Abstract
Newer generation drug-eluting stents are the most commonly inserted stent in the setting of percutaneous coronary intervention. This narrative review focuses on the evidence underpinning the perioperative management of patients with newer generation drug-eluting stents undergoing non-cardiac surgery. Six studies reported the incidence of major adverse cardiovascular events according to the time interval from percutaneous coronary intervention to non-cardiac surgery, and the comparative risks of newer and first generation drug-eluting stents. No study demonstrated an increased risk of major adverse cardiovascular events once three months had elapsed between stent implantation and non-cardiac surgery. Only one study included patients with third and fourth generation drug-eluting stents. Seven studies analysed the relationship between antiplatelet therapy, major adverse cardiovascular events and perioperative bleeding. The risks of major adverse cardiovascular events do not appear to be increased if antiplatelet therapy is ceased for less than seven days but are increased if it is discontinued for more than seven days. Most studies reported no differences in the incidence of major bleeding associated with antiplatelet therapy. The risk of perioperative major adverse cardiovascular events in non-cardiac surgery does not appear to be increased after three months following implantation with newer generation drug-eluting stents. However, the possibility of increased risk cannot be excluded as most studies were inadequately powered. The thrombotic risk is substantially reduced in patients with fourth (polymer free) generation drug-eluting stents, and urgent non-cardiac surgery can be considered one month after percutaneous coronary intervention. Larger multicentre studies are needed to define the optimal window for non-cardiac surgery after percutaneous coronary intervention and provide definitive perioperative strategies for patients presenting for non-cardiac surgery after the implantation of newer generation drug-eluting stents.
Introduction
Drug-eluting stents (DESs) were introduced to attenuate neointimal hyperplasia and in-stent re-stenosis, a complication occurring in up to 30% of bare-metal stents (BMSs). 1 There are three components to a DES: (a) the antiproliferative agent eluted; (b) the polymer matrix that provides controlled release of the antiproliferative agent; and (c) the stent platform. 2 The antiproliferative agents first used were sirolimus and paclitaxel. 3 Examples of these first generation stents were Cypher™ (Cordis, Miami Lakes, FL, USA) and Taxus® Express (Boston Scientific, Maple Grove, MN, USA). 4 Second generation DESs were developed because of the relatively high rates (0.4%–0.6% per year) of very late (>12 months) stent thrombosis and late in-stent re-stenosis associated with the first generation. 5 Biocompatible polymers were introduced to minimise delayed hypersensitivity reactions, endothelial dysfunction, delayed arterial healing and stent thrombosis. 5 Everolimus or zotarolimus (sirolimus derivatives) are the antiproliferative agents used to reduce neointimal hyperplasia. These newer mammalian target of rapamycin (mTOR) inhibitors are more lipophilic and have a longer duration of action and less systemic toxicity. 6 The metallic stent platform was changed from stainless steel to cobalt chrome or platinum chrome with thinner struts (<100 µm) and enhanced radial strength.3,7 Examples of second generation DESs include Endeavor Resolute® (Medtronic, Santa Rosa, CA, USA), Xience V® (Abbott, Santa Clara, CA, USA), and PROMUS Element™ (Boston Scientific, Maple Grove, MN, USA). 5 Second generation DESs are now the most commonly used stent in Australia in percutaneous coronary intervention (PCI). 1 However, interventional cardiologists are increasingly implanting third and fourth generation DESs. Third generation DESs, such as SYNERGY™ (Boston Scientific, Maple Grove, MN, USA), have biodegradable polymers and effectively become BMSs over time. This is advantageous because chronic exposure to a durable polymer is associated with endothelial dysfunction and thrombosis.6,8 Polymer-free DESs, such as BioFreedom™ (Biosensors, Newport Beach, CA, USA), are fourth generation DESs and the risk of polymer-induced inflammation is eliminated. 8 We will use the term ‘newer generation’ DESs to include all DESs from the second generation onward.
