Abstract
In general anaesthesia, early collapse of poorly ventilated lung segments with low alveolar ventilation–perfusion ratios occurs and may lead to postoperative pulmonary complications after abdominal surgery. An ‘open lung’ ventilation strategy involves lung recruitment followed by ‘individualised’ positive end-expiratory pressure titrated to maintain recruitment of low alveolar ventilation–perfusion ratio lung segments. There are limited data in laparoscopic surgery on the effects of this on pulmonary gas exchange. Forty laparoscopic bowel surgery patients were randomly assigned to standard ventilation or an ‘open lung’ ventilation intervention, with end-tidal target sevoflurane of 1% supplemented by propofol infusion. After peritoneal insufflation, stepped lung recruitment was performed in the intervention group followed by maintenance positive end-expiratory pressure of 12–15 cmH2O adjusted to maintain dynamic lung compliance at post-recruitment levels. Baseline gas and blood samples were taken and repeated after a minimum of 30 minutes for oxygen and carbon dioxide and for sevoflurane partial pressures using headspace equilibration. The sevoflurane arterial/alveolar partial pressure ratio and alveolar deadspace fraction were unchanged from baseline and remained similar between groups (mean (standard deviation) control group = 0.754 (0.086) versus intervention group = 0.785 (0.099), P = 0.319), while the arterial oxygen partial pressure/fractional inspired oxygen concentration ratio was significantly higher in the intervention group at the second timepoint (control group median (interquartile range) 288 (234–372) versus 376 (297–470) mmHg in the intervention group, P = 0.011). There was no difference between groups in the sevoflurane consumption rate. The efficiency of sevoflurane uptake is not improved by open lung ventilation in laparoscopy, despite improved arterial oxygenation associated with effective and sustained recruitment of low alveolar ventilation–perfusion ratio lung segments.
Keywords
Introduction
Lung gas exchange is impaired in patients under general anaesthesia (GA) for a variety of reasons, including collapse of poorly ventilated lung segments early after anaesthetic induction, arising from a reduction in muscle tone and resting lung volume. This manifests in reduced dynamic lung compliance and increased driving pressure (the end-expiratory to peak inspiratory pressure difference required to achieve a set tidal volume) during positive pressure ventilation (IPPV). This lung atelectasis is thought to contribute to the development of postoperative pulmonary complications (PPCs),1–10 and studies have suggested an association between high driving pressure and PPCs.11,12
Ventilatory strategies to reduce atelectasis and improve arterial oxygenation under GA include positive end-expiratory pressure (PEEP) and lung recruitment manoeuvres (RMs).13–16 A combination of RMs with high PEEP (12 cmH2O) to maintain lung recruitment has been recommended but data in large trials suggesting any possible protection from PPCs have mainly been seen in laparoscopic surgery, in which pneumoperitoneal insufflation promotes basal lung compression and further impairs dynamic lung compliance.13,15
A refinement of this strategy (‘open lung’) involves lung recruitment by an automated stepped lung recruitment or ‘PEEP-stairway’ manoeuvre to increase dynamic lung compliance, followed by ‘individualised’ high PEEP titrated to maintain recruitment of low alveolar ventilation–perfusion ratio (Va/Q) lung segments in that patient, which is monitored by breath-to-breath measurement of lung compliance and driving pressure. A PEEP-stairway recruitment tool (Auto-RM) is available in the Flow-i anaesthesia machine (Getinge Maquet, Solna, Sweden), but there are limited data on its effect on either lung mechanics, gas exchange or PPC risk during laparoscopy. 16
We measured the effect of an open lung ventilation strategy using this recruitment tool on pulmonary gas exchange, as well as on driving pressures and lung compliance, during laparoscopic surgery. Furthermore, while better arterial oxygenation due to reduced Va/Q mismatch is expected with lung recruitment and PEEP, it is possible that it may also be accompanied by more efficient alveolar–capillary uptake of anaesthetic agent. This would be expected to result in smaller arterial–alveolar (a–A) gradients for both respiratory gases and volatile agents and possibly with reduced volatile agent consumption due to the requirement for delivery of a lower inspired concentration to achieve a given anaesthetic partial pressure in arterial blood. We measured these variables in a randomised controlled trial (RCT) of open lung ventilation in laparoscopic surgery patients.
