USA).
Eight of the 40 BVM devices (three adult BVM devices and five paediatric BVM devices) evaluated failed to deliver a FiO2 above 0.85 for at least one tidal volume, and five models failed to achieve this at all tidal volumes tested (Table 3). Of the poorly performing BVMs, one device utilised a disc-type non-rebreather valve and seven utilised a duckbill non-rebreather valve. Six of these eight poorly performing devices delivered reducing concentrations of inspired oxygen with increasing tidal volumes. Variability in performance between different BVMs of the same model was typically small.
Underperforming bag-valve-mask devices (fraction of inspired oxygen <0.85): average fraction of inspired oxygen following 2 min of simulated spontaneous ventilation.
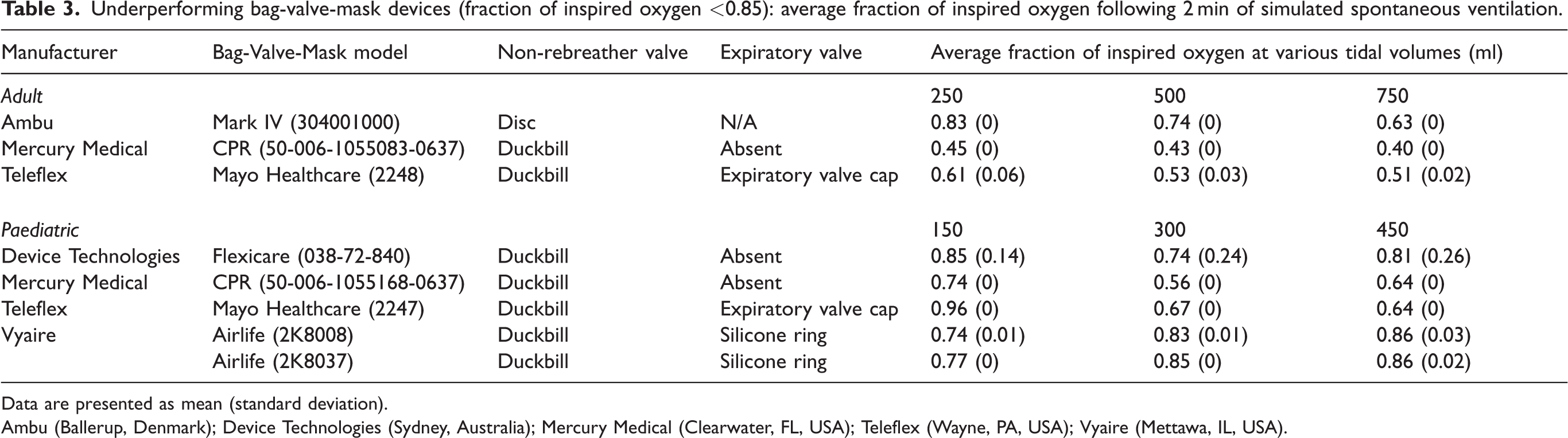
Data are presented as mean (standard deviation).
Ambu (Ballerup, Denmark); Device Technologies (Sydney, Australia); Mercury Medical (Clearwater, FL, USA); Teleflex (Wayne, PA, USA); Vyaire (Mettawa, IL, USA).
Ten of the 11 (91%) BVM devices with a disc-type non-rebreather valve (Figures 1 and 2) consistently delivered a FiO2 of 1. In contrast, only 22 of the 29 (75.9%) devices manufactured with a duckbill valve delivered a FiO2 above 0.85, and with greater variability in the concentration delivered across tidal volumes (Tables 2 and 3). Of the 29 BVM devices with a duckbill non-rebreather valve, six did not have a dedicated expiratory valve (Table 1) and three failed to deliver a FiO2 above 0.85 (Table 3).
