Abstract

Sharp-needle ophthalmic blocks (retrobulbar and peribulbar blocks) have been the mainstay of ophthalmic regional anaesthesia for decades. Despite the increasing popularity of the blunt cannula (sub-Tenon’s block) technique in recent years, 1 sharp-needle blocks are still performed widely around the world. The possible reasons for the slow uptake of sub-Tenon’s block in some countries have been discussed in a recent publication. 1
Teaching and training peribulbar block can be a daunting task. Traditionally, it is first taught on real patients, with the trainee observing the block performed by the trainer and thereafter attempting it under direct supervision. This style of training is not ideal as inexperienced learners may not comprehend or appreciate ocular anatomy and optimal needle placement (i.e. insertion site, trajectory, depth of insertion), thus increasing the risk of serious life- and sight-threatening complications. The use of a practice model, system or simulator, for initial learning of sharp-needle block techniques without involving patients would be beneficial.
The authors (AWYC and CMK) have conducted many hands-on workshops for ophthalmic block techniques, hosted at local hospitals, function rooms and convention centres. We have used different systems with various success in several countries for anaesthesia and ophthalmic organisations, including the Australian and New Zealand College of Anaesthetists.
The authors have been fortunate to use human cadaver heads with intact orbital structures for training in sharp-needle blocks (Sight Foundation Clinical Skills Laboratory, Sydney Eye Hospital, Australia). Cadavers offer the best and closest anatomical model for training but they are not ideal. The tissues are fragile and lose their elasticity after the application of preservatives, and frozen storage with subsequent thawing for use. The globes, with hypotony, sink deeper into the orbital cavity and lack normal tactile feedback (but this can be improved by re-inflating the cadaver eye). Limitations of cadaver-based teaching methods include the potential risk of transmitting unknown diseases in the event of needlestick injury, availability only in facilities licensed to handle human tissue, and above all, expense and difficulty of acquisition.
Animal heads with intact eyes (sheep, goat and pig) have been used for ophthalmic block workshops in Australia, New Zealand and South-East Asian countries. While the tissues are fresh, their elasticity is maintained. However, they are anatomically different to humans, with altered thickness of orbital tissues, and shape and depth of the orbital cavity. In addition, animal specimens may be unacceptable due to religious and ethical beliefs.
