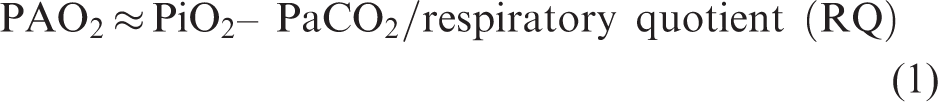Abstract
Opioid-induced ventilatory impairment is the primary mechanism of harm from opioid use. Opioids suppress the activity of the central respiratory centres and are sedating, leading to impairment of alveolar ventilation.
Respiratory physiological changes induced with acute opioid use include depression of the hypercapnic ventilatory response and hypoxic ventilatory response. In chronic opioid use a compensatory increase in hypoxic ventilatory response maintains ventilation and contributes to the onset of sleep-disordered breathing patterns of central sleep apnoea and ataxic breathing. Supplemental oxygen use in those at risk of opioid-induced ventilatory impairment requires careful consideration by the clinician to prevent failure to detect hypoventilation, if oximetry is being relied on, and the overriding of hypoxic ventilatory drive. Obstructive sleep apnoea and opioid-induced ventilatory impairment are frequently associated, with this interrelationship being complex and often unpredictable. Monitoring the patient for opioid-induced ventilatory impairment poses challenges in the areas of reliability, avoidance of alarm fatigue, cost, and personnel demands. Many situations remain in which patients cannot be provided effective analgesia without opioids, and for these the clinician requires a comprehensive knowledge of opioid-induced ventilatory impairment.
Introduction
Anything in excess is a poison.
Theodore Levitt
Opioids are licenced as restricted medications in part because of the desirable feelings they induce.1,2 When this is combined with the desire of a good clinician to act with beneficence 3 and be in line with examples of their peers, then the principles of lowest risk prescribing may be unwittingly circumvented. An understanding of patient and clinician behaviours may explain the witnessing of several anomalous opioid prescribing practices. For example, in patients with chronic pain who were prescribed high opioid doses (of greater than 200 mg morphine oral equivalents per day), the risk of death tripled compared to those prescribed less than 20 mg morphine oral equivalents per day.4 Again in chronic pain, there was a tendency to prescribe high-dose opioids to patients with mental health problems. For these patients, if an opioid use disorder subsequently developed, a poor ability to cope with this additional life-stressor was predictable.5 In acute pain, opioids were frequently on-prescribed well past the initially intended time period of opioid use.6 In response to examples such as these, opioid risk-reduction strategies in chronic pain and opioid-sparing strategies perioperatively are now encouraged to minimise opioid harms.7,8
There will remain circumstances in which opioid-sparing strategies are unachievable and there is an appropriate need to administer opioids. The major burns, multitrauma or emergency surgery patient often cannot be provided with effective analgesia without opioids. For these patients and many others, the clinician requires a comprehensive knowledge of opioid-induced ventilatory impairment (OIVI).
Opioid-induced death
The central nervous system depressant actions of opioids leading to OIVI and death are of greatest concern to the clinician utilising opioids for pain management. 9 Opioids have effects on many other organ systems, with the gastrointestinal tract effects being perhaps the next most problematic; however, these other system effects are in the most troublesome rather than life-threatening. Less frequently, cardiac effects such as QT interval prolongation as seen with methadone are of concern and need to be monitored to prevent sudden deaths. 10
Pathophysiology of opioid-induced ventilatory impairment
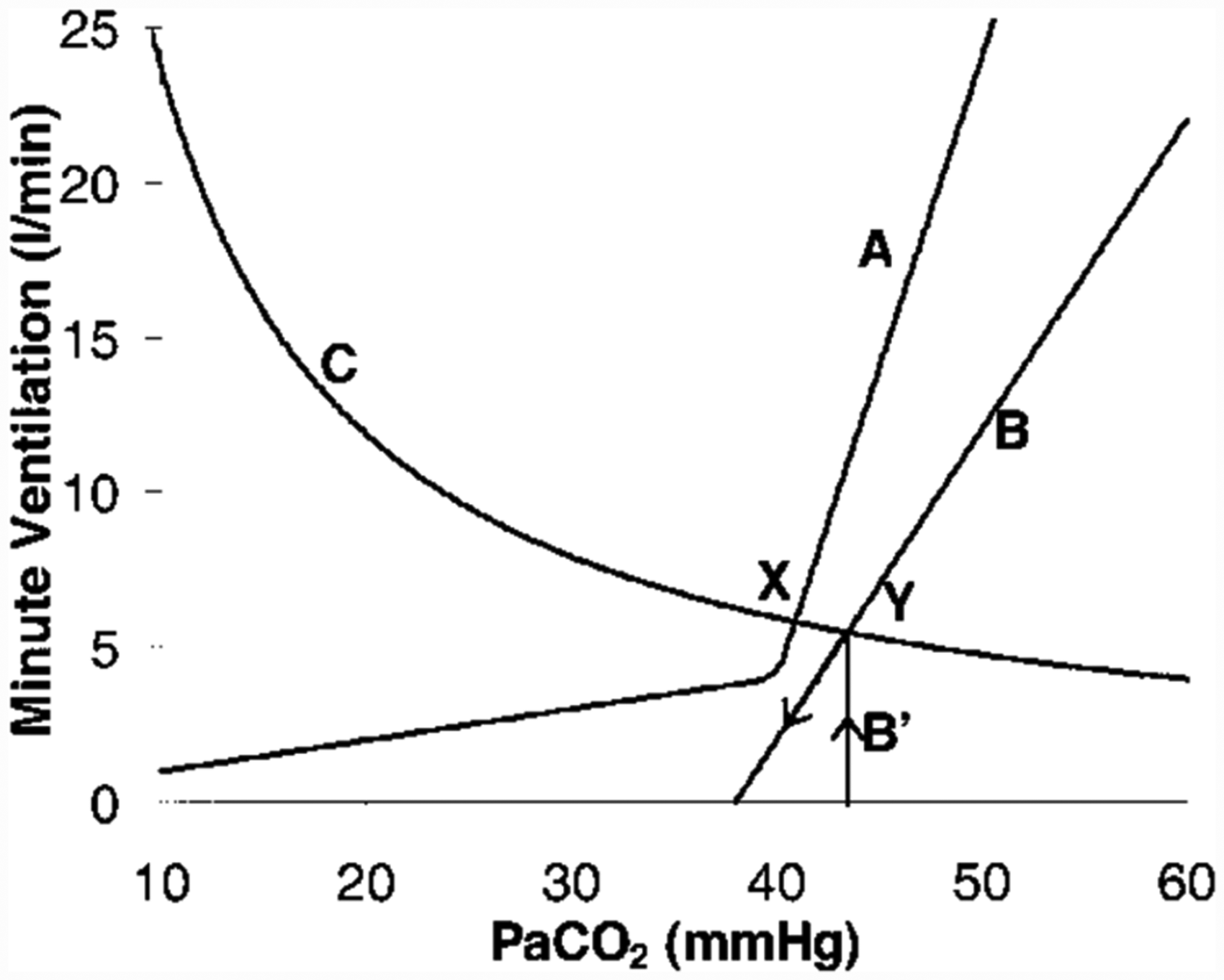
In the normal state there is a basal tone of endogenous opioid suppression of respiration. 11 The key respiratory control sites affected by opioids include chemoreceptors in the pontine parabrachial and Kölliker-Fuse nuclei, and rhythm control in the medullary pre-Bötzinger complex. 11 Opioid-induced respiratory depression (OIRD) culminates from both suppression of chemoreceptor response and respiratory rhythm generation leading to hypoventilation or apnoea with hypercarbia and hypoxia. 12 Changes in respiratory rhythm can be measured by respiratory rate and tidal volume. The changes in chemoreceptor response are measured physiologically as decreases in both hypercapnic ventilatory response (HCVR) and hypoxic ventilatory response (HVR) and an increase in ventilatory response threshold (VRT).13,14 Administering exogenous carbon dioxide (CO2) allows for the HCVR and VRT to be determined (Figure 1). 15 Lowering the arterial oxygen tension (PaO2) allows for the HVR to be determined (Figure 2).
