Abstract
Vasopressor dependence is a common problem affecting patients in the recovery phase of critical illness, often necessitating intensive care unit (ICU) admission and other interventions which carry associated risks. Midodrine is an orally administered vasopressor which is commonly used off-label to expedite weaning from vasopressor infusions and facilitate discharge from ICU. We performed a single-centre, case-control study to assess whether midodrine accelerated liberation from vasopressor infusions in patients who were vasopressor dependent. Cases were identified at the discretion of treating intensivists and received 20 mg oral midodrine every eight h from enrolment. Controls received placebo. Data on duration and dose of vasopressor infusion, haemodynamics and adverse events were collected. Between 2012 and 2019, 42 controls and 19 cases were recruited. Cases had received vasopressor infusions for a median of 94 h versus 29.3 h for controls, indicating prolonged vasopressor dependence amongst cases. Midodrine use in cases was not associated with faster weaning of intravenous (IV) vasopressors (26 h versus 24 h for controls, P = 0.51), ICU or hospital length of stay after adjustment for confounders. Midodrine did not affect mean heart rate but was associated with bradycardia. This case-control study demonstrates that midodrine has limited efficacy in expediting weaning from vasopressor infusions in patients who have already received relatively prolonged courses of these infusions.
Introduction
Persistent hypotension without evidence of end-organ dysfunction is commonplace in the post-resuscitation phase of critical illness and may necessitate the use of invasive haemodynamic monitoring and intravenous (IV) vasopressor agents. These interventions often necessitate admission to an intensive care unit (ICU) 1 or delay discharge from the ICU for patients who are otherwise clinically stable. Administration of IV vasopressors may also prolong need for venous and arterial access devices with attendant (albeit low) risks such as bloodstream infection and vasopressor extravasation.2–4 Therefore, there is a need to establish whether patients with persistent hypotension can be liberated from IV vasopressors through the use of alternative, enterally-administered vasopressor agents.
Midodrine is an enterally-administered prodrug, which is converted to the alpha adrenoceptor agonist desglymidodrine, is highly bioavailable, and reaches peak plasma concentrations within one h after oral administration. 5 Midodrine’s alpha-agonist activity is thought to increase systemic vascular resistance and blood pressure, and has been shown in multiple unblinded, non-randomised studies to expedite liberation from IV vasopressor therapy.1,6
The MIDAS study 7 was a multicentre, placebo-controlled, randomised clinical trial which assessed whether the use of oral midodrine accelerated liberation from IV vasopressor therapy in otherwise resuscitated ICU patients with hypotension. The MIDAS trial did not demonstrate a reduction in duration of vasopressor infusion, ICU or hospital length of stay with midodrine in patients who received the drug relatively soon after commencement of IV vasopressors (median 35.5 h). 7 However, there is a distinct patient group—those who have been on low-dose vasopressors for several days without any apparent weaning—in whom midodrine may facilitate discontinuation of the IV vasopressors. In our ICU at the time of the study it was common practice for treating intensivists to commence oral midodrine to facilitate weaning from vasopressor infusions. In this case-control study, we explored whether midodrine, when used in patients who had required prolonged IV vasopressor infusions, facilitated more rapid weaning than placebo. We also present analysis of detailed haemodynamic and vasopressor dosage data for these patient groups.
Materials and methods
Study design
This single-centre, case-control study was conducted between 2012 and 2020 at Sir Charles Gairdner Hospital in Perth, Western Australia, parallel to the MIDAS study. The study was approved by the Sir Charles Gairdner Osborne Park Health Care Group Human Research Ethics Committee (#2015-098) for the control arm, and retrospective review of the data from the cases was approved as quality improvement (# 35731). The MIDAS study was registered with the Australian Therapeutic Goods Administration (CT-2016-CTN-00226-1 v3). All participants or their medical treatment decision-maker provided written informed consent.
Study participants
Cases comprised patients commenced on oral midodrine at Sir Charles Gairdner Hospital ICU during the period 2012–2019. Cases were commenced on midodrine solely at the discretion of the treating intensive care consultant, where weaning from IV vasopressors had been prolonged and midodrine judged useful in expediting weaning. Cases were not included in the MIDAS trial. Data collection for cases was by retrospective chart review. Control patients were prospectively enrolled in the MIDAS study over the same period at Sir Charles Gairdner Hospital ICU, and as part of that trial were randomised to the placebo arm. Following unblinding and publication of the MIDAS study, data for control patients became available for use in this case-control study.
Eligibility criteria were age 18 years or older, admitted to an ICU or high dependency unit, low-dose single agent IV vasopressor treatment (<8 µg/min norepinephrine (noradrenaline), or <60 µg/min metaraminol) and a requirement for IV vasopressors to achieve clinically determined blood pressure targets for 24 h or more. Participants had to be otherwise resuscitated with reversible causes of hypotension treated.
