Abstract
Lung transplantation is limited by a lack of suitable lung donors. In Australia, the national donation organisation (DonateLife) has taken a major role in optimising organ donor identification. However, the potential outside the DonateLife network hospitals remains uncertain. We aimed to create a prediction model for lung donation within the DonateLife network and estimate the untapped lung donors outside of the DonateLife network. We reviewed all deaths in the state of Victoria’s intensive care units using a prospectively collected population-based intensive care unit database linked to organ donation records. A logistic regression model derived using patient-level data was developed to characterise the lung donors within DonateLife network hospitals. Consequently, we estimated the expected number of lung donors in Victorian hospitals outside the DonateLife network and compared the actual number. Between 2014 and 2018, 291 lung donations occurred from 8043 intensive care unit deaths in DonateLife hospitals, while only three lung donations occurred from 1373 ICU deaths in non-DonateLife hospitals. Age, sex, postoperative admission, sepsis, neurological disease, trauma, chronic respiratory disease, lung oxygenation and serum creatinine were factors independently associated with lung donation. A highly discriminatory prediction model with area under the receiver operator characteristic curve of 0.91 was developed and accurately estimated the number of lung donors. Applying the model to non-DonateLife hospital data predicted only an additional five lung donors. This prediction model revealed few additional lung donor opportunities outside the DonateLife network, and the necessity of alternative and novel strategies for lung donation. A donor prediction model could provide a useful benchmarking tool to explore organ donation potential across different jurisdictions, hospitals and transplanting centres.
Introduction
Lung transplantation (LTx) is a crucial life-saving treatment for patients with end-stage lung diseases. In Australia, LTx numbers have increased over the past decade, not only because of developments in transplantation techniques and strategies, but also improved organ donor identification, referral processes, and donor management.1–3 In 2008, the Australian National Organ Donation Reform programme established the DonateLife network of hospitals. These hospitals were predominantly public metropolitan and tertiary hospitals considered to have the greatest capacity for organ donation, and a programme of onsite medical and nursing donation specialist staff, dedicated educational programmes, and auditing of all potential and actual donors was instituted. 4 In 2014, the Australian Electronic Donor Record was introduced to facilitate operational aspects of organ donation throughout Australia, and to record referral, organ offer, and acceptance information.
As a result, overall organ donation rates nationwide have increased from 11.4 per million population (PMP) in 2009 to 21.6 PMP in 2019. 5 In the state of Victoria, donation strategies have been particularly successful, with an overall organ donor rate of 24.6 PMP in 2019. In combination with an established proactive approach to the use of extended criteria donor lungs, this has translated to LTx rates that are among the world’s highest at 11.3 LTx PMP in 2019. Notwithstanding this success, the number of recipients waiting for LTx still continues to exceed the number of lung donors, and it is therefore necessary to explore all potentially transplantable donor lungs. Although it has been reported that few missed potential donors are seen inside the DonateLife network, 6 the size of the pool of untapped donor opportunities outside the network remains uncertain.7,8
All intensive care units (ICUs) in Victoria also contribute individual de-identified patient data to the Australian and New Zealand Intensive Care Society (ANZICS) adult patient database (APD), one of four clinical quality registries run by the ANZICS Centre for Outcome and Resource Evaluation (CORE).
We hypothesised that there is a significant pool of donors potentially suitable for LTx in non-DonateLife hospitals. The current study aimed to estimate the expected number of lung donors in Victorian hospitals outside the DonateLife network, through linkage of patient data on deaths at all hospitals listed in the ANZICS APD to their donation outcomes recorded in the DonateLife electronic donor record.
Materials and methods
Study design
We retrospectively reviewed all deaths in ICUs in the state of Victoria between April 2014 and December 2018 using two clinical databases.
Setting and data source
De-identified data on all deceased patients (including actual lung donors) after ICU admission were extracted from the following data sources:
The ANZICS APD covers all patients admitted to Victorian adult ICUs (which includes 21 DonateLife network hospitals and 22 non-DonateLife hospitals during the study period). This high-quality binational database records patient admission diagnosis, chronic health information, physiological and biochemical variables within the first 24 hours of admission as well as ICU and hospital outcomes. The Glasgow Coma Scale score (GCS) used is the one prior to sedation before ICU admission or during the first 24 hours. Data are entered by data collectors in participating ICUs and sent to the ANZICS CORE where they undergo both manual and automated validation checks. No information about consent discussions, assessments of suitability for donation or outcome of donation is included in this database. The electronic donor record has detailed demographic, historical and contemporary data on actual and potential organ donors worked up for donation, including consecutive donor ICU management, as well as the donation outcomes.
