Abstract
There is a lack of data to support either continuation or interruption of non–vitamin K oral anticoagulants for cataract and vitreoretinal surgery. A prospective audit was undertaken of 291 patients undergoing cataract surgery or vitreoretinal surgery, predominantly under sub-Tenon’s block, while continuing these agents. The median time from last non–vitamin K oral anticoagulant dose to the insertion of sub-Tenon’s block was five hours. No patient required emergency reversal of anticoagulation. There were no sight-threatening complications in the immediate perioperative period, although two vitreoretinal patients (3.8%) had a moderate haemorrhagic complication on day five, and two cataract patients (0.8%) had a minor haemorrhagic complication on days one and 14 postoperatively. Despite continuing their non–vitamin K oral anticoagulants, three (1%) cataract patients had a moderate thromboembolic complication within the 30-day postoperative period. The risk of haemorrhagic complications associated with continuation of anticoagulation with non–vitamin K oral anticoagulants for cataract and vitreoretinal surgery is low, and this audit supports the continuation of non–vitamin K oral anticoagulants for our patients having cataract and vitreoretinal surgery.
Keywords
Introduction
The age and severity of comorbidities of the ophthalmic surgical population is increasing. A study of the National Cataract Dataset in the United Kingdom in 2009 indicated that more than 5% of cataract patients took an anticoagulant. 1 Since that time, the introduction of non–vitamin K oral anticoagulants (NOACs) and a widening of the indications for anticoagulation have led to a further increase in the number of anticoagulated patients presenting for cataract surgery. 2 For example, current statistics indicate that nearly 6% of patients over the age of 65 years in New Zealand are now taking dabigatran.3,4
Over the past decade several NOACs have emerged. These include: direct thrombin inhibitor, dabigatran (Pradaxa; Boehringer Ingelheim Pharmaceuticals, Ingelheim am Rhein, Germany), and factor Xa inhibitors, rivaroxaban (Xarelto; Bayer Healthcare, Leverkusen, Germany) and apixaban (Eliquis; Bristol-Myers Squibb, New York, NY, USA). Their advantages over older anticoagulants include: more predictable pharmacological action negating the need for coagulation monitoring, greater efficacy, fewer drug–drug interactions and a similar or lower risk of intracranial bleeding. The number of patients on these drugs has increased steadily in many countries over the past years. 5
The continuation of these drugs in the perioperative period may, potentially, be associated with bleeding which may be sight-threatening. However, the risk of interruption is known to be significant and may be life-threatening: in the RE-LY study by Healy and colleagues in 2012, 6 427 patients had anticoagulation (either dabigatran or warfarin) interrupted for cataract surgery. Of those, 1.4% had a major thrombotic event (cardiovascular death, ischaemic stroke, non–central nervous system embolism or pulmonary embolism) in the first 30 days postoperatively. 6
Similarly, in a study by Vene et al. in 2016, interruption of dabigatran increased the short-term thromboembolic risk more than 20-fold. 7
In patients who present for ophthalmic surgery while on these medications, it is necessary to balance the thromboembolic risk associated with stopping anticoagulation against the haemorrhagic risk of surgery while continuing anticoagulation. Although many centres continue these agents in the perioperative period, there is very little evidence in the literature to support this decision. Previous studies of patients on warfarin indicate that it is most likely safe to continue this agent for cataract surgery, 8 vitreoretinal surgery 9 and for anaesthesia under sub-Tenon’s block. 10 Currently, there are limited data regarding the safety of continuing or stopping NOACs in these situations; what data are available are predominantly retrospective and involve small sample sizes.11,12 A retrospective literature review by Makuloluwa et al. 13 in 2019 recommended withholding NOACs for two days preoperatively and restarting one or two days postoperatively for cataract and vitreoretinal surgery. However, the authors acknowledged that this recommendation was made because of the lack of data to evaluate the haemorrhagic risk of continuing these agents. 13 A Clinical Excellence Commission guideline in 2017 suggests that the continuation of NOACs for cataract surgery may be appropriate but does not mention vitreoretinal surgery. 14
In discussion with the ophthalmologists in our institution, it was decided to conduct a prospective audit with the aims of:
Quantifying the perioperative incidence and severity of haemorrhagic complications associated with cataract and vitreoretinal surgery in patients continuing NOACs. Quantifying the incidence and severity of haemorrhagic complications associated with sub-Tenon’s block in patients continuing NOACs. Quantifying the incidence and severity of thromboembolic complications that occur in this group even if NOACs are continued.
