Abstract
Vasopressors are ubiquitous in intensive care units. While central venous catheters are the preferred route of infusion, recent evidence suggests peripheral administration may be safe for short, single-agent courses. Here, we identify risk factors and develop a predictive model for patient central venous catheter requirement using the Medical Information Mart for Intensive Care, a single-centre dataset of patients admitted to an intensive care unit between 2008 and 2019. Using prior literature, a composite endpoint of prolonged single-agent courses (>24 hours) or multi-agent courses of any duration was used to identify likely central venous catheter requirement. From a cohort of 69,619 intensive care unit stays, there were 17,053 vasopressor courses involving one or more vasopressors that met study inclusion criteria. In total, 3807 (22.3%) vasopressor courses involved a single vasopressor for less than six hours, 7952 (46.6%) courses for less than 24 hours and 5757 (33.8%) involved multiple vasopressors of any duration. Of these, 3047 (80.0%) less than six-hour and 6423 (80.8%) less than 24-hour single vasopressor courses used a central venous catheter. Logistic regression models identified associations between the composite endpoint and intubation (odds ratio (OR) 2.36, 95% confidence intervals (CI) 2.16 to 2.58), cardiac diagnosis (OR 0.72, CI 0.65 to 0.80), renal impairment (OR 1.61, CI 1.50 to 1.74), older age (OR 1.002, Cl 1.000 to 1.005) and vital signs in the hour before initiation (heart rate, OR 1.006, CI 1.003 to 1.009; oxygen saturation, OR 0.996, CI 0.993 to 0.999). A logistic regression model predicting the composite endpoint had an area under the receiver operating characteristic curve (standard deviation) of 0.747 (0.013) and an accuracy of 0.691 (0.012). This retrospective study reveals a high prevalence of short vasopressor courses in intensive care unit settings, a majority of which were administered using central venous catheters. We identify several important risk factors that may help guide clinicians deciding between peripheral and central venous catheter administration, and present a predictive model that may inform future prospective trials.
Introduction
Vasopressors are a pharmacological cornerstone in the treatment of shock.1,2 The timing and duration of vasopressor interventions are active areas of research and have been shown to affect patient outcomes significantly in intensive care unit (ICU) settings.3–5 Vasopressors are also associated with adverse outcomes of varying severity including tissue ischaemia and cardiac dysrhythmias.6–8 One common mechanism for adverse outcomes is tissue extravasation from peripheral intravenous catheters (PIV). 9 For this reason, among others, the central venous catheter (CVC) is currently the standard route of vasopressor infusion. 10
CVCs are associated with complications including infection, bleeding and pneumothorax.10–15 Recent literature has begun to address the safety of prolonged peripheral administration.9,16–19 One single-centre ICU study found that peripheral administration of norepinephrine (noradrenaline), dopamine, or phenylephrine had low adverse event and subsequent CVC insertion rates. 18 This study and others suggest that the duration of vasopressor infusion is associated with PIV adverse events.9,20 Taken together, there is a growing body of evidence suggesting PIV administration of vasopressors may be safe and effective for limited courses.
CVC placement is a time - and skill-intensive procedure, often requiring multiple providers at the bedside to maintain appropriate sterility and safety protocols. As a result, provider bandwidth is likely a key factor in the timing of CVC insertion, implying that the procedure may be performed at a time of convenience rather than when a patient has failed a PIV protocol. For these reasons, accurate patient risk stratification for vasopressor duration may facilitate appropriate utilisation of CVCs. To date, there have been no reports addressing demographic and physiological risk factors associated with the length of vasopressor infusion.
Predictive analytics have become increasingly common in the critical care literature 21 and promise to improve the accuracy of patient prognosis as well as provide early identification of disease. 22 Prior studies have focused on acute respiratory distress syndrome, 23 sepsis24,25 and ICU mortality.26,27 We propose that the development of a new predictive model addressing the duration of vasopressor treatment may augment clinical decision-making in determining which patients will benefit from a CVC. Moreover, a predictive model may provide a framework for prospective trials of peripheral vasopressor administration.
In this study, we sought to address vasopressor knowledge gaps using the large, open-access Medical Information Mart for Intensive Care (MIMIC-IV) database. 21 Using prior studies of peripheral vasopressor safety, we identified 24 hours as a conservative cut-off for peripheral administration.9,16–19 In addition, as patients requiring multiple vasopressors are likely to be more ill and the use of multiple vasopressors is a common indication for CVC placement, we also identified patients requiring multiple agents as more likely to benefit from a CVC.28,29 From these observations, we created a composite study endpoint of vasopressor administration for 24 hours or longer or the need for multiple agents for any duration. In this study, we complete three related aims: to (a) characterise patterns of short vasopressor administration using CVCs; (b) evaluate relationships between vital signs and other risk factors such as the type of ICU and diagnosis prior to vasopressor administration and the composite endpoint; and (c) develop a predictive model with an associated application estimating the likelihood of a patient requiring a CVC based on the composite endpoint.