Carbon dioxide (CO2) response curve changes following administration of an opioid. Curve A represents the normal ventilatory response to changes in arterial carbon dioxide partial pressure (PaCO2), line B the changes following opioid administration and curve C the CO2 excretion hyperbola (PaCO2 response to changes in ventilation). The inflection point on curve A is the ventilatory response threshold beyond which (to the right) is the normal hypercapnic ventilatory response. The X-intercept of line B illustrates that apnoea can be induced, particularly with potent rapid onset opioids. Reproduced, with permission, from Anesthesiology.
Oxygen response curve following sub-apnoeic opioid dosing. A indicates the line slope equating to hypoxic ventilatory response.
As distinct to depression of respiration, ventilation can be impaired by upper airway obstruction secondary to sedation from the direct pharmacodynamic action of the opioid. 16 Hypercarbia is commonly thought also to contribute to the sedation (narcosis) seen with opioids. This belief is based largely on historical animal experiments in which an anaesthetising effect of CO2 was purported, although this effect was more likely to be due to hypoxia.17,18 The notion of CO2 narcosis is challenged by case reports of markedly elevated PaCO2 of 130–175 mmHg in respiratory failure patients without depression of consciousness.18,19 Clinical observations of patient somnolence with hypercarbia after application of oxygen to relieve hypoxaemia have been suggested to be simply circumstances of exhaustion and sleep deprivation. 20
In one experimental animal study, opioids were demonstrated to reduce the tone of upper airway muscles; however, clinical correlates of impaired ventilation through obstruction by this mechanism have not been demonstrated in humans.21–23 Nonetheless, the ability of opioids to impair ventilation by obstruction due to sedation provides reason for the more encompassing term of OIVI 24 (Figure 3).
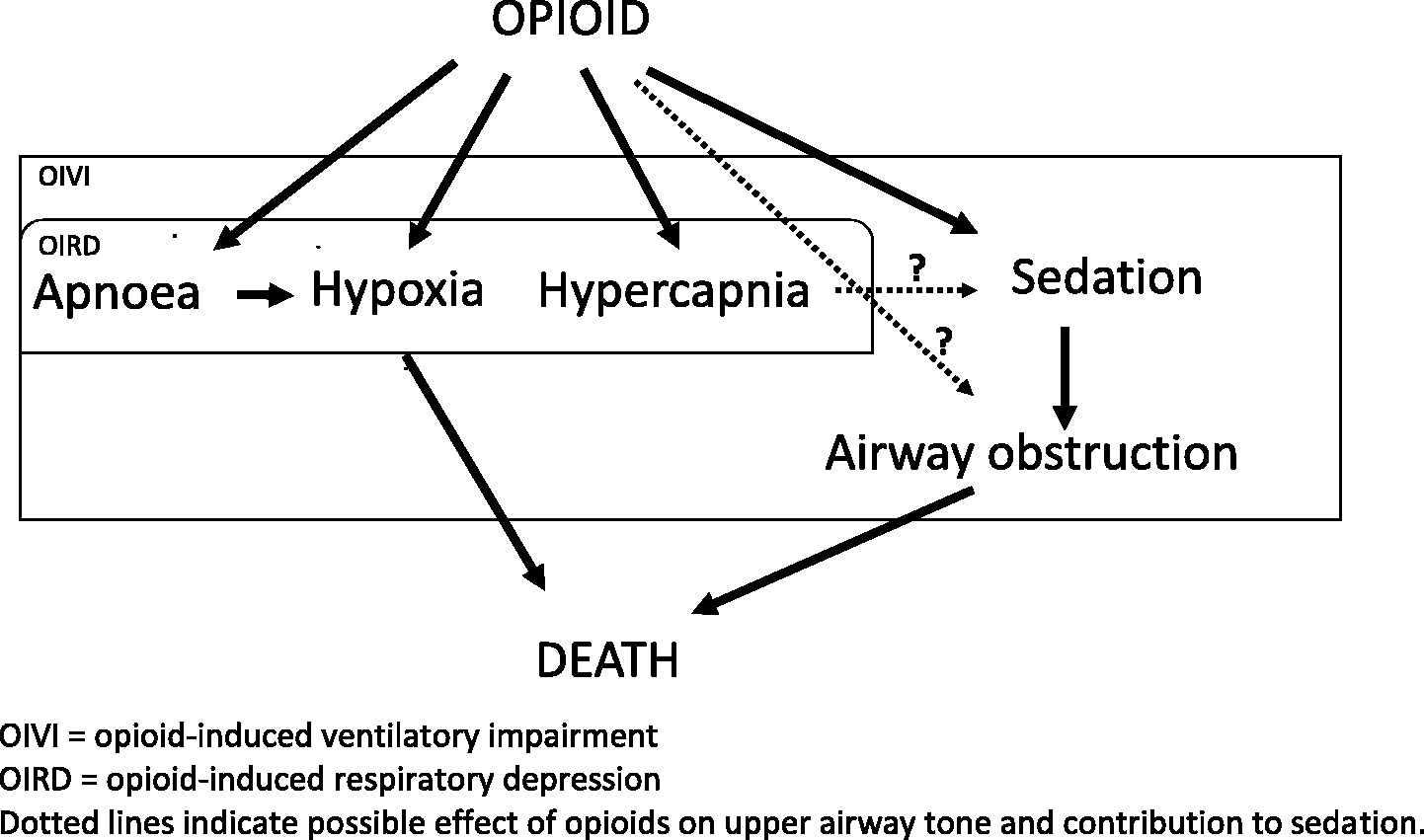
Schematic diagram of relationship between opioids and ventilatory changes. OIVI: opioid-induced ventilatory impairment; OIRD: opioid-induced respiratory depression. Dotted lines indicate possible effect of opioids on upper airway tone and contribution to sedation.
Sleep and the effect of opioids
Normal adult sleep leads to clinically relevant changes in respiratory physiology parameters. 13 Minute ventilation declines by up to 1.6 l/min from a combination of reduction in tidal volume and respiratory rate. During rapid eye movement (REM) sleep stage R, respiratory rhythm becomes irregular with episodes of apnoea and a pattern similar to Cheyne–Stokes breathing. Chemoreceptor responsiveness is dampened, seen as decreases in the HVR and HCVR, leading to a small fall in oxygen saturation of up to 2% and a rise in PaCO2 of 2–8 mmHg. These sleep-related changes are likely to contribute to the peak of respiratory depression events occurring between 0200 and 0600 hours. 25 REM sleep, accounting for 20%–25% of sleep duration, is the period of lowest tone of the pharyngeal musculature, and therefore the time when obstructive sleep apnoea (OSA) obstruction episodes typically worsen.13,22 The lighter sleep stages of N1 and N2 are easiest from which to rouse, while deep sleep stages, N3 and N4, are the restorative phases and most resistant to arousal. 26
Opioids can significantly alter the normal sleep architecture: REM sleep decreases substantially, even being eliminated; time in stages N1 and N2 increases; and time in stages N3 and N4 decreases. 22 For OSA sufferers these sleep architecture changes with opioids can paradoxically result in benefit for some. 22 Cessation of opioids leads to a rebound effect on sleep pattern with increased time in REM and deep sleep and thus the potential for worsening OSA symptoms at these timepoints; however, there has been no experimental evidence of this occurring. 22
Opioid-induced ventilatory impairment and risk of death
The increased risk of death for inpatients receiving opioids, reasonably assuming this is due to OIVI, is very difficult to determine with accuracy, instead an estimate can be derived by several means.