Methods
The Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines were adhered to in manuscript preparation. 15 Ethical approval for this study (17/NTA/4) was given by the New Zealand National Health and Disability Ethics Committee.
The study was undertaken in the Ophthalmic Surgical Centre, Greenlane, which constitutes the tertiary ophthalmic centre for the Auckland District Health Board. The study period comprised a three-year period from February 2017 to February 2020.
Prior to commencement of the audit period, in collaboration with the Haematology Department, an emergency reversal protocol was created. This included immediate access to idarucizumab to enable rapid reversal of dabigatran, and clotting factors appropriate for the reversal of rivaroxaban and apixaban in the event of any sight-threatening bleeding in the perioperative period that could not be controlled by standard surgical measures.
All patients on the waiting list for cataract and vitreoretinal surgery who were taking NOACs were instructed to continue with these drugs as usual in the preoperative and postoperative phases. This was irrespective of the anticipated surgical difficulty of the case.
On arrival in the operating room, the standard anaesthetic technique consisted of a classic sub-Tenon’s block with a blunt cannula, with insertion of an indwelling intravenous line.10,16 Topical anaesthesia with 2% lidocaine jelly or general anaesthesia was also permitted if considered appropriate. Anxiolysis, with a small (1–2 mg) dose of intravenous midazolam was permitted, if required.
For each patient, an audit form was completed, which included the medical indication, surgery type, interval between surgery and the last dose of NOAC, anaesthetic type, the presence and degree of subconjunctival bleeding and any anaesthetic or surgical complications relating to haemorrhage, including whether or not emergency reversal was required. The presence of subconjunctival bleeding was graded on a four-point scale as none, mild (limited to one quadrant), moderate (two quadrants) or severe (three or more quadrants).
The electronic record of each patient was followed up for a 30-day period postoperatively, to determine if there were any haemorrhagic or thromboembolic complications. This was performed via the Regional Clinical Portal, which includes access to ophthalmic follow-up and any outpatient or inpatient referral/admission in the Auckland Region District Health Boards.
Patients were excluded from the study if they had not taken their NOAC in the past 24 hours.
Sample size calculation was performed using the 95% confidence interval, an estimated outcome occurrence of 3% and a margin of error of 2%. This gave a sample size of 280 patients.
Results were de-identified and entered on to a database for analysis.
Statistical analysis was performed using GraphPad Prism, version 6.02 (San Diego, CA, USA). Fisher's exact test was used to compare discrete variables. The level of statistical significance was set at two-sided P<0.05.
Results
Data were obtained from 239 patients having cataract surgery and 52 patients who underwent vitreoretinal surgery who all continued their NOAC.
Indications for anticoagulation were predominantly for atrial fibrillation and are outlined in Table 1. Four patients were taking dabigatran as prophylaxis for mechanical heart valves due to an inability to tolerate warfarin.
Participant characteristics.
