Abstract

We report an otherwise uncomplicated total pancreatectomy and autologous islet cell transplant (TPAIT) procedure in which the patient suffered from severe type B lactic acidosis. The authors utilised unproved therapies with good effect, and the purpose of this report is to raise awareness of not only type B lactic acidosis but also to ignite a conversation about potential therapies. Written consent for this case presentation was obtained from the patient.
A 48-year-old, 100 kg man with recurrent acute on chronic pancreatitis secondary to pancreatic divisum presented for a TPAIT procedure. He had a medical history significant for hypertension, anxiety, and chronic pain. His complete blood count, chemistry, renal and hepatic profiles were all within the normal range. After an uneventful anaesthetic induction, central venous and arterial lines were placed. A baseline arterial blood gas (ABG) was obtained which showed a slightly elevated lactate of 2.3 mmol/l (see Table 1). One hour into the procedure, an insulin infusion was initiated at 2 units/hour to maintain blood glucose (0.8 to 1.2 g/l). Laboratory values were checked throughout the 12-hour case and the haemoglobin was maintained between 100 and 125 g/l. Electrolytes remained stable. It was noted that the lactate level was steadily climbing despite a stable haemoglobin, aggressive volume administration, and a relatively stable pH (Table 1). The surgeons meticulously inspected the viscera and ruled out any obvious surgical causes of acidosis, such as bleeding or ischaemia. Doppler ultrasound confirmed adequate blood flow in the hepatic vessels. Eight hours from incision, blood-tinged urine was noted and was attributed to retractor compression on the kidney. Creatine kinase concentrations were evaluated, and normal values ruled out rhabdomyolysis but the lactate level continued to rise despite no obvious cause (Table 1). Hepatic enzymes were repeated intraoperatively with normal values. Throughout the case, the patient maintained adequate (>0.5 ml/kg/hour) urine output and remained haemodynamically stable. No vasoactive support was needed, and the patient was adequately fluid resuscitated. Other causes of ischaemic acidosis were ruled out. Ten hours into the case the lactate climbed to 8.9 mmol/l, at which point 500 mg of levocarnitine (L-carnitine) and 15 g of N-acetylcysteine (NAC) were administered. Within 15 minutes, ABG showed a decline in lactate levels (8.9 mmol/l to 5.8 mmol/l). The majority of islet cells (80%) were reinfused into the liver via a portal infusion and the remaining 20% into the peritoneal cavity due to elevated portal pressures. The lactic acidosis continued to improve over the next several hours. By the next day, lactate levels had normalised.
Perioperative arterial blood gases.
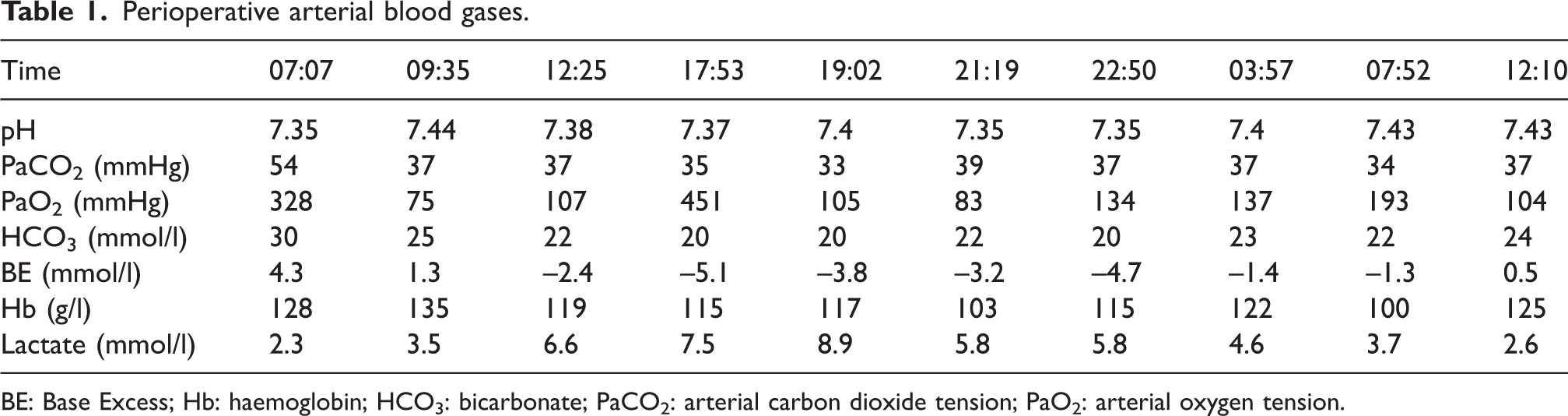
BE: Base Excess; Hb: haemoglobin; HCO3: bicarbonate; PaCO2: arterial carbon dioxide tension; PaO2: arterial oxygen tension.
