Abstract

Cricothyroidotomy is considered the preferred emergency front-of-neck airway access technique, as it has shown a significantly reduced incidence of late complications compared to emergency tracheostomy. 1 Even so, when attempting cricothyroidotomy during the acute phase of a ‘Can’t oxygenate’ crisis, failure to achieve an airway in adequate time is still possible, and more likely in patients with difficult necks where the cricothyroid membrane (CTM) is poorly palpable. Difficult necks may occur in obese patients and in conditions associated with neck oedema or altered airway anatomy.
When the CTM is impalpable, current Difficult Airway Society (DAS) guidelines recommend performing an 8–10 cm incision to aid in locating it (plan D). 2 Such length incisions could act as a psychological barrier in the transition to cricothyroidotomy. 3 Also, anaesthetists performing plan D in difficult neck simulations achieved only low cricothyroidotomy success (<46%) despite long skin incisions (up to 14 cm). 4 Although most achieved ventilation (>96%), this appears not infrequently to have been via airways placed through the laryngeal cartilages or trachea, and times to ventilation were relatively long (median ≥105 seconds). 4
Cannula cricothyroidotomy remains a possible clinical option even in difficult necks (with support from United States and Australian guidelines) and it can avoid long incisions, but it has shown low success (40%) in difficult neck simulations.5–7 Furthermore, when a cannula is used for infraglottic access in the difficult neck, it may kink or migrate out of the infraglottic airway more easily than an endotracheal tube.
We hypothesised that point-of-care ultrasound guidance may facilitate minimally invasive (single-stab) scalpel cricothyroidotomy during ‘Can’t oxygenate’ emergencies in patients with difficult necks. Ultrasound-guided (US) single-stab scalpel-bougie cricothyroidotomy has shown high success (95%) in cadavers with body mass index (BMI) ranging from 12.2 to 44.9 kg/m2. 8 However, many may not have had difficult necks (mean BMI only 21.9 kg/m2), and there was no control group. 8 We performed a simulation experiment to assess the accuracy and speed of US single-stab scalpel-bougie cricothyroidotomy in simulated difficult neck models compared to non-US cannula cricothyroidotomy.
After ethics committee approval (UTas H0018256), this experiment was conducted at the Launceston General Hospital on 16 December 2019 and 10 June 2020. The sole participant was the first author (KG, the proceduralist, an anaesthetist experienced in airway ultrasonography). Sixty-two bespoke phantom models were pre-produced to simulate difficult adult human necks, with invisible and poorly palpable airway landmarks, see Figure 1. We have named these DiffNeckModels. The airway of each model used Microfoam (3M, North Ryde, NSW) to simulate the CTM, and this was covered with enough opaque, thickened jelly to produce CTM depths similar to that found in patients with a BMI of 45 kg/m2 or greater. 9 The CTM position within each DiffNeckModel was randomly distributed among nine possible locations (all within 1 cm laterally or longitudinally from the model’s midpoint, which was the default position to simulate the assumed normal transverse level of the CTM in the midline). This was to mimic human anatomical variations and was double-blinded. Each DiffNeckModel was also randomly assigned to receive either US or non-US cricothyroidotomy. The sample size (62 DiffNeckModels: 31 per cricothyroidotomy technique group) was based on pre-study estimation of US cricothyroidotomy success to be 90% and non-US 50%, with α = 0.05 and power of 80%.