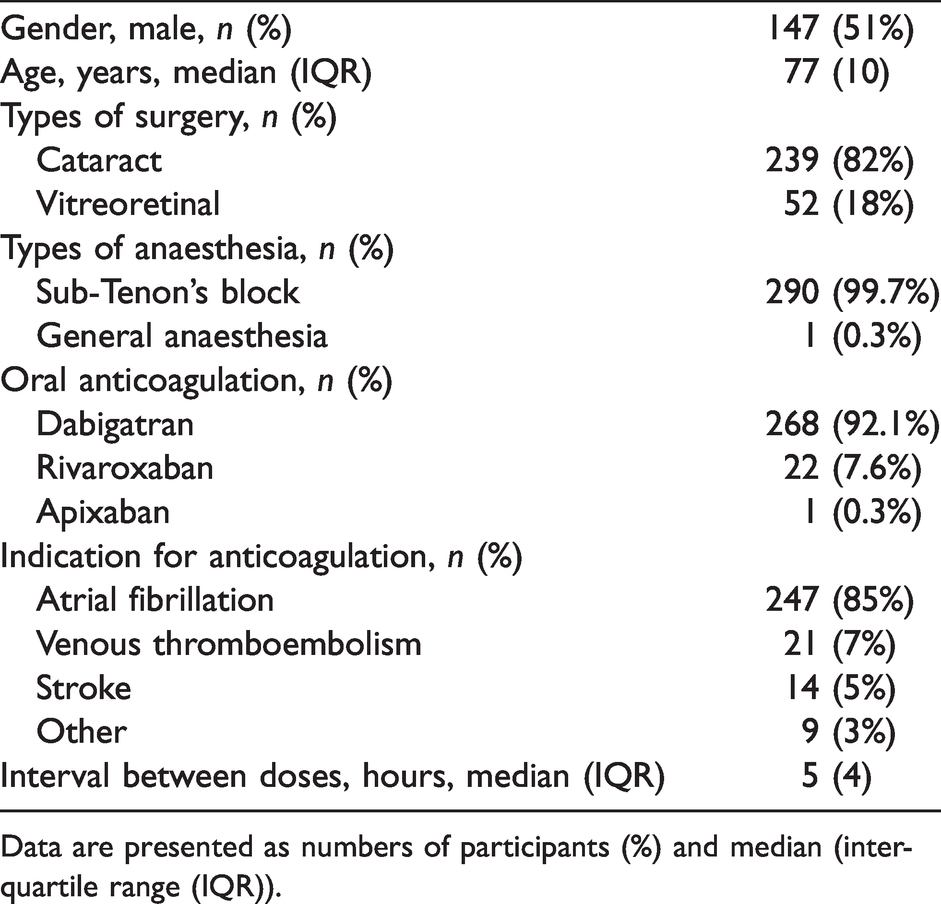
Data are presented as numbers of participants (%) and median (interquartile range (IQR)).
Cataract procedures
There were 121 (51%) men and 118 (49%) women. The median age was 78 years, with an interquartile range (IQR) of ten years. The majority of patients, 95% (228 patients), were taking dabigatran and 5% (11 patients) were taking rivaroxaban; no patients were taking apixaban.
The median time from the last dose of NOAC to arrival in the operating theatre was five (IQR 4) hours, and with a range of 1–20 hours.
The anaesthetic technique was blunt-cannula sub-Tenon’s block using a standard technique in 238 of the 239 cases. One patient had a general anaesthetic for their surgery.
In the perioperative period, in two patients, the surgeon noted increased ooze from corneal incisions but this did not require additional treatment. There were no intraoperative haemorrhagic issues that required surgical intervention. Three patients received a posterior capsule tear requiring anterior vitrectomy. All patients went home on the day of surgery.
In the 30-day postoperative period, two patients had a minor haemorrhagic complication (see Table 2). On day 1, one patient (on dabigatran) was noted to have a small disk haemorrhage, which was associated with a high intraocular pressure requiring acetazolamide, and which cleared spontaneously. On day 14, one patient (on dabigatran) developed a minor spontaneous subconjunctival haemorrhage, which also cleared spontaneously.
Incidence of outcome between cataract and vitreoretinal procedures.