Materials and methods
Study design and setting
We performed a retrospective secondary analysis of the MIMIC-IV dataset, a single-centre database from Beth Israel Deaconess Medical Center in Boston, MA, USA. 30 This database contains over 40,000 hospital admissions from 2008 to 2019 for patients from adult ICUs, and includes nurse-verified physiological monitoring, continuous intravenous drips, procedure documentation and patient outcomes data.
Data collection and processing
Data were extracted using the tidyverse v.1.2.1 package from R v.3.6.0. For vital signs, we extracted the mean heart rate (HR), mean arterial pressure (MAP; computed from systolic and diastolic pressures), respiratory rate (RR), temperature, pulse oximetry, and blood glucose using hour increments preceding the initiation of vasopressors. We also extracted information on the start and end time of each ICU stay, vasopressor course, vasopressor type, admission information and patient demographics. 31 The code for data extraction and analysis is available on GitHub (https://github.com/ruoyijiang/haimovich_pressors).
Filters and outcome definitions
We included adult patients (≥18 years of age) on their first vasopressor course of an ICU stay. A vasopressor course was defined as a grouping of one or more vasopressor administrations interrupted by less than 24 hours. Interruptions for greater than 24 hours were considered separate vasopressor courses and only the first was included in our analysis. In order to exclude patients with uncertain vasopressor durations due to vasopressor initiation prior to ICU arrival, we excluded patients with vasopressor infusions documented in the first hour of their ICU course. We excluded vasopressors administered by intermittent bolus dosing (i.e. push doses) by removing epinephrine (adrenaline) or phenylephrine courses lasting less than 30 minutes. To avoid skewing our dataset towards shorter vasopressor courses, we also excluded individuals who died while on vasopressors as defined by a vasopressor course terminating within five minutes of death. We filtered the study population into four groups for comparison based on proposed cut-offs for peripheral vasopressor usage from Loubani and Green: single agents for less than six hours, six to 24 hours, or greater than 24 hours, or multiple agents for any duration. 9 Using this literature, we define patients likely to require a CVC as those meeting the composite outcome of single vasopressor administration for greater than 24 hours or the use of multiple agents.
Covariate definitions
We selected covariates in the MIMIC-IV dataset that would be generalisable across critical care settings with an emphasis on patient characteristics (age, kidney function, intubation status, emergency department (ED) admission), vital signs (MAP, HR, RR, oxygen saturation (SpO2)), diagnosis and patient setting (ICU type). Age, MAP, HR, RR and SpO2 are studied as continuous variables while the remaining variables are included as categorical variables. After manually reviewing the available diagnoses in the MIMIC-IV dataset, we grouped patient diagnoses into the following categories: ‘infection’, ‘cardiac’, or ‘other’ (supporting information). To identify CVC-related procedures, a set of MIMIC procedure codes were used for assignments (Supplementary Methods). Because baseline kidney function was not available in this cohort, we set a renal impairment threshold using an initial creatinine of 106 µmol/l. For relative risk analysis, patients were grouped into ages at an interval of ten years for easier visualisation of the association between age and relative risk compared to other categorical variables.
Data analysis
All statistical analysis was performed in R v.3.6.0. For univariate analysis, relative risk ratios were calculated for demographic variables using the epitools package v0.5-10. To identify significant univariate vital sign trends prior to vasopressor initiation, we used joint models implemented with JM v1.4-8. The timepoint for the survival model was how far into the admission vasopressors were required, with censoring for course duration (more than 24 hours) or required multiple vasopressors; the linear model used a random effect for the specific course and a fixed effect for the length of time into the admission. Multivariate logistic regression models for each agent individually and in a combined vasopressor model were implemented using the generalised linear model (glm) function in base R to examine the association between vasopressor administration and covariates identified by univariate analysis, including demographic, diagnosis, care unit, intubation, sepsis severity scores, vasopressor type, mean vitals data the hour preceding the initiation of therapy and, for the composite model, vasopressor choice. For multiple logistic regression models, missing data were imputed using random forest imputation implemented in the randomForest v4.6-14 package. Multicollinearity was checked by variance inflation factor analysis implemented in the car v3.0-3 R package. Five-fold cross-validation was used to assess model accuracy and overfitting—data were split into 80%/20% training/testing sets five times and the average of the area under the receiver operating characteristic curve across splits was reported. pROC v1.15.3 was used for computing area under the receiver operating characteristic curves (AUROCs) and boot v1.3-22 was used for computing confidence intervals for plotted AUROCs. A P-value of less than 0.05 was used to indicate statistical significance.
Risk assessment application and data sharing
An app was developed by implementing the logistic regression model developed in this study using the shiny v1.5.0 package in R. This app is available for download at https://github.com/ruoyijiang/haimovich_pressors/app. After inputting the parameters needed for the model, this app then reports the likelihood (probability) that the vasopressor course will last for more than 24 hours or involve multiple vasopressors.