Comparative studies
It is not feasible to randomly assign a population of patients needing pain relief into one half who receive an opioid and another who do not, for ethical reasons. Instead we can only study opioids with an active analgesic comparator, which then adds a confounding influence of the active comparator on mortality. An example of such an approach is to compare mortality outcomes between systemic opioid and epidural analgesia.
The 2002 MASTER (Multicentre Australian Study of Epidural Anaesthesia) trial compared epidural with intravenous opioid analgesia and failed to demonstrate a difference in mortality outcome at 30 days. 27 The robustness of the findings was influenced by several factors including the substantial epidural failure rate of 46% and the lack of standardised practice across study sites.
A retrospective analysis of 55,556 postoperative patients with lumbar or thoracic epidural analgesia found a small mortality benefit compared with alternative analgesia, which although not stated was likely to be opioid based, with a number needed to treat (NNT) of minimal clinical relevance of 477. 28
A meta-analysis of predominantly thoracic epidurals reported a clinically important mortality benefit deriving a NNT of 56. 29
NNT defines the mortality benefit of an intervention, in this case an epidural, but these same values can be used to quantify the number needed to harm of the comparator, in this circumstance the opioid. The number needed to harm for opioids is therefore in the range of 56 to 477, reflecting the net effect on mortality between opioids and an epidural. Epidural analgesia is known to moderate the neuroendocrine stress response to surgery thought to contribute to morbidity and mortality. 30 These figures therefore are not the true risk of death due to opioids alone. They are the relative risk of death when systemic opioids are used in preference to epidural analgesia.
Monitoring studies
One in-hospital observational study designed to test continuous central respiratory monitoring reported one OIVI death for almost every 5000 unmonitored patients. 31 These patients were on a general care ward and their rates of opioid use were not reported so this denominator value is likely to be an underestimate of risk.
Opioid-related adverse events
Another approach taken to determine opioid-related death has been to compare outcomes in patients who are coded as having opioid-related adverse events (ORADEs) with those who have not. Using this methodology, a recently reported study in the surgical literature reviewed 135,000 postprocedural patients, of whom 11% were classified as having an ORADE. 32 The risk-adjusted inpatient mortality was substantially higher if an ORADE was reported at 3.0% versus 0.1% in the no-ORADE group. The authors appropriately qualified their findings as being associations and not causality. There was no difference in the median daily opioid dose between groups, with these doses considered very low clinically at only 15 mg oral morphine equivalents, and the time relationship between the event and opioid dosing was not recorded—all indicating an absence of a dose–response effect as might be expected. Further caution is needed in interpreting these data by noting that many of the events classified as an ORADE had causalities other than an opioid, and these contributed substantially to the total ORADE counts. For example, mechanical ventilation contributed 23%, ileus 14%, and acute respiratory failure 9%. While open abdominal and cardiac surgery contributed to 25% of the ORADE count, these surgeries were also highly likely to have an ORADE-qualifying event not directly due to an opioid.
Opioid-induced ventilatory impairment risk associations
An extensive list of factors has been reported to be associated with increased rates of OIVI, and they vary depending on the criteria set for the definition of OIVI. 9
The recently reported PRODIGY (Prediction of Opioid-induced respiratory Depression In patients monitored by capnoGraphY) study utilised a broadly encompassing definition of respiratory depression (RD) of any one of: respiratory rate less than five breaths per minute for more than three min, peripheral oxygen saturation (SpO2) less than 85% for more than three min, end-tidal carbon dioxide (EtCO2) less than 15 mmHg or greater than 60 mmHg for more than three min, or apnoea for longer than 30 s. 33 Out of an exhaustive list of 31 possible associations, some previously reported, five factors with the highest odds risk correlation with RD were identified: age over 60 years, male sex, opioid naivety, sleep-disordered breathing, and chronic heart failure. At least one of these five factors was present in 74% of patients deemed to have had an episode of RD after surgery. The daily oral median morphine equivalent in this study was 21 mg in those with RD versus 25 mg in those without, raising the possibility that RD events were not opioid induced, and as such identifying only those patients who are potentially at greater risk. 34
It can seem logical to focus attention on patients with known associated risk factors for OIVI. Even limiting focus to just the five factors identified in the PRODIGY study will identify large numbers of patients as being at risk, 50% based on age alone. Flagging large numbers will, however, still miss many as all five factors were absent in 26% of patients who experienced RD. If the aim is one of preventing all deaths due to OIVI then all patients must be assumed to be at risk.
Clinical considerations for opioid-induced ventilatory impairment
Opioid naivety and tolerance
The first exposure to opioids in the truly naive, meaning no prior opioid experience, presents a risk for OIVI because their response is unknown. A common misconception is that risk in the naive is due to a lack of opioid tolerance. However, it is largely due to the recommended initiating doses of opioid being those that provide effective analgesia in a high percentage of patients—rather than being the dose at which OIVI is prevented in all. The first exposure to an opioid should therefore be of a short-acting agent so the response is readily witnessed and is short-lived. Slow-release (SR) opioid formulations and those with a prolonged duration of action, for example, methadone and transdermal fentanyl, are not recommended in the naive for this reason. 35 The risk posed for the opioid naive to be unwittingly ‘overdosed’ on their first exposure to an opioid is highlighted by the association between opioid naivety and OIVI. 33
Tolerance to the analgesic action of opioids can be profound; in contrast, tolerance to the RD effect is much slower to develop and is less marked. 36 The tolerant patient can require markedly increased acute opioid dosing to achieve equianalgesia, with the risk being that such doses will be more likely to be associated with RD. Consequently, prescribing equi-analgesic doses to the opioid tolerant requires a similar level of vigilance as with the opioid naive.
Differential tolerance to analgesia and RD has been demonstrated in animal studies. Rhesus monkeys, after a 12-week period of high-dose twice daily morphine injections, when tested for the analgesic response to a single morphine injection showed significant tolerance across a range of doses. 37 No tolerance to the RD effect was seen and even the low doses showing no analgesic effect produced the same degree of RD as seen in the baseline control measurements.
Human research comparing methadone-maintained subjects with non-tolerant controls demonstrated some tolerance to the RD actions of an acute morphine dose. 38 In the methadone- maintained, a four-fold greater dose of intravenous morphine achieving a seven-fold higher morphine blood concentration had no analgesic effect, as measured by cold pressor testing. As in the animal studies, the chronically dosed subjects demonstrated similar RD to controls, but only at higher acute opioid doses—indicating some tolerance to RD in humans. Of note, RD was greatest in the subgroup with the highest maintenance dose of methadone of 81–115 mg/day.