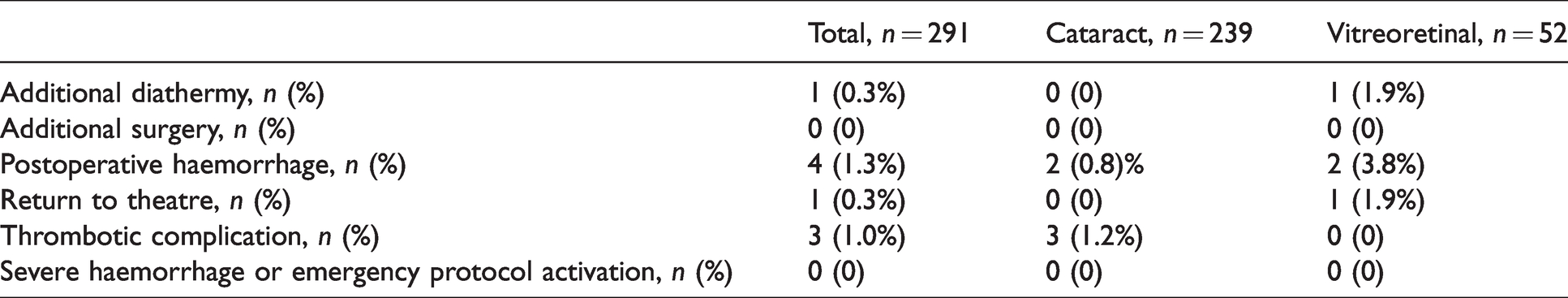
Three patients had a thromboembolic episode (see Table 2). On day 1, one patient developed a symptomatic extension of their recent deep vein thrombosis: dabigatran was discontinued and low molecular weight heparin was commenced. On day 3, one patient was diagnosed with a repeat minor transient ischaemic attack (TIA) despite continuing dabigatran, requiring no further treatment. On day 21, one patient (on dabigatran) developed an occlusion in the superotemporal branch of the retinal vein with macular oedema, which resolved spontaneously.
No patient required emergency reversal of anticoagulation throughout the perioperative period.
Vitreoretinal procedures
There were 26 (50%) men and 26 (50%) women. The median (IQR) age was 75 (12.5) years. In terms of anticoagulant, 77% (40 patients) were taking dabigatran and 23% (11 patients) were taking rivaroxaban. One patient was taking apixaban.
The median (IQR) time from the last dose of NOAC to arrival in the operating theatre was 6 (IQR 5.25) hours hours, range of 1–20 hours.
All vitreoretinal surgery patients had their procedure under sub-Tenon’s block.
Intraoperatively, one of the cases was noted by the surgeon to be more ‘oozy’ than would normally be expected, which required additional diathermy but this was minor in nature. There were also two patients who were noted to be slightly ‘oozy’, but did not require any additional treatment. There were no other surgical issues related to bleeding.
Postoperatively, two patients (3.8%) had problems related to the NOAC: one patient required a return to theatre five days postoperatively for recurrence of vitreous haemorrhage from a retinal artery aneurysm, requiring permanent cessation of rivaroxaban. The second patient developed a recurrent hyphaemia from iris/lens chafing requiring long-term cessation of dabigatran, which resolved the problem without further intervention. There were no thromboembolic events.
No patient required emergency reversal of anticoagulation at any time in the perioperative period.
Sub-Tenon’s block
A total of 290 patients had their surgery under sub-Tenon’s block.