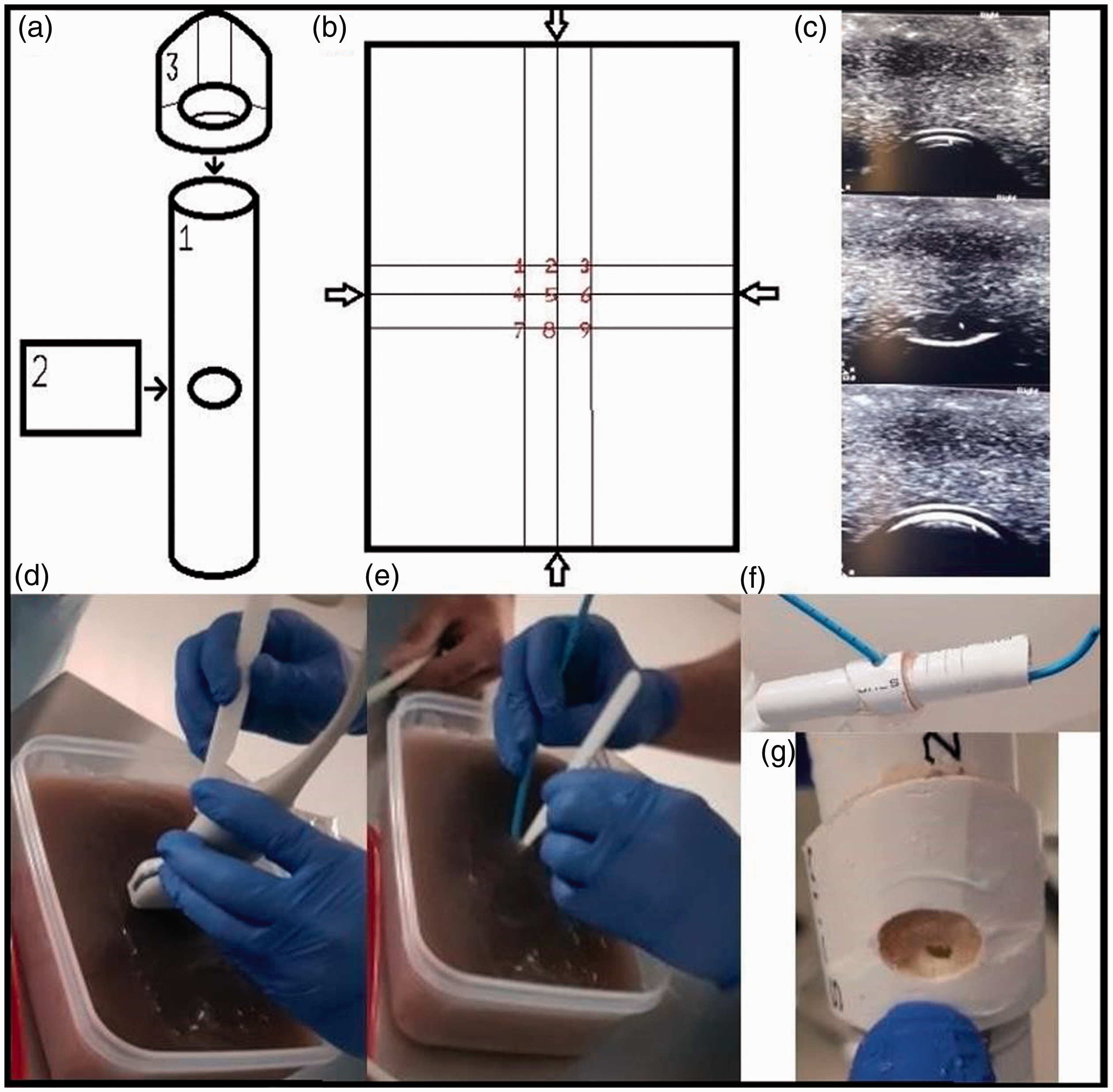
DiffNeckModels and ultrasound-guided (US) cricothyroidotomy. From top left to bottom right: (a) The simulated airways embedded within DiffNeckModels were constructed of two different sized and shaped polyvinyl chloride pipe pieces (1 and 3) fitted together with Microfoam (3M, North Ryde, NSW) (2) in between to simulate the cricothyroid membrane (CTM). (b) The nine possible positions within a DiffNeckModel’s container where the simulated CTM could be positioned depending on randomisation, with arrows indicating the container’s middle point. (c) Transverse ultrasonographic views of a pre-study DiffNeckModel’s thyroid cartilage (top), cricothyroid membrane (middle) and cricoid cartilage (bottom). (d) US cricothyroidotomy technique involves inserting the scalpel blade at the midpoint of the cephalad aspect of the transducer once CTM is ultrasonographically located. (e) Blade rotated after cricothyroidotomy to aid bougie placement. (f) Successful bougie placement (airway shown after removal of jelly). (g) Cricothyroidotomy puncture site.