Chronic opioid dosing dampens HCVR, which can be further exacerbated by acute dosing. The observation of chronically dosed patients’ apparent lack of a RD ill effect to sometimes extraordinarily large opioid doses and an impression of tolerance is due to a compensatory increase in the HVR acting to normalise alveolar ventilation 39 (Figure 4). The effect of this increased HVR is a state of heightened chemoreceptor reflex–mediated control of ventilation commonly presenting during sleep. A florid presentation of this is central sleep apnoea (CSA), defined as apnoea of greater than ten seconds in the absence of respiratory effort. CSA has been reported in up to 60% of chronic opioid users. 40 The respiratory pattern in CSA consists of clustered breaths followed by apnoea due to HVR-triggered hyperventilation driving the PaCO2 to the apnoea point. A different type of HVR-associated sleep-disordered breathing, that of ataxic breathing, is reported in up to 92% of chronic opioid users. 40 Currently CSA and ataxic breathing are not in themselves thought to pose a risk of increased death, rather they should be seen as positive adaptive changes to chronic opioid exposure. 39
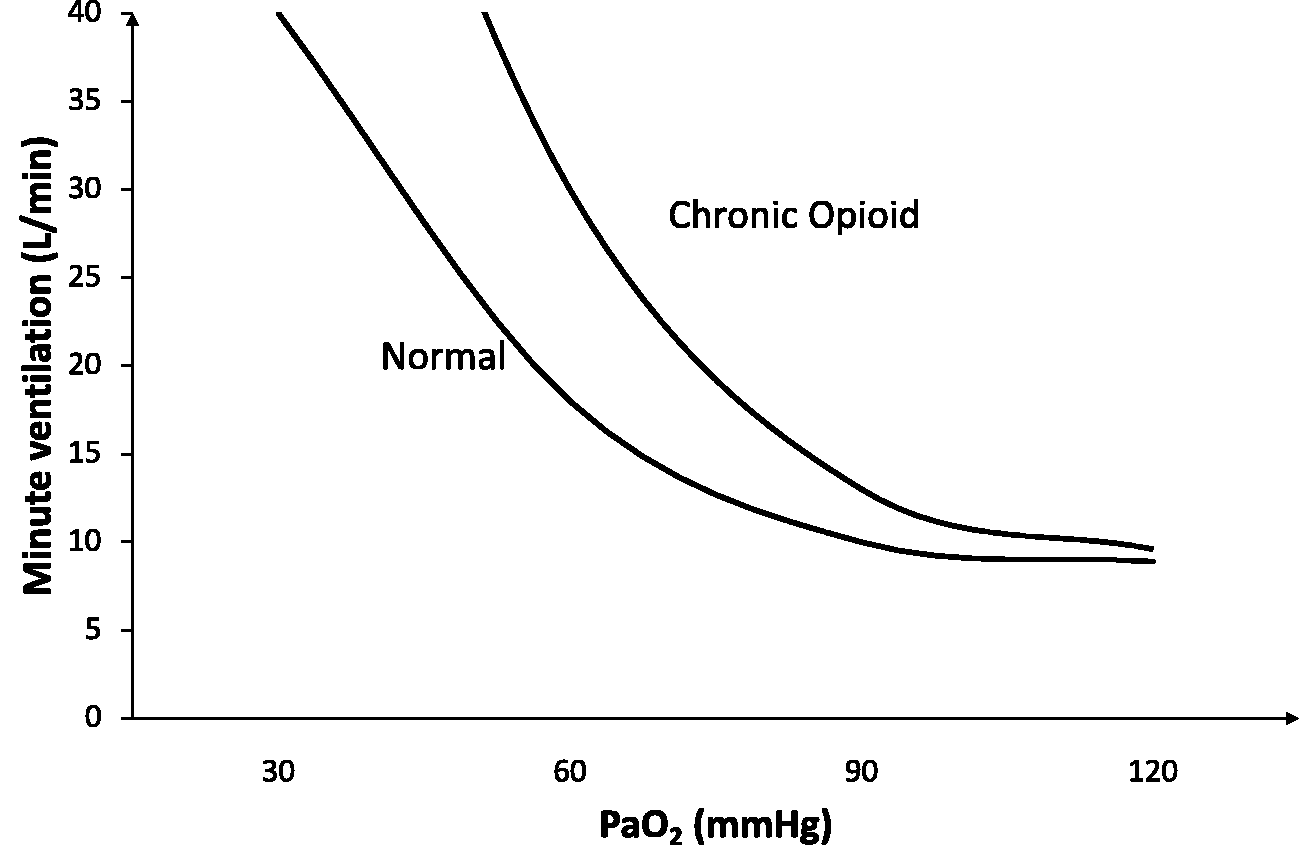
Oxygen response curve with chronic opioid use demonstrating the increase in hypoxic ventilatory response.
Hence the opioid-naive as well as the tolerant patient should raise concerns for OIVI. The naive feature in studies such as those of PRODIGY as they are in essence inadvertently overdosed on their first exposure. At the other end of the spectrum, the tolerant also feature in adverse event data, accounting for 8% of OIVI critical events, possibly due to clinicians up-sizing opioid doses to achieve analgesia without understanding the pathophysiology of chronic opioid dosing. 41
Supplemental oxygen
As one of the readily identifiable manifestations of OIVI is hypoxaemia the use of supplemental oxygen (SO), typically with nasal prongs or a Hudson mask at 2–6 l/min, has been historically advocated to limit harm. This was based on a belief that remedying the hypoxaemia caused by hypoventilation would prevent harm. It is now more apparent that it is the apnoea induced directly by the opioid, or by obstruction of the airway from sedation, which leads to harm. The application of SO to prevent harm from OIVI is not evidence-based and the only benefit yet identified is to reduce the frequency of hypoxaemia.
The addition of SO via nasal prongs with patient-controlled intravenous analgesia (PCA) was shown by Stone et al. to reduce the frequency of nocturnal desaturations (SpO2 <90%) from 56% to 12%. 42 The authors noted no harm resulted from hypoxaemia. There was one adverse event in 32 patients while SO was in use due to the normalising of the patients’ SpO2 delaying the diagnosis of significant OIVI.
Several beliefs about the benefits of SO can pervade but which have been discredited. For example, the addition of postoperative SO does not reduce the frequency of postoperative myocardial ischaemia or of associated hypoxaemia in at-risk patients. 43 Similarly, surgical site infection rates are not altered by the use of postoperative SO. 44 A Cochrane meta-analysis reported on the impact of increased SO utilisation when hypoxaemia was detected by continuous SpO2. 45 Applying SO to hypoxaemic patients did not alter mortality, intensive care transfers, cognitive dysfunction or length of hospital stay.
The unreliability of SO as a failsafe to prevent deaths or other critical events was highlighted in a closed claims analysis of 92 cases judged to be OIVI-related. 41 In this, one in six patients had been receiving SO prior to the event. When OSA critical events including death are analysed, the use of SO preceding the event triples to being one in two patients. 46
These findings reinforce the fact that the mechanism for opioid-induced death with opioids is primarily failure of ventilation and not secondary hypoxaemia. In the absence of ventilation, it is self-evident that the application of SO to the front of the mouth or nose is unable to rectify the resultant hypoxaemia.
Postoperative hypoxaemia
There can be an underappreciation by clinicians of how commonly postoperative hypoxaemia occurs. Sun et al. used continuous SpO2 monitoring for the first 48 hours on 833 postoperative patients to record oxygen desaturations below 90% in 42% for at least 30 min, and a remarkable 27% of patients for at least two hours. 47 The high frequency of hypoxaemia was recorded despite SO use in 66% of patients. Complications were not measured, but given the commonality of hypoxaemia, the authors reasonably commented, ‘It remains possible that hypoxaemia is largely an indicator of underlying disease rather than a treatable mechanism’.