Manikin-based simulation technology has been developed, but remains in its infancy. The ophthalmic anaesthesia simulation system (OASiS) incorporates anatomically accurate orbital and ocular structures, with an integrated sensing system to provide feedback on the block performed. 2 The sensing mechanism detects the needle entry point and provides alerts when the globe or extraocular muscles are approached or damaged by the needle. However, the OASiS is currently not commercially available.
We have developed an in-house modified life-size human skull replica for training in the peribulbar block technique. The model is cheap, lightweight, simple, easy to assemble, and transportable across states and countries. It is appropriate to all participants, and acceptable to be used in any venue or facility. It does not carry unknown communicable diseases. It allows the participants of our workshops to familiarise themselves with the relevant anatomy, needle trajectory and insertion depth, ergonomics and hand positioning. The model is particularly useful for practitioners new to peribulbar blockade. The cost is approximately A$70. We have used this model for conducting workshops in ophthalmic regional blocks, both in home countries and overseas, with positive reviews from participants of our workshops. Materials required and stepwise preparation of the model are included in Figures 1 and 2, respectively.
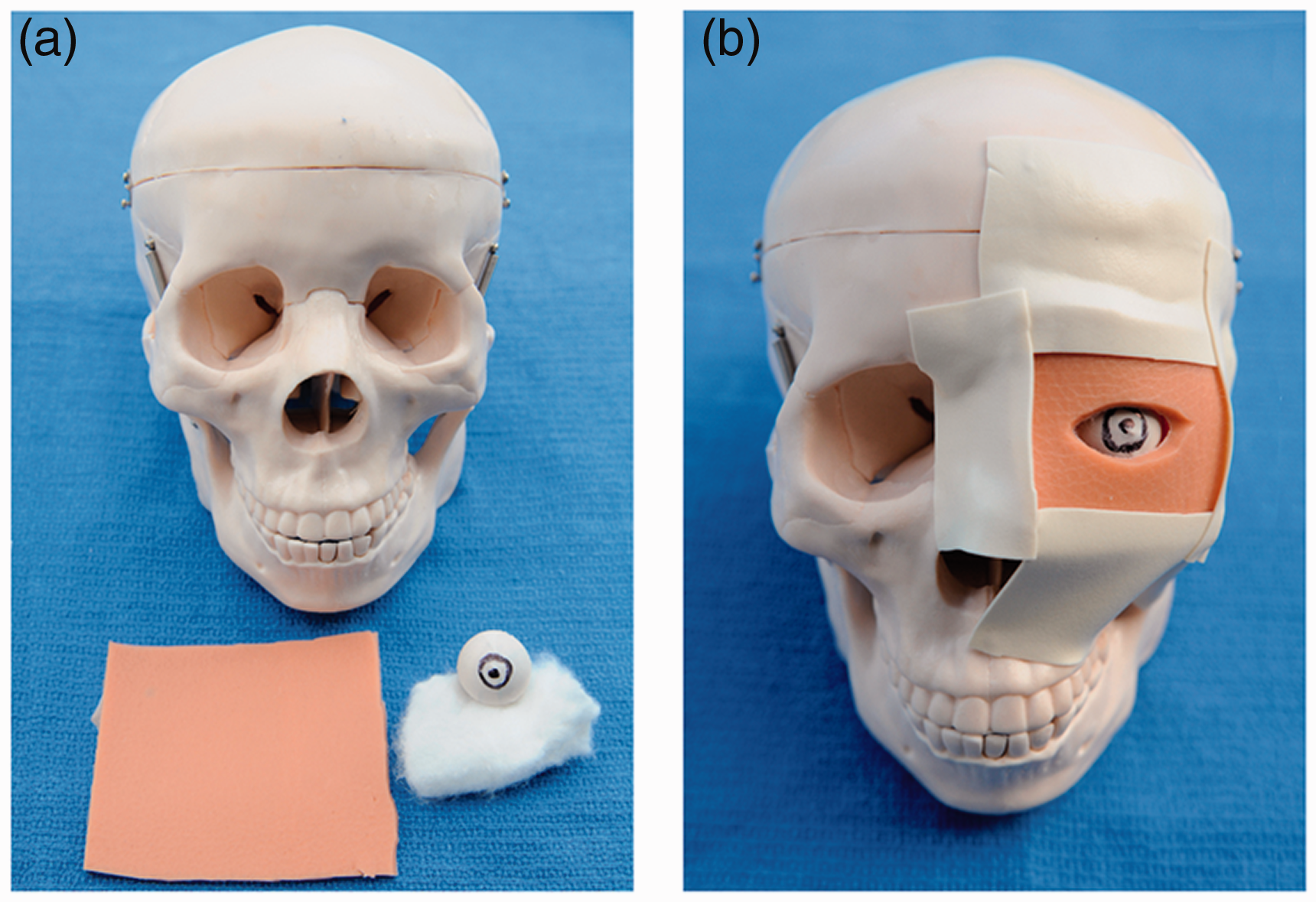
(a) Materials required: a life-size human skull replica (Mentone Educational, code MEN104, Victoria, Australia), cotton wool, a circular object (textile material) diameter 23 mm, one disposable surgical (nitrile) glove and three-dimensional (3D) printed latex sheet, approximately 2 mm thickness (alternative: a few combined layers of disposable nitrile gloves). (b) Assembling steps: Trimmed disposable glove material is wrapped round the circular object. Pupil and corneal limbus are sketched on the surface to replicate the ‘globe’. It is placed inside the orbital cavity and the surrounding area is lightly packed with cotton wool to keep the globe in the centre. A layer of non-transparent skin replica (3D printed latex sheet), with an opening cut to resemble eyelids, is attached to cover and extend beyond the entire surface of the orbital rim.
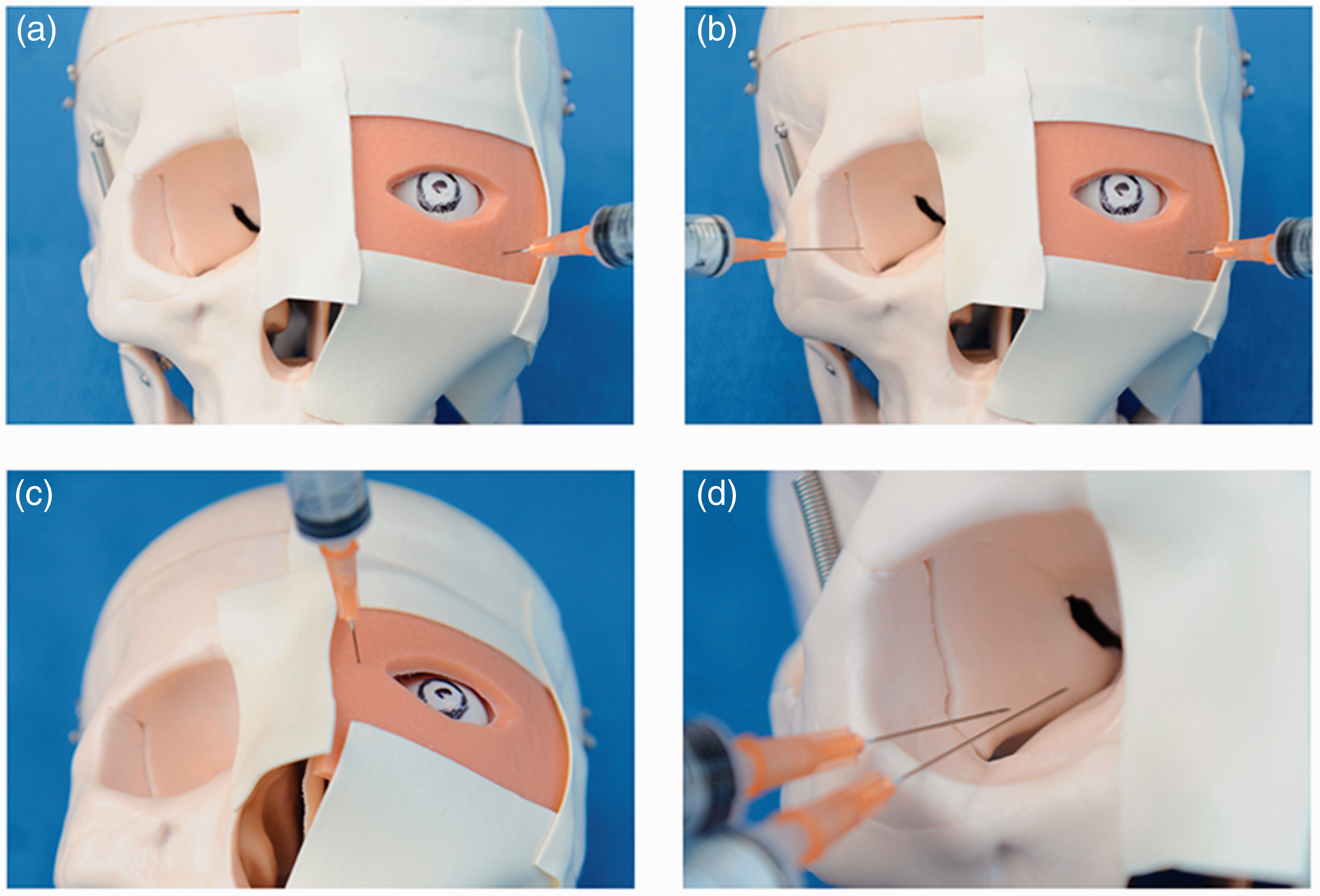
(a) Extreme inferotemporal peribulbar block; (b) Simultaneous demonstration of extreme inferotemporal peribulbar block in the modified and unaltered orbital cavities; (c) Medial peribulbar block; (d) Demonstrating the relative risks of 25 mm and 38 mm length needles.