Results
A total of 69,619 ICU stays are recorded in the MIMIC-IV database, of which 21,903 cases included the use of vasopressors (31.5%), 29,447 stays involved intubation (42.3%) and 17,032 stays had both vasopressors and intubation (24.5%). A total of 17,053 vasopressor courses met our inclusion criteria (i.e. first vasopressor course of an ICU stay, vasopressors initiated ≥one hour after ICU arrival) with 35 (0.2%) of courses excluded because the patient died while vasopressors were infusing (Table 1) (Supplementary Table 1). Of the included vasopressor courses, 22.3% (3807) lasted less than six hours, while 46.6% (7952) lasted less than 24 hours (Figure 1(a)). These patterns of vasopressor usage were consistent across vasopressor types (Figure 1(b)) (Supplementary Table 1). We describe the mortality rates for the various vasopressor cohorts in the supplement (Supplementary Table 2). The rate of CVC use increased with vasopressor duration (Figure 1(c)). We noted that 80.0% (3047) of single vasopressor courses less than six hours involved a concomitant CVC, while 80.8% (6423) courses less than 24 hours involved a CVC. By comparison, 90.4% (3023) of courses greater than 24 hours and 92.6% (5333) involving multiple vasopressors included a CVC.
Comparison of group demographics for single vasopressor courses less than six hours, between six and 24 hours, greater than 24 hours, or multi-agent courses of any duration.
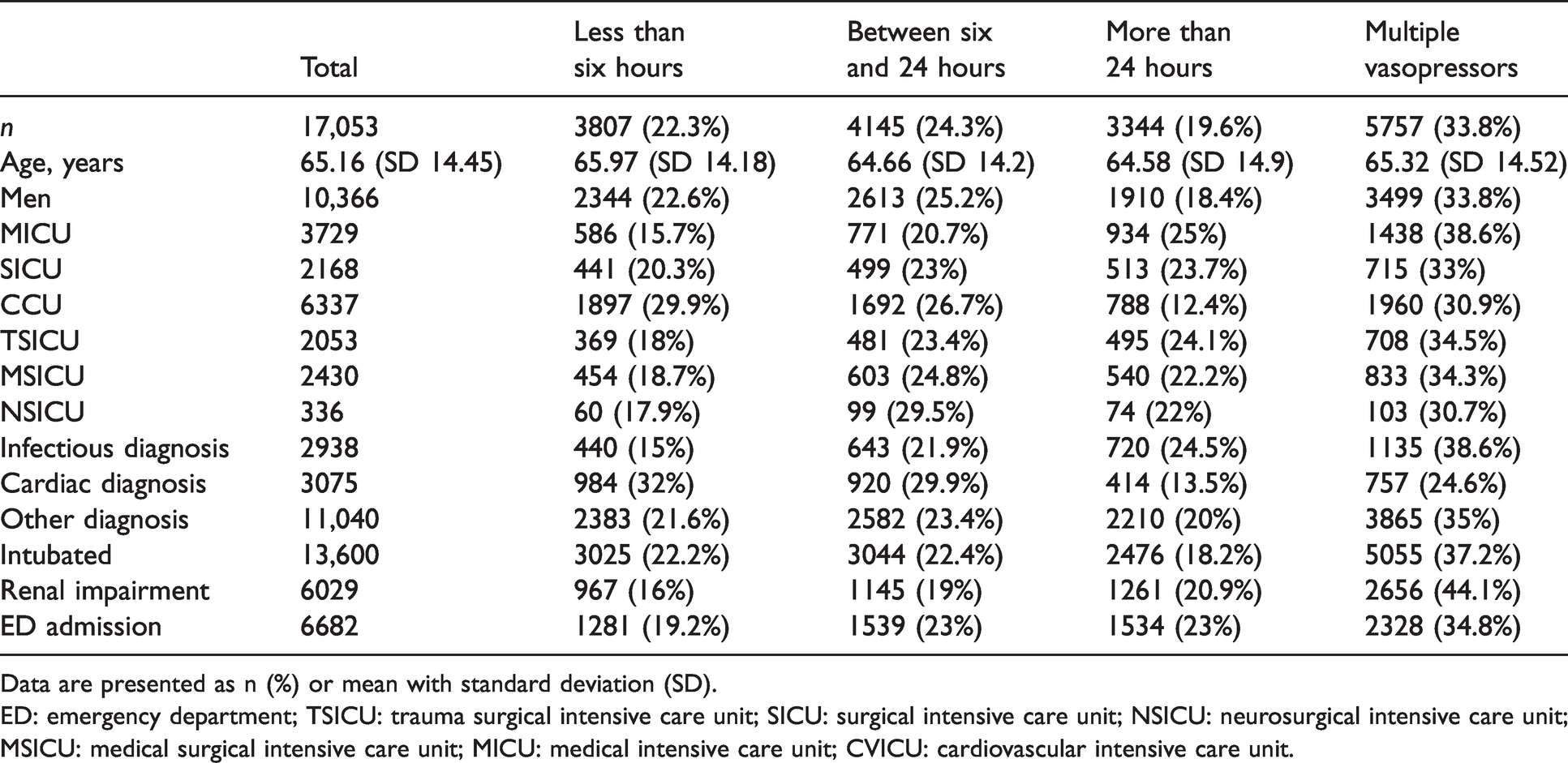
Data are presented as n (%) or mean with standard deviation (SD).