Oxygen delivery
In the resting healthy state there is an excess of oxygen delivery to the periphery such that blood returns to the right side of the heart with only 25% of oxygen content extracted. 14 As arterial oxygen saturation (SaO2) falls and oxygen delivery with it, the extraction percentage increases to maintain tissue oxygen delivery. For this reason, critical oxygen delivery, determined as the point when the lactate rises due to additional anaerobic glycolysis, is generally considered to occur in the healthy person at approximately an SaO2 of less than 55%. 14
Healthy human volunteers undergoing laboratory research on hypoxaemia have recorded SpO2 as low as 45% and periods of SpO2 50%–70% for 10–30 min without acidosis or cardiovascular compromise. 48 High altitude mountain climbers, after a period to acclimatise to altitude sickness, experience lengthy and considerable hypoxaemia, with prolonged SpO2 in the 40% range not infrequent. 48 Elite breath-hold divers frequently record similarly low SaO2 of around 50% without ill effect. 48 These findings challenge the common misconceptions of harm from hypoxaemia such as an SaO2 of 70% in the absence of decreased blood flow leading to brain damage. 48
Repeated short exposure to hypoxaemia or ischaemia can realise benefits from preconditioning allowing for subsequent more profound hypoxaemia and ischaemia to be tolerated. Mediators thought to be important in hypoxaemia preconditioning include hypoxia-inducible factor 1 (HIF-1). HIF-1 increases in hypoxic conditions and acts to up-regulate genes which promote survival in low oxygen conditions. 49
Hypoventilation, hypoxaemia and the impact of SO
Under the influence of opioids, alveolar ventilation declines with resultant hypercarbia and hypoxaemia. When breathing room air, the SpO2 can be predicted to fall below 90% once alveolar ventilation is reduced by approximately one-third of normal
50
(Figure 5). As alveolar ventilation decreases even further to 2 l/min, approximately half normal, the alveolar partial pressure of carbon dioxide (P
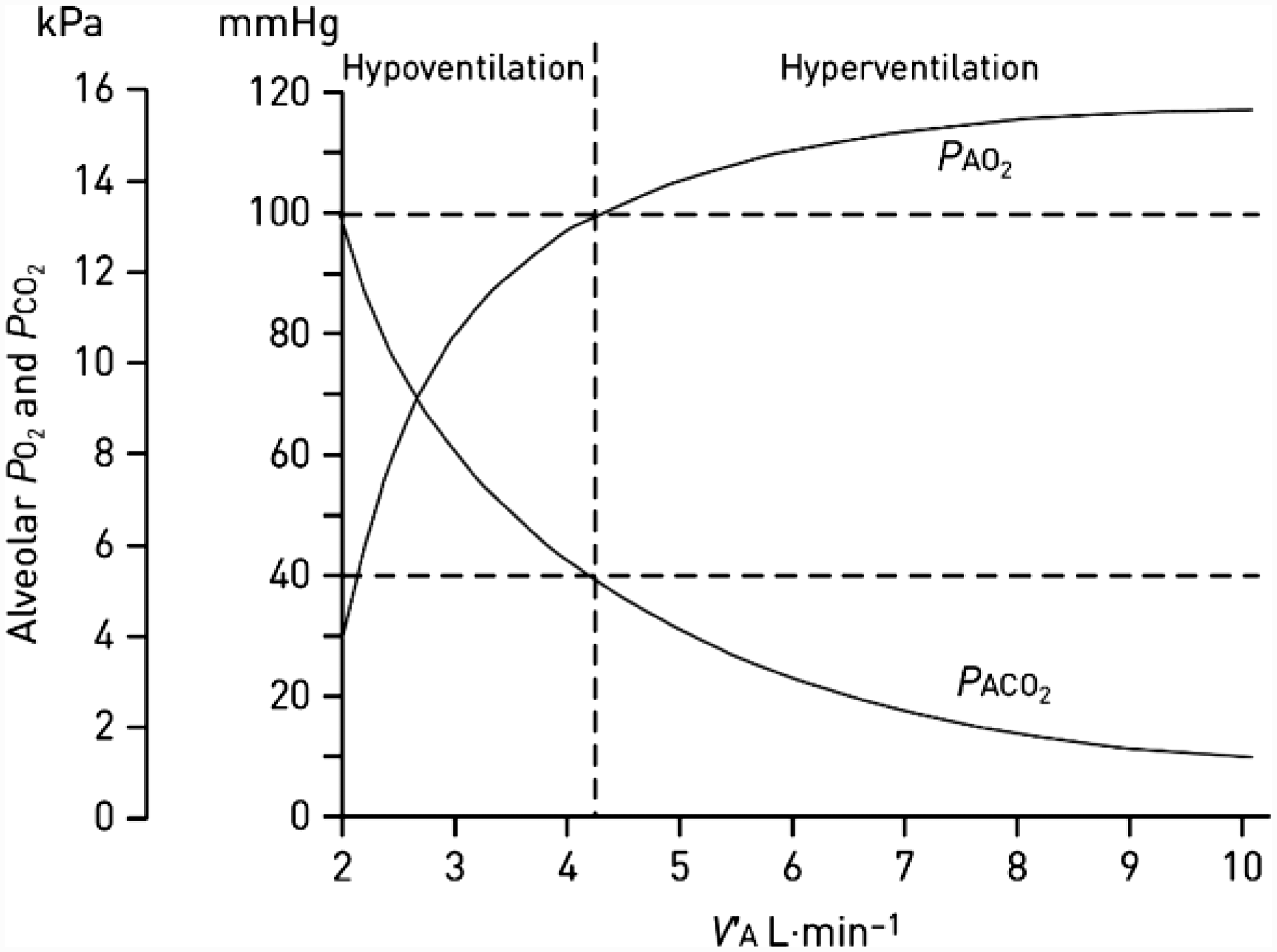
Relationship between alveolar ventilation and alveolar PO2 and PCO2 Reproduced, with permission, from European Respiratory Journal.
Supplemental oxygen at 2–6 l/min via nasal prongs or a Hudson mask elevates the FiO2 to at least the required 0.3 and is therefore capable of normalising SpO2 in the presence of significant alveolar hypoventilation. 52 Consequently when SO is in use, a normal SpO2 should not be used to judge the adequacy of ventilation. 50 At a certain point alveolar ventilation is so impaired that delivery of oxygen into the alveoli, even with supplementation, is insufficient to match consumption and only then will SpO2 fall. With reserves exhausted, desaturation occurs very rapidly.
Lack of awareness of the ability of SO to normalise SpO2 in the presence of significant hypoventilation is a frequent contributor to adverse outcomes in the unsuspected OIVI patient. Typically, ward staff are lulled into a false sense of security by a normal SpO2 even when witnessing patient sedation, only being alerted to a problem when apnoea finally intervenes and a precipitous and profound desaturation occurs. 53 It is for this reason the routine use of SO with opioids is not recommended. 54 SO is applied if desaturations are identified on room air as supportive therapy while definitive diagnosis and management of the cause of hypoxaemia is undertaken.
Effect of SO on other causes of hypoxaemia
The two most common causes of postoperative new onset hypoxaemia are hypoventilation or venous admixture. 50 Pathological causes of venous admixture, otherwise referred to as shunt, include: pulmonary embolus, pulmonary oedema, sepsis, pneumonia, and atelectasis.50,51 Hypoxaemia due to hypoventilation, as discussed, is readily rectified by a small increase in the FiO2. Venous admixture in comparison responds poorly to oxygen supplementation. 50 The FiO2 increase reasonably achieved by SO to approximately 0.4 is capable of only normalising the PaO2 of mild (up to 15%) venous admixture.51,52 An FiO2 requirement greater than 0.4 is hence an indicator for senior clinician review, and above 0.5 for an intensive care review. 55
SO is ineffective at correcting hypoxaemia in the critically unwell, and if being applied in the context of hypoxaemia due to mild venous admixture can delay the diagnosis until the disease process is far more advanced, and the critical importance of early detection and management is missed. 53
Overriding of hypoxic ventilatory drive
Although opioids depress the HVR, the underlying hypoxic drive is intact and current evidence indicates this remains crucial to maintaining alveolar ventilation. This was demonstrated by Niesters et al. who administered a short remifentanil infusion to 20 volunteers first on room air and then with oxygen supplementation to 50%. 56 On room air, 10% of volunteers became apnoeic but this increased to 50% with oxygen supplementation. The clinical implication of these findings is that in addition to masking hypoventilation-induced hypoxaemia, SO will inadvertently worsen OIVI. 54
Mandatory use of supplemental oxygen
In the time course of typical OIVI, prior to the point of apnoea there is a short period in which SO will prevent the breaching of the SaO2 below the critical oxygen delivery point of 55%. This equates with a PaO2 of 30 mmHg, and at this extent of RD a commensurate deep level of opioid-induced sedation would be expected. Mandatory use of SO as a mechanism to prevent life-threatening hypoxaemia due to concerns about ward staff inability to detect the deteriorating patient is counter to good patient care. If deeply sedated patients are unable to be detected by ward staff, the clinician, acting as a patient advocate, may prefer to defer providing opioids in this setting. The finding of one in six critical OIVI-related events occurring while SO is in use attests to the danger of relying on SO as a fail-safe. 41
Oxygen use guidelines
Guidelines for oxygen use are summarised in Table 1.54,55,57 These recommend a minimum SpO2 to provide a margin of safety ensuring tissue oxygen demands are met by generally recommending commencing SO at a threshold SpO2 of 92% or less. A recent randomised study into oxygen use in adult respiratory distress syndrome identified worsened 90-day mortality and the onset of mesenteric ischaemia in patients in whom a lower SpO2 of 88%–92% was targeted, underlying the importance of adhering to these guidelines particularly in at-risk populations. 58
Postoperative oxygen therapy recommendations applicable to all routes of opioid delivery.

