Abstract

N95 filtering facepieces (FFPs) are an important component of personal protective equipment (PPE) to mitigate the risk of severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) transmission in healthcare workers. Fit testing is conducted to ensure that a seal can be achieved between the user and the FFP so that the FFP provides the intended level of protection. Without an adequate seal, an N95 FFP offers the same level of protection as a surgical mask. 1
In June 2020, after obtaining human research ethics approval (ethics approval no.: 13453) and registering with the Australian New Zealand Clinical Trials Registry (no.: 12620000702910), we conducted a single-centre, prospective cohort study in a university teaching hospital. Anaesthetists and anaesthesia registrars and nurses who were part of the coronavirus disease 2019 (COVID-19) airway team were invited to participate in a fit testing study to evaluate the pass rate of the BSN Medical ProShield® N95 (size medium TN01-11 and small TN01-12, thereafter called ProShield N95; BSN Medical Australia, Mulgrave, Victoria) using quantitative fit testing with a PortaCount 8048 machine (TSI Incorporated, Shoreview, MN, USA). The ProShield N95 was the most commonly used FFP in our hospital at the time of the ethics application. We hypothesised that users who passed a fit check, as described by the Department of Health and Human Services (State of Victoria, Australia), would also pass the fit test.
As part of the study, participants selected the Proshield N95 size that they would usually wear and performed a fit check. They were then immediately fit tested using the modified Occupational Safety and Health Administration (OSHA) protocol. An overall fit factor of 100 or greater was considered a pass in line with Australian standards and OSHA legislation. Participants who did not pass were provided education using the ‘real time mode’ on the PortaCount machine. This mode allowed participants to modify their FFP placement to observe directly how this affected the fit factor in real time. If participants could consistently achieve a fit factor or 100 or greater in the ‘real time mode’, they were asked to doff and re-don the FFP for repeat testing. If the overall fit factor remained less than 100 during the retest, the fit test was considered failed. If appropriate, the test was repeated with a different size ProShield N95.
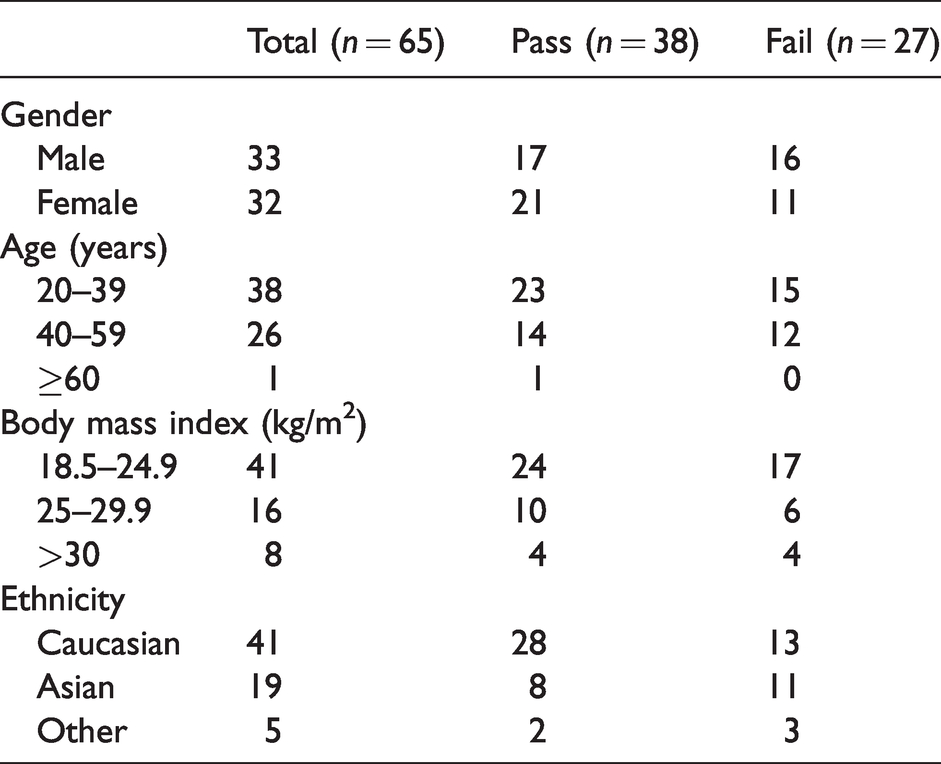
A total of 65 participants were tested in our study. All the participants passed a fit check but only 22/65 (34%) passed the subsequent fit test. The difference in the pass rate between the fit check and fit test was statistically significant with a P<0.0001 using a two-sided Fisher’s exact test. A further 16 participants passed after either receiving education using the ‘real time mode’ function or changing the size of the FFP resulting in an overall pass rate of 38/65 (58%). There were no statistically significant differences observed in the pass versus fail rates between gender, age and ethnicity (Table 1).
ProShield® N95 (BSN Medical Australia, Mulgrave, Victoria) filtering facepieces quantitative testing results.
Our study is consistent with previous studies which have also found that fit checking alone is insufficient to assess the adequacy of a N95 FFP seal.2–4 However, our study found a higher fail rate in participants who passed a fit check compared to other studies.3–5 This could be due to the construction of the duckbill-style FFP, which is less rigid than a cup or three-panel FFP. It is therefore more likely to inflate or deflate with a positive or negative fit check, which falsely reassures the participant that the seal is adequate. Previous studies have also found that the duckbill design may be more likely to fail fit testing compared to other designs.4–6 Our study further highlights that fit checking alone is insufficient in identifying users who do not adequately fit this brand of N95 FFP.
Footnotes
Author Contribution(s)
Declaration of conflicting interests
The author(s) have no conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Department of Anaesthesia, Austin Health, Victoria, Australia.