Criteria for exclusion were clinical evidence of inadequate tissue oxygenation, hypotension due to adrenal insufficiency, liver failure, chronic renal failure (baseline serum creatinine >177 µmol/l) or severe organic heart disease (left ventricular ejection fraction <30%). Patients with acute urinary retention, phaeochromocytoma, thyrotoxicosis, bradycardia (heart rate (HR) <50/min), pregnancy, known allergy to midodrine, or who were unable to receive medications enterally were similarly excluded.
Intervention
Cases received 20 mg of oral midodrine every eight h from enrolment until IV vasopressors had been discontinued for 24 h or more, at which point midodrine was ceased. Control participants from the MIDAS trial received oral placebo prepared by study pharmacists.
Baseline variables and outcomes
Baseline information and data on potential sources of bias collected for all participants included age, sex, body mass index, Acute Physiology and Chronic Health Evaluation II (APACHE II) score, admission reason (medical, surgical, sepsis), use of propofol, dexmedetomidine, hydrocortisone, epidural analgesia and choice and dose of IV vasopressor agent. The pre-specified primary outcome was median time to discontinuation of IV vasopressor therapy after commencement of midodrine or placebo. Exploratory outcomes included ICU and hospital length of stay (LOS), IV vasopressor dose, HR, and mean arterial pressure (MAP), as well as adjustment of these variables for likely confounders such as APACHE II score and pre-enrolment MAP. Data on adverse events such as hypertension (systolic blood pressure >160 mmHg or an increase by ≥20% above goal) and bradycardia (HR < 40/min or ≥20% below goal) were also collected.
Statistical analysis
Data analysis was performed using a modified intention-to-treat approach, including all patients who received at least one dose of either midodrine (cases) or placebo (controls). Continuous data are expressed as mean (standard deviation) or median (interquartile range (IQR) or 95% confidence interval (CI)), as appropriate, based on normality of distribution. Case and control groups were compared using t-tests for continuous data and chi-square or Fisher’s exact tests, as appropriate, for categorical data. Linear mixed models, incorporating random subject effects and group–time interactions, were used to compare longitudinal outcomes (HR, MAP, dosage) between groups, with results summarised using estimated marginal means and 95% CIs. Vasopressor treatment duration, ICU and hospital LOS were estimated using Kaplan–Meier survival probabilities and compared using log-rank tests. Cox proportional hazards regression models were used to examine the effect of midodrine compared with placebo on vasopressor treatment duration and LOS, with results summarised using hazard ratios and 95% CIs. All models were adjusted for APACHE II scores and pre–study drug MAP. A two-sided P < 0.05 was considered as statistically significant. Statistical analyses were performed using Stata version 15 (StataCorp LLC, College Station, TX, USA) and RStudio 8 with the ggpubr 9 , rstatix 10 and survminer 11 packages.
Results
Baseline characteristics
A total of 61 participants were recruited (19 cases and 42 controls) between January 2016 and March 2020. Age was similar between cases and controls (64.6 years versus 68.1 years respectively) as were anthropometric measurements with the exception of weight (cases mean 71.05 kg versus controls mean 84.15 kg, P = 0.047). There were no differences between cases and controls with respect to admission diagnosis, the use of hydrocortisone, or enrolment MAP. Of note, the mean (standard deviation) APACHE II score of 20 (5) was higher in cases, compared with 14 (5) for controls (P < 0.001), indicating a higher predicted ICU mortality amongst cases. As expected, median duration of IV vasopressor use prior to commencement of the study drug was higher in cases at 94 h versus 29.3 h in controls (P < 0.001), which also resulted in a higher total IV vasopressor duration amongst cases (P < 0.001). Baseline characteristics of both groups are shown in Table 1.
Baseline characteristics by treatment group.
