Abstract
Peripheral venous cannulation (PVC) is a commonly performed invasive medical procedure. Topical treatments such as the eutectic mixture of local anaesthetics (EMLA®, Aspen Pharmacare Australia Pty Ltd, St Leonards, NSW) attenuate the associated pain, but are limited by requiring up to one hour of application before becoming effective. The Coolsense® (Coolsense Medical Ltd., Tel Aviv, Israel) pain numbing applicator is a new device using a cryoanalgesic means to anaesthetise skin within seconds. Coolsense is being increasingly used for cannulation, but comparative studies are lacking. We recruited 64 healthy adult volunteers to this open-label two sequence, two period randomised crossover trial. Participants had two 20 gauge venous cannulae inserted, one on the dorsum of each hand. Each cannulation attempt was preceded by treatment with Coolsense or an EMLA patch containing 2.5% lidocaine and 2.5% prilocaine. The primary outcome was participant pain using the 0–10 numerical pain rating scale. Secondary outcomes were participant satisfaction scores on a 0–10 scale, treatment preference, and failed cannulation attempts. Participants were randomly assigned to either the Coolsense EMLA (n = 32) or EMLA Coolsense (n = 32) sequence. All participants completed the trial. The pooled mean paired difference of the numerical pain rating scale was –1.84 (95% confidence intervals –1.28 to –2.41; P < 0.001) in favour of EMLA. The pooled mean paired difference for satisfaction score was 2.26 (95% confidence intervals 1.46 to 3.07; P < 0.001) higher with EMLA. Most participants preferred EMLA over Coolsense (P < 0.001). There was no significant difference regarding failed cannulation between the two treatments (P = 0.14). Among healthy individuals undergoing elective PVC, EMLA was associated with reduced pain, increased satisfaction, and was the preferred treatment compared to Coolsense.
Introduction
Peripheral venous cannulation (PVC) is performed in up to 70% of all patients during their hospital stay1,2 and is a routine procedure to administer anaesthesia. The pain from PVC causes anxiety among many patients. 3 Needle phobia is a recognised psychological condition with physiological responses, and has an estimated prevalence of up to 10%. 4 Negative patient experiences with PVC may have a deleterious impact on subsequent engagement with healthcare provision. 5
The effectiveness of various interventions in reducing the pain of PVC in both adults and children has been extensively studied. These methods have included: eutectic mixture of local anaesthetics (EMLA®, Aspen Pharmacare Australia Pty Ltd, St Leonards, NSW); amethocaine (Ametop®, Smith & Nephew Healthcare Ltd, Hull, England); lidocaine/tetracaine (Rapydan®, Eurocept International BV, Ankeveen, Netherlands); transdermal diclofenac; intradermal lidocaine; ethyl chloride; nitrous oxide; communication; and distraction.6–19
A network meta-analysis of local anaesthetic interventions for PVC in an adult population 20 concluded that all topical and injected local anaesthetics were beneficial to reduce pain and no particular intervention within this class appeared unequivocally superior. The authors advocated the routine use of such treatments for PVC among adults, in keeping with common paediatric practice, and suggested it should be used as a care quality marker. 20
EMLA is the most widely studied topical local anaesthetic for PVC. 20 It has a similar efficacy to intradermal local anaesthesia; 20 benefits from not requiring infiltration with a needle for administration making it more appropriate for individuals with needle phobia or in paediatric practice. A pertinent limitation of EMLA is the time required to achieve a therapeutic effect, which is upwards of one hour of application. 21 The Coolsense® pain numbing applicator (Coolsense Medical Ltd., Tel Aviv, Israel) is a recently developed handheld cryoanalgesia device originally designed for needle-based cosmetic procedures. It can be used multiple times and in all ages. Coolsense uses an electronic means to control the temperature of the applicator head, which is applied directly to cool and anaesthetise an area of skin to produce the desired effect within seconds.
Comparative studies of Coolsense to date are limited in scope to those with diabetes for needle-prick–based glucose sampling compared to a non-cooled device 22 and for therapeutic botulinum toxin administration compared to a different cooling technique. 23 Despite this, increasing numbers of hospitals are adopting it as an analgesic device for PVC. A single-centre case series consisting of 100 patients who were six–18 years of age requiring PVC for anaesthesia or radiological contrast administration at a major Australian tertiary paediatric hospital reported high levels of satisfaction with Coolsense among both patients and their carers. 24 It concluded by calling for a randomised controlled trial (RCT) against topical local anaesthetic treatments for PVC, which appear to be absent from the medical literature.
We designed this RCT with the aim of examining the efficacy of Coolsense on reducing pain from PVC compared to EMLA, a widely used standard of care. Secondary aims were to examine participant satisfaction, treatment preference and failed cannulation attempts between the two treatments.
Materials and methods
We conducted an open-label two sequence, two period randomised crossover trial in line with Consolidated Standards of Reporting Trials (CONSORT) guidelines.25,26 These are commonly referred to as 2×2 or AB/BA trials. 27 This study was deemed to be outside the scope of review by the Health and Disability Ethics Committee of New Zealand after submission of the protocol because it involved a low-risk medical device. Local approval was granted by the University of Otago Research Office (RO#18115) and Māori consultation through Te Komiti Whakarite of Canterbury District Health Board. The RCT was registered with the Australian New Zealand Clinical Trials Registry (ACTRN12618001354279). Eligible participants were healthy adults aged 18 years or over and able to provide written informed consent. Exclusion criteria were refusal, current antiplatelet or anticoagulant therapy, allergy to chlorhexidine or isopropyl alcohol, or needle phobia. The trial was undertaken at Christchurch Public Hospital between September and December 2018. Participants were directly approached among healthcare professionals working in theatres at the hospital by a study investigator. There were no financial incentives.