Discussion
Our study showed that several currently available BVM devices failed to deliver sufficient oxygen for safe pre-oxygenation with spontaneous ventillation. Eight of the 40 BVM devices tested failed to deliver a FiO2 above 0.85 for at least one level of tidal volume, and five models failed to achieve this at any measured tidal volume. Concerningly, three of these devices delivered a FiO2 below 0.55. Devices which performed the worst were those with a duckbill non-rebreather valve and without a dedicated expiratory valve to prevent air entrainment. These findings are consistent with and expand on the findings of Grauman et al. 1 and Mills et al. 3
The effectiveness of pre-oxygenation can be influenced by patient, operator, situational and technical factors. 1 The commonest factor affecting pre-oxygenation is an incomplete seal between the facemask and the patient which allows entrainment of ambient air. This occurs more commonly in the hands of inexperienced operators and in patients who are edentulous, have facial hair or have a nasogastric tube in situ. 15 The effect of modifiable technical factors (including positive end-expiratory pressure (PEEP), FiO2 and oxygen flow rate) on pre-oxygenation are well understood.6,16,17 Information about the oxygen delivery performance between devices is often not readily available, nor mandated under regulatory requirements. Earlier studies have reported variability in the delivery of oxygen among BVM devices as a result of design flaws; however, these studies evaluated a small number of devices.1– 5 It is suggested that devices without a dedicated expiratory valve may entrain air into the device through the expiratory port, diluting the concentration of delivered oxygen.1,2,4,9,18,19 However, even the inclusion of an expiratory valve may not prevent air entrainment if it does not function properly. 3 Larger tidal volumes appeared to cause greater air entrainment in our study, further decreasing the concentration of delivered oxygen. The effect of tidal volume on oxygen delivery among BVM devices in spontaneously breathing patients has not been reported in current models of BVM devices. Because BVM devices are commonly used for resuscitation and pre-oxygenation outside the operating theatre, it is essential that they can reliably deliver a FiO2 above 0.85 in spontaneously breathing patients with variable ventilatory patterns.
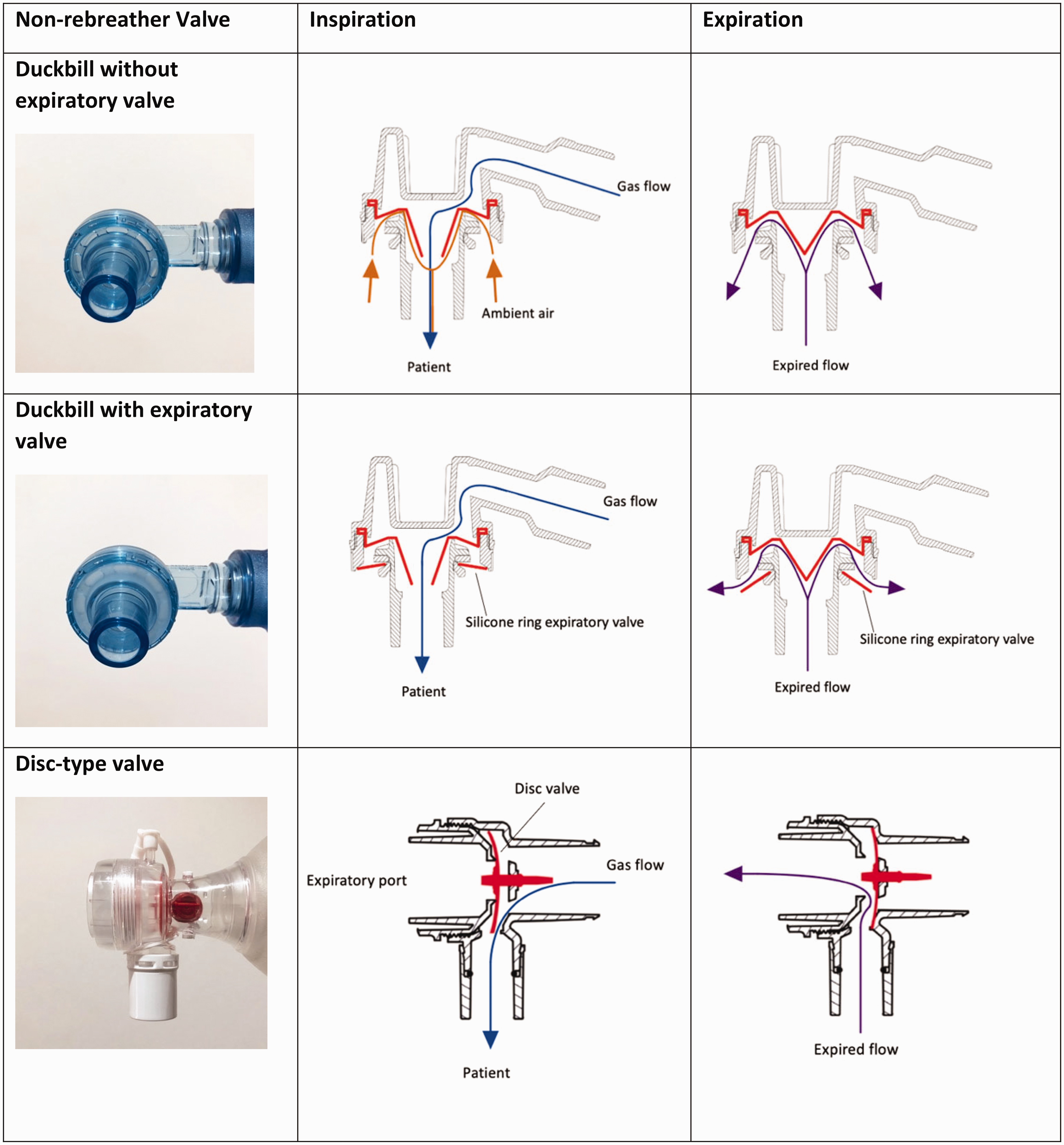
To understand the performance and limitations of BVM devices, it is necessary to be familiar with their components, especially the valves. The non-rebreather valve, most commonly a disc-type or duckbill valve, is a unidirectional valve that prevents rebreathing. The disc-type valve is made of a single silicone disc. During inspiration, the disc retracts against the expiratory port to allow the passage of gas from the device through to the patient whilst simultaneously sealing the expiratory port to prevent the entrainment of ambient air. During expiration, the disc returns to its original position, which allows expired gas to escape through the expiratory port, preventing re-entry of expired air into the device (Figures 1–3). This valve functions in the same manner during both spontaneous and manual ventilation.