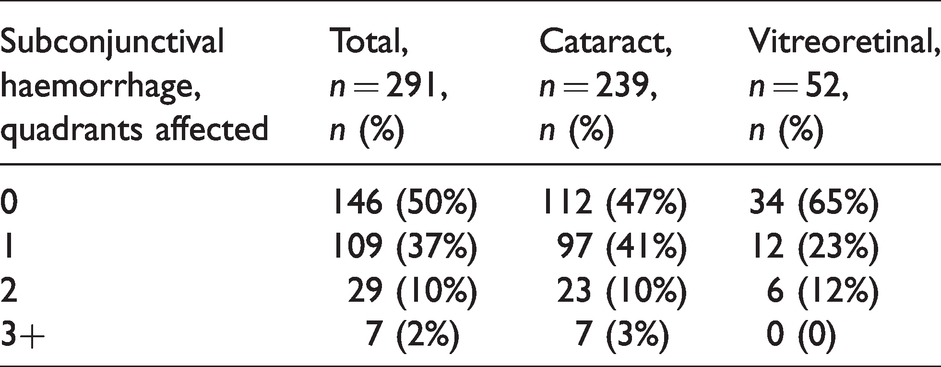
Following insertion of the sub-Tenon’s block, but prior to commencement of surgery, the eye was examined for the presence of subconjunctival bleeding. There was no subconjunctival haemorrhage in 50% of cases (Table 3). The presence of subconjunctival haemorrhage was noted in the remainder: this was noted to affect one quadrant of the conjunctiva in 37%, two quadrants in 10%, and three or more quadrants in 2% of patients. There was no statistical correlation between the degree of subconjunctival bleeding and the time since the last dose of NOAC (P=0.07). None of the bleeding was considered to be sight-threatening and there was no interference with surgery.
Incidence of subconjunctival haemorrhage following sub-Tenon’s block.
There were no other complications associated with sub-Tenon’s block.
Overall, there were no major haemorrhagic events in this cohort of patients and no patient required emergency reversal of anticoagulation.
Discussion
This is a prospective study looking at the perioperative events associated with continuing NOACs for cataract and vitreoretinal surgery under sub-Tenon’s block. Although there are data to support the continuation of traditional anticoagulant and antiplatelet agents for cataract and vitreoretinal surgery,8,17 the current literature contains very little guidance to support either continuing or interrupting NOACs in these patients. The data in this study indicates that continuing NOACs throughout the perioperative period for cataract and vitreoretinal surgery is associated with a low incidence of mild to moderate haemorrhagic events requiring little, if any, additional surgical intervention: 0% for cataract surgery, 3.8% for vitreoretinal surgery and 0% for sub-Tenon’s block. There were, however, no major haemorrhagic events in this series and no patient required emergency reversal of anticoagulation. Furthermore, there were no major thrombotic events within the 30-day postoperative period, although three cataract patients (1.2%) had a moderate thrombotic complication despite continuing with full anticoagulation.
By way of comparison, a retrospective survey of electronic records by Cheung et al. in 2019 noted 42 patients who had continued their NOAC for cataract surgery and 24 patients in whom anticoagulation was interrupted. 11 There were no haemorrhagic complications in the group who continued with their NOAC but two postoperative periorbital bleeds in the group in which the NOAC was withheld. The bleeding occurred on resumption of the NOAC and both patients had received a retrobulbar block. There were no thromboembolic complications in either group. 11 The study of Cheung et al. 11 differs from ours in several ways: the study was retrospective and the sample size was small. Also, the anaesthetic technique was predominantly topical anaesthesia (82%) or retrobulbar block (7.5%), with only one patient receiving a sub-Tenon’s block. It supports the continuation of NOACs for cataract surgery, and highlights the potential danger of the rapid resumption of anticoagulation that occurs once these drugs are restarted postoperatively. It also illustrates the potential for orbital trauma with sharp-needle techniques, such as retrobulbar block, in anticoagulated patients.
A small prospective study by Barequet et al., of 25 patients who continued NOACs for cataract surgery under topical anaesthesia, noted five patients who had increased conjunctival bleeding, but no major haemorrhagic issues. 18 However, the authors included only cataracts that were considered to be surgically straightforward, whereas our study related to the entire cataract population.
With regard to the vitreoretinal patient group, the 3.8% incidence of postoperative haemorrhagic issues including a 1.9% return to theatre compares well with previous historical analyses of patients on NOACs. Grand and Walia, 12 in a retrospective study of 33 patients, compared these to a historical control of 66 patients not on anticoagulants. They found an 11% incidence of postoperative haemorrhage, with a 5.5% incidence of repeat surgical intervention in the three-month period following surgery. This compared to an 8.3% incidence of postoperative haemorrhage in the control group, with a similar (5.5%) incidence of repeat surgery. 12
A study by Meillon and colleagues in 2018 looked at 63 patients on anticoagulants including 18 patients on NOACs who continued them during the perioperative period. 19 The authors reported a 6.6% incidence of haemorrhagic complications overall, but there was no statistical difference in haemorrhagic complications between the NOAC group and a group who stopped anticoagulants. They recommended that all anticoagulants be continued perioperatively.