Participants were informed they would have two 20 gauge venous cannulae inserted, one on the dorsal aspect of each hand. The proposed site of vascular access on one hand was anaesthetised with the application of a topical EMLA patch containing 2.5% lidocaine and 2.5% prilocaine for a minimum of 60 minutes. EMLA was chosen as the control arm for this RCT because if the main target of Coolsense were children or those with needle phobia, which are where it has gained most traction for its use in recent years, then intradermal injection of local anaesthesia was not considered an appropriate comparator. EMLA is also ubiquitously available in hospitals compared with other topical treatments and is often applied by nursing staff using a standing order rather than a prescription. The other hand was anaesthetised using Coolsense in line with the instructions of the manufacturer. This involved removal of the device from a freezer just prior to the PVC attempt, confirmation of the correct operating temperature for use by a displayed green light, use of the alcohol gel applicator, application of the device to the desired area of skin for ten seconds and PVC attempted within six seconds.
A crossover design was suitable due to the stable conditions among participants and each intervention being of short duration. This was coupled with the short interval between intervention and outcome that would avoid issues such as participant dropout between treatment periods. The design resulted in an assessment of both interventions for each participant who would act as their own control. The differences were therefore based on within-subject comparisons rather than between-subject comparisons as would be the case in a parallel arm design. 27 This meant less variability and increased precision, meaning far fewer participants were required. A lengthy washout period was deemed not to be required given the lack of any systemic pharmacological carryover between treatment periods and different sites of cannulation. Adult volunteers were chosen as the population in this first-time comparison study due to easier recruitment and because a crossover design was inappropriate among children.
Block randomisation ensured an even distribution of participants who were primarily allocated to one of two treatment sequences, namely Coolsense EMLA or EMLA Coolsense. They were also randomly allocated as to which of the left or right hand would undergo PVC first. This resulted in four blocks: Coolsense EMLA left; Coolsense EMLA right; EMLA Coolsense left; and EMLA Coolsense right. The block allocation was written on an EMLA patch and all were placed in an opaque container. The enrolled participant picked his or her own patch from the container that would be used for the study and provided the allocation.
Once the EMLA patch had been applied to the designated hand over a site determined by a study proceduralist for at least 60 minutes, the participant was then seated in a hospital treatment or anaesthetic room. The Coolsense device was checked to ensure suitability of its use as previously outlined. The same neutral phrase (‘procedure’) was said by the proceduralist to alert the participant moments before each PVC attempt. There was one attempt at penetrating the skin and cannulating the chosen vein. Multiple needle passes under the skin were not permitted. All data were collected from participants once both PVC attempts had been completed and recorded on a data collection sheet. Both cannulae were then removed and pressure dressings applied. The application of each treatment and attempts at PVC were performed by the same proceduralist for each participant. There were two proceduralists, one an anaesthesia Fellow the other an anaesthesia specialist.
The primary outcome was participant rated pain for each treatment using the 0–10 numerical pain rating scale (NPRS). 28 This was chosen due to its simplicity, speed and because it could be administered verbally. The potential advantage of a visual analogue scale (VAS) being more sensitive to smaller changes was not deemed to be essential. Secondary outcomes were participant satisfaction score on a 0–10 scale, participant treatment preference and failed cannulation attempts. Any complications and qualitative feedback were also recorded.
For the primary outcome, the sample size calculation was based on an estimate of Coolsense having a mean NPRS of 3 and a standard deviation (SD) of 2, based on the only observational data of Coolsense for PVC. 24 The within-subject correlation coefficient for repeated measurements of this endpoint was conservatively assumed to be 50%. At a power of 90%, two-sided alpha value of 0.05 and allowing for up to 20% dropout or missing data, a total sample size of 63 participants was required to detect a mean difference NPRS of 1 with EMLA, which was deemed to be the minimum clinically relevant effect. 29
Data were entered into an Access database (Microsoft Corp, Redmond, WA, USA). Anonymised datasets were analysed using Stata, version 14 (StataCorp, College Station, TX, USA) and SAS, version 9.4 (SAS Institute, Cary, NC, USA).
There appears to be no firm consensus regarding the analyses of pain scores. 30 Some argue that measures such as the NPRS should be described and analysed using an ordinal scale 31 and others that measures such as the VAS represent a linear scale.32,33 There appears to be a very strong correlation between NPRS and VAS for acute pain. 29 As a result, data were described according to the distribution observed 34 and to minimise the loss of information, 35 particularly if the sample size appeared large enough to approximate a normal distribution.
Proportions and percentages were used to describe categorical data. Data were assessed for normality by their graphical distribution and the Shapiro–Wilk test. Paired differences between treatments for each sequence were calculated. Means, SDs and 95% confidence intervals (CIs) or medians, ranges and interquartile ranges (IQRs) were used to describe continuous data, depending on their distribution. The possibility of a period effect was also analysed. Parametric and non-parametric tests were used where appropriate to test hypotheses.
Results
Seventy-five individuals were approached to participate in the study, of whom 11 (14.7%) declined, resulting in 64 participants recruited (Figure 1). The overall median age was 30.5 years (range 21–58) and 42/64 (65.6%) were women. Baseline characteristics of participants by sequence are summarised in Table 1. Participants randomly assigned to either the Coolsense EMLA or EMLA Coolsense sequence resulted in 32/64 (50%) in each treatment arm. The first side to be cannulated was evenly divided among left or right in both sequences. All participants received the allocated treatments. There were no protocol violations, no dropouts and no missing data.