There are two main groups used to categorise the aetiology of lactic acidosis (Table 2). 1 Causes of type A lactic acidosis include any mechanism leading to globally decreased oxygen delivery, such as reduced cardiac output, hypovolaemia, arterial hypoxaemia and anaemia. This is the type most often considered when interpreting ABG results as an endpoint in volume resuscitation. Type B lactic acidosis, however, occurs despite adequate oxygen delivery and may be an underappreciated contributor to the declining status of critically ill patients. Mechanisms that contribute to type B acidosis include decreased regional or microcirculatory blood flow, increased oxygen utilisation and decreased hepatic and/or renal lactate clearance. Unfortunately, clinical contexts may involve multiple overlapping mechanisms that lead to lactate accumulation, making the diagnosis and management challenging.
Aetiologies of lactic acidosis.
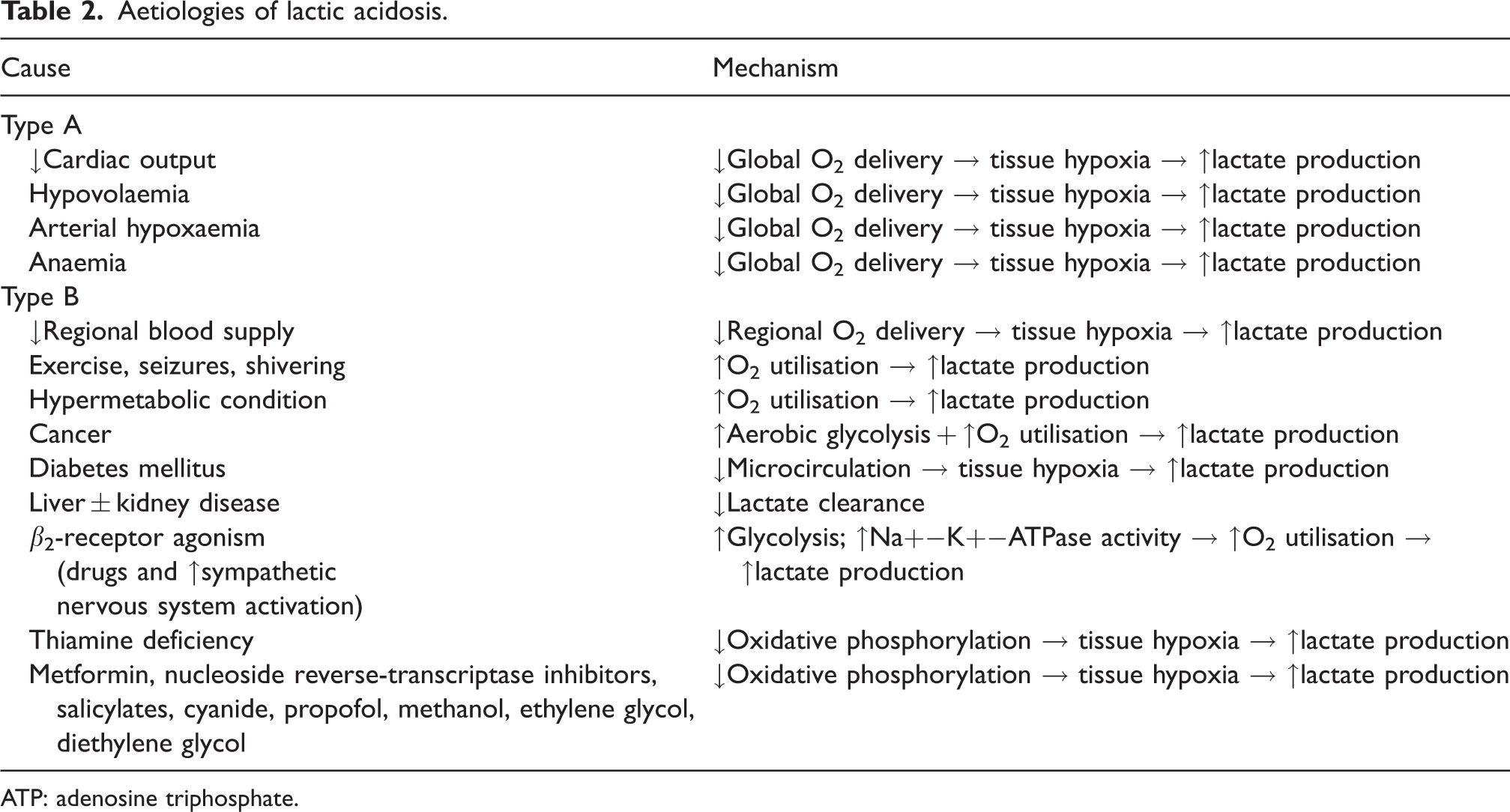
ATP: adenosine triphosphate.