OIVI: opioid-induced ventilatory impairment; SpO2: peripheral oxygen saturation.
There is increasing concern about the deleterious effects of hyperoxia in many disease states including stroke and myocardial infarction. 57 Therefore, there is a recommended target upper limit with oxygen therapy of SpO2 of 96%. 57
Obstructive sleep apnoea and opioid-induced ventilatory impairment
Patients with OSA feature in discussions on OIVI and are a cause of much clinician consternation.
Obstructive sleep apnoea pathophysiology
OSA is a complex disease with heterogeneous subgroups, all however sharing some commonalities. 59 Upper airway tone decreases especially in stages R (REM) and the deep sleep stages of N3 and N4, leading to obstructed breathing with either hypopnoea, or apnoea with the potential for rapid desaturation.
Termination of obstruction is dependent on a neural-hormonal response involving relay of chemoreceptors and pharyngeal pressure sensors to bring about airway opening, which is usually accompanied by a cortical arousal from sleep, one that does not necessarily result in a return of full wakefulness.60,61 After reopening of the airway, obstruction-induced hypercarbia causes hyperventilation and a rapid restoration of SaO2. When arousal is cortically mediated, a ventilatory overshoot is more likely at this stage which eliminates the stimuli required to keep the airway open and this has a greater destabilising effect on breathing. 61 Deeper sleep stages eventually return and, in this way, cycling of obstructions terminated by airway openings and/or arousal continues. These cycles are reflected in the apnoea–hypopnoea index (AHI) in which the number of apnoeas and hypopnoeas lasting more than ten seconds are counted over an hour. Loop gain is a term often used in OSA and refers to the stability and responsiveness of ventilatory control; high loop gain states are unstable but also very responsive. 62
OSA sufferers have been described as being in a constant state of arousal-dependent survival. 53 Factors believed to affect arousal adversely include surgery, pain, sedatives, anaesthetic agents, and opioids (discussed further in a following section). 63 For many years, cortical arousal has been believed to be crucial to the effective termination of obstruction; so much so that administration of sedatives (which can impair cortical arousal) was believed to be anathema to good care of the OSA patient; recent research has challenged this long-held belief. 61
In the event of cortical arousal failure upper airway patency may not be restored. The ensuing sustained hypoxaemia leads to cerebral hypoxia further preventing cortical arousal. Hypoxaemia then worsens and death eventuates. In the period between obstruction and death, CO2 continues to be produced by the tissues but is not eliminated. A blood gas taken in the immediate post-morbid phase typically shows a markedly elevated PaCO2 with acidosis for this reason, leading to many of these OSA deaths being misdiagnosed as a typical OIVI death. 53 Such deaths are in contrast unheralded, being devoid of forewarning with the sedation or sustained hypoxaemia of typical OIVI. Indeed patients are usually wide awake with vital signs recorded in the normal range preceding the event. 53 Sudden nocturnal deaths reported in OSA sufferers peak between midnight and 0600 hours and have been referred to as ‘found-dead-in-bed syndrome’, and require appreciation and distinction from deaths of typical OIVI.53,64–66
OSA is a significant disease burden in the general population, with diagnosis peaking at 8% for middle-aged Australian men. 67 There is significant concern that the majority of OSA sufferers are undiagnosed. An Australian study revealed 49% of a random sample of middle-aged men qualified for a diagnosis of OSA (AHI >10) by polysomnography. 68 Preoperative testing of a mixed surgical population with polysomnography similarly identified disconcertingly high rates of previously undiagnosed OSA; mild (AHI >5–15) in 30%, moderate (AHI >15–30) in 21%, and severe (AHI >30) in 18%. 69
High arousal thresholds, and hence greater vulnerability to arousal failure, are commonly observed in moderate and severe OSA. 70 With the high rates of OSA presentation it can seem logical to focus attention on these seemingly higher-risk patients with moderate and severe OSA. However, the AHI cannot be relied on to predict all events, as even mild OSA was a feature of 18% of OSA critical events in one series. 46 So while the use of OSA screening tools such as STOP-Bang are widely advocated to indicate likely OSA severity, consensus on how best to apply the findings is lacking.69,70
Clinical manifestations
The relationship between OSA and OIVI is complicated and contentious. Studies on OIVI risk factors may unintentionally flag OSA sufferers due to the commonality of the defining respiratory endpoints present for both OSA and OIVI. 34 Apnoeas, low respiratory rates and even desaturations (nadirs of 85%–90%), the ‘baseline’ features of OSA, can be misinterpreted as diagnostic of OIVI leading to potential overemphasis of the OIVI risk carried by OSA. 9 A recent meta-analysis of OIVI concluded that OSA was a risk factor, with an odds ratio of 1.4. 9 This analysis relied on naloxone use predominantly in the postoperative care unit (PACU) to define OIVI. In one of the included studies, 98.3% of patients received only a single dose of naloxone in the PACU—indicating that administration was likely for a transient event having causality not limited solely to opioids. 71 Further, the timing immediately after anaesthesia belies the contribution of sedating agents in the pathogenesis of OSA events.
Case series of OIVI critical events in a similar vein report OSA is an association. 64 The use of the terms ‘association’ and ‘risk’ indicates likelihood, and misinterpreting this as proof of causality should be avoided.
Published case series of postoperative critical events in known or suspected OSA patients can aid with identifying which factors may provoke a destabilisation of the precarious obstruction/arousal balance. In two published case series of 66 and 60 OSA events, respectively, the only unifying factor was that all were following surgery.46,72 In the one series, 25% were anaesthesia related, and of the remaining 75% related to opioid use, morphine intravenous equivalent was less than 10 mg/day in 81%. 72 Consistent across the findings were most commonly surgery and the associated pain, followed by sedative anaesthetic agents, and opioids. The events while perhaps being opioid related are not pathognomonic of typical OIVI with sedation, and are more in keeping with the sudden arousal failure type of OSA events.
Other features of these series also aid in identifying the pathological process causing harm. The findings related to the use of monitoring are suggestive of the pathological process being of the sudden OSA type. 46 Continuous bedside SpO2 was in use in 14% while continuous centrally monitored SpO2 was similar at 17% of events. The rate of death or brain damage was noticeably different depending on the continuous monitoring type, 78% if bedside versus a much lower 27% if centrally monitored. Bedside monitoring is frequently unattended whereas central monitoring allows for alarms to be noted earlier and response initiated rapidly. Further, despite 47% of events being directly witnessed, death or brain damage occurred in 37% of these events. These findings point clearly to a sudden and dramatic deterioration in the patient as seen with obstruction rather than the slow decline of typical OIVI with sedation.