The duckbill valve comprises a thin and flexible diaphragm with two protruding leaflets that form the valve. The duckbill valve is typically situated horizontally in the device and functions differently in spontaneous compared with manual ventilation. During manual inspiration, positive pressure generated by compression of the self-inflating bag causes the leaflets of the valve to open, allowing gas to flow from the device to the patient. During expiration, the duckbill leaflets return to their original position and the valve relaxes off the expiratory port, allowing expired gas to exit. During spontaneous inspiration, the leaflets of the duckbill valve are opened by negative pressure generated by the patient. The forward flow of fresh gas assists with the opening of the valve (Figures 1–3).1,3 The degree of air entrainment from the expiratory port is dependent on the opening pressure of the valve and the relative resistances to airflow through the valve and the expiratory port. 3
A number of methods can be employed to prevent air entrainment via the expiratory port. The most common is a silicone ring expiratory valve typically located directly below the duckbill non-rebreather valve, or at the end of the outflow tract in some devices fitted with a flow diverter. Less commonly, a removable expiratory valve cap which attaches to the flow diverter is supplied with some BVM models. A PEEP valve will also function to prevent air entrainment.
We demonstrated that all but one BVM device with a disc-type non-rebreather valve consistently delivered a FiO2 of 1 in spontaneously breathing patients. Previous studies have demonstrated similar efficacy in oxygen delivery in BVM devices of this design.1,4
Greater variability in oxygen delivery was found in BVM devices with a duckbill non-rebreather valve. Our study demonstrated that BVM devices with a duckbill non-rebreather valve without a dedicated expiratory valve performed the worst. Devices with a dedicated expiratory valve were able to deliver a safe concentration of oxygen suitable for use in pre-oxygenation for 19 out of 23 models. The observed variability in oxygen delivery among several devices with a dedicated expiratory valve is likely the result of an incomplete seal at the expiration port. Greater reliability in oxygen delivery was seen in devices with a silicone ring expiratory valve compared with a flow diverter expiratory valve cap. Our data support previous studies that demonstrated that devices fitted with an expiratory valve delivered oxygen more reliably during spontaneous ventilation compared with BVM devices without an expiratory valve.1,3 Given the variability we observed between devices, and the difficulty in identifying the valves, we recommend that manufacturers provide this information in the product information.
All but one device fitted with the disc-type valve consistently delivered a FiO2 of 1 at all tidal volumes. We found that FiO2 significantly reduced with increasing tidal volumes in some BVM devices, including those with an expiratory valve cap at the end of a flow diverter, and especially those with a duckbill valve without an expiratory valve. We suggest that this is likely because a larger tidal volume resulted in a greater volume of ambient air to entrain into the device, thus diluting the FiO2. This may represent an additional risk for patients with an increase in minute ventilation in whom adequate pre-oxygenation is particularly important. 16 Additionally, because tidal volume increases with height, 20 and increased body mass is associated with greater metabolic requirements, 21 inadequate pre-oxygenation is likely to have the greatest effect on larger adults. Finally, during the pre-oxygenation process it is common for healthcare providers to instruct patients to take slow, deep breaths. In situations where a BVM device without a dedicated expiratory valve is used, this instruction, which many believe improves pre-oxygenation, could predispose to hypoxaemia.