In the case above, the patient developed severe lactic acidosis in the setting of adequate tissue perfusion and normal global hepatic function. Instant blood-mediated inflammatory response is a known phenomenon in cases of islet cell transplantation; 2 however, as the acidosis began prior to islet cell infusion, this was an unlikely aetiology. By process of elimination, type B lactic acidosis was established as the presumptive diagnosis. As the treatment for lactic acidosis, regardless of the type, is to treat the underlying cause, this case presented a unique challenge. The most likely explanations were speculated to be underlying mitochondrial dysfunction and impaired regional oxygen delivery, although this was not proved. Acknowledging that there is no support in the literature for this treatment, the decision was made to give NAC to increase hepatosplanchnic perfusion and L-carnitine to reverse the impaired production of mitochondrial adenosine triphosphate (ATP).
Serving as an endogenous antioxidant, NAC is a precursor of glutathione, and has been studied as a treatment modality in multiple disease processes. Since the 1960s, NAC has been available for commercial use and has most commonly been used to treat patients with paracetamol overdose. It is safe and has minimal side-effects. 3 It has been studied extensively as an antioxidant, but the role of NAC in the treatment of lactic acidosis is minimal. However, NAC has been reported to have positive effects on liver perfusion due to its ability to improve splanchnic blood flow. Hein et al. 4 assessed five septic patients, measuring lactate signal intensities in the liver both before and after NAC administration. The authors found that, following NAC infusion, median lactate signals in liver tissue decreased 89%, with an increase in liver perfusion by 41%. They presumed that NAC may have a role in patients with compromised hepatosplanchnic function in improving hepatic function and lactate processing. 4 Ammar and Abdou 5 looked at 50 patients following hepatectomies and the potential benefit of NAC. Patients were randomly assigned either to receive NAC intraoperatively and postoperatively or lactated Ringer’s throughout. The authors found that the group receiving NAC had a significant reduction in serum lactate at 0, 24 and 48 hours compared to the control. 5 Our patient did not undergo hepatic resection, but his pathology showed 20% macrosteatosis, which may have been a contributing factor. Our patient did not have obvious signs of hypoperfusion, and type B lactic acidosis was assumed. Without signs of metabolic overproduction, it was suspected that the patient had some degree of mitochondrial dysfunction and thus NAC was administered.
L-carnitine is a naturally occurring amino acid that is used to support energy metabolism and maintain normal mitochondrial function. It has multiple functions, including transporting long-chain fatty acids from the cytosol across the inner mitochondrial membrane into the mitochondrial matrix. It is a necessary cofactor in fatty acid oxidation and has been shown to possess anti-apoptotic activity. 6 It is used for patients with known carnitine deficiency, and more recently as a potential therapy for patients with lactic acidosis due to nucleoside analogue medications which induce cell death due to mitochondrial dysfunction. Multiple case reports show that L-carnitine has beneficial effects on patients with HIV with type B lactic acidosis from nucleosidic reverse transcriptase inhibitors.7,8
The role of L-carnitine supplementation in patients with hepatic injury or hepatic dysfunction remains unclear. A recent meta-analysis of 18 randomised controlled trials reported the effect of L-carnitine on hepatic enzymes. They found that supplementation reduced hepatic enzyme levels, an effect which was more pronounced with higher doses over longer periods of time. 9 Patients with baseline hepatic disease were likely to have the greatest benefit. We administered L-carnitine to our patient with the assumption that mitochondrial dysfunction led to profound type B lactic acidosis. Like NAC, L-carnitine is well tolerated and was administered without complication. 9
Our patient developed intraoperative type B lactic acidosis during TPAIT. Therapy with NAC and L-carnitine had a temporal relationship with decreasing lactate levels in our patient and represents a unique potential therapeutic option. The authors acknowledge that it is possible the acidosis may have improved on its own with time, but the literature surrounding the treatment of type B lactic acidosis in the surgical population remains sparse and is worthy of future research. Type B lactic acidosis is a diagnosis of exclusion, and it is the authors’ recommendation that in scenarios in which hepatosplanchnic flow or mitochondrial dysfunction are suspected, NAC and L-carnitine should be considered as potential therapies.
Footnotes
Author Contribution(s)
Declaration of conflicting interests
The author(s) have no conflicts of interest to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