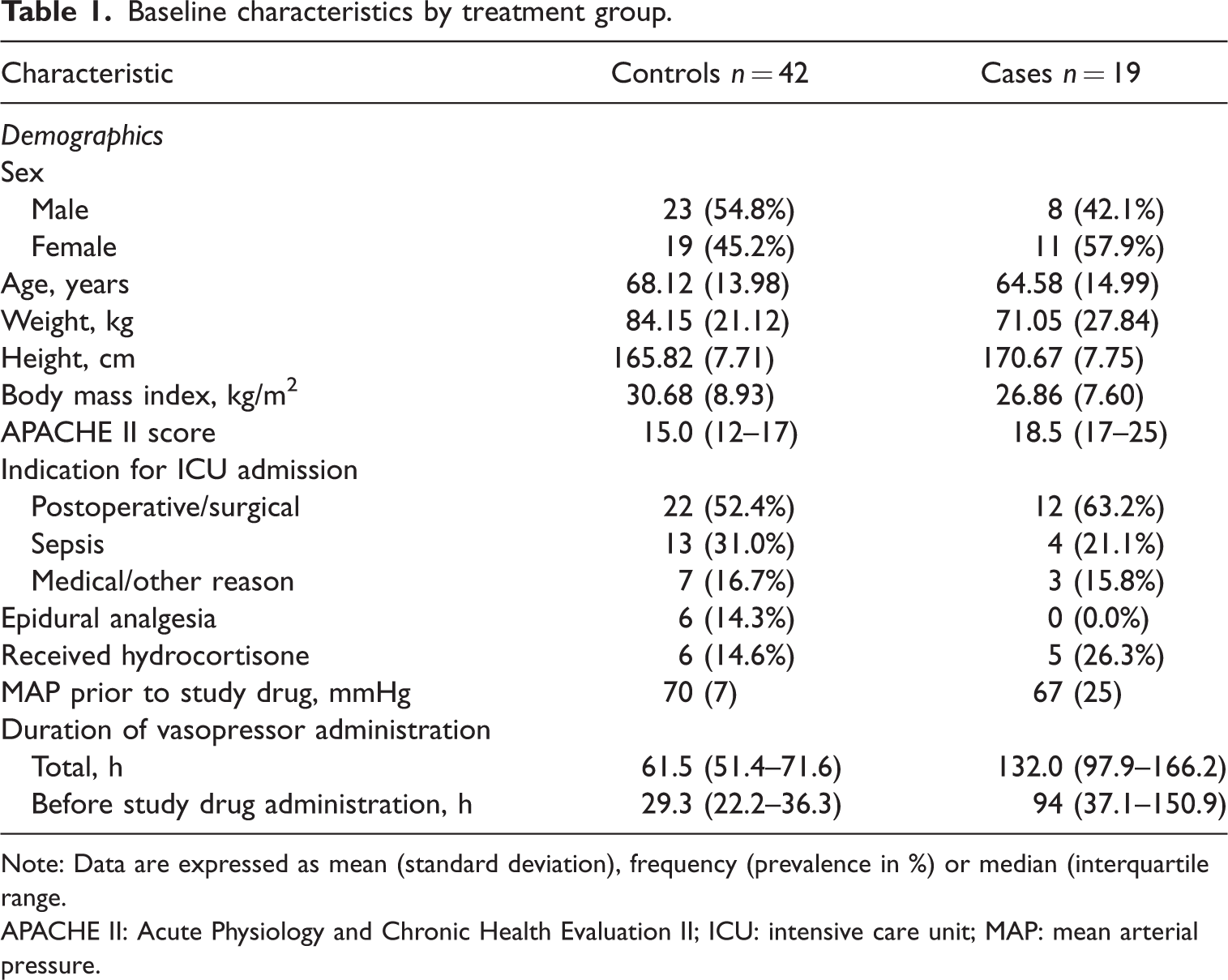
Note: Data are expressed as mean (standard deviation), frequency (prevalence in %) or median (interquartile range.
APACHE II: Acute Physiology and Chronic Health Evaluation II; ICU: intensive care unit; MAP: mean arterial pressure.
Primary outcome
Median (IQR) time to discontinuation of IV vasopressor was 26 (22–36) h for cases and 24 (17–93) h for controls, a difference which did not reach statistical significance (P = 0.511). The Kaplan–Meier plot for time to IV vasopressor discontinuation is shown in Figure 1. There was no difference between median time to discontinuation of vasopressor between cases and controls when adjusted for APACHE II score and pre-intervention MAP (Table 2).
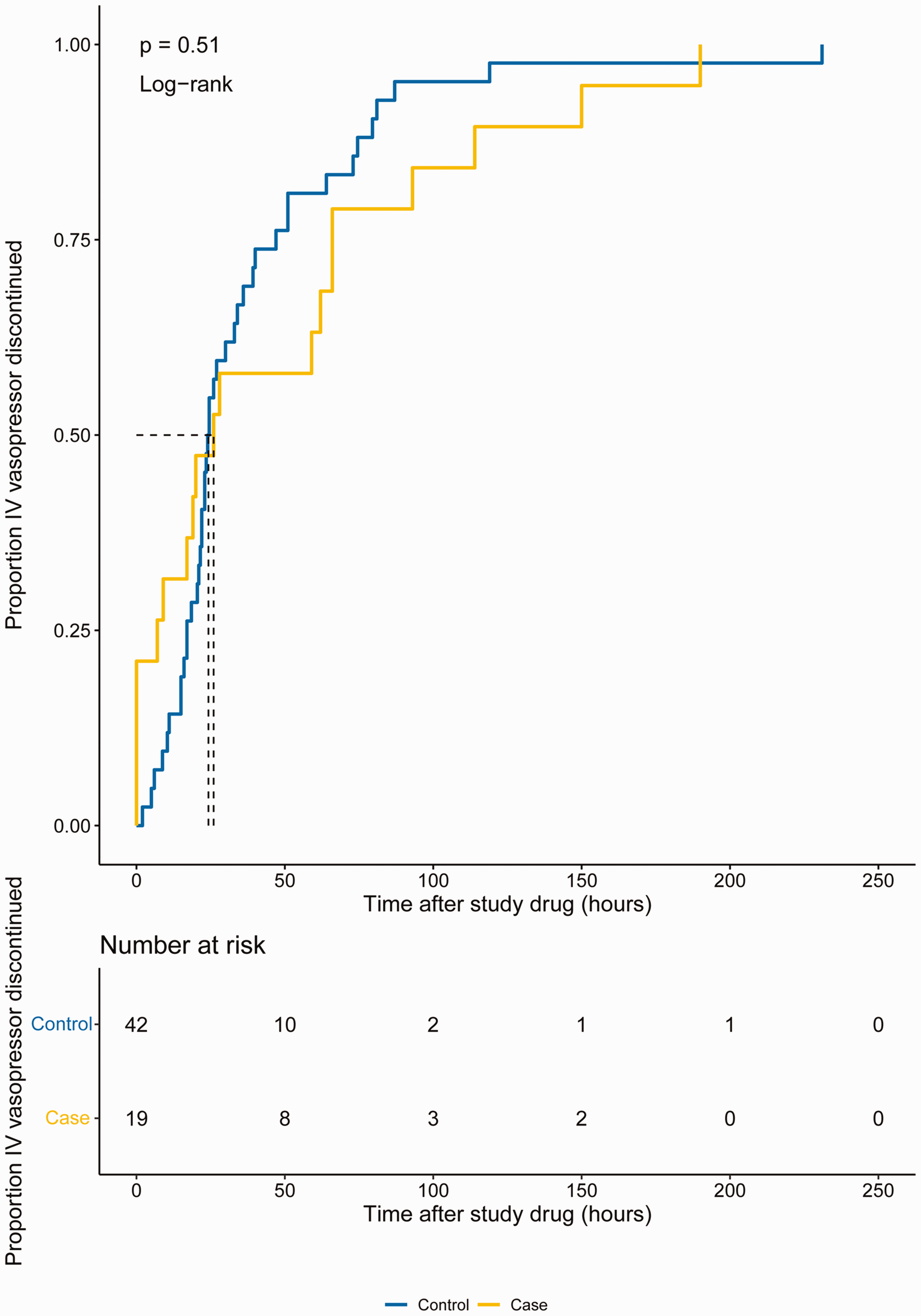
Midodrine use was not associated with accelerated liberation from intravenous (IV) vasopressor therapy. Kaplan–Meier cumulative incidence plot of time to discontinuation of IV vasopressor post commencement of midodrine or placebo. Median (95% confidence interval) time to discontinuation 26.0 (22 to 36) h for cases, 24.0 (17 to 93) h for controls, P = 0.511 by log-rank test.