The study was approved by the Alfred Hospital ethics committee (project no. 28/19) and the Australian Red Cross Blood Service ethics committee. Access to ANZICS APD was granted by the ANZICS CORE management committee in accordance with standing protocols.
Data linkage
The study group of interest was lung donors defined as patients for whom the retrieval procedure was performed and the lung graft transplanted. To identify actual lung donors in ANZICS APD, matching of individual patient records between ANZICS APD and the electronic donor record was performed by a probabilistic linkage. 9 As both databases contained only de-identified information and no unique personal identifiers were available, the linkage used five indirect identifiers: (a) hospital identification number; (b) age; (c) sex; (d) date of death; and (e) date of admission, which were required to be an exact match. We allowed a ±one-day window for date to address potential data inaccuracy. If a lung donor in the electronic donor record matched multiple patient records in ANZICS APD, the true match was then determined based on the patient’s diagnosis. The datasets were merged and linked by a single researcher (SO) and additionally audited by two researchers (SO, DVP) before analyses.
Management of datasets and prediction model
To create a prediction model for lung donation, we initially only examined data from hospitals inside the DonateLife network, which was randomly split into mutually exclusive derivation (67%) and validation (33%) datasets. The derivation dataset was used for the construction of prediction models, whereas the validation dataset was used to test the performance of the developed models. The final prediction model was then applied to deaths at hospitals outside the DonateLife network, which allowed comparison of the expected number of lung donors with the actual number of lung donors.
Statistical analysis
Univariate comparisons of lung donors and non–lung donors were performed. Continuous variables were compared using Wilcoxon rank sum tests and reported as medians (interquartile range). Categorical data were compared using chi-square tests and reported as number (
Co-linear variables were explored using a correlation matrix and then assessed using univariate logistic regression to select the one with greater discrimination for inclusion in the prediction model. Continuous variables were converted to categorical variables dichotomised at relevant cut-off points, with an additional category for missing value.
The prediction model was developed by multivariate logistic regression with both stepwise selection and backward elimination techniques derived using all patient-level variables with a univariate
Finally, if the prediction model derived was reliable (i.e. good or excellent) in the validation dataset, we applied the prediction model to the non-DonateLife hospitals’ dataset, to estimate the untapped lung donors outside of the DonateLife network. All analyses were performed using R version 3.6.1 (R Foundation for Statistical Computing, Vienna, Austria). A two-sided
Results
Overall, there were 14,358 patients who died after an admission to an ICU in Victoria recorded in the ANZICS APD, of whom 9416 died in the ICU. Among them, 8043 patients died in the DonateLife network hospitals and 1373 patients died in non-DonateLife hospitals (Figure 1). The characteristics of the deceased patients inside and outside the DonateLife network are summarised in the Supplementary Appendix (Supplementary Table 1). Patients in non-DonateLife hospitals tended to be older and less likely to be admitted to the ICU due to cardiac or neurological disease or trauma. The results of data linkage are shown in the Supplementary Appendix (Supplementary Table 2). We identified 291 actual lung donors inside the DonateLife network and only three outside of the network. The summary of numbers of deaths and donors by hospital type is shown in Table 1.