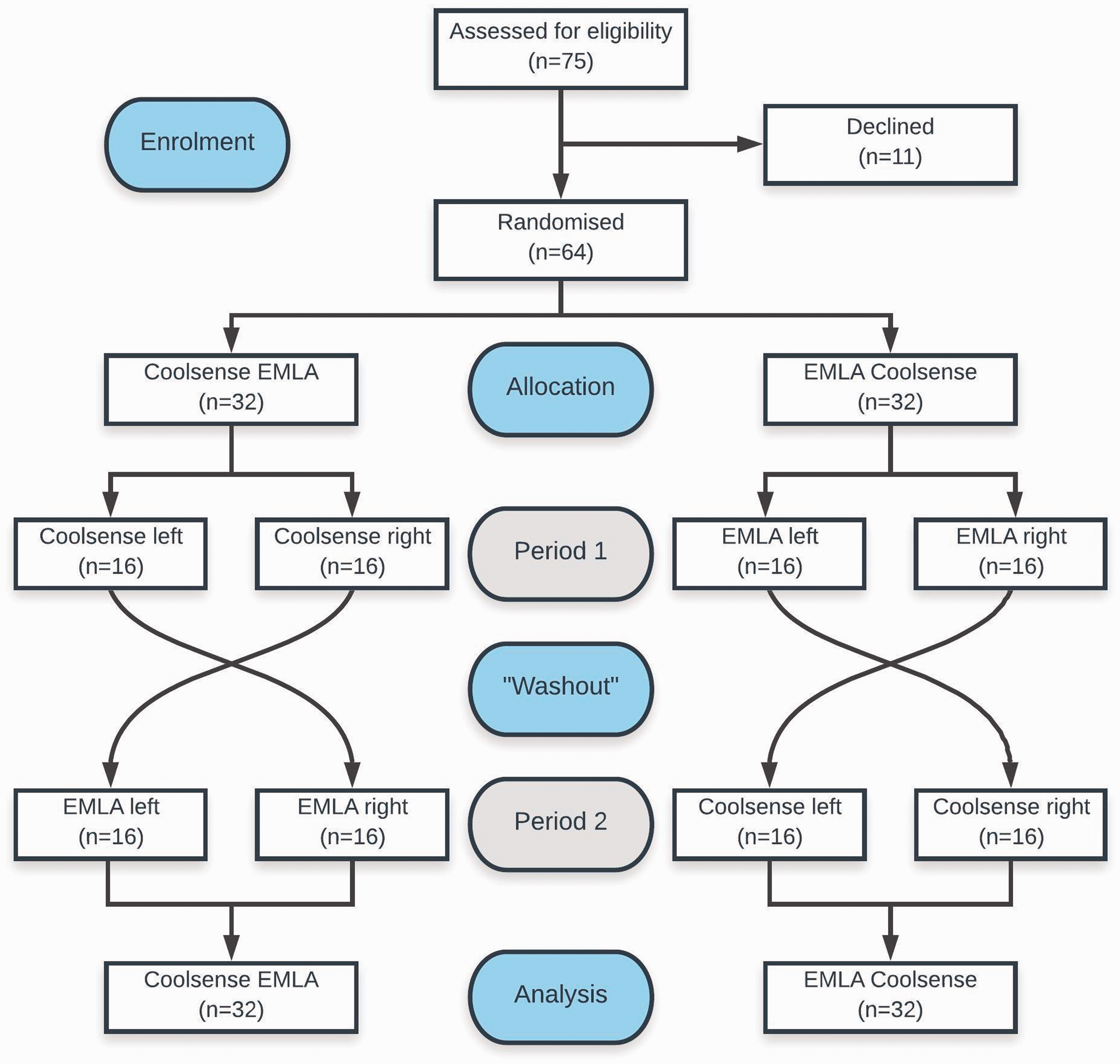
Consolidated Standards of Reporting Trials (CONSORT) diagram of participant recruitment and allocation. EMLA® (Aspen Pharmacare Australia Pty Ltd, St Leonards, NSW): eutectic mixture of local anaesthetics; Coolsense® (Coolsense Medical Ltd., Tel Aviv, Israel).
Baseline participant characteristics by sequence.
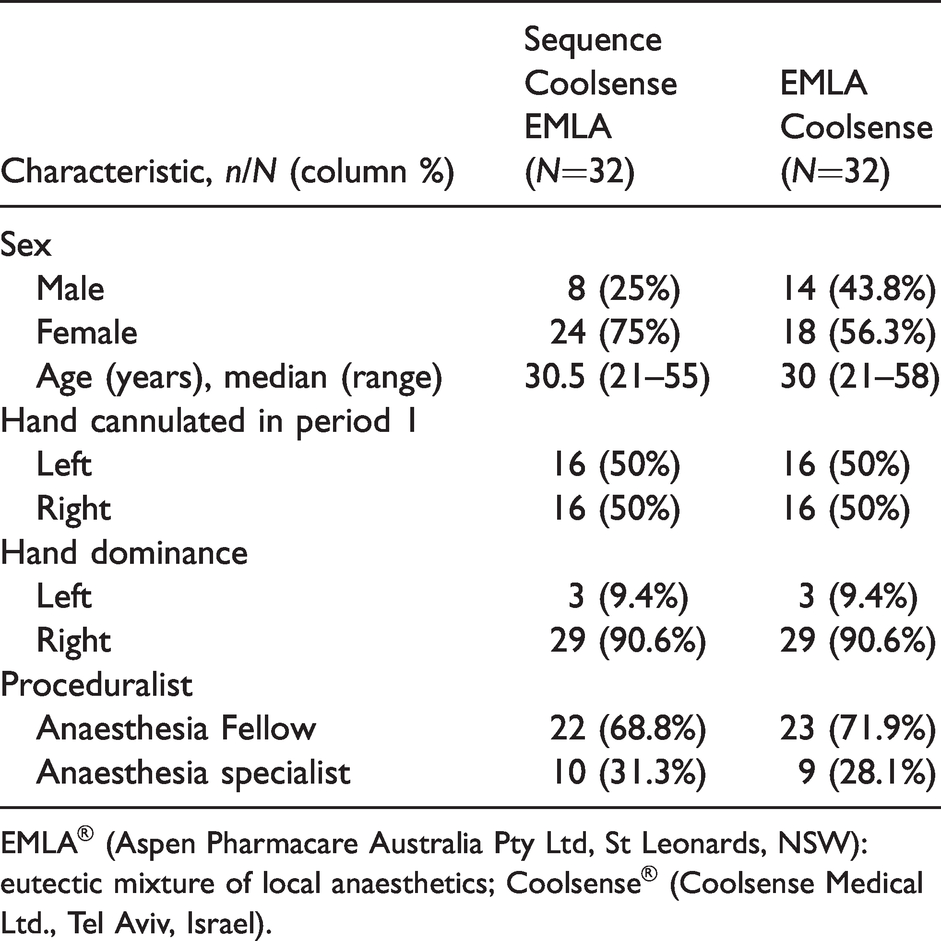
EMLA® (Aspen Pharmacare Australia Pty Ltd, St Leonards, NSW): eutectic mixture of local anaesthetics; Coolsense® (Coolsense Medical Ltd., Tel Aviv, Israel).