Cox regression by cases, adjusted for (1) APACHE II score, (2) APACHE II score and pre-study drug mean arterial pressure.
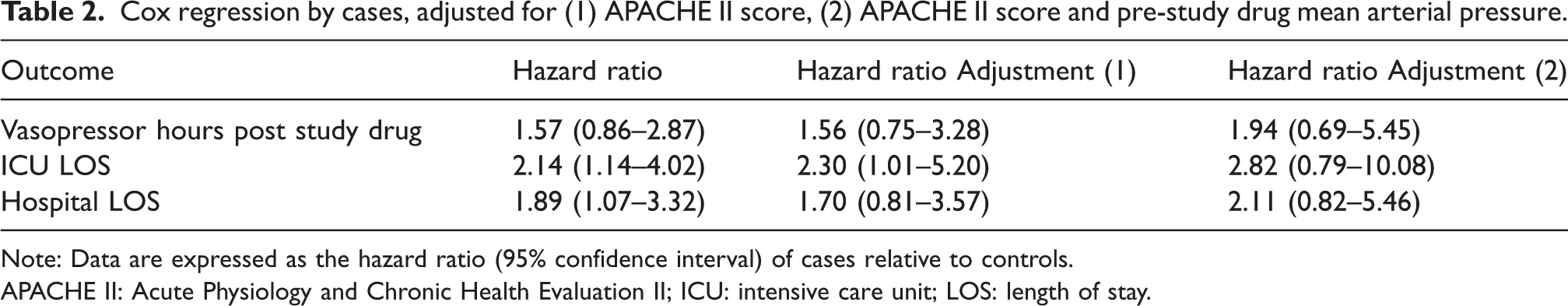
Note: Data are expressed as the hazard ratio (95% confidence interval) of cases relative to controls.
APACHE II: Acute Physiology and Chronic Health Evaluation II; ICU: intensive care unit; LOS: length of stay.
Exploratory outcomes
Duration of intensive care and hospital admission
Duration of ICU admission amongst cases was longer than in controls, with a median (95% CI) length of seven (six to 13) and six (five to six) days respectively, P = 0.0058. The difference in duration of hospital admission was more pronounced than ICU admission, with cases being hospitalised for a median (IQR) of 26.0 (14–51) versus 14.0 (10–17) days for controls, P = 0.022. However, when adjusted for baseline differences in APACHE II score and pre-intervention MAP, there were no differences between cases and controls for duration of ICU or hospital admission (Table 2).