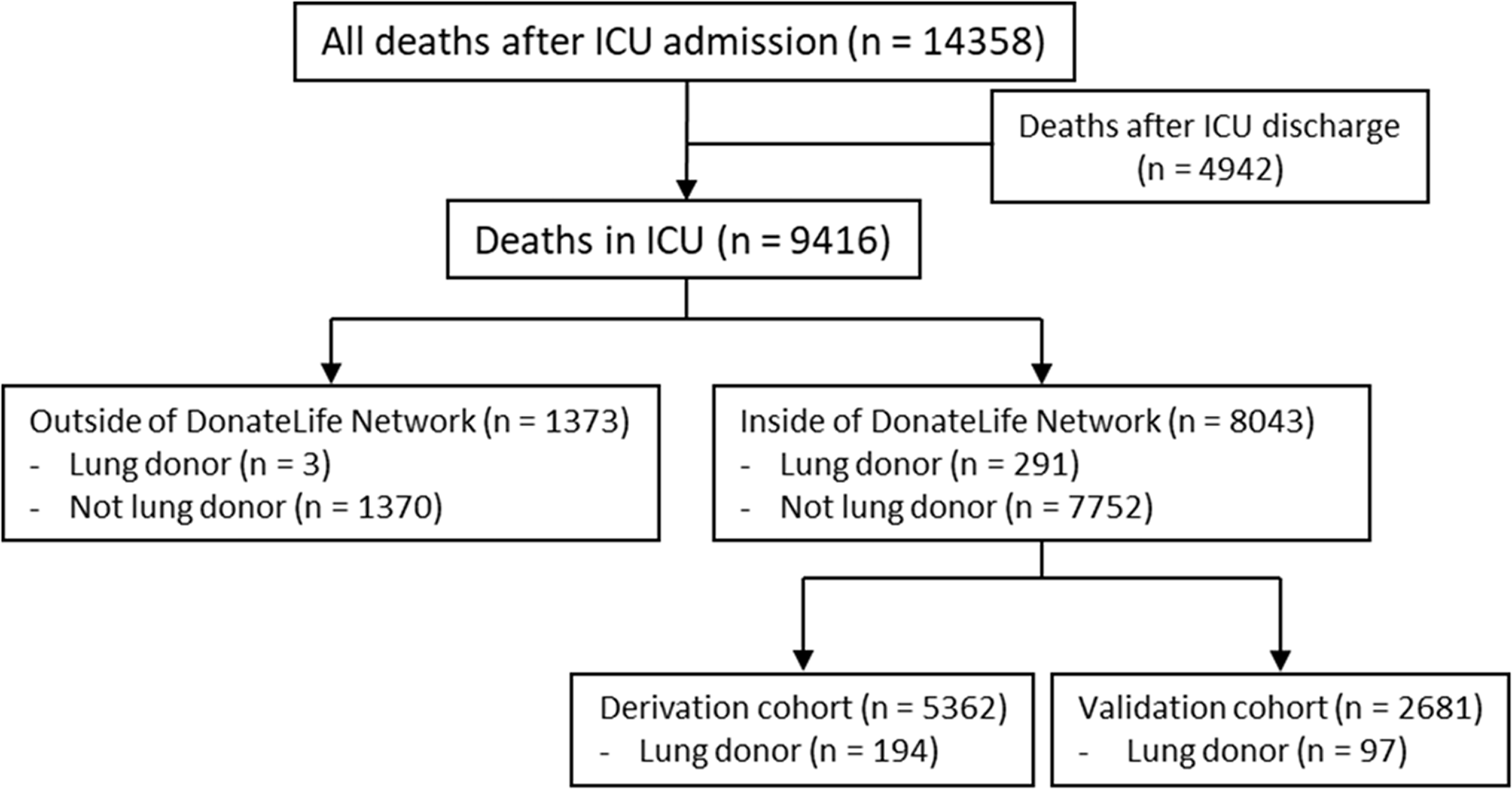
Flow of participants through the study to estimate the expected number of lung donors. ICU: intensive care unit.
Summary of numbers of deaths and lung donors by hospital type.