The participants of our workshops were given a didactic presentation on how to perform a peribulbar block in accordance with the ‘modern’ technique described,3,4 followed by hands-on demonstration on the skull model. The optimal placement of needle insertion, angle of trajectory and needle insertion depth were demonstrated. Intentional needle deviation to replicate globe perforation was also demonstrated so the trainees could learn to avoid the same trajectory. The different textile feedbacks from different materials used (opaque latex outer layer (skin), cotton wool (orbital fat) and circular object (globe)) enabled easy detection of globe perforation. An uncovered contralateral orbital cavity was used as a reference to highlight the potential risk of longer needles (>31 mm long) entering orbital openings. The trainee(s) then practised on the skull model under the supervision and guidance of the instructor. For more advanced training, a larger circular object (diameter >26 mm) could be used to resemble a myopic globe. In addition, a small amount of dye could be injected after needle insertion to confirm the location of the dye and site of needle placement, after removal of the opaque outer layer and the ‘globe’.
The skull replica model is not perfect, and will never replace clinical experience. It lacks the tactile sensation of real tissue planes, despite layers of different synthetic materials used. It does not alert the very close proximity of the needle to the globe or extraocular muscle damage. An average human globe has similar equatorial horizontal width (EHW) and axial length (AL); however, the increase in EHW is relatively smaller compared with the increase in AL in myopic globes. 5 Therefore, larger circular objects do not truly represent human myopia. In addition, the model does not compensate for tight orbital septae and shallow orbits—these variations can only be appreciated in real clinical cases.
Further modification can also be made to the above skull model. A fibreoptic camera, connected to a computer for screening and recording, can be placed near the superior orbital fissure. Real-time needle placement is possible or it can be watched subsequently from the recorded video playback. This involves multiple drillings in the skull and substitutes cotton wool with transparent material. The skull model, however, becomes quite cumbersome, technical and expensive. A peribulbar block training simulator manikin or 3D printed skull replica with anatomically comparable orbital tissue planes might be the way forward and worth exploring. These simulating models might be helpful as learning tools in potentially reducing the risk of sharp-needle–related serious ophthalmic complications; however, trainees will still require supervision when they perform the block clinically on patients.
Footnotes
Author Contribution(s)
Declaration of conflicting interests
The author(s) have no conflicts of interest to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