ED: emergency department; TSICU: trauma surgical intensive care unit; SICU: surgical intensive care unit; NSICU: neurosurgical intensive care unit; MSICU: medical surgical intensive care unit; MICU: medical intensive care unit; CVICU: cardiovascular intensive care unit.
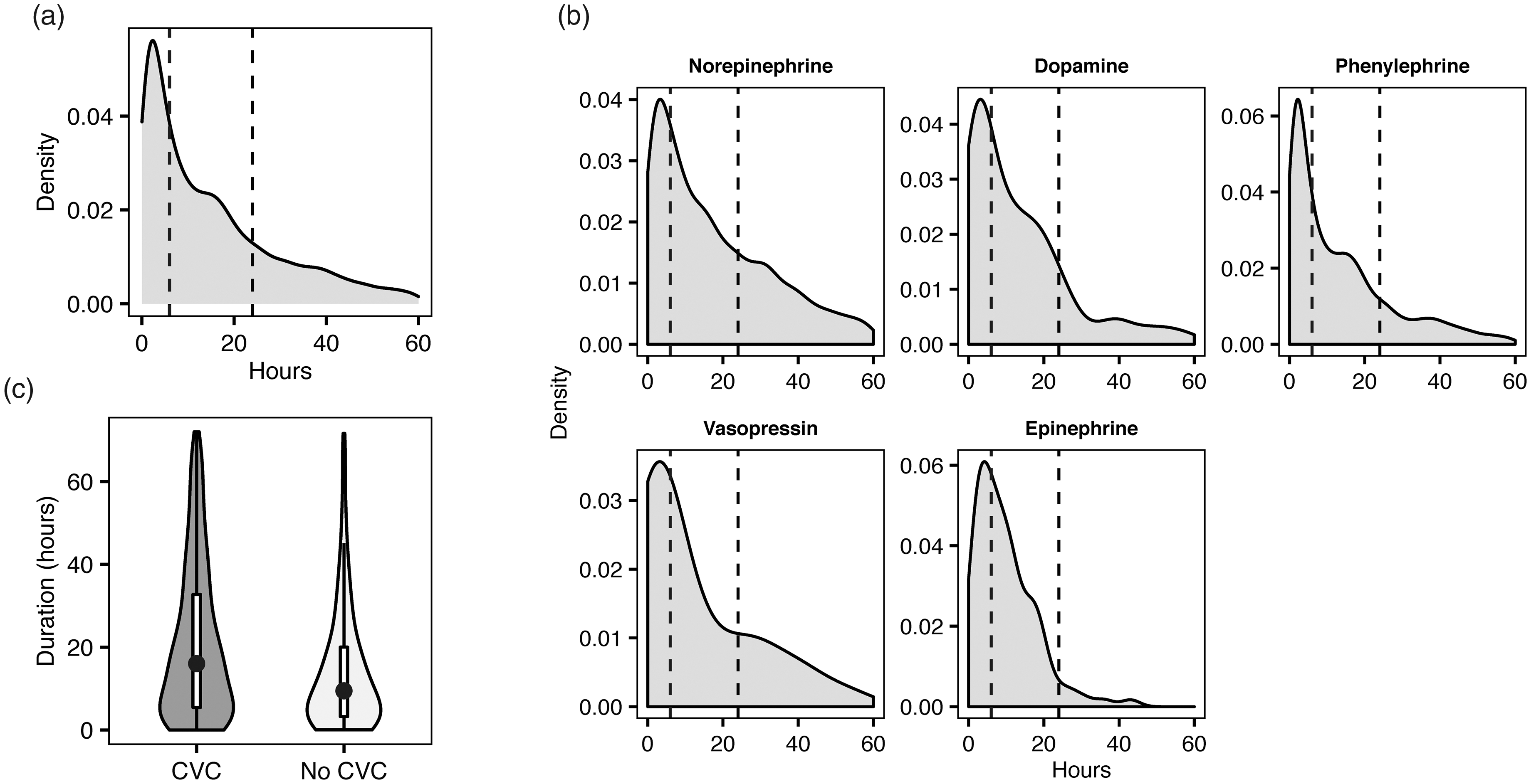
Grouped analysis of 17,053 vasopressor courses contained in the Medical Information Mart for Intensive Care (MIMIC-IV) dataset meeting the inclusion criteria. Durations of all vasopressors (a) and individual durations for each vasopressor (b) are shown as a histogram (smoothed with a Gaussian kernel density estimate). Dashed lines reflect six and 24-hour timepoints. (c) Violin plot of vasopressor durations up to 72 hours associated with a central venous catheter (CVC) or not associated with a CVC. Embedded box plots represent upper and lower quartiles and whiskers represent 1.5 times the interquartile range. Dots represent median vasopressor durations for each status.