Approximately 15% of patients will require non-cardiac surgery (NCS) within 12 months after placement of a DES. 9 NCS in patients following PCI is associated with major adverse cardiovascular events (MACEs) such as myocardial infarction (MI), stent thrombosis, the need for repeat revascularisation, and death. Stent thrombosis, although relatively uncommon, carries a mortality of approximately 30%. 1 Acute stent thrombosis has been linked to interruption of dual antiplatelet therapy (DAPT). 10 With abrupt discontinuation of antiplatelet therapy, a ‘rebound phenomenon’ with increased levels of cyclooxygenase 1 (COX-1) and thromboxane A2 (TXA2) occurs. 11
The 2014 American College of Cardiology/American Heart Association (ACC/AHA) guidelines recommended delaying non-urgent NCS for 12 months after DES placement. 12 This was based on data from first generation DESs which showed a higher risk of MACEs when NCS was performed within six to 12 months of stenting.13–16 As a result of the improved safety profile of second generation DESs, the ACC/AHA guidelines were updated in 2016. 17 The recommended minimum time for performing elective NCS following second generation DES placement was reduced from one year to six months, and NCS could be considered after three months if the risk of delaying surgery was considered to be greater than the risk of stent thrombosis.18,19 This recommendation was based on multiple non-inferiority studies,20–26 which demonstrated that six months of DAPT following second generation DES placement was not associated with a higher risk of ischaemic events compared to 12–24 months of DAPT, with a lower risk of major bleeding. If the DES had been placed for the treatment of an acute coronary syndrome (ACS), the guideline suggested delaying elective surgery for at least 12 months. 20 These recommendations, however, were not primarily based on studies that had specifically recruited patients with new generation DESs undergoing surgery.
This narrative review evaluates the evidence underpinning the perioperative management of patients with newer generation DESs undergoing NCS. The aims were to identify the optimal time interval from PCI to NCS, and the role of antiplatelet therapy with respect to MACEs associated with NCS in patients with newer generation DESs. The objective was not to replace existing established consensus guidelines, but rather systematically to evaluate the evidence or lack thereof pertaining to this patient population that is of clear clinical relevance to the anaesthetist.
Materials and methods
Study design and search strategy
Searches of MEDLINE and EMBASE databases were performed using the keywords and medical subject headings: drug-eluting stents, second generation, new generation, percutaneous coronary intervention, coronary stent, surgery, non-cardiac surgery and perioperative. The full search strategy is detailed in Supplementary Appendix 1. The search was limited to articles up to May 2020.
Inclusion criteria
Studies were included if they consisted of patients with new generation DESs undergoing NCS. In two studies identified, the inclusion criteria were not strictly limited to NCS, although only a minority of patients underwent cardiac surgery.21,22 Only human studies were included.
Exclusion criteria
Studies were excluded if patients with new generation DESs in the study population were not included or specified. Non–English language articles were excluded.
Selection of studies
Two reviewers screened the titles and abstracts of identified articles. The full manuscripts of selected articles were then screened by the same two reviewers to assess for eligibility. We manually searched the reference list of articles to identify further studies. A total of 1159 articles was identified. One hundred and eighty-three duplicate articles were identified using the software EndNote (X8 version; Clarivate, Philadelphia, PA, USA). After applying exclusion criteria, a total of ten studies was included in this review (Figure 1).