Heart rate and mean arterial pressure
Haemodynamic data for the 36-h period encompassing the intervention (T–12 to T+24) are shown in Figure 2. Complete data were available for 60 patients. There was a trend to lower mean HR amongst cases, though this trend did not reach statistical significance (Figure 2(a)). Mean MAP was higher amongst cases receiving metaraminol; however, this difference was present prior to commencement of the study drug, though not at enrolment (Figure 2(b) and Table 1).
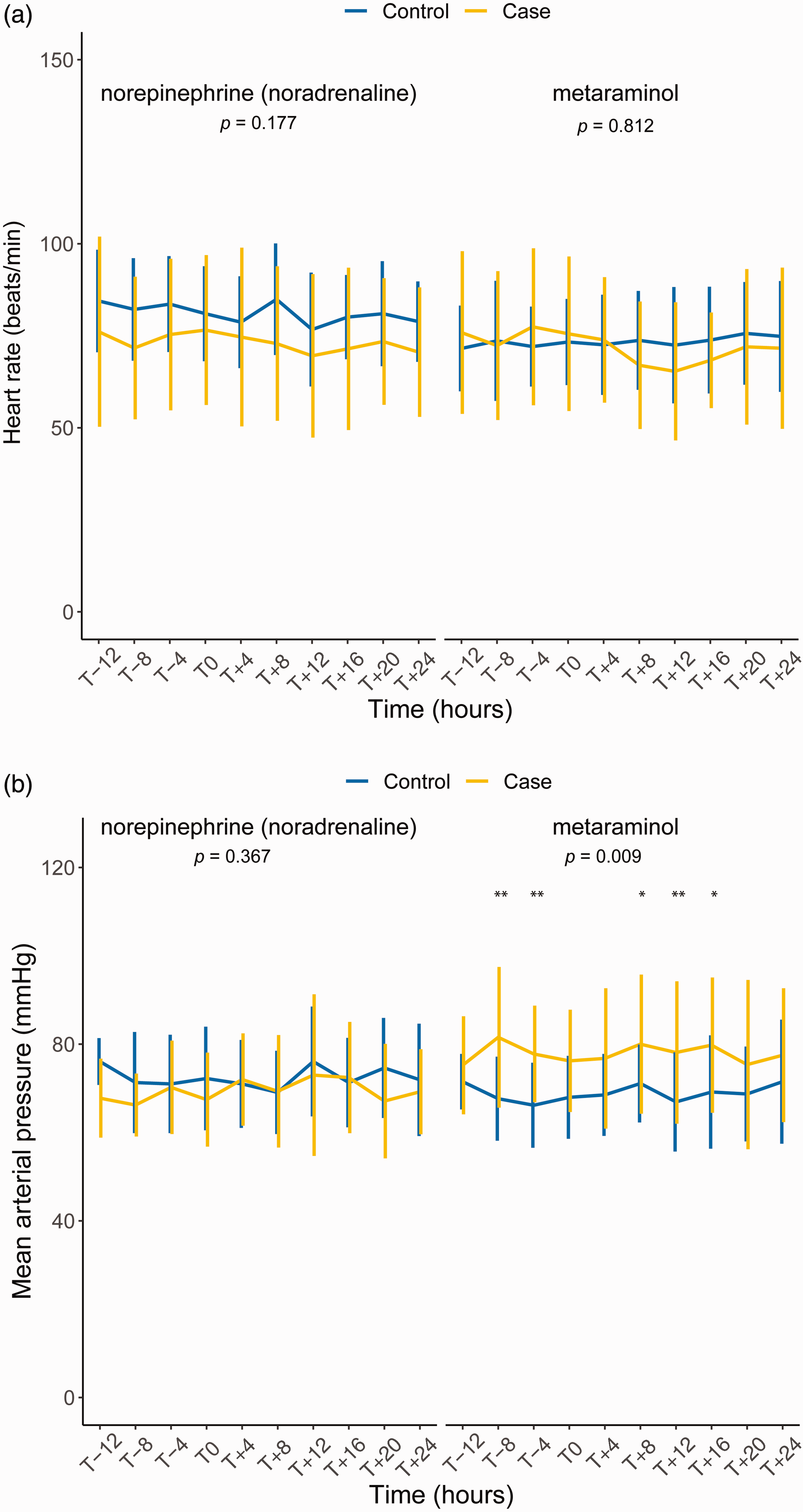
Midodrine did not affect mean heart rate and cases receiving metaraminol had higher mean arterial pressure prior to commencement of the study drug. (a) Mean heart rate was no different between cases and controls, irrespective of intravenous vasopressor and (b) Mean arterial pressure was significantly higher amongst cases receiving metaraminol, though this difference was present prior to introduction of study drug. Data are shown as mean (standard deviation). Pairwise comparisons at individual timepoints are shown where significant; * and ** indicate Bonferroni
Intravenous vasopressor dose
IV vasopressor dose for the 36-h period encompassing the intervention (T–12 to T+24) is shown in Figure 3 for 57 patients for whom complete data were available. There was an overall reduction in IV vasopressor dose with time, though this reduction was less pronounced in cases receiving metaraminol. Cases received lower doses of IV vasopressors, though, importantly, these dose differences were present prior to the commencement of study drug and disappeared with time (see Figure 3 and caption for P-values for respective timepoints). Cases receiving metaraminol demonstrated a smaller reduction in IV vasopressor dose than controls or cases receiving norepinephrine (noradrenaline).