Relative risk analysis was used to identify factors related to patient demographics, diagnosis and intubation status associated with the composite endpoint prolonged vasopressor duration or need for multiple agents (Figure 2) (Supplementary Table 3). In a grouped analysis of all single vasopressor courses, patients with cardiac diagnoses (relative risk 0.67, 95% confidence intervals (CI) 0.64 to 0.70), male patients (relative risk 0.95, 95% CI 0.92 to 0.97) were less likely to meet the composite endpoint. Conversely, we observed that intubated patients (relative risk 1.22, 95% CI 1.17 to 1.27), patients with infectious diagnoses (relative risk 1.23, 95% CI 1.19 to 1.27), patients admitted through the ED (relative risk 1.14, 95% CI 1.11 to 1.18) and patients with renal impairment (relative risk 1.38, 95% CI 1.34 to 1.42) were more likely to meet the endpoint. We replicated this analysis at the individual vasopressor level for the three most commonly used agents in our dataset: dopamine, norepinephrine (noradrenaline) and phenylephrine (Supplementary Figure 1, Supplementary Table 4). Across these agents, intubation and renal impairment were strong risk factors for the composite endpoint, while associations with diagnosis classes varied.
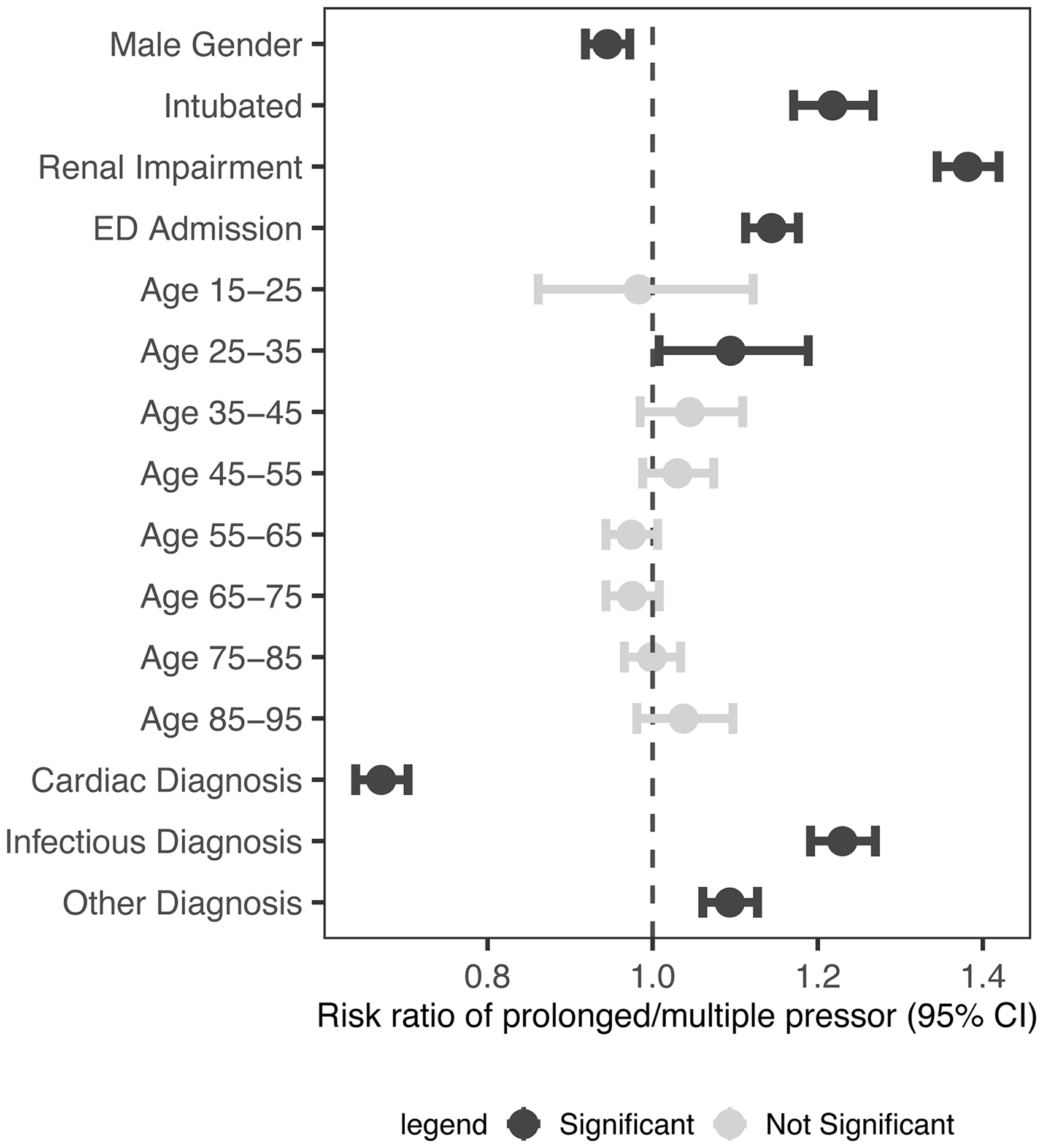
Relative risk ratios for composite endpoint receiving more than 24 hours of vasopressors or multiple agents. 95% confidence intervals (CI) are shown with error bars. Dark-coloured risk factors represent categorical variables found to be significant (P ≤0.05). ED: emergency department.