Of the underperforming BVM devices evaluated in this study, only the product information provided by Teleflex (Wayne, PA, USA) specified the use of their device in spontaneously breathing patients. The product information for the remaining underperforming seven devices reported the use of their device for ‘resuscitation’, which could reasonably include the delivery of supplemental oxygen to a spontaneously breathing patient. No device specified against the use of their product for this purpose. It is necessary that manufacturers clearly outline the intended use and limitations of their device in the product information.
Additional considerations in bag-valve-mask device design
There are two other aspects of BVM design that improve their function and safety for patients: a PEEP valve and a pressure release valve.
In our review, we found that some BVMs came with a PEEP valve attached, while others required attachment when included. For those BVMs with a PEEP valve, the default PEEP valve setting varied among devices, even of the same model. Applying a PEEP valve to a BVM device allows for extrinsic PEEP to be applied during BVM ventilation. This can improve ventilation/perfusion matching by reducing atelectasis and is particularly useful in ventilating patients with a significant shunt. 22 The PEEP valve can also attenuate air entrainment in devices without a dedicated expiratory valve; 1 however, PEEP valve use is not considered safe in all clinical situations. A pre-attached PEEP valve set to ‘on’ has the potential to increase respiratory work in children and adults who are already in respiratory distress. 23 We suggest that manufacturers should provide PEEP valves that come unattached and set to zero for all BVM devices.
Pressure release valves are intended to reduce the risk of barotrauma with BVM devices. The pressure at which these valves open is predetermined by the manufacturer. Of those BVMs evaluated with a pressure release valve, we found them set at 40 cmH2O in paediatric devices and 40–60 cmH2O in adult devices. Sixteen of the devices we tested did not have a pressure release valve. Despite being considered a desirable component of the BVM device, their presence is not mandated in adult devices by the ISO guidelines.
Strengths and limitations
Our study expands on the work by Grauman et al. 1 and Mills et al. 3 and confirms the increased variability in oxygen delivery of BVM devices with a duckbill valve. As this is a simulation study, the performance of each BVM device in the clinical setting where facemask seal will also influence oxygen delivery was not evaluated. Moreover, the absence of a facemask reduces the dead space, which may also influence FiO2, particularly at small tidal volumes. However, this study evaluated a large variety of commercially available BVM devices currently used in clinical practice in Australia and eliminated the influence of operator and patient factors on BVM performance. Therefore, this enabled an objective and repeatable assessment of each variety of BVM based on design characteristics. Although we studied the effect of tidal volume on FiO2 at a fixed respiratory rate, we did not evaluate the effect of changes in respiratory rate with a fixed tidal volume on BVM performance, nor did we evaluate the effect of different inspiratory flow patterns. The role of resistance to breathing during spontaneous ventilation is an important characteristic of BVM functionality that was not assessed as part of this study and requires further investigation.
Conclusion
We demonstrated that several BVM devices available in Australia do not deliver a reliable FiO2 for pre-oxygenation in spontaneously breathing patients. Devices with a duckbill non-rebreather valve and without a dedicated expiratory valve performed the worst, while devices fitted with disc-type non-rebreather valves were the most reliable in delivering high concentrations of oxygen. Further, BVM devices without a pressure release valve and those with pre-attached PEEP valves set to ‘on’ may present a risk to patients. To improve the safety of BVM devices, we recommend that the ISO guidelines be updated. Moreover, we recommend that manufacturers be required to report the performance of the BVM device for pre-oxygenation in spontaneous ventilation in the product information or include a safety warning if their device should not be used for this purpose. BVM devices not providing an adequate FiO2 without a clear safety warning should be removed from clinical use. It is imperative that clinicians using BVM devices to deliver oxygen to spontaneously breathing patients are familiar with the characteristics and limitations of the BVM device used.
Footnotes
Author Contribution(s)
Acknowledgements
We would like to thank Laerdel (Stavanger, Norway) and Ambu (Ballerup, Denmark) for kindly providing permission to use and modify the schematic diagrams of the patient port used in this paper. Laerdal (Stavanger, Norway) also supplied the ASL 5000 Mechanical Lung which was used in this study. Thank you to Professor Peter Kam for his review of the manuscript. Equipment: BVM devices were provided by HealthShare New South Wales who requested samples directly from suppliers.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Not required as this was a benchtop study not involving patients or animals.