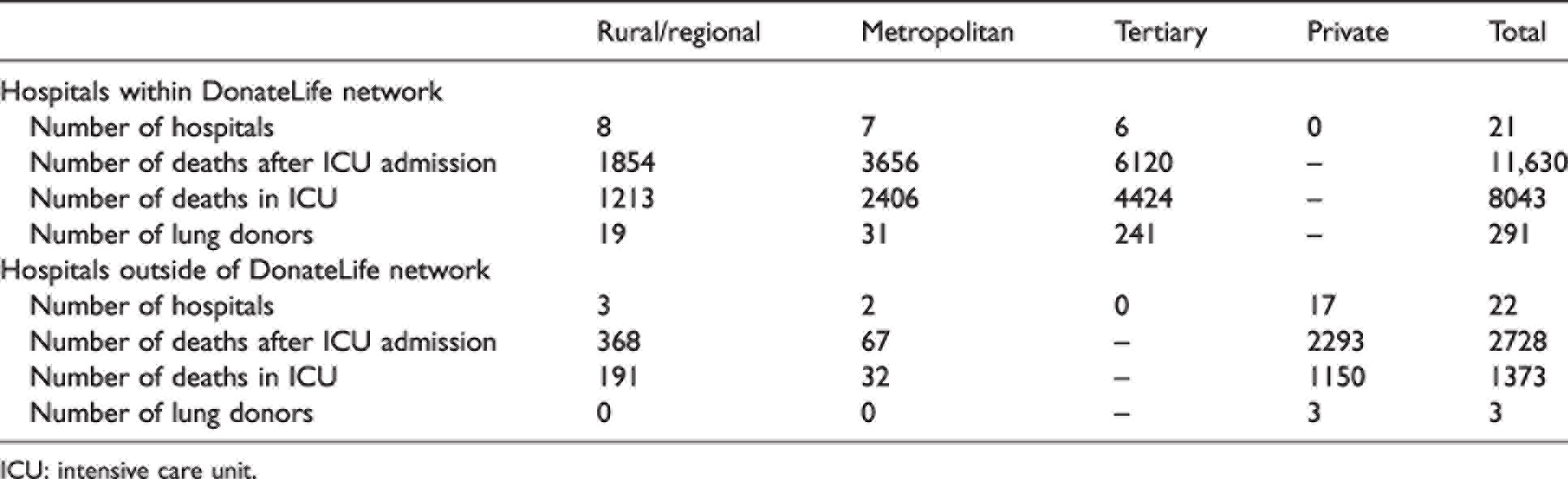
ICU: intensive care unit.
Table 2 shows the characteristics of patients who became lung donors compared to those who did not in DonateLife network hospitals. Lung donors were younger and more often women, were more likely to be admitted to the ICU due to neurological disease or trauma, had a lower prevalence of chronic medical conditions (cardiovascular, lung, liver, and renal) and a higher ratio of arterial partial pressure of oxygen to fraction of inspired oxygen (P/F ratio).
Comparison of lung donors to non-lung donors.
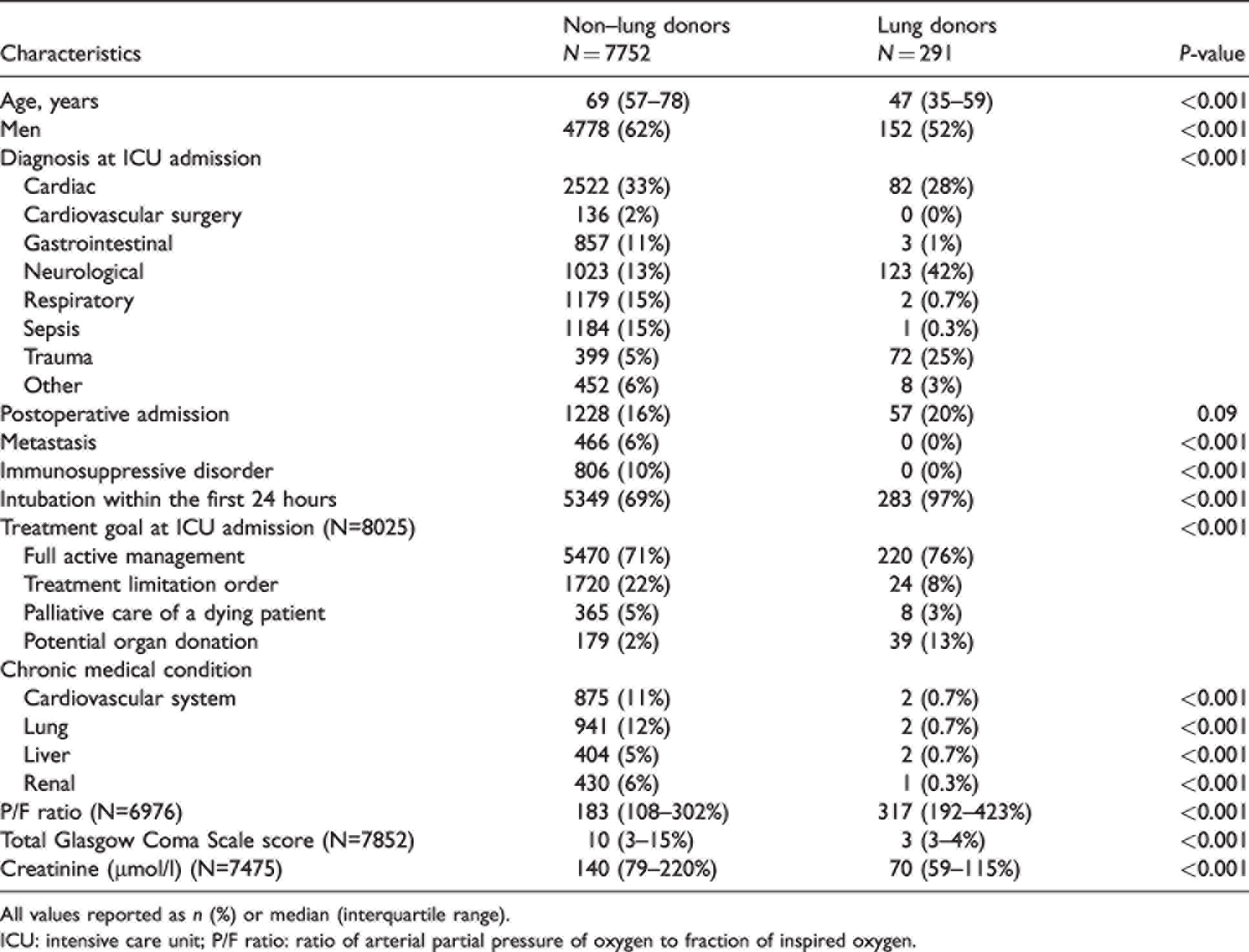