We then examined associations with vital sign trajectories in our grouped vasopressor analysis and observed that elevated HR (log hazard ratio 0.0061, CI 0.0051 to 0.0071) was significantly associated with our endpoint. Associations were also observed for lower MAP (log hazard ratio –0.028, CI –0.029 to –0.026), higher RR ((log hazard ratio 0.008, CI 0.007 to 0.009) and lower SpO2 (log hazard ratio –0.021, CI –0.022 to –0.020) (Figure 3). These trends varied in the vasopressor subgroups for the three most commonly used vasopressors (Supplementary Figure 2).
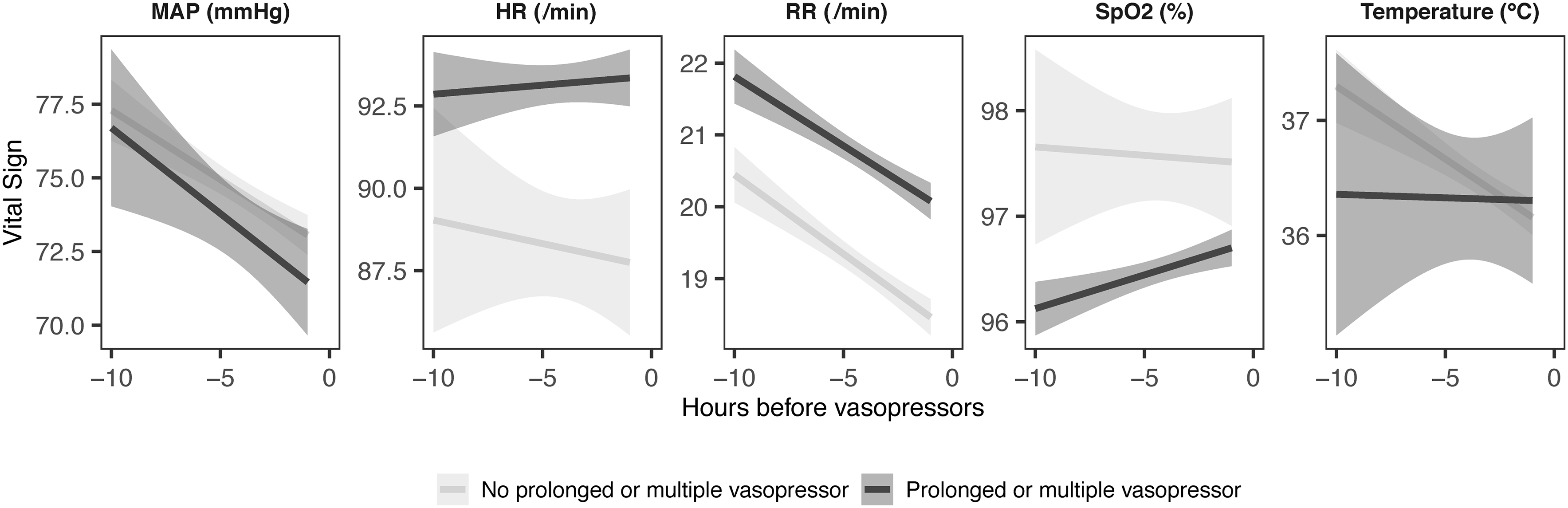
Plots of linear estimates for continuous vital signs for up to ten hours before the initiation of vasopressors. Shaded areas represent 95% confidence intervals for estimates of linear regression lines. Lines are plotted for either vital signs associated with eventual courses that lasted more than 24 hours or involved multiple agents (dark) or not (light).
We developed a multivariate logistic regression analysis using demographic covariates, ICU type, care unit, intubation status and diagnosis, as well as vital signs before vasopressor initiation to predict patients requiring a CVC as determined by the composite endpoint (Figure 4(a), (b)) (Supplementary Table 5). We observed that intubation status (odds ratio (OR) 2.36, CI 2.16 to 2.58) was predictive of the composite endpoint, as was renal impairment (OR 1.61, CI 1.50 to 1.74). Higher HR (OR 1.006, CI 1.003 to 1.009), higher RR (OR 1.006, CI 1.001 to 1.011) and lower SpO2 (OR 0.996, CI 0.993 to 0.999) in the hour prior to the induction of vasopressors were also significant variables. We repeated this analysis for the single vasopressors norepinephrine (noradrenaline) and phenylephrine, finding that intubation status (CI 2.94 to 3.64, CI 1.53 to 1.99) and renal impairment (CI 1.56 to 1.89, CI 1.89 to 2.29 respectively) were also positively associated with prolonged or multi-agent courses, while the opposite was observed for cardiac diagnoses hazard ratio (CI 0.63 to 0.94, CI 0.56 to 0.70 respectively) (Supplementary Table 6). Multivariate analysis was not performed for dopamine due to the small number of samples (n=96).