A recent study by Louison et al. in patients having macular hole surgery predominantly under peribulbar block identified 24 patients who continued NOACs and found no significant increase in ocular bleeding complications with this group compared to a group that was not anticoagulated. 20
An editorial by McClellan and colleagues in 2014 raised the possibility that vitreoretinal surgery in patients on new antithrombotics could be reclassified as moderate or low risk for haemorrhagic complications if further studies were conducted to confirm this. 21 Our study showed that there was no statistical difference in the occurrence of haemorrhagic and thrombotic complications between those patients who underwent cataract surgery and those who had vitreoretinal surgery, further supporting this assertion.
With regard to sub-Tenon’s block, the continuation of anticoagulation was associated, in this study, with a 49.7% incidence of predominantly minor, non–sight-threatening, conjunctival haemorrhage. These data are consistent with previous studies of sub-Tenon’s block in anticoagulated patients on traditional anticoagulants, which indicate that the incidence of subconjunctival bleeding is higher than in patients not taking anticoagulants. 10 A study by Kumar et al. in 2006 also noted an increased incidence of minor subconjunctival haemorrhage after sub-Tenon’s block in patients on warfarin of 35%, compared to a non-anticoagulated control group of 19%. 22
The absence of postoperative major thromboembolic complications in this study contrasts with historical data in which anticoagulation was interrupted for ophthalmic surgery, which reported a 1.4% incidence of major thromboembolic events in the first 30 days postoperatively. 6
The strengths of this study are that it is prospective and has a larger sample size that previous studies and is inclusive of all cataract and vitreoretinal cases independent of anticipated surgical complexity. It also has clear data on the time interval between the last dose of NOAC and surgery to show that the patient was actually therapeutically anticoagulated at the time of surgery. A potential weakness is that although the cases included in the study were sequential, a small number of cases that were on NOACs could have passed undetected and not been entered in the study by the procedural anaesthetist. However, this would be a random event and should not affect the results.
The study does not include patients taking novel antiplatelet agents (P2Y12 inhibitors), and further studies of patients on these medications are warranted.
In summary, this study indicates that the risk of haemorrhagic complications associated with the continuation of anticoagulation with NOACs for cataract and vitreoretinal surgery is low and likely to be less, in incidence and severity, than the thrombotic complications associated with their interruption. It also supports the low potential for sight-threatening bleeding with sub-Tenon’s block in anticoagulated patients. It therefore lends further support to the continuation of NOACs for patients having cataract and vitreoretinal surgery.
Supplemental Material
sj-pdf-1-aic-10.1177_0310057X211057136 - Supplemental material for Haemorrhagic complications following cataract and vitreoretinal surgery with sub-Tenon’s block in patients receiving non–vitamin K oral anticoagulant agents: A prospective audit
Supplemental material, sj-pdf-1-aic-10.1177_0310057X211057136 for Haemorrhagic complications following cataract and vitreoretinal surgery with sub-Tenon’s block in patients receiving non–vitamin K oral anticoagulant agents: A prospective audit by Guise Philip A Conceptualization Data curation Methodology Project administration Supervision Writing original draft Writing review editing Wang Kailun Data curation Formal analysis Software Writing original draft Writing review editing in Anaesthesia and Intensive Care
Footnotes
Author Contribution(s)
Acknowledgements
The author(s) wish to acknowledge Dr Douglas Campbell, specialist anaesthetist, Auckland City Hospital, for his advice on preparing this manuscript.
Declaration of conflicting interests
The author(s) have no conflicts of interest to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