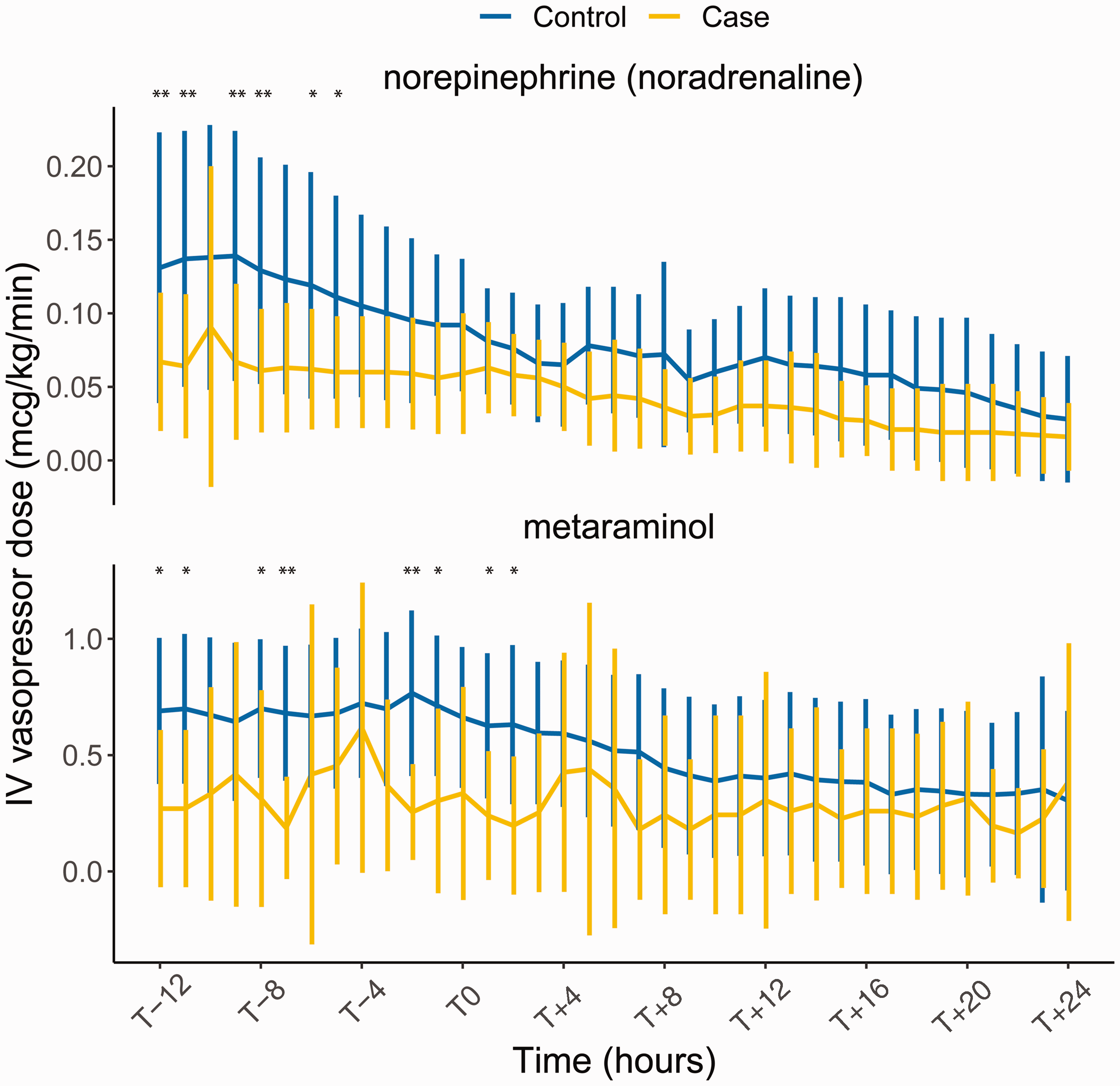
Intravenous (IV) vasopressor dose decreased markedly with time, and midodrine did not appear to affect IV vasopressor dose. (a) Mean norepinephrine (noradrenaline) dose was initially lower amongst cases (prior to commencement of study drug) and this difference was lost over time as IV vasopressor dose decreased. n = 6 cases, 13 controls, and (b) Mean metaraminol dose was initially lower amongst cases (prior to commencement of study drug) and this difference was also lost over time. n = 9 cases, 29 controls. Data are shown as mean and standard deviation. Pairwise comparisons at individual timepoints are shown where significant; * and ** indicate Bonferroni-adjusted P < 0.05 and <0.01 respectively.