Regarding participant NPRS, responses are described in Figure 2. The NPRS for Coolsense may have approximated a normal distribution for both the Coolsense EMLA (P = 0.27, Shapiro–Wilk test) and EMLA Coolsense sequences (P = 0.31, Shapiro–Wilk test). However, for EMLA such individual responses were skewed for both the sequences (Coolsense EMLA P = 0.04 and EMLA Coolsense P < 0.001, Shapiro–Wilk test). These independent data were therefore described using non-parametric techniques. Paired responses for each participant are described in Figure 3. For such repeated measures, which are the true measures of interest for this crossover trial, these appeared to fit a normal distribution (Figure 4) for both the Coolsense EMLA (P = 0.84, Shapiro–Wilk test) and EMLA Coolsense sequences (P = 0.81, Shapiro–Wilk test). Data for the paired differences of NPRS were therefore analysed using parametric techniques.
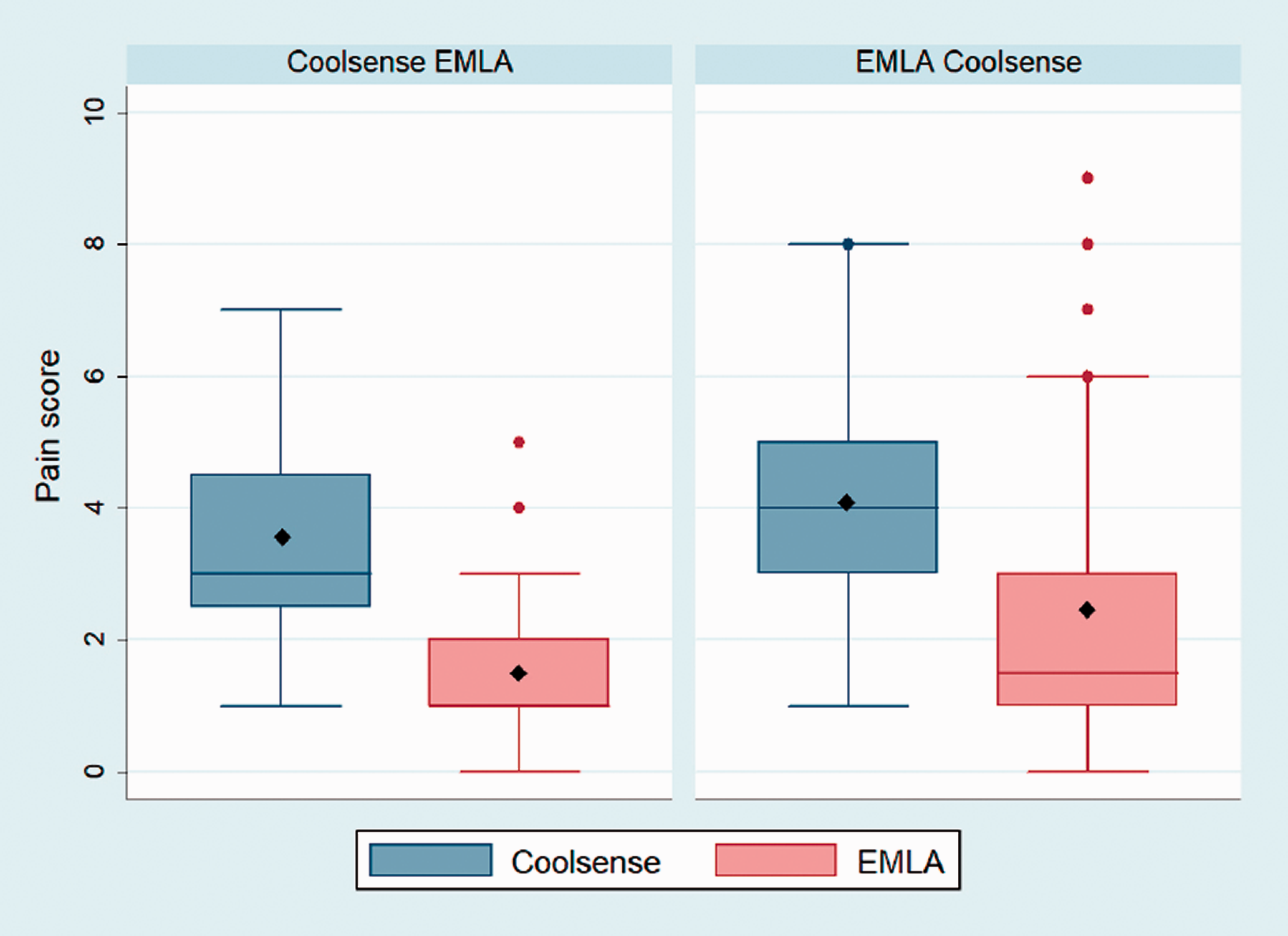
Box plots of pain scores by sequence. Data are median (horizontal line), mean (solid diamond), interquartile range (box), 1.5× interquartile range of nearest quartile (whisker) and outliers (solid circles). EMLA® (Aspen Pharmacare Australia Pty Ltd, St Leonards, NSW): eutectic mixture of local anaesthetics; Coolsense® (Coolsense Medical Ltd., Tel Aviv, Israel).