All values reported as
ICU: intensive care unit; P/F ratio: ratio of arterial partial pressure of oxygen to fraction of inspired oxygen.
The prediction model was developed from the derivation dataset (
Logistic regression of factors used in prediction model for lung donation.
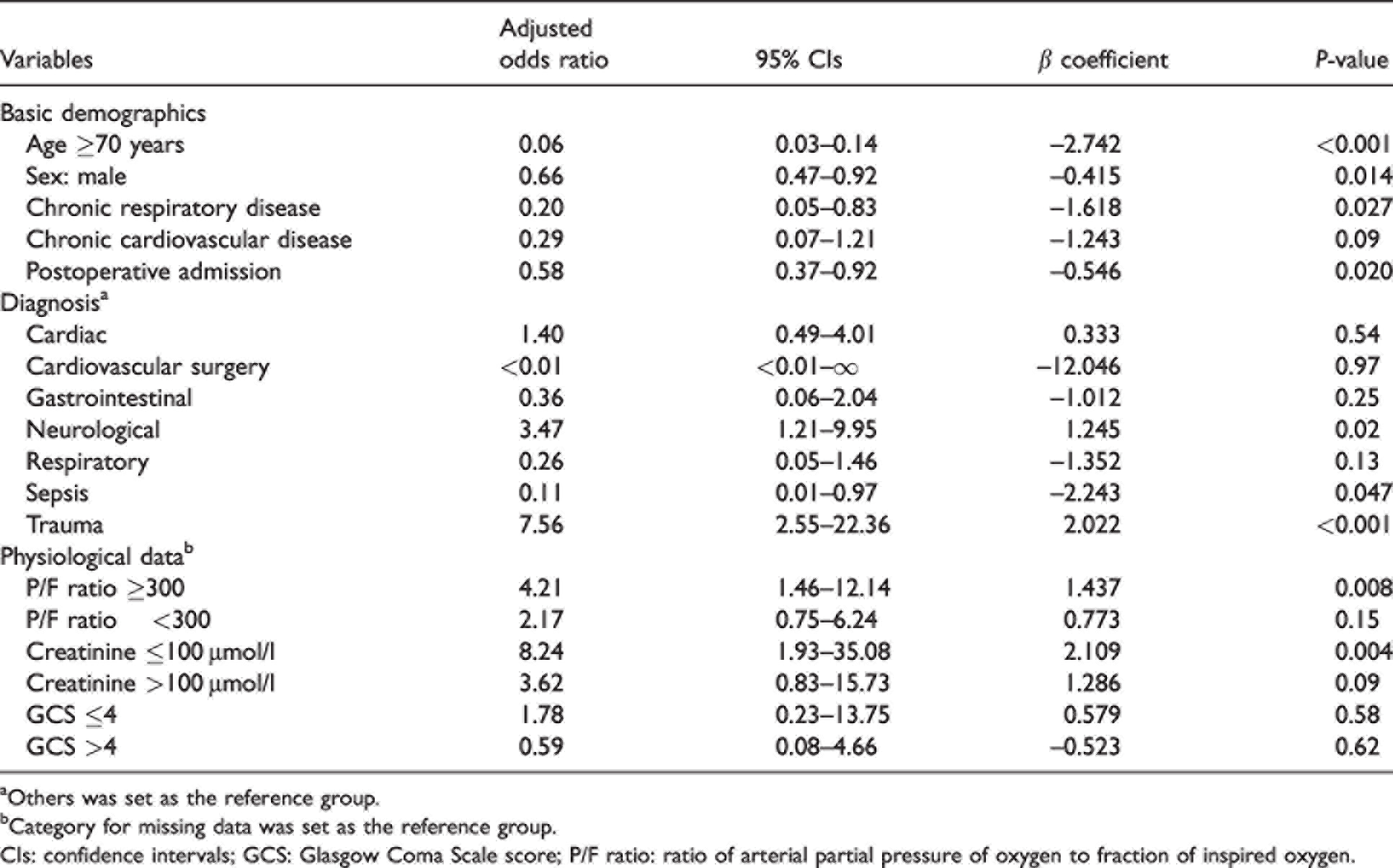
aOthers was set as the reference group.
bCategory for missing data was set as the reference group.
CIs: confidence intervals; GCS: Glasgow Coma Scale score; P/F ratio: ratio of arterial partial pressure of oxygen to fraction of inspired oxygen.