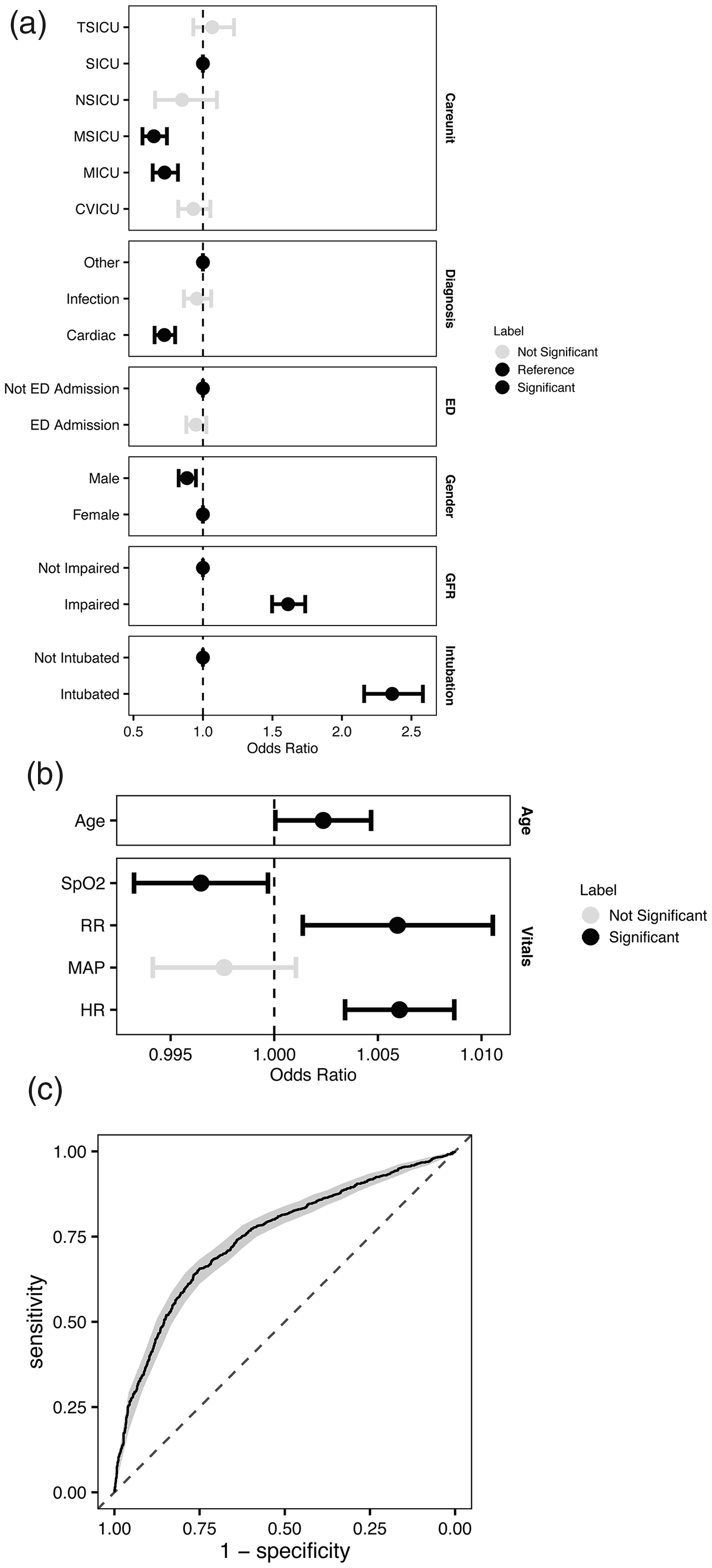
Predictive, multivariable logistic regression model of composite endpoint of single-agent courses longer than 24 hours or multi-agent courses of any duration using (a) categorical covariates, and (b) age or vital signs. For the age and vital sign analysis, odds ratios reflect the unit of measurement (e.g. a single year for age, or a single point for MAP). (c) Model performance as reflected by a receiver operator characteristic curve. AUROC (standard deviation) for this composite model was 0.747 (0.013). Error bars indicate a 95% confidence interval and significance was determined using P ≤0.05. Dark-coloured confidence intervals represent variables found to be significant (P ≤0.05). TSICU: trauma surgical intensive care unit; SICU: surgical intensive care unit; NSICU: neurosurgical intensive care unit; MSICU: medical surgical intensive care unit; MICU: medical intensive care unit; CVICU: cardiovascular intensive care unit; GFR: glomerular filtration rate; MAP: mean arterial pressure; AUROC: area under the receiver operator characteristic curve; ED: emergency department.
We assessed for model overfitting and internal generalisability of the multivariate logistic regression model using five-fold cross-validation. Logistic regression across the composite dataset including dopamine, norepinephrine (noradrenaline) and phenylephrine resulted in an AUROC (standard deviation, SD) of 0.747 (SD 0.013) and accuracies of 0.691 (SD 0.012) for predicting single vasopressor infusions greater than 24 hours or multi-agent infusions of any duration (Figure 4 (c)) (Supplementary Table 7). Multivariate logistic regression models for the single vasopressors norepinephrine (noradrenaline) and phenylephrine performed with AUROCs of 0.694 (SD 0.040) and 0.710 (SD 0.029), respectively (Supplementary Figure 3). This model was used to develop an application that can be used to predict a patient’s risk of meeting the composite endpoint of multiple vasopressors or a single-agent infusion for greater than 24 hours (Supplementary Figure 4).