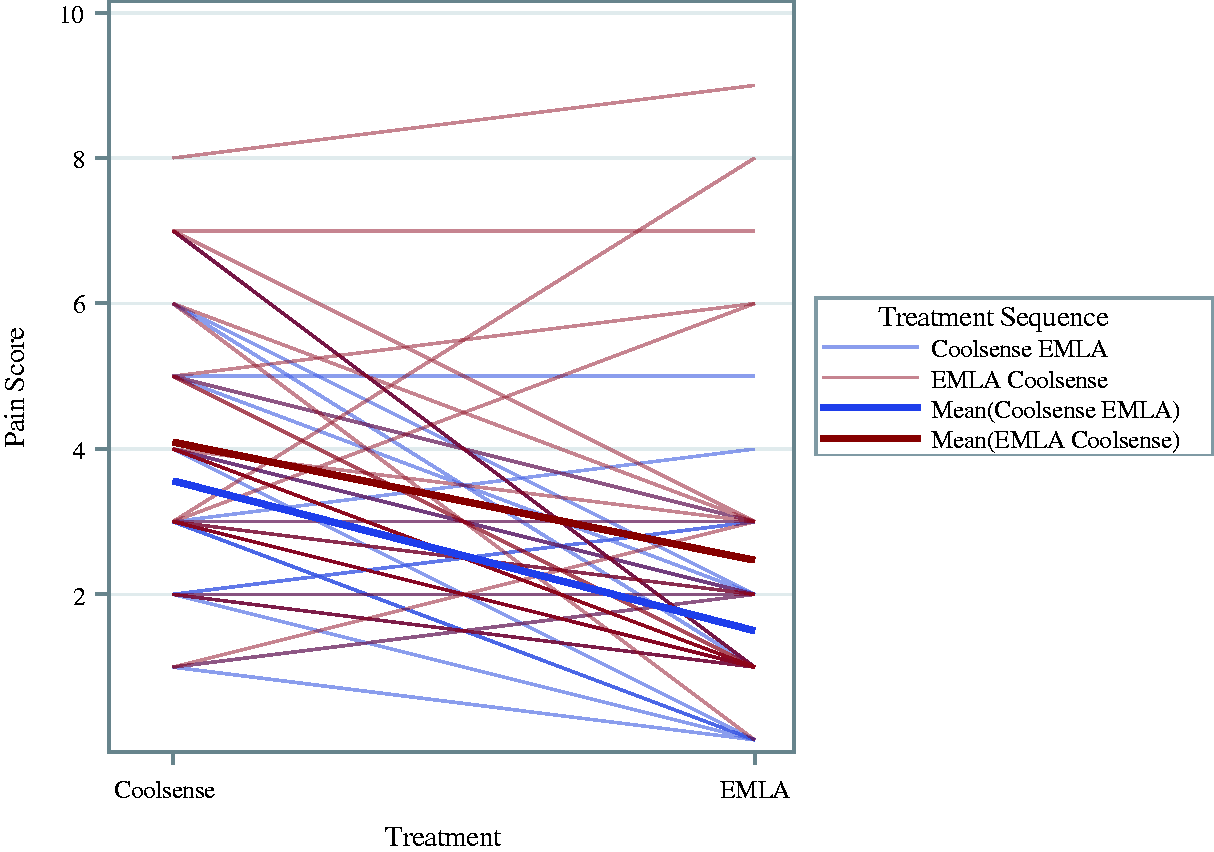
Profiles over treatment plot of pain scores. Data are paired responses (thin lines) and overall mean response (thick lines). EMLA® (Aspen Pharmacare Australia Pty Ltd, St Leonards, NSW): eutectic mixture of local anaesthetics; Coolsense® (Coolsense Medical Ltd., Tel Aviv, Israel).