Opioid studies in obstructive sleep apnoea
Three studies so far have directly studied the effect of opioids on OSA. Bernards et al. monitored ten OSA patients during sleep while administering a remifentanil infusion at analgesic dosing rates. 22 Compared to controls and in contrast to previously held beliefs, remifentanil halved the mean number of obstructive apnoeas, reducing the number of obstructive events in nearly all subjects. Somewhat unexpectedly the central apnoea index was affected by remifentanil; while it was unchanged in six subjects, in four there was a marked increase with the baseline mean of 0.8 rising to 43 per hour. Central apnoeas are identified by the absence of respiratory muscle activity, indicating they are not due to obstruction, and can be a feature of opioid administration in non-OSA patients. 39 Remifentanil notably increased the time spent in stage N1 sleep (the stage of dozing when arousal can easily occur) and virtually eliminated time in REM sleep—the time of greatest loss of pharyngeal muscle tone and characteristically when the longest periods of apnoea are seen in OSA.22,73 It was these changes in sleep architecture which likely contributed to the reduction in obstructive apnoeas with remifentanil. The findings of this study do suggest some OSA sufferers are vulnerable to the effects of opioids, but not as expected due to worsening obstruction but rather by provoking central apnoeas.
Rowsell et al., utilising a crossover design, studied 60 OSA sufferers after administering SR morphine 40 mg prior to sleep. 63 As seen in the preceding study a separation in response subtypes was evident. For the primary endpoint of sleep time with a SpO2 less than 90% (T90): morphine resulted in 50% having a small worsening (of less than 5%), 20% had no change, but in 27% an improvement was seen. Although some patients spent more time with an SpO2 less than 90%, their SpO2 nadir was essentially unchanged at 86% versus 87%. Variability in the effect of morphine may have been genetically related as a distinct separation of allelic subtypes for the opioid receptor gene OPRM1 was found between the five worst and five best responders to morphine. There was the expected increase in VRT and decrease in HCVR with morphine, leading the authors to propose the mechanism for improvement in some OSA patients was by advantageously dampening the chemoreceptor responsiveness in those with a high loop gain. Reducing the loop gain for these patients reduces the frequency of chemoreceptor-driven cortical arousals and by doing so allows the breathing pattern to stabilise. In others in which the T90 values worsened, it was thought likely to be due to the central neurodepressant opioid effect predominating to prevent the cortical arousal needed for these patients to reopen the upper airway.
Martins et al., in a follow-on study to the previously described study, found upper airway collapsibility measured using pharyngeal pressure monitoring was unaffected by morphine. 23 Loop gain was found to be reduced after morphine administration, supporting the concept that this is a possible mechanism for improved oxygenation in some OSA patients by stabilising breathing.
In summary, of primary importance is that the research on opioids in OSA demonstrates upper airway obstructions are not worsened. Opioids can be beneficial in some patients: first, there are advantageous changes in sleep architecture which can reduce the frequency of obstructions; and second, central actions of the opioids can reduce loop gain or reduce cortical arousal-associated ventilatory overshoot, both acting to stabilise breathing and allow oxygenation to improve. In others, and this is likely to be dose related, a deleterious neurodepressant effect may predominate by at least one and possibly all three mechanisms: impaired rhythm generation to induce central apnoea; chemoreceptor impairment of ventilatory drive; or cortically preventing arousals. 23
Mortality
The effect of OSA on postoperative mortality is reported in 14 large retrospective or prospective studies.74,75 In nine studies, OSA had no significant impact on mortality; in three OSA was associated with a reduction in mortality and in only two did OSA worsen mortality. The intermittent hypoxaemia by preconditioning seen in OSA has been proposed as the mechanism for the association with a reduction in severity of myocardial infarction and ischaemic brain injury, and may similarly provide protection against OIVI-related postoperative mortality.76–78
Postoperative OSA-related adverse outcomes are reported in two case series. Subramani et al. reviewed 60 cases predominantly from case reports spanning 1986 to 2016. 72 Twelve of the 27 deaths reported had a known diagnosis of OSA. Four deaths occurred in the PACU, one in the intensive therapy unit and the remaining 22 on the ward while receiving opioids. Men accounted for 62% with a mean age of 46 years.
Bolden et al. reported on 66 registry cases of critical events occurring over a timespan of two decades. 46 Opioids had been administered in the previous 24 hours in all but one case, at a median oral morphine equivalent of 120 mg/day. Deaths were categorised along with brain damage and occurred in 65%. All events occurred within 3.5 days of anaesthesia completion, with 76% occurring within the first 24 hours. Of deaths and brain damage, 28% occurred post-discharge in the home. The mean age was 53 years and 65% occurred in men.
The presence of OSA reduces life expectancy although a significant increase in mortality is limited to men under the age of 50 years.79,80 Patients with these demographic features can be expected to be overrepresented, as they are, in reported postoperative mortality cases.
Case series of OSA critical events provide crucial details on circumstances allowing the opportunity for lessons to be learnt. Misinterpretation of the impact of OSA on mortality due to confirmation bias may occur if these small case series are viewed in isolation and without acknowledging the important findings of large comparative studies as well as the known natural history of the OSA disease state.
Recommendations
Current research undoubtedly adds additional complexity to our decision-making when caring for the postoperative OSA patient. Notwithstanding this and as we cannot be certain of an individual OSA patient’s response to opioids, opioid-sparing is an appropriate universal OIVI prevention strategy, irrespective of the presence of reported risk factors.
Postoperative management of OSA patients based on published recommendations, current evidence and knowledge are summarised in Table 2.70,81,82
Recommended management of the postoperative obstructive sleep apnoea patient receiving opioids.
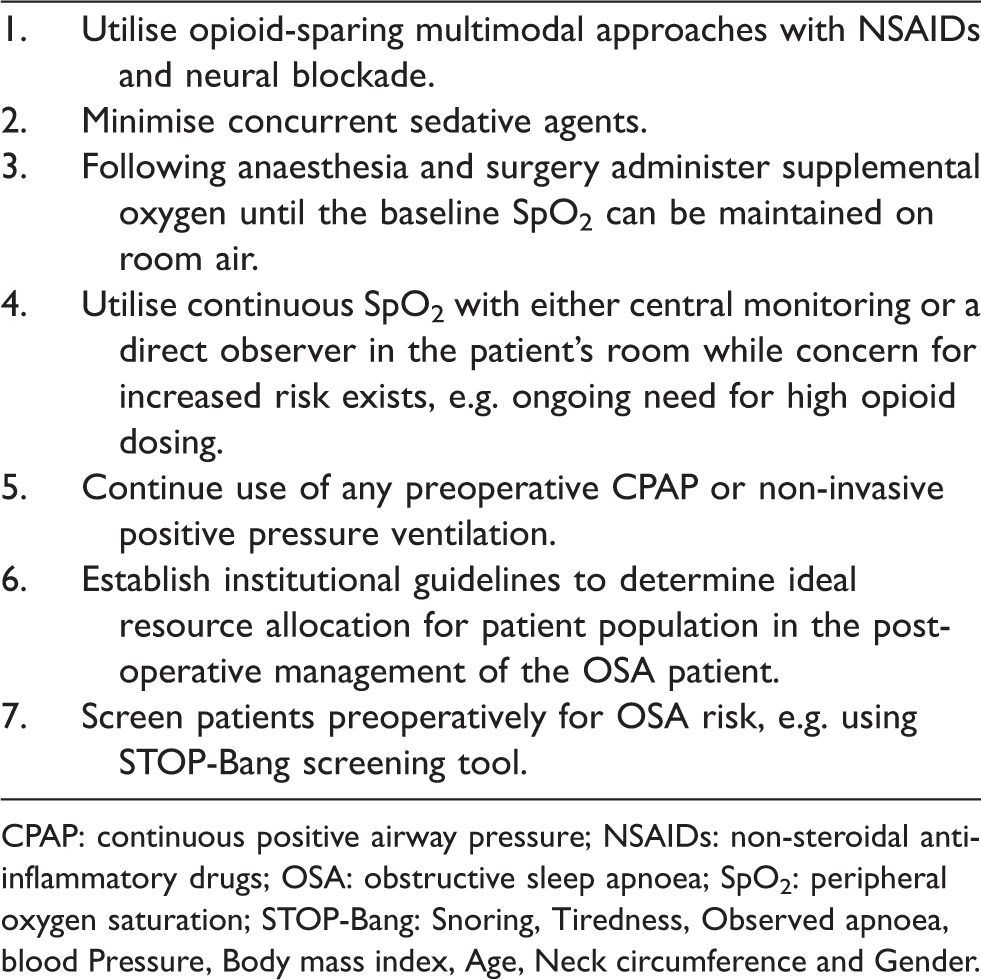
CPAP: continuous positive airway pressure; NSAIDs: non-steroidal anti-inflammatory drugs; OSA: obstructive sleep apnoea; SpO2: peripheral oxygen saturation; STOP-Bang: Snoring, Tiredness, Observed apnoea, blood Pressure, Body mass index, Age, Neck circumference and Gender.