Methods
Local institutional human research ethics committee approval (HREC/18/Austin/164 12 November 2018) and trial registration were obtained (Trial ID: ACTRN12619000298112, 27/2/2019). Written informed consent was obtained from all recruited patients. Eligible patients were those consented for elective laparoscopic bowel surgery, which was expected to take more than an hour, was booked on the first author's regular operating list, and planned for relaxant general anaesthesia and intra-arterial pressure monitoring. Exclusion criteria were: pregnancy, body mass index (BMI) greater than 35 kg/m2, age under 18 or over 85 years, inability to provide informed consent, or documented severely impaired lung function (respiratory function parameters less than 30% of predicted).
Patients were randomly assigned using a preprepared, shuffled and sequentially numbered series of sealed envelopes to either of two groups: standard ventilatory management (‘control’) group, or an intervention group in which a dynamic lung recruitment protocol consistent with an open lung ventilatory strategy was administered, as outlined below.
Intravenous (IV) access was established with standard intraoperative monitoring including electrocardiography (ECG) and arterial line connected to a Vigeleo/FloTrac pulse contour continuous cardiac output monitor (Edwards Lifesciences, Irvine, CA, USA) with bispectral index (BIS) monitoring of anaesthetic depth. Following preoxygenation with fractional inspired oxygen (FiO2) 80%, induction of anaesthesia and neuromuscular blockade was performed with intravenous propofol and a non-depolarising neuromuscular blocker. Intraoperative analgesia was provided with fentanyl 3–5 µg/kg at induction and subsequent 1 µg/kg boluses in response to surgical stimulation. Insertion of an endotracheal tube was performed, with confirmation of correct position. Intravenous propofol infusion was continued, as was neuromuscular blockade with regular maintenance boluses.
The patient was connected to a Flow-i anaesthesia machine with circle breathing system, and controlled positive pressure ventilation of the lungs was commenced using pressure control mode, at a breath frequency of 10–12 /min with zero PEEP, with peak inflation pressure set to achieve a tidal volume of 7–10 ml/kg measured body weight, and to maintain an end-tidal partial pressure carbon dioxide (PE’CO2) of 28–33 mmHg as measured by capnography. FiO2 was set at 30% or as near as possible to this for maintenance of a peripheral oxygen saturation (SpO2) of not less than 96%, and sevoflurane administration was commenced for maintenance of anaesthesia. This was done by enabling automatic gas control (AGC) mode, which is the low fresh gas flow, target control mode on the Flow-i system. AGC was set to a speed setting of 6/10, with a target end-tidal sevoflurane concentration of 1.0%. Intravenous propofol infusion was continued to provide the balance of required anaesthetic depth, targeting a BIS of 35–50. Body temperature maintenance was performed with a forced air warming device to target a core body temperature, monitored using a nasopharyngeal temperature probe, of 36°C or higher.
In addition to standard sidestream tidal gas concentration monitoring, a Datex Capnomac Ultima anaesthetic agent monitor (GE/Datex-Ohmeda, Helsinki, Finland) was also connected, which was calibrated according to the manufacturer’s specifications. This was interfaced to a notebook computer (Macbook Air, Apple Corp, Cupertino, CA, USA) via an analog-digital converter (USB 900, National Instruments, Austin, TX, USA) running LabVIEW 2011 (National Instruments, Austin, TX, USA) to allow real-time download and capture on hard disk of all raw gas concentration waveforms at the measurement timepoints for the study. Tidal volume and frequency, PEEP, peak inflation pressures and dynamic lung compliance were also recorded along with heart rate, blood pressure and cardiac output estimated by the Vigeleo/FloTrac.
The first (baseline) set of measurements was taken after establishment of pneumoperitoneum and positioning of the operating table for surgery for a minimum of ten minutes, with stability (less than 5% relative variation) in all tidal gas concentrations and in systolic blood pressure and cardiac output (less than 20% variation) over the previous five minutes. Tidal gas concentrations were recorded while 10 ml arterial blood samples were simultaneously collected in two heparinised 20 ml gas-tight glass syringes. Oxygen (O2) and carbon dioxide (CO2) partial pressures were measured at point of care on an ABL 800 blood gas analyser (Radiometer, Odense, Denmark), and measurement of sevoflurane partial pressure in blood at body temperature was done by the double headspace equilibration technique using a previously described method. 15 This consisted of equilibration of blood with an equal volume of room air in a heated shaking water bath for a minimum of 45 minutes, followed by decanting of the headspace gas into a second glass syringe for injection into the same Datex Capnomac Ultima gas analyser used to measure tidal gases at the time of blood sampling, with waveform capture. This technique allows capture of a clear gas partial pressure waveform plateau to identify headspace gas concentrations accurately. 17 All gas partial pressure measurements were adjusted to body temperature and pressure saturated (BTPS) for subsequent calculations.