The prediction model showed excellent discrimination with AUROC of 0.91 and good calibration in both the derivation and validation (
Discrimination and calibration of the prediction model and the predicted number of lung donors.
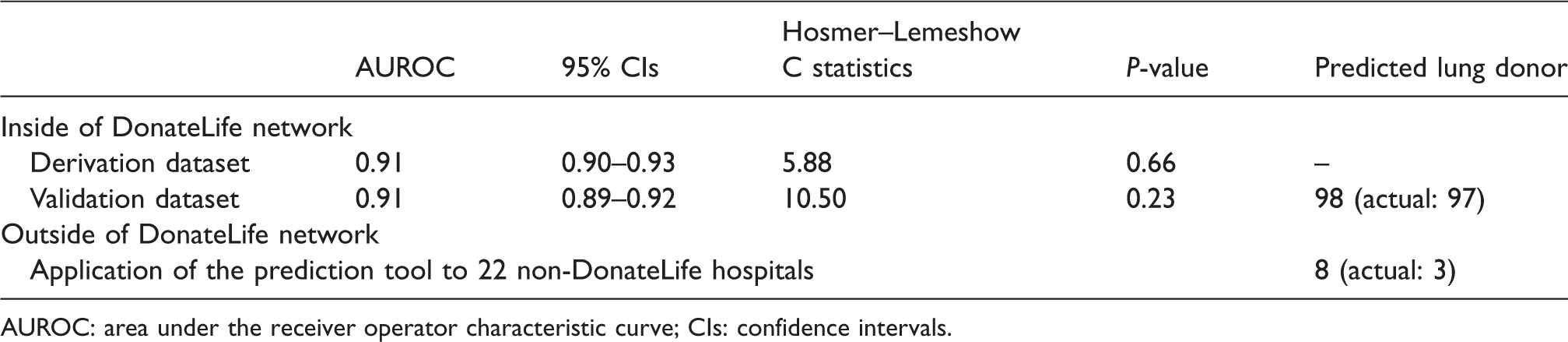
AUROC: area under the receiver operator characteristic curve; CIs: confidence intervals.
Case examples
A 64-year-old woman who died in the ICU with cerebral hypoxia after cardiac arrest, had chronic respiratory disease, P/F ratio of 233, a creatinine of 464 μmol/l, and GCS of 3 had a 1.3% possibility of becoming a lung donor.