Schematic diagram of a typical bag-valve-mask device with a duckbill (a) and a disc-type non-rebreather valve (b). The effect of non-rebreather valve design on gas flow during spontaneous inspiration and expiration. Cross-sectional view of the duckbill versus disc-type non-rebreather valve.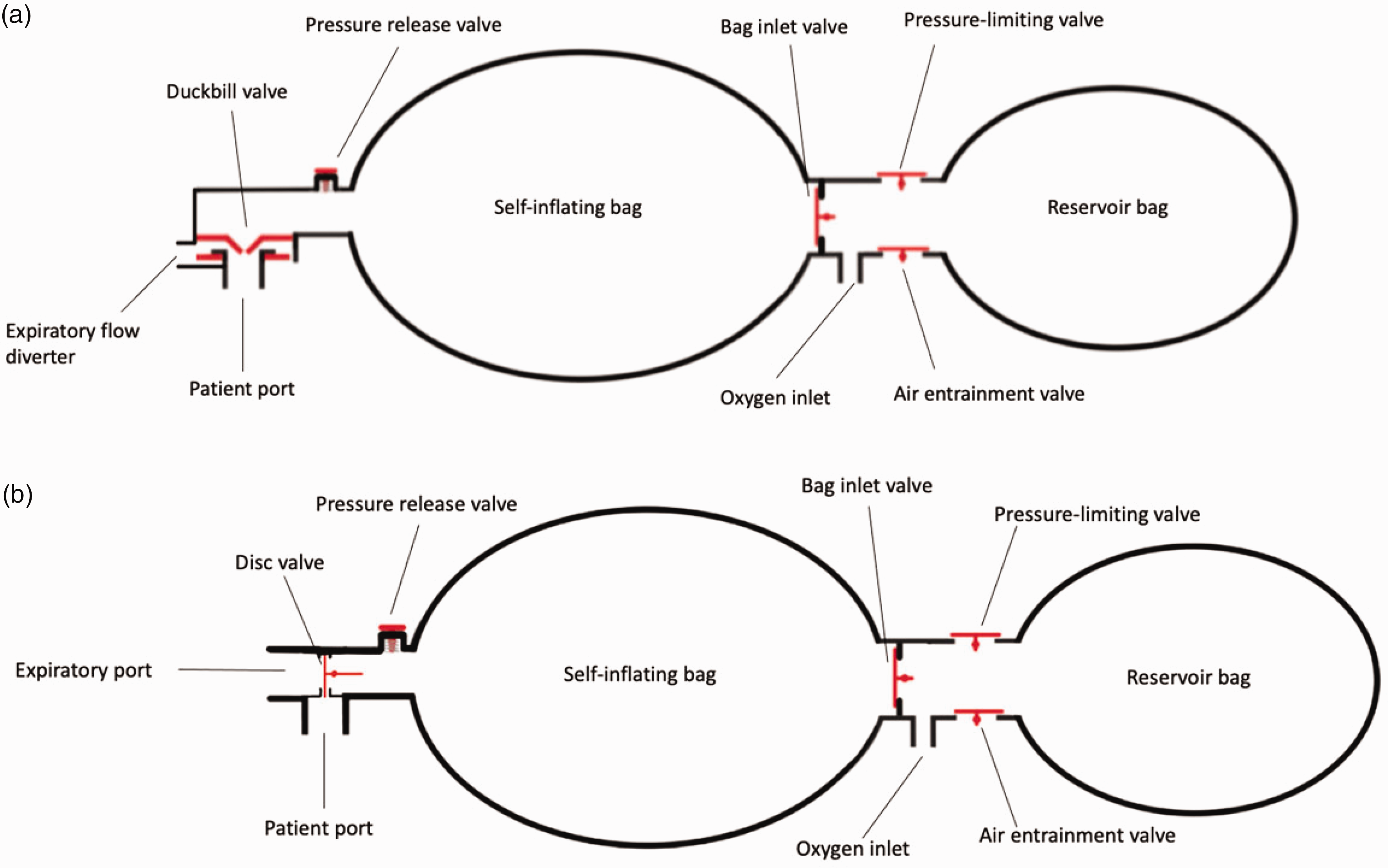

Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