Immediately following the collection of baseline samples, the ventilation intervention to which the patient was randomly assigned was commenced. In the control group this was simple continuation of the same ventilation settings as at baseline. In the intervention group, an automated stepped lung RM was performed using the automated RM (auto-RM) function embedded in the Flow-i system. This manoeuvre involved stepwise increases in PEEP and peak inflation pressure, while remaining in pressure control ventilation mode, to a target of 20 cmH2O and 40 cmH2O, respectively, over a period of 75–90 seconds. Following this, PEEP was continued starting at an arbitrary setting of 12–14 cmH2O. Achievement of optimal individualised PEEP for the patient was evaluated by monitoring breath-to-breath dynamic lung compliance (Cdyn) following the RM. If this subsequently declined to a threshold 20% below the maximal post-recruitment value, a repeat automated RM was performed, and PEEP subsequently continued at 2 cmH2O higher than previously to seek the threshold for individualised PEEP for the patient.
After a minimum of 30 minutes and a maximum of 90 minutes, during a similar period of stability in monitored variables and with the patient in the same position as at the first measurement point, a second set of measurements was collected using the same protocol as at baseline. The randomised ventilatory intervention was then continued throughout surgery. At the end of wound closure, neuromuscular blockade was reversed with neostigmine and an antimuscarinic agent and sevoflurane and propofol administration terminated, according to standard practice, while AGC mode continued. Positive pressure ventilation was ceased and the patient was converted to spontaneous breathing with pressure support. Extubation occurred when the patient spontaneously opened their eyes to verbal command repeated every 30 seconds. Time to extubation from end of surgery (last stitch), and total sevoflurane consumption throughout the whole case by the Flow-i data logging system, were both recorded.
The trial endpoints were variables representing efficiency of alveolar–capillary lung gas exchange. The primary trial endpoint was comparison between the two groups in measured (a–A ratio) for sevoflurane at the second timepoint.
Secondary endpoints which were also compared between the two groups at the second timepoint were measured: arterial oxygen partial pressure (PaO2)/FiO2 concentration ratio (mmHg), driving pressure, alveolar deadspace fraction (Vda/Va) calculated from the modified Bohr–Enghoff equation using arterial and end-expired CO2 partial pressures. Because of the relatively small sample size of the trial, the change from baseline in these variables was also compared to control for any baseline differences between the two groups. The mean sevoflurane consumption (ml per hour) over the course of the anaesthetic was also compared.
For the sample size calculation, we nominated a relative difference of 20% in the a–A ratio for sevoflurane as clinically significant. From previously published data, a range of 20% either side of this is expected. Using a t-test for unpaired data, at an alpha of 0.05 and a beta of 0.8, 32 patients were required. Allowing for protocol deviations and data attrition, enrolment of 40 patients was planned. Summary data for measured variables used the mean and standard deviation (SD) for normally distributed data, or median and interquartile range (IQR) of difference for non-normal data. 25 Statistical significance testing was done with the paired t-test for normally distributed data confirmed by the Shapiro–Wilk test, and the Wilcoxon rank sum test for non-normal data. The significance of comparison of groups in change in variables from baseline was determined using analysis of variance (ANOVA) examining group–time interaction. All statistical tests of significance were performed using Stata 12 (Stata Corp, College Station, TX, USA), using a significance threshold of P = 0.05 for the primary endpoint, with Bonferroni adjustment for multiple comparisons for the secondary endpoint variables.