Subgroups and adverse events
Intensive care admission reason
Table 3 demonstrates duration of IV vasopressor therapy after study drug commencement stratified by ICU admission reason (sepsis, postoperative or other) for cases and controls. A postoperative/surgical indication for admission to the ICU was associated with prolonged IV vasopressor requirement amongst cases (median 129 h versus 66.3 h) though no differences were observed for other admission indications. Data on adverse events including bradycardia and hypertension were collected. The prevalence of bradycardia was higher amongst cases (four participants, 22%) versus controls (one participant, 2.4%, P = 0.025). There was no difference in prevalence of hypertension between cases and controls (1, 5.6% versus 2, 4.8%, P > 0.9). Data on other adverse events were not collected in the case-control study.
Duration of intravenous vasopressor post commencement of study drug amongst cases and controls stratified by admission indication.
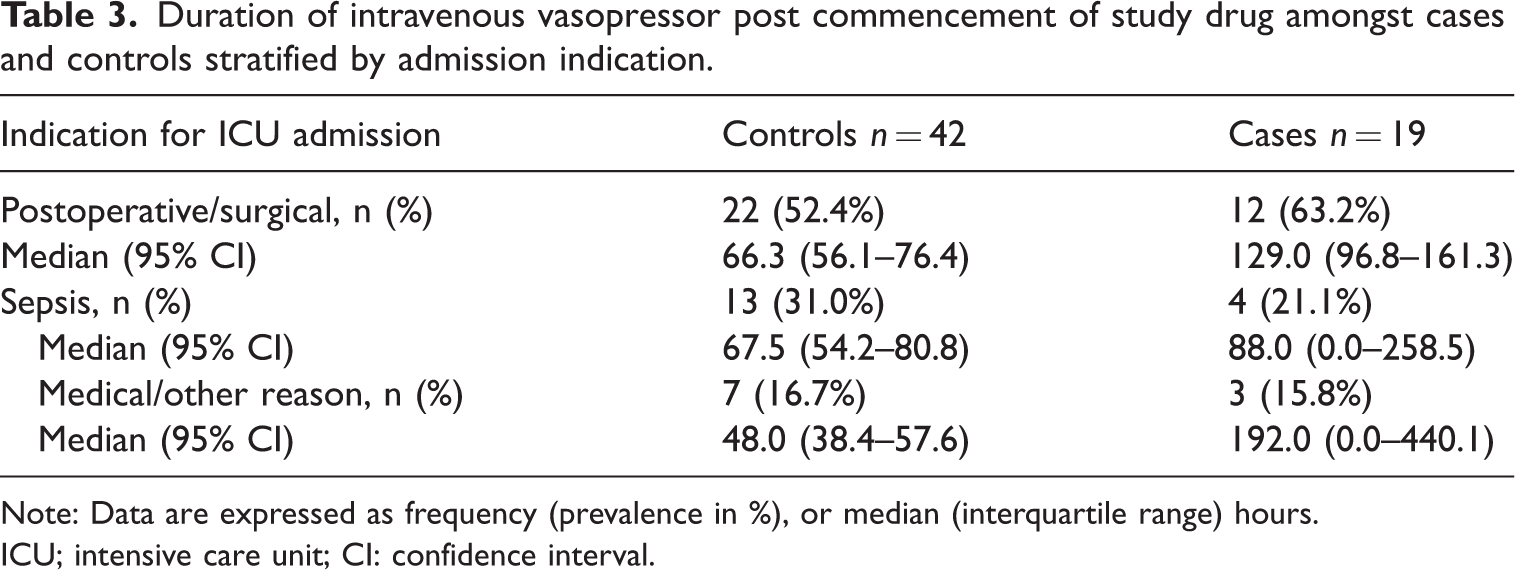
Note: Data are expressed as frequency (prevalence in %), or median (interquartile range) hours.
ICU; intensive care unit; CI: confidence interval.
Discussion
This case-control study sought to understand whether the oral vasopressor midodrine expedited weaning of IV vasopressors in patients who had already received prolonged courses of these medications, often at low doses. This aim is in contrast to the randomised controlled MIDAS trial 7 and other randomised studies, 12 which evaluated the early use of oral midodrine to accelerate liberation from IV vasopressors. This is a controversial and important issue, as expedited weaning from IV vasopressors may accelerate ICU discharge and reduce complications related to their use. Prior to the publication of the MIDAS trial, midodrine was used in this ICU as the treating intensivists believed that it assisted with weaning of patients who appeared ‘stuck’ on low-dose IV vasopressors for several days. The median duration of IV vasopressor prior to commencement of study drug was 94 h amongst cases versus 23.4 h in controls, indicating that our study captured this group of patients.