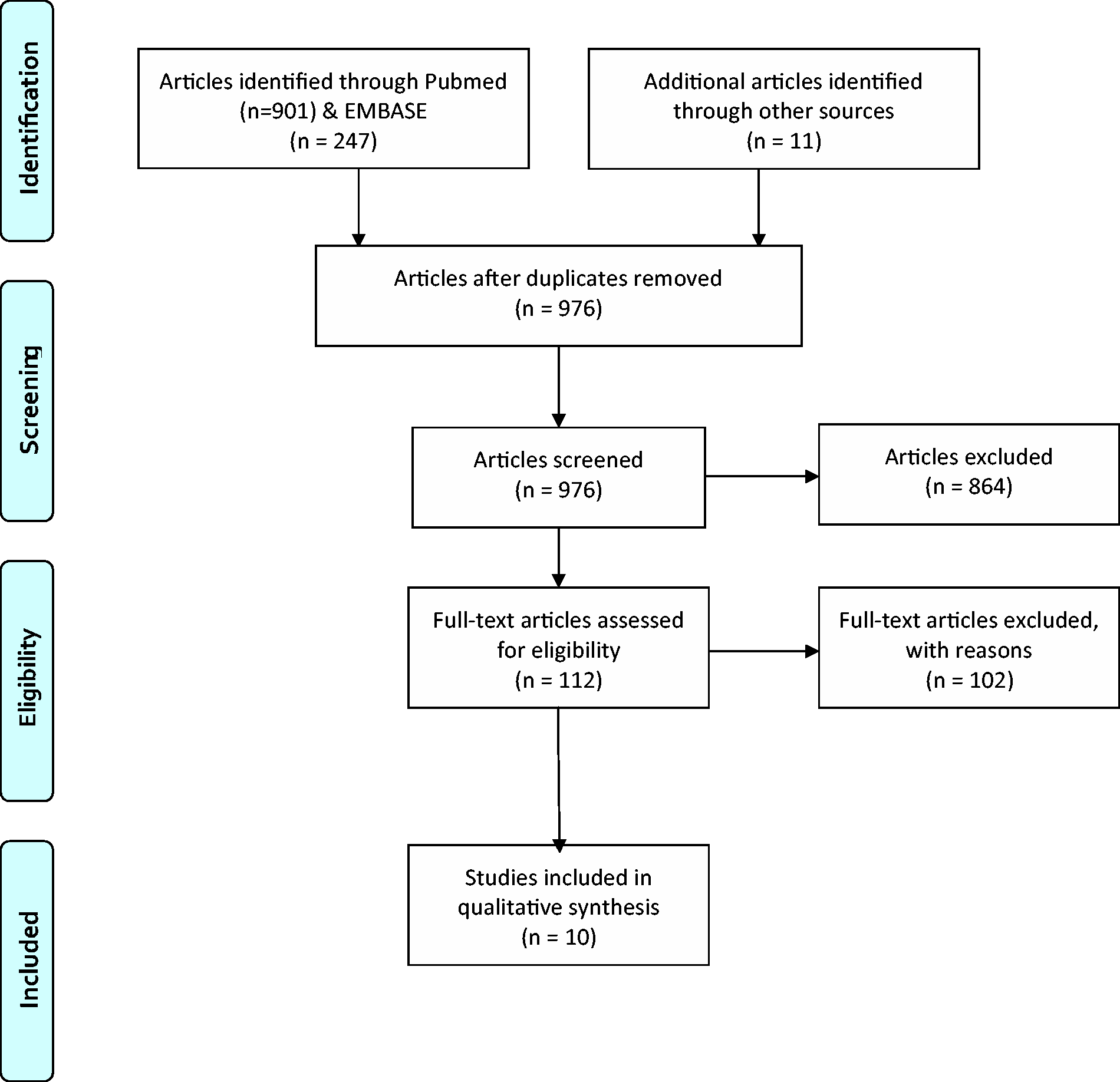
Preferred Reporting Items for Systematic Review and Meta-Analyses (PRISMA) flow chart.
Data extraction
Data were extracted by one reviewer. The following data were extracted from the studies: study design, number of subjects, type of NCS, type/generation of DES, indication for DES, incidence of MACEs, stent thrombosis, MACE according to time from PCI to NCS, MACE associated with first versus second generation DESs and MACE and major bleeding associated with antiplatelet therapy.
Results
Perioperative MACEs following deployment of second generation DESs
Six studies analysed the incidence of MACEs in relation to the time from PCI to NCS, and compared first and newer generation DESs (Table 1).9,22–26 Only one study included patients with third and fourth generation DESs. 22 No study demonstrated a significantly increased risk of MACEs once three months had elapsed between stent implantation and NCS.
MACE incidence according to interval from PCI to NCS; 2nd generation versus 1st generation DESs.