Results
Of all eligible patients approached to participate in the trial, four declined. Forty consenting patients were enrolled over an 18-month period. In four cases, the protocol was unable to be administered in full because of surgical decision intraoperatively to abandon laparoscopy and convert to open laparotomy to complete bowel resection before the second set of data could be collected. The final dataset therefore consisted of complete data from 17 control group and 19 intervention group patients. There were three active and three former smokers in the control group, and eight active and one former smoker in the intervention group. In the control group the mean (SD) age was 59.4 (21.9) years and BMI 26.7 (4.3) kg/m2, and in the intervention group age was 53.2 (17.6) years and BMI 27.3 (5.9) kg/m2. The mean (SD) time between commencement of laparoscopic insufflation and the second set of measurements was 66 (14) minutes in the control group and 61 (15) minutes in the intervention group, and the mean (SD) time between the first and second sets of measurements was 46 (12) minutes and 41 (15) minutes, respectively. Total duration of surgery was 254 (74) minutes in the control group, and 272 (77) minutes in the intervention group.
Mean tidal volume, PE’CO2 and driving pressure (peak inspiratory pressure (PIP) minus PEEP) were similar between the two groups at the first timepoint, as were the median respiratory rate, FiO2 and dynamic lung compliance (Cdyn).
At the second timepoint, the mean tidal volume, PE’CO2, median respiratory rate and FiO2 were unchanged in both groups. Median (IQR) PEEP in the intervention group was 14 (13.5–14) cmH2O. Driving pressure was lower compared to the control group (mean (SD) = 13.6 (3.1) versus 21.5 (5.2) cmH2O, P < 0.001). Cdyn was higher in the intervention group compared to the control group (median (IQR) = 50 (40–68.5) versus 25 (20–29) ml/cmH2O, P < 0.001). These ventilatory variables are summarised in Table 1.
Measurements of ventilatory variables at the first and second timepoints in each group.
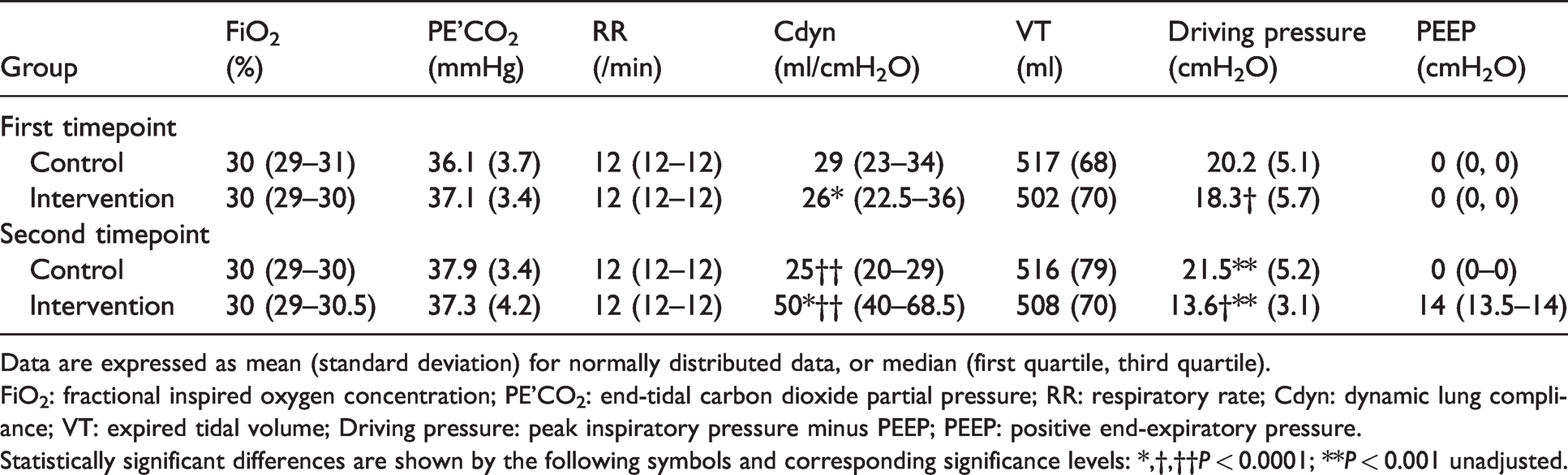
Data are expressed as mean (standard deviation) for normally distributed data, or median (first quartile, third quartile).FiO2: fractional inspired oxygen concentration; PE’CO2: end-tidal carbon dioxide partial pressure; RR: respiratory rate; Cdyn: dynamic lung compliance; VT: expired tidal volume; Driving pressure: peak inspiratory pressure minus PEEP; PEEP: positive end-expiratory pressure.