Utility of monitoring and clinical signs
The major challenge for monitoring is enabling the early detection of deterioration when successful reversal is most likely and at the same time avoiding false positives. All single measurable thresholds for detecting OIVI prior to a critical event fail to reach 100% reliability either because they are prone to error, or the thresholds chosen lack confirmed sensitivity and specificity. 53 For this reason, no single threshold should be over-relied on, and instead optimal combinations for the clinical circumstance utilised with endorsement by institutional guidelines recommended.
Respiratory rate
Opioids affect respiratory centre rhythm generation resulting in a rate reduction that may eventually reach a critically low point indicating the presence of OIVI. To be used as an effective, reliable warning mechanism, the minimum respiratory rate threshold needs to accommodate a wide range of normal or safe circumstances without triggering false alarms. Respiratory rate normally decreases during sleep and can even become irregular during REM with the onset of apnoeas and so the patient safely receiving opioid analgesia during sleep may readily breach a lower-limit respiratory rate threshold if set too high. 13 Hence the lowest limit threshold of five breaths per minute is commonly utilised clinically to minimise false alarms. However, breaching this lower-limit threshold is a late feature of OIVI with sedation already advanced, particularly when other sedatives such as benzodiazepines are administered concomitantly. 83 Bedside measurement of the respiratory rate by ward staff can frequently fail to detect periods of apnoea possibly due to verbal and tactile stimulation leading to wakening. 84 The use of chest plethysmography respiratory rate measurement using electrocardiographic electrodes can circumvent such errors but introduces the problem of the excessive medicalisation of every patient receiving an opioid. 83 Episodes of apnoea, particularly as seen in OSA and CSA, are common and will be detected by oversensitive monitoring and do not necessarily need intervention.33,84 Respiratory rate measurement has been reported to be the vital sign most frequently omitted by ward staff. 85 For all these reasons, the respiratory rate is commonly combined with a sedation score to determine response to an opioid. 86
Sedation scores
The sedating action of opioids limits the upper range of safe opioid dosing. A five-point scoring system based on the Pasero opioid-induced sedation scale (POSS) is commonly utilised in Australian practice. 87 The POSS was developed for opioid prescribing and has greater reliability compared with alternative scales having a larger number of possible scores. 86 The POSS allows for sleep (S) to be recorded. The scores from lowest to highest originally described were of: (1) awake; (2) slightly drowsy easily aroused; (3) frequently drowsy, arousable; and (4) somnolent, minimal or no response to verbal and physical stimulation.
Assessment of sedation may be problematic during sleep because of ward staff reluctance and patient resistance to being woken every one to two hours, leading to OIVI potentially being unrecognised during sleeping hours.
In the analysis of OIVI-related adverse events of Lee et al., preceding sedation was identified in only 62%. 41 This reflected several clinical problems, including failure of ward staff to recognise the significance of sedation, and that not all cases were likely to be preceded by the sedation of typical OIVI in the circumstance of an OSA patient with arousal failure.
Pulse oximetry
SpO2 measurement is a useful tool to detect OIVI in the absence of SO use. Once SO is applied, only the very advanced stages of OIVI are detected, so other measures of OIVI, most commonly sedation and respiratory rate, acquire greater importance. The ideal lower threshold for SpO2 alarm triggering is not known. 53 The use of 90% is largely empirical and many OSA sufferers will breach this threshold without necessarily requiring intervention. Ideally thresholds should be patient specific, although the additional time demands of such an approach in a busy environment can add clinical complexity.
Intermittent SpO2 measurements conducted by ward staff have the benefit of direct observation enabling immediate recognition of desaturation and an opportunity for timely intervention; however, some desaturation events may be missed. Continuous SpO2 is advocated for detecting desaturations frequently missed by intermittent checks. 47 Evidence of benefit from continuous oximetry is limited to reducing harm only when combined with relay to a central monitoring station or a pager alert to ward staff.31,46,88 Routine use of centrally relayed oximetry does, however, present other challenges including alarm fatigue and how best to optimise resource allocation given the cost of the equipment, need for additional training of staff, and greater demand on medical response teams. 89
Capnography
Capnography is in the early stages of clinical application in this setting and while it has been recommended to improve the detection of OIVI when SO is in use, research to show benefit is pending. 83 The main challenges to be overcome are unreliability driven by impracticalities through physical restriction, patient intolerance of tubing, and alarm fatigue from false positive alarm triggers due to interruptions from removal or misplacement of sampling lines. 84 The findings of the PRODIGY study in which 1335 patients were monitored with continuous oximetry and capnography provides useful insights into these clinical challenges. 33 The average number of any alarm trigger per patient was 4.3 over a median duration of monitoring of 24 hours. An apnoea alarm was triggered in 97% of patients without the need for intervention in any. Although the allure of capnography is the ability to detect the hypercarbia of OIVI by way of an elevated EtCO2 there were no incidents of breaching the upper EtCO2 threshold. Capnography detected the one patient requiring intervention for a low respiration rate. Most interventions were for hypoxaemia detected by oximetry.
Transcutaneous CO2 monitors are available predominantly in the neonatal setting, with clinical utility limited by technical challenges including regular need for calibration and re-siting. 90
Conclusions
‘Opioid-induced ventilatory impairment’ (OIVI) is the preferred term for the major cause of death from opioids as it emphasises the broader failure of alveolar ventilation and the contribution of opioid-induced sedation. Use of the alternative term ‘opioid-induced respiratory depression’ (OIRD) can carry with it an insinuation that concern need only be for depressed respiration rate.
Risks and associations for OIVI are reported but do not encompass all who may experience harm. Care is required when prescribing both for the opioid naive as they have no previous exposure by which opioid responsiveness can be judged, and for the opioid tolerant as they remain vulnerable to OIVI when acute opioid doses are upsized to provide adequate analgesia.
SO should be used with caution recognising the risks posed by rectifying the hypoxaemia of opioid-induced hypoventilation up to the advanced stages and over-riding hypoxic ventilatory drive to worsen OIVI. Postoperative hypoxaemia is also caused by venous admixture (shunt) and application of SO in the mild stages may delay the diagnosis of cause.
The presence of OSA should not carry greater concern for typical OIVI but rather for rare unpredictable sudden death due to failure of arousal and/or release of upper airway obstruction which may be exacerbated by opioids.
Pulse oximetry can be utilised as a measure to detect early OIVI when intervention has the greatest chance of success only if SO is not being applied. Staff education about this aspect is paramount. If continuous oximetry is chosen it should be centrally monitored by skilled personnel able to respond rapidly to provide any benefit.
As there is no absolute failsafe to eliminate OIVI deaths, the clinician has, in essence, a duty of care to aim to minimise risk through optimising all the possible opioid-sparing multimodal strategies, thereby reducing opioid exposure.