Our findings show that even in this group of patients, where clinicians felt midodrine was likely to have a beneficial effect, there was no effect of midodrine on expediting IV vasopressor weaning relative to placebo (Figure 1). This finding is in keeping with the primary outcome of MIDAS, the only appropriately-powered, randomised study of this question, 7 as well as several other studies and meta-analyses which showed no effect of midodrine on similar outcomes.12–15 Earlier retrospective or observational studies demonstrated that midodrine accelerated either liberation from vasopressors or discharge from the ICU.1,6,16 These differences are likely due to heterogeneous study populations, retrospective or observational designs and differing dose regimens.
Of note, corticosteroid use in our study was modest (14.6% controls, 26.3% cases; Table 1). This finding is likely driven by multiple factors. First, clinicians may have limited steroid use due to concerns over their effect on wound healing in a predominantly postsurgical cohort. Importantly, the majority of the study was conducted prior to the publication of the ADRENAL trial 17 in 2018, which may have resulted in intensivists electing to use steroids based on positive secondary outcomes of that study. Furthermore, international guidelines recommend corticosteroids be used only when adequate MAP cannot be maintained with norepinephrine (noradrenaline) doses of 0.25 µg/kg per min 18 or more, which was a different group of patients from the one studied here. An effect of midodrine on secondary outcomes such as ICU and hospital LOS initially appeared but was not significant after adjustment for likely confounders such as APACHE II score (Table 2).
Our study also assessed the haemodynamic effects of midodrine on HR, MAP and IV vasopressor dose in high resolution (four-hourly for MAP and HR, hourly for vasopressor dose). There was a trend toward a lower mean HR amongst cases, though this trend did not reach statistical significance (Figure 2). Consistent with this observation, bradycardia was noted with greater frequency amongst cases, further supporting data from MIDAS and other studies.1,16 MAP was higher amongst cases who received metaraminol but not norepinephrine (noradrenaline). This finding was present prior to the initiation of midodrine (Figure 2), indicating that it was likely a baseline difference in the patient groups rather than an effect of midodrine itself.
IV vasopressor dose over 36 h for each participant is shown in Figure 3, demonstrating that time was the predominant factor which affected weaning from vasopressor infusions. Cases received lower doses of IV vasopressor, though, like MAP, this difference was present at baseline and therefore unlikely to be due to midodrine use. A key potential determinant of these findings is the duration of IV vasopressor use prior to commencement of the study drug, which was significantly higher amongst cases. In combination with higher APACHE II scores, this may indicate that cases exhibited a multifactorial vasoplegic state which, once established, was unlikely to respond to selective vasopressor therapy.
Acute and severe vasoplegia has been extensively studied in the setting of sepsis and cardiopulmonary bypass, though it is likely a feature of almost all shock states. 19 The pathophysiology is heterogeneous and involves both intrinsic factors such as nitric oxide, endothelin 1 and prostanoids (the production of which is driven by inflammatory responses to damage and pathogen-associated molecular patterns), as well as extrinsic factors including catecholamine and vasopressin responsiveness and relative corticosteroid deficiency, as reviewed elsewhere.19–21 The maturation of these severe perturbances in vasoreactivity is less well understood, though there is some evidence for relative corticosteroid deficiency playing a role in prolonged vasopressor dependence similar to that noted in the current study. 22
Shock and vasopressor dependence in the acute phase of critical illness is associated with a mortality of up to 50%.23–25 In contrast, the relationship between prolonged, low-dose vasopressor dependence and prognosis is less well studied, though a developing literature suggests prolonged vasoplegia is not a benign entity. Data recently published by Chotalia and colleagues 26 demonstrate that IV vasopressor use on day 4 of ICU admission for septic shock, even at relatively low doses, is associated with a substantial increase in 90-day mortality. Similarly a large, retrospective, US-based cohort study demonstrated a higher 90-day and one-year mortality amongst patients who received continuous or late vasopressor therapy relative to those weaned within the first three days of ICU admission. 27 Such a complex and prognostically significant state affecting a substantial proportion of ICU patients warrants further investigation, particularly of the temporal pattern of the condition and targeted therapies which may address the underlying pathophysiology.
Strengths of the current study include a clear, prospectively defined study protocol, inclusion and exclusion criteria and a controlled study design which captured the patient cohort of interest. A significant limitation was imbalance between the case and control groups at baseline with respect to the APACHE II score. Attempts were made to control for APACHE II score through Cox regression which did not significantly alter the conclusions. This study provides further evidence against the routine use of midodrine to accelerate liberation from IV vasopressor infusions in critically ill patients and adds detailed haemodynamic data in support of this point.
Footnotes
Availability of data and material
All study data and statistical analysis code available on request after deidentification.
Author Contribution(s)
Acknowledgement
The author(s) would like to thank Ryan Mahony, Jade Jackson and Darragh Fitzgerald for their assistance with data collection.
Declaration of conflicting interests
The author(s) have no conflicts of interest to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