Statistically significant differences are shown by the following symbols and corresponding significance levels: *,†,††P < 0.0001; **P < 0.001 unadjusted.
There was no significant difference in the a–A ratio for sevoflurane at the first timepoint (mean (SD) control group = 0.752 (0.061) versus intervention group = 0.741 (0.070), P = 0.637). This persisted at the second timepoint (mean (SD) control group = 0.754 (0.086) versus intervention group = 0.785 (0.099), P = 0.319 unadjusted), see Table 2. There was no significant difference between groups in change from baseline in a–A ratio for sevoflurane on ANOVA examining group–time interaction (P = 0.081 unadjusted).
Measured gas exchange variables at the first and second timepoints in each group.
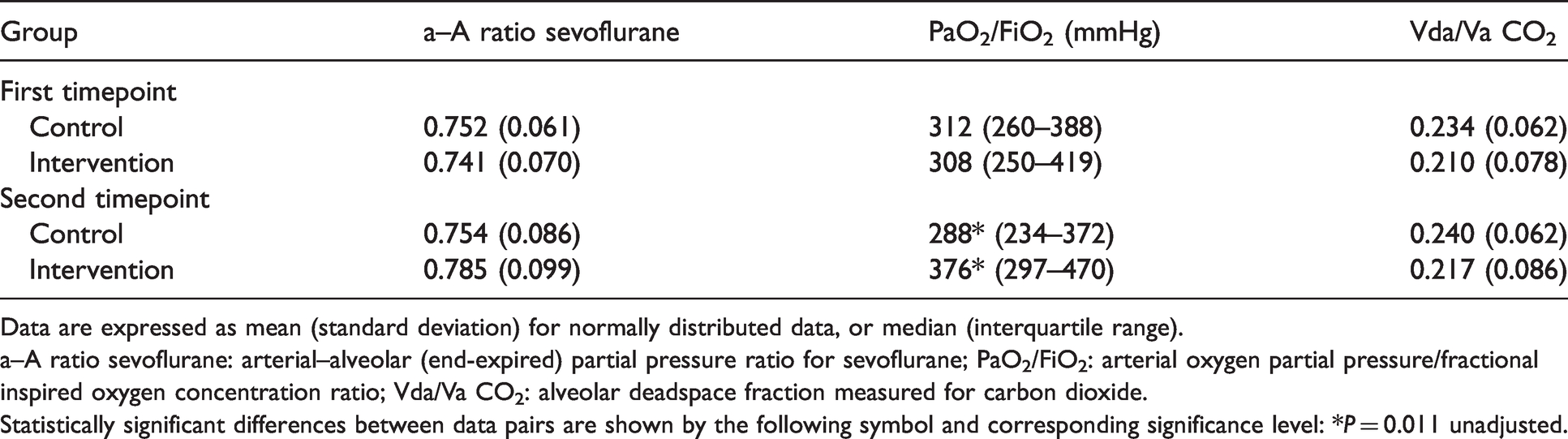
Data are expressed as mean (standard deviation) for normally distributed data, or median (interquartile range).
a–A ratio sevoflurane: arterial–alveolar (end-expired) partial pressure ratio for sevoflurane; PaO2/FiO2: arterial oxygen partial pressure/fractional inspired oxygen concentration ratio; Vda/Va CO2: alveolar deadspace fraction measured for carbon dioxide.
Statistically significant differences between data pairs are shown by the following symbol and corresponding significance level: *P = 0.011 unadjusted.
The PaO2/FiO2 ratio was similar in the two groups at baseline (median (IQR) 312 (260–388) mmHg in the control group versus 308 (250–419) mmHg in the intervention group, P = 0.837), and increased in the intervention group at the second timepoint (median (IQR) 288 (234–372) mmHg versus 376 (297–470) mmHg, P = 0.011 unadjusted), see Table 2. This difference was not statistically significant after Bonferroni adjustment for multiple comparisons. However, the increase from baseline in the PaO2/FiO2 ratio in the intervention group compared to the control group remained significant on ANOVA examining group–time interaction (P < 0.001 adjusted).