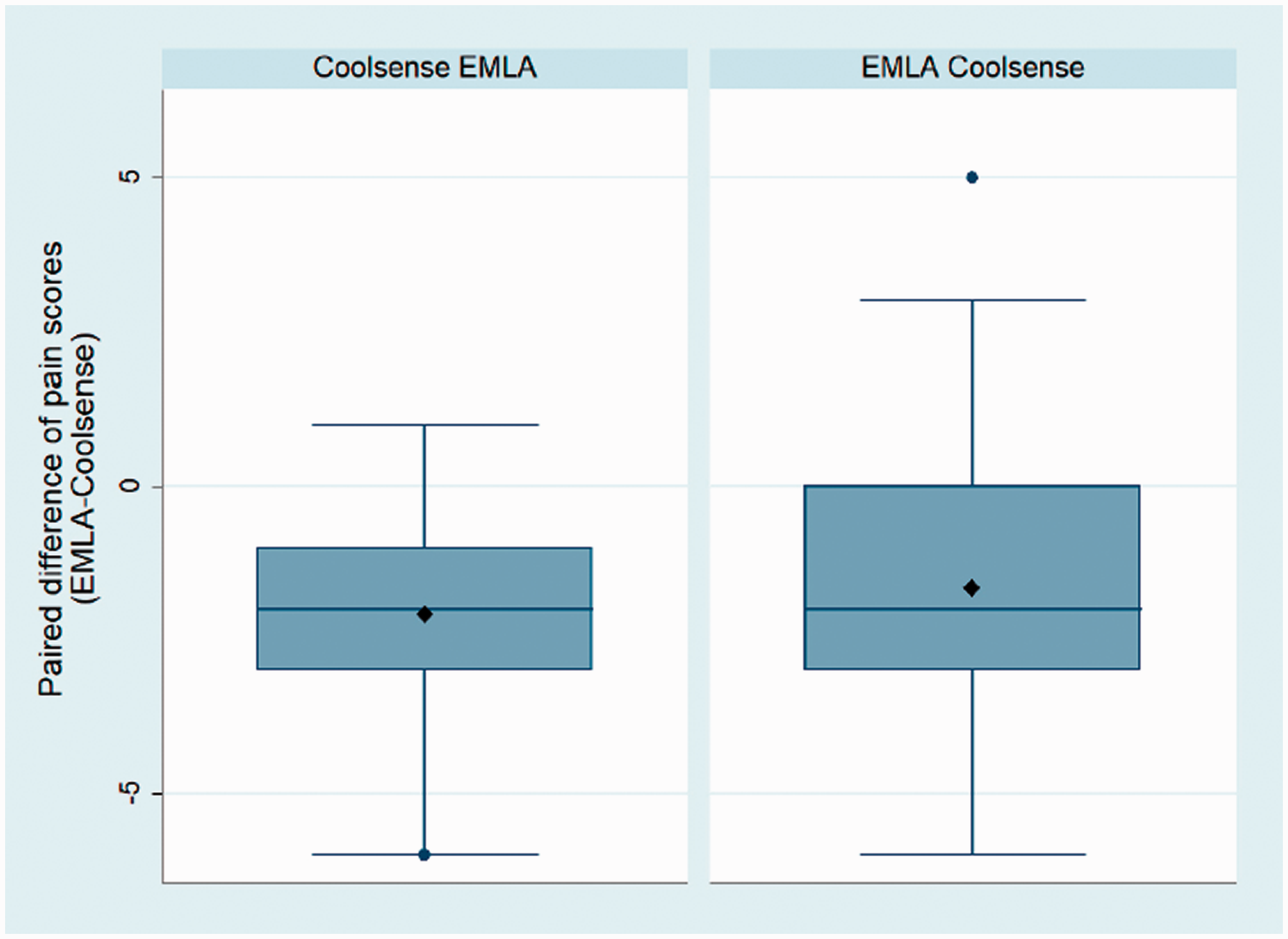
Box plots of paired difference of pain scores by sequence. Data are median (horizontal line), mean (solid diamond), interquartile range (box), 1.5× interquartile range of nearest quartile (whisker) and outliers (solid circles). EMLA® (Aspen Pharmacare Australia Pty Ltd, St Leonards, NSW): eutectic mixture of local anaesthetics; Coolsense® (Coolsense Medical Ltd., Tel Aviv, Israel).
Within the Coolsense EMLA sequence the median NPRS was 3 (IQR 2.5–4.5) for Coolsense and 1 (IQR 1–2) for EMLA (Figure 2). The mean paired difference of NPRS in this sequence was –2.06 (95% CI –1.35 to –2.78) with EMLA (Figures 3 and 4). The EMLA Coolsense sequence median NPRS was 1.5 (IQR 1–3) and 4 (IQR 3–5) for EMLA and Coolsense, respectively. The mean paired difference of NPRS within the EMLA Coolsense sequence was –1.63 (95% CI –0.72 to –2.53) with EMLA. There was no evidence of a period effect (P = 0.44, Student’s t-test) and therefore the pooled mean paired difference of NPRS was –1.84 (95% CI –1.28 to –2.41) in favour of EMLA (Table 2).
Participant pain and satisfaction by sequence.
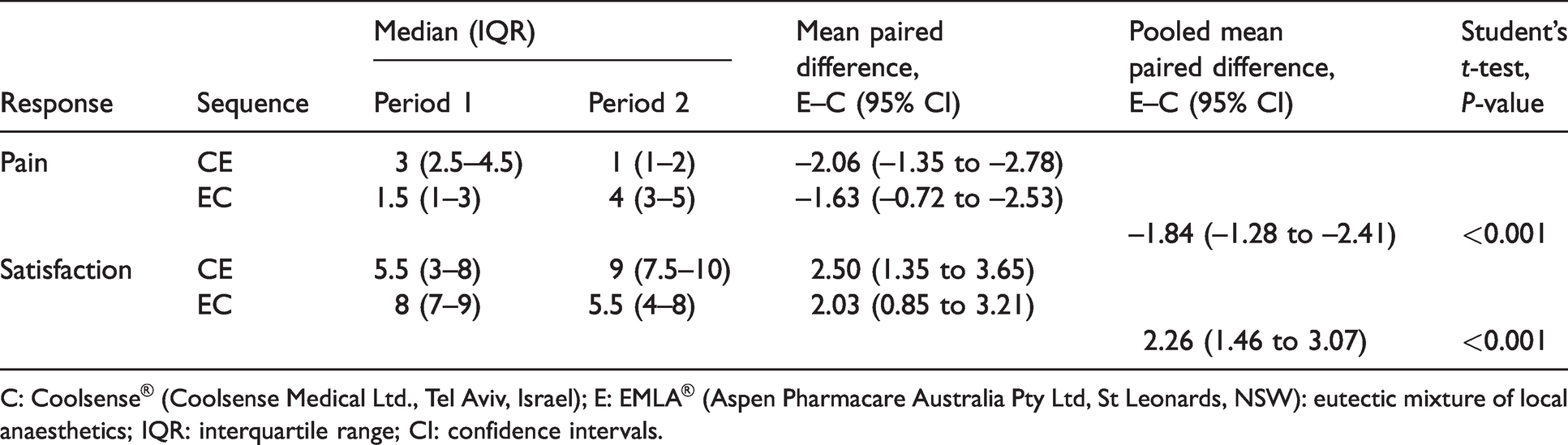
C: Coolsense® (Coolsense Medical Ltd., Tel Aviv, Israel); E: EMLA® (Aspen Pharmacare Australia Pty Ltd, St Leonards, NSW): eutectic mixture of local anaesthetics; IQR: interquartile range; CI: confidence intervals.