Discussion
Vasopressors are a cornerstone of ICU care and there is a growing body of evidence to suggest that short durations of peripherally administered vasopressor usage may be safe. In this study, we examined whether it is possible to predict a composite endpoint of prolonged vasopressor duration or the need for multiple agents prior to medication initiation. To accomplish this, we set out to complete three related aims using the open-access MIMIC-IV database. First, we described the prevalence of short vasopressor durations in various ICU settings, finding that a significant fraction (46.6%) of vasopressor courses lasted for less than 24 hours and involved a single agent. This finding is consistent with previous literature showing low rates of conversion from peripheral administration to CVCs; short single-agent vasopressor courses are more common than those that eventually require multiple agents or prolonged infusions.18,20 We also note that 72.4% of single vasopressor courses lasting less than six hours involved a CVC. Second, we hypothesised that vital signs preceding administration may be significantly associated with prolonged/multiple vasopressor courses. We used repeated measures linear regression analysis to show univariate correlations with elevated HR and RR, as well as lower MAP and SpO2 prior to administration. Several studies on complications from peripheral vasopressor administration have shown that prior vitals status represents an important safety factor. This finding also supports the use of trends in vital signs prior to vasopressor initiation to identify situations in which peripheral vasopressor use may be sufficient.
We then developed a multivariate logistic regression model using the mean vital signs during the hour prior to vasopressors in addition to readily accessible patient demographic covariates to predict which patients were likely to need prolonged vasopressors or require multiple agents. Here, our work mirrors prior efforts in the critical care literature including those addressing vasopressor weaning in the ICU and fluid responsiveness in acute kidney injury.32,33 We demonstrated the predictive value of vital signs combined with categorical information of patient demographics, diagnosis, care unit and intubation status. Moreover, our work suggests that the prediction of vasopressor duration and the need for multiple agents prior to initiation is technically feasible and may identify safe contexts for the utilisation of peripheral vasopressors. We present an application to enable other researchers to probe the findings of this study.
Taken together, we conclude that vasopressors are often required for less than six hours and, in this dataset, these short courses are typically associated with CVCs. We provide proof of concept about the predictability of these courses, highlighting demographic, vital signs and ICU setting data associated with vasopressor duration and the multiplicity of agents. The combination of these observations suggests that studies of peripheral vasopressor safety be prioritised in order to minimise the potential risks of CVCs.
Our study has a number of limitations. First, the MIMIC-IV database is a powerful platform for clinical research, but is limited to a single tertiary care centre. As a result, the patterns of vasopressor usage may not match current standards of practice and models generated may not be generalisable. For example, hospital policies around patient placement into varied ICUs may differ between institutions. This study does not include vasopressor doses as covariates or endpoints, which may be relevant for both modelling and CVC placement policies. Our predictive models also have limitations, including the use of an imputation step to account for missing data. While this practice is common in model development, it is associated with biases. We are also not able to include potentially important variables because of their availability in the MIMIC-IV database, including illness severity markers such as sequential organ failure assessment (SOFA) scores and pre-vasopressor fluid resuscitation. Finally, we cannot capture the nuance of individual patient care, including the opportunity cost of a CVC placed while a patient is stable, rather than needing to do so urgently during active decompensation. We anticipate that algorithms incorporating multiple timepoints and more complex data streams such as clinical notes will show improved benchmarks in terms of predictive performance, and future studies are needed to assess whether the temporal trend of vital signs before vasopressor initiation will predict the prolonged use of vasopressors.
Conclusions
In this retrospective study, we used the open-source MIMIC-IV database to identify settings in which peripheral vasopressor administration may be appropriate based on anticipated vasopressor need. We generated predictive models for a composite endpoint of multiple vasopressor administration or single-agent infusions for greater than 24 hours. We identified demographic and physiological risk factors associated with the length of vasopressor infusion that could be used by providers to choose the most appropriate route of vasopressor administration. These data also support expanded prospective studies of peripheral vasopressor use in which patients are stratified to CVCs or peripheral vasopressor administration using a model such as the one described here; doing so would confirm and translate our findings in a more robust study context.
Supplemental Material
sj-pdf-1-aic-10.1177_0310057X211024258 - Supplemental material for Risk factor identification and predictive models for central line requirements for patients on vasopressors
Supplemental material, sj-pdf-1-aic-10.1177_0310057X211024258 for Risk factor identification and predictive models for central line requirements for patients on vasopressors by Adrian D Haimovich in Anaesthesia and Intensive Care
Footnotes
Author Contribution(s)
Data availability
The MIMIC-IV dataset is publicly available from the authors at https://mimic.physionet.org. The code used for our analysis is available online at ![]() .
.
Declaration of conflicting interests
The author(s) declare that they have no competing interests or disclosures to report.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary materials
The supplementary materials document contains Supplementary methods (I), Supplementary tables (II) and Supplementary figures (III) corresponding to the results and methods described in this study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