The alveolar deadspace fraction for CO2 was similar between the two groups at the first timepoint (mean (SD) control group = 0.234 (0.062) versus intervention group = 0.210 (0.078), P = 0.314 unadjusted), and remained so at the second timepoint (mean (SD) control group = 0.240 (0.062) versus intervention group = 0.217 (0.086), P = 0.373 unadjusted), see Table 2. There was no significant difference between groups in the change from baseline in alveolar deadspace fraction for CO2 from baseline on ANOVA examining group–time interaction (P = 0.081 unadjusted).
The sevoflurane consumption rate adjusted for body weight was not statistically different between the two groups (median (IQR) control group = 0.059 (0.057–0.070) versus intervention group = 0.075 (0.062–0.087) ml/hour/kg, P = 0.065 unadjusted). Time to eye opening from end of surgery was similar in both groups (mean (SD) control group = 20 (10) versus intervention group = 16 (7) minutes, P = 0.204 unadjusted).
Discussion
We did not find any effect of the open lung intervention on the a–A gradient for sevoflurane, nor on other possible indicators of sevoflurane gas exchange efficiency, such as the overall volatile agent consumption rate. In principle, a recruited lung will have an increased diffusion area and reduced shunt fraction, leading to more efficient overall gas exchange. We found a 30% increase in intraoperative PaO2 compared with standard ventilation involving zero PEEP, at identical FiO 2 and similar levels of overall alveolar ventilation and CO2 clearance. This occurred in parallel with a doubling of Cdyn, confirming that the RM achieved and maintained an increased end-expiratory lung volume (EELV). Our 30% improvement in PaO2/FiO2 contrasts with an approximately 10% increase found with a similar open lung strategy compared to standard ventilation in the iPROVE trial. 16 This difference may be due to a mixture of open and laparoscopic surgery, as well as the delivery of PEEP at 5 cmH2O in the standard ventilation group in iPROVE, but further indicates that the open lung strategy achieved a significant difference in lung mechanics and oxygenation efficiency between the two groups in our study. This is also demonstrated by the fact that in addition to improved oxygenation and doubling of compliance, there was a significant reduction in mean driving pressure in the intervention group of 7.9 cmH2O compared to the control group. As the tidal volume was the same in both groups, this reflects a larger EELV in the intervention group, placing this group on a different part of the lung pressure–volume (compliance) curve compared to the control group.
However, our findings indicate that the specific effect of open lung management on arterial oxygenation is not generalisable to other gases which undergo alveolar–capillary exchange. The likely reasons for this lie in the relationship of lung gas exchange with the distribution of Va/Q ratios across the lung during general anaesthesia, which is different for gases of differing solubility in blood. This was recently demonstrated in a study measuring the partial pressure cascade and alveolar deadspace for four different volatile anaesthetic agents and CO2 in anaesthetised, ventilated cardiac surgery patients. Physiological distributions of alveolar ventilation and blood flow in the lung can be characterised using the multiple inert gas elimination technique (MIGET),18,19 and application of this to anaesthetised patients has demonstrated an increase in Va/Q mismatch.20–25 Multicompartment computer modelling of predicted gas exchange simulating typical physiological distributions of alveolar ventilation and blood flow during anaesthesia has shown how the distribution of CO2 elimination closely follows that of alveolar ventilation, while the distribution of O2 uptake follows that of pulmonary blood flow, differences which are driven by their widely different blood solubilities. The distribution of sevoflurane, with intermediate solubility lies in between, but relies relatively little on low Va/Q lung segments and is therefore not expected to be affected by recruitment of these lung units. 26 Our study design, which measured gas exchange efficiency for all three gases before and after lung recruitment, therefore provides support for the predictions of this physiological model of lung gas exchange. Consistent with this, alveolar deadspace was not increased by the intervention, suggesting that the recruitment of collapsed low Va/Q ratio lung units that resulted in improved arterial oxygenation had minimal effect on the effective alveolar ventilation of high Va/Q lung regions and accompanying a–A gradients for CO2.
Results from the RCTs published in recent years on intraoperative ventilation strategies have in many ways been contradictory. There are several reasons for that. Some trials have used manual continuous positive airway pressure (CPAP) manoeuvres and others manual PEEP step-based recruitment, which may improve the effectiveness of the recruitment process while minimising associated lung barotrauma and circulatory compromise. Two large trials have used a standardised high PEEP setting (e.g. 12 cmH2O) after manual lung recruitment, while others have sought an individualised PEEP level, which achieves maintenance of a measured increase in dynamic lung compliance without the need for repeated further RMs.14–16 Implementation and standardisation of this open lung protocol is made easier by automation of the process, such as is made available on the Flow-i anaesthesia machine used in the current study.