Participant satisfaction followed a similar pattern to NPRS in terms of the distribution of independent and paired data. Independent data were likely skewed, whereas paired data appeared normally distributed. As a result, similar techniques were used to describe and analyse these data as with NPRS. The Coolsense EMLA sequence median satisfaction score was 5.5 (IQR 3–8) for Coolsense and 9 (IQR 7.5–10) for EMLA (Table 2). The mean paired difference of satisfaction score was 2.50 (95% CI 1.35 to 3.65) higher with EMLA for this sequence. The EMLA Coolsense sequence median satisfaction score was 8 (IQR 7–9) and 5.5 (IQR 4–8) for EMLA and Coolsense, respectively. The mean paired difference of satisfaction score within this sequence was 2.03 (95% CI 0.85 to 3.21), also in favour of EMLA. There was again no evidence of a period effect (P = 0.56, Student’s t-test). The pooled mean paired difference of satisfaction score was 2.26 (95% CI 1.46 to 3.07) higher with EMLA.
Regarding participant preference, within the Coolsense EMLA sequence there were 3/32 (9.4%) who preferred Coolsense, 28/32 (87.5%) who preferred EMLA and 1/32 (3.1%) with no preference (Table 3). For the EMLA Coolsense sequence, there were 24/32 (75%) who preferred EMLA, 7/32 (21.9%) who preferred Coolsense and 1/32 (3.1%) with no preference. There was strong evidence of participant preference for EMLA (P < 0.001, Mainland–Gart test).
Participant treatment preference and failed cannulation.
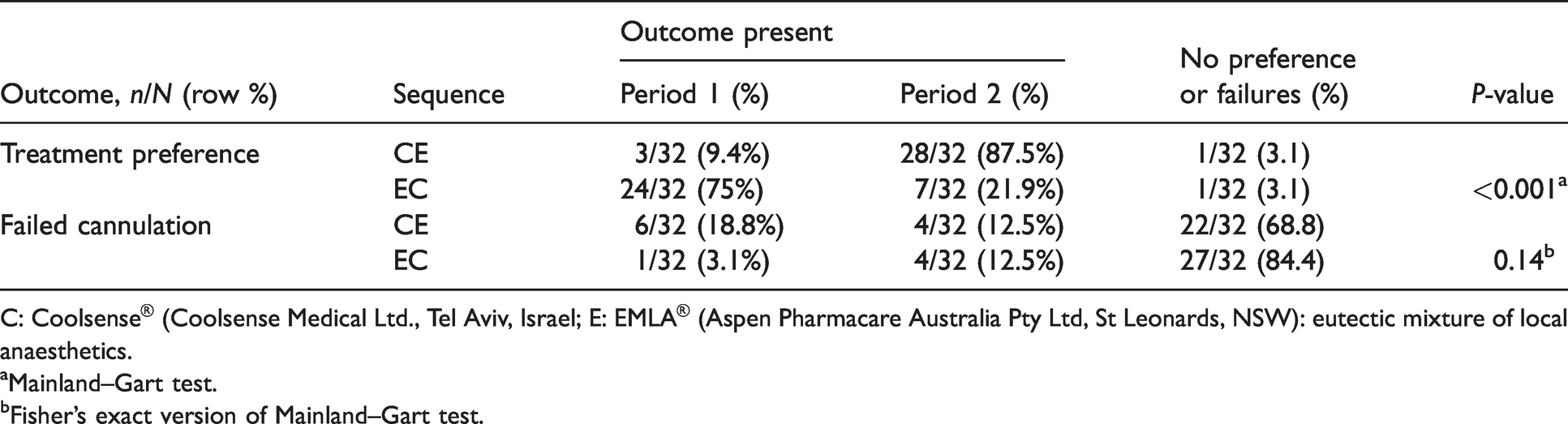
C: Coolsense® (Coolsense Medical Ltd., Tel Aviv, Israel; E: EMLA® (Aspen Pharmacare Australia Pty Ltd, St Leonards, NSW): eutectic mixture of local anaesthetics.
aMainland–Gart test.
bFisher’s exact version of Mainland–Gart test.
There were 6/32 (18.8%) failed cannulation attempts for Coolsense and 4/32 (12.5%) for EMLA within the Coolsense EMLA sequence (Table 3), whereas in the EMLA Coolsense sequence, there was 1/32 (3.1%) failed attempt with EMLA and 4/32 (12.5%) for Coolsense. There was no significant difference between each treatment and failed cannulation attempts (P = 0.14, Fisher’s exact version of the Mainland–Gart test).