A 31-year-old woman, who died after ICU admission due to trauma, had no chronic respiratory/cardiovascular disease, P/F ratio of 465, a creatinine of 52 μmol/l, and GCS of 3 had a 62.2% possibility of becoming a lung donor.
Estimation of lung donor opportunities outside of the DonateLife network
Applying the above prediction model, we predicted an estimate of eight theoretical lung donors in 22 non-DonateLife hospitals, as compared with three actual lung donors during the corresponding time frame.
Discussion
Using ANZICS APD data from 21 DonateLife network hospitals in Victoria, we developed an excellent prediction model which adequately estimated the number of lung donors within an ICU population. Application of the prediction tool to 22 non-DonateLife hospitals’ data identified only eight expected lung donors in these hospitals over the five-year study period. This would translate to an additional five extra lung donors (i.e. one per year) beyond the actual lung donors.
In this study, most donor lung grafts (99%) were donated at the DonateLife network hospitals, which would indicate that the network covers the vast majority of all lung donors. The prediction model was developed from this cohort, but can be used to optimise potential lung donor opportunities in other cohorts. The relevant factors selected in the prediction model could be roughly divided into two types. First, donor age and diagnosis on ICU admission (neurological disease or trauma) are related to ‘the likelihood to become organ donors’. 10 In particular, massive intracranial haemorrhage and traumatic brain injury are representative conditions that lead to death and potential for organ donation. Second, ‘lung quality’ such as P/F ratio and the absence of chronic lung disease are also relevant in terms of lung donation.11,12 A donor P/F ratio less than 300 mmHg does not always directly correlate with post-transplant graft function and this specific threshold is controversial. 13 Nevertheless, the P/F ratio would be one of the primary factors in any consideration of lung donation. These components seem to be reasonable from the donation offer process and clinical point of view.
Audits performed at Victorian DonateLife hospitals suggested that few potential donors were missed at the network hospitals. 6 In this study focusing on the possibility of lung donation outside the network, we would have expected only five additional lung donors at the 22 non-DonateLife hospitals over a five-year timeframe. As the main reason, in the non-DonateLife cohort, there were more older patients and fewer ICU admissions due to cardiac disease, neurological disease or trauma. O’Brien et al. previously suggested that there appeared to be a small but underutilised pool of potential organ donors in non-DonateLife hospitals throughout Australia, using linear regression modelling and examining overall hospital characteristics. 7 Their study relied on a prediction model which was not constructed on individual patient-level data. Put together with our findings based on patient-level data, it is apparent that there are few untapped lung donors in current practice and an alternative strategy is required if we are to expand the lung donor pool to improve the outcomes for those on the LTx waiting list.
The use of donation after circulatory death (DCD) lungs for LTx has significantly and safely expanded the donor pool. In addition, there is emerging evidence that the current 90 minutes time frame from withdrawal of cardiorespiratory support to the declaration of death may not be the true limit for lung grafts.1,14,15 During the five-year time frame of the current study, 59 intended DCD lung donors did not progress to death within the 90 minutes, of whom 53 died at some point within 24 hours. 16 Potentially, a sizeable portion of these otherwise acceptable donors could be retrieved by a ‘time-extended’ DCD pathway with optional ex vivo lung perfusion assessment or optimisation of logistics. 17
The strength of this large study is the completeness of five-year death data in the ANZICS APD, which does allow us to view the whole state potential donor pool including the established DonateLife network and also non-DonateLife hospitals, which lack the same processes for donor identification. Our highly discriminatory and well calibrated model is based on patient-level information. This validated model could be applied to other state or individual hospital donor populations throughout Australia or elsewhere, to explore and benchmark untapped lung donation opportunities. This model is best focused on public hospitals–acknowledging the different patient mix of the private hospital system. The same approach and methodology could be used to assess untapped opportunities for other solid organ donation, or indeed assess ‘big picture’ changes to the donor pool during events such as the current coronavirus disease 2019 (COVID-19) crisis.