There are some other conceivable causes for the conflicting results in those RCTs investigating different lung protective concepts and PPC. First, the nature of the RMs employed have been variable, including manual CPAP manoeuvres, few breaths with larger tidal volumes (sighs) and PEEP-stairways, with different inflation pressures, volumes and durations. Second, very few RCTs have presented any continuous measure of lung compliance or driving pressure demonstrating a lasting positive effect of the manoeuvre on lung mechanics and association with improved oxygenation. Third, most large trials have for practical reasons involved a variety of anaesthesia machines, with different performance, especially at higher inflation pressures, and some with limited ability to set higher PEEP levels.
Furthermore, most large trials have studied a mixed population undergoing either open or laparoscopic abdominal surgery, which may further cloud interpretation of their findings. Laparoscopic surgery presents particular challenges to ventilatory management and homeostasis intraoperatively, with substantial reductions in lung compliance produced by peritoneal insufflation and steep Trendelenburg positioning of the patient for prolonged periods of time.1–10 In contrast, postoperative factors such as acute postoperative pain are likely to contribute less to PPCs than in open surgery. For this reason, it may be the case that the potential benefits of an intraoperative open lung approach are most likely to be found in the laparoscopic surgery population. Data from the PROBESE trial suggest a strong trend towards fewer complications with recruitment and high PEEP when the minority of patients in the trial who underwent open abdominal surgery are excluded from the analysis. 15 This is supported by data from a recent meta-analysis from Serpa Neto et al. of 17 RCTs including 2250 patients, who found that driving pressure was the main determinant for PPCs. 11 They concluded that intraoperative high driving pressure and changes in the level of PEEP that result in an increase of driving pressure are associated with more PPCs. Their results are also in alignment with the findings of Amato et al. after recalculation of ARDSnet (ARDS, acute respiratory distress syndrome) data. 27
In our study the RM reduced driving pressure significantly to a suggested protective level for normal lungs and dynamic lung compliance (Cdyn) doubled. 12 Improved oxygenation and reduced driving pressure were maintained at least through to the second measurement timepoint by individualised high PEEP, which ranged between 12 and 15 cmH2O, with a median of 14 cmH2O. The high PEEP level was well tolerated and none of the studied patients in the intervention group needed any vasoactive support following the commencement of the RM.
Data on the effects of open lung ventilatory management on gas exchange in laparoscopic surgery have been lacking. As far as we know our study is the first investigating the effect of open lung ventilatory management on anaesthetic agent uptake as well as on the respiratory gases. Its limitations are that it was a small trial with a primarily mechanistic focus designed to measure intraoperative gas exchange variables, and measurement of lung mechanics in a laparoscopic surgical population. More direct methods for the demonstration of lung segment recruitment, such as imaging techniques for instance, were beyond the limitations of our study and not adaptable to an intraoperative environment. In common with other trials in the field, the intervention was not able to be double-blinded. No attempt was made to collect data on postoperative outcomes including PPCs, which would require a much larger randomised trial to achieve adequate statistical power. Arguably, any potential pharmacoeconomic advantage we sought from the effect of improved gas exchange efficiency from RMs on sevoflurane uptake would be relatively small compared with overall operative costs.
In conclusion, we found no evidence of increased efficiency of gas exchange for sevoflurane from open lung ventilatory management in laparoscopic surgical patients, despite clear evidence of improved arterial oxygenation associated with recruitment of poorly ventilated, low Va/Q lung segments. The PEEP-stairway lung recruitment decreased ventilatory driving pressure to lung protective levels without any demonstrable increase in pulmonary alveolar deadspace. Despite no evident pharmacoeconomic advantage, the implications of this for PPC risk are worthy of subsequent further investigation in a larger clinical trial.
Footnotes
Data sharing statement
Data collected in this study is available on application to the corresponding author.
Author Contribution(s)
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest: Philip Peyton received a paid research consultancy from Maquet/Getinge for this project. Mats Wallin was an employee of Maquet/Getinge at the time the study was conceived and conducted.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article. Philip Peyton received a paid research consultancy from Maquet/Getinge for this project. Mats Wallin was an employee of Maquet/Getinge at the time the study was conceived and conducted.