US cricothyroidotomy involved transverse ultrasonography to locate the CTM. Once the transducer was positioned over the CTM, the scalpel blade was passed into the jelly surface adjacent to the middle of the cephalad side of the transducer (similar to the technique described in Curtis and colleagues), 8 aiming for cricothyroidotomy. Scalpel advancement was originally intended to be further aided with real-time guidance. 10 However, the proceduralist did not routinely employ this, rather relying on embedded airway feedback when required. Once through the CTM the transducer was put aside, the scalpel turned 90 degrees to aid bougie insertion, after which, the scalpel was removed.
In the non-US cricothyroidotomy group, up to five strategic attempts were permitted to place a 14 G Insyte (Becton Dickinson, Macquarie Park, NSW) cannula through the CTM while aspirating an attached 5 ml syringe containing 2 ml of 0.9% isotonic saline. The initial attempt was performed at the midpoint of the model. Then, if required, further attempts were performed 1 cm each side of the first attempt, then at 2 cm each side of the first attempt. Once the CTM was traversed and air aspirated, the cannula was railroaded and the needle was then removed.
Independent observers assessed cricothyroidotomy accuracy (if either the bougie or cannula was successfully placed in the airway via the CTM) and speed (duration from starting an attempt until completion or failure). The two cricothyroidotomy groups were compared for success rate using Fisher’s exact test, and for mean procedural speed using Student’s t-test.
On first skin puncture, US cricothyroidotomy was overall more accurate than non-US (68%, 21/31 versus 16%, 5/31; P < 0.001). The accuracy of US cricothyroidotomy was higher again (81%, 21/26), with an acceptable mean (standard deviation, SD) speed of 76 (30) seconds, in our DiffNeckModels that had ultrasound visualisable airways. However, in our DiffNeckModels that unexpectedly did not have ultrasound visualisable airways, US cricothyroidotomy was completely ineffective (0%, 5/5). Non-US cricothyroidotomy, even with up to five strategically placed skin puncture attempts, still had low accuracy compared to all first skin puncture US cricothyroidotomy attempts (29%, 9/31; P=0.002), despite faster mean (SD) speed of 34 (13) seconds.
This study had limitations which included a single proceduralist. Assessing the success of first pass, real-time guided, US cricothyroidotomies (as originally intended) was not possible as it was not consistently employed by the proceduralist; instead first skin puncture was reported (as was considered usually recorded). 10 Airway injuries were noted in the models in both groups (US 6%, 2/31 versus non-US 10%, 3/31; P=0.57). However, their material was harder than human cartilage and we cannot comment on the likelihood or otherwise of airway injuries in this simulation study.
In conclusion, in this study of a single experienced proceduralist, a modified US-guided minimally invasive scalpel cricothyroidotomy technique was more accurate than non-US cannula cricothyroidotomy in bespoke phantom difficult neck models. Further research into the safety and efficacy of US-guided minimally invasive cricothyroidotomy techniques for difficult necks is warranted.
Supplemental Material
sj-pdf-1-aic-10.1177_0310057X211042387 - Supplemental material for A comparison of a modified ultrasound-guided minimally invasive scalpel cricothyroidotomy technique with non–ultrasound-guided cannula cricothyroidotomy using phantom difficult neck model simulations
Supplemental material, sj-pdf-1-aic-10.1177_0310057X211042387 for A comparison of a modified ultrasound-guided minimally invasive scalpel cricothyroidotomy technique with non–ultrasound-guided cannula cricothyroidotomy using phantom difficult neck model simulations by Karl J Gadd: on behalf of Anaesthesia Quality Improvement New Zealand in Anaesthesia and Intensive Care
Footnotes
Author contributions
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Launceston General Hospital’s Anaesthetic Department Trust Fund funded the cost of some externally purchased materials used in this research.
Acknowledgements
The authors would like to than I Robertson, Clifford Craig Foundation, Launceston, for study design advice.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