We recognise this study has several limitations. First, this is a retrospective study reflecting current practices in our state. However, both databases used in this study are prospectively collected and do reflect the clinical reality of an experienced large LTx centre and donation network. In the ANZICS APD, 15% of cases had missing value in the P/F ratio, serum creatinine, or GCS. To ensure the inclusion of all patients and thus all potential donors, missing values were assigned to their own category in this model. This method has also been used in a previous study. 4 Missing values were mostly negatively associated with lung donation. These were typically patients who had a very short length of ICU stay, high levels of physiological disturbance or comorbidities which would have made them unlikely to be organ donors. Second, as the prediction developed from the DonateLife network cohort would be strongly related to national or local state donor processes including consent rate, the accurate estimation in other states or countries may require recalibration of this model. Finally, without direct patient identifiers, the accuracy of the data linkage and potential for false linkage cannot be definitively determined. To minimise the chance of false linkage, five indirect identifiers including high discriminatory variables such as date were required to be an exact match. Indeed, we had most cases with an exact match and only one case requiring an individual inspection. This method has been validated in other studies using data matching.9,18,19
In conclusion, a prediction model using routinely collected data can accurately estimate the expected numbers of lung donors within an ICU population. Additional lung donor opportunities outside of the established DonateLife network were also limited, implying that we need to continue exploring alternative and novel strategies for expanding lung donation and transplantation opportunities. Donor prediction modelling provides a useful benchmarking tool with potential applications across all solid organ transplant areas.
Supplemental Material
sj-pdf-1-aic-10.1177_0310057X211070011 - Supplemental material for A prediction model to determine the untapped lung donor pool outside of the DonateLife network in Victoria
Supplemental material, sj-pdf-1-aic-10.1177_0310057X211070011 for A prediction model to determine the untapped lung donor pool outside of the DonateLife network in Victoria by Okahara Shuji Conceptualization Data curation Formal analysis Investigation Methodology Software Validation Visualization Writing – original draft Writing – review & editing Snell Gregory I Conceptualization Data curation Formal analysis Methodology Project administration Supervision Validation Writing – review & editing Levvey Bronwyn J Conceptualization Data curation Methodology Project administration Validation Writing – review & editing McDonald Mark Conceptualization Data curation Methodology Writing – review & editing Rohit D’Costa, Opdam Helen Conceptualization Formal analysis Methodology Project administration Resources Supervision Validation Writing – review & editing Pilcher David V Conceptualization Formal analysis Methodology Project administration Supervision Validation Writing – review & editing in Anaesthesia and Intensive Care
Footnotes
Author Contribution(s)
Acknowledgements
The author(s) acknowledge DonateLife Victoria and the Australian Organ and Tissue Authority for providing electronic donor record and the Lungitude Foundation for their general lung transplant research support.
The author(s) and the ANZICS CORE management committee would like to thank clinicians, data collectors and researchers at the following contributing sites: Albury Wodonga Health ICU; Austin Health ICU; Ballarat Health Services ICU; Barwon Health ICU; Bendigo Health Care Group ICU; Box Hill Hospital ICU; Cabrini Hospital ICU; Central Gippsland Health Service ICU; Dandenong Hospital ICU; Epworth Eastern Hospital ICU; Epworth Geelong ICU; Epworth Hospital ICU; Frankston Hospital ICU; Freemasons Hospital ICU; Goulburn Valley Health ICU; Holmesglen Private Hospital ICU; John Fawkner Hospital ICU; Knox Private Hospital ICU; Latrobe Regional Hospital ICU; Maroondah Hospital ICU; Melbourne Private Hospital ICU; Mildura Base Hospital ICU: Monash Medical Centre ICU; Peninsula Private Hospital ICU; Peter MacCallum Cancer Institute ICU; Royal Melbourne Hospital ICU; Saint John of God Bendigo ICU; South West Healthcare ICU; St John of God Health Care Geelong ICU; St Vincent’s Hospital Melbourne Ltd. ICU; St Vincent’s Mercy Private ICU; Sunshine Hospital ICU; The Alfred Hospital ICU; The Bays Hospital ICU; The Northern Hospital ICU; The Valley Private Hospital ICU; Wangaratta District Base Hospital ICU; Warringal Private Hospital ICU; Werribee Mercy Hospital ICU; Western District Health Service ICU; Western Hospital ICU; Western Private ICU; Wimmera Health Care Group ICU.
Declaration of conflicting interests
The author(s) have no conflicts of interest to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