There was only one noteworthy complication within the EMLA Coolsense sequence in which Coolsense application subsequently led to a large area of local blanching for over 12 hours and was described as ‘uncomfortable’, but with no lasting consequences.
Discussion
This single-centre randomised crossover trial among healthy adult volunteers demonstrated that EMLA provided a clinically relevant reduction in the pain associated with PVC compared to Coolsense. EMLA also provided a higher level of satisfaction and was the preferred treatment between the two options among most participants. There appeared to be no difference in failed cannulation attempts between each treatment.
Coolsense is a relatively new device, but EMLA has been frequently studied. Examination of how the independent measurements of NPRS with EMLA for this study fit with others is complex for several reasons. Firstly, the population in this case were volunteers willing to undergo PVC solely for the benefit of the RCT. Other studies have often involved individuals about to undergo a medical intervention, such as women for day case surgery6,7 or patients for regular haemodialysis. 16 Some studies have included premedicated patients. 12 Heterogeneity among these studies was increased by factors such as cannulation sites used that are not just limited to the dorsum of the hand, a variety of needle sizes ranging from 23G up to 16G and different clinical settings ranging from wards to anaesthetic rooms. 20 The methodology for most studies had involved a parallel arm design and the use of a crossover design appeared to be uncommon. 16 The design of this RCT involved repeated observations within the same individual and analyses were duly made as paired data. As this study did not have a treatment arm with a placebo or no treatment, we could only comment on the efficacy of Coolsense compared with EMLA. Pain was also measured in ways other than the NPRS for some of these studies. These all mean direct comparisons of such absolute measurements require caution. There were a handful of outliers for whom EMLA appeared to have little efficacy, possibly due to a failure of the patch. Anecdotally, such treatment failures are occasionally noted in day-to-day practice.
The aforementioned case series of Coolsense for paediatric patients that initially called for an RCT between Coolsense and topical local anaesthesia 24 stated that 28% reported the NPRS was zero, 66% as 1–3 and the remaining 6% as 4 or more. The median NPRS was not reported. In this RCT, the median NPRS for Coolsense was 3 or 4, depending on the sequence, which are likely slightly higher than the case series. Other than different study populations, particularly with regard to age, these differences may be because the case series used smaller 22G or 24G cannulae and the cubital fossa was the site of PVC for three-quarters of patients. Given Coolsense was initially developed for cosmetic procedures that use small needles, it may have more efficacy for PVC when used for smaller cannulae, rather than the larger 20G used in this RCT. The case series also reported 95% first-time success for PVC24. Our success rate was slightly lower than this. We did not find a significant difference for failed PVC between Coolsense and EMLA, however it is possible this may represent a type II error.
The limitations of this study include that it was restricted to adult volunteers so there should be some prudence before generalising the results to paediatric groups or hospital attendees. Participant blinding of these two different treatments would have been extremely difficult and was not attempted, particularly as Coolsense works by actively cooling the skin. Needle phobia was an exclusion criterion and while found to be relatively uncommon during enrolment, it is arguable that this group could benefit most from investigating the attenuation of pain associated with PVC potentially to improve their experience of many hospital processes. Aspects of this RCT may not represent typical operational conditions of hospital care. The EMLA treatments were allowed a full 60 minutes and applied to an identified vein by the same anaesthetist performing the PVC. This is often not feasible in usual clinical practice and may contribute to why EMLA was clearly the superior treatment in this study. There was also no acuity or specific time pressure in the need for successful PVC. Clinical pain is also a more complex concept than experimental pain as it is associated with a multitude of factors including expectations, emotions, education and socioeconomic status. 36
Examining other interventions may have been considered helpful, such as a treatment arm with placebo or no treatment as outlined above; however, it would have increased the methodological complexity. It would have meant more treatment sequences requiring additional PVC sites beyond the dorsum of each hand. Given EMLA and other local anaesthetic treatments are clearly known to be superior to no treatment with little to differentiate between them, 20 such additional complexity would have been of little use. Alternative control arms, such as intradermal local anaesthesia, were not considered appropriate choices for this study as outlined earlier. There was no assessment of participant vascular access quality or body mass index, but these were deemed less important because comparisons would be made within each participant. These results may be less applicable to patients with difficult vascular access necessitating several possible PVC sites to be identified.
The Coolsense device and cartridge can be used many times, which reduces the cost to NZ$0.20–0.30 per application. This is in contrast to EMLA, which costs our institution NZ$9.31 for a 5 g tube or NZ$5.95 for a patch. These only reflect the direct costs, whereas indirect costs would include the necessary staff and time to administer EMLA appropriately. Despite the perceived advantages of Coolsense that have likely led to its increasing use, particularly its speed of onset, it appears inferior in attenuating the associated pain of PVC compared to EMLA. Future studies could compare the efficacy of Coolsense for cannulae smaller than 20G, or in higher acuity patients who cannot wait the time necessary for topical local anaesthetic treatments to work. It is possible these situations are when Coolsense may have greatest utility in clinical practice, if such a role for it exists. Such studies could also examine paediatric populations or cannulation sites other than the dorsum of the hand.
In conclusion, among healthy adults undergoing elective PVC, EMLA was superior to Coolsense in reducing the associated pain. EMLA was also associated with higher satisfaction and was the preferred participant treatment choice over Coolsense. Based on these findings, our recommendations are that EMLA or interventions shown to have a similar efficacy 20 should remain the preferred choice for attenuating the associated pain from PVC over Coolsense. It is plausible that the superiority of EMLA over Coolsense will also be the case for children undergoing PVC, although a prospective study comparing the efficacy of these two interventions in this specific population may be warranted.
Footnotes
Author contributions
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