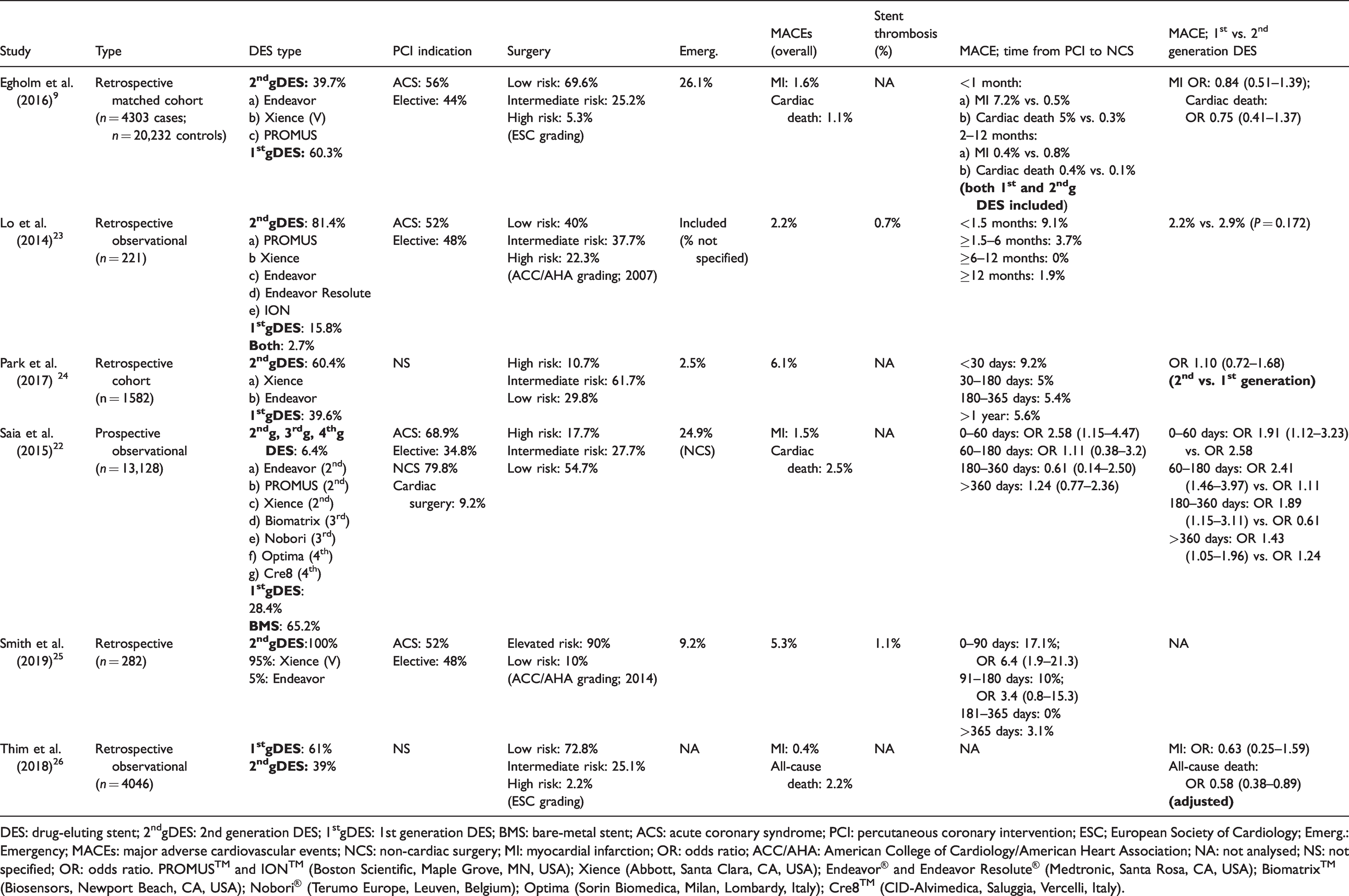
DES: drug-eluting stent; 2ndgDES: 2nd generation DES; 1stgDES: 1st generation DES; BMS: bare-metal stent; ACS: acute coronary syndrome; PCI: percutaneous coronary intervention; ESC; European Society of Cardiology; Emerg.: Emergency; MACEs: major adverse cardiovascular events; NCS: non-cardiac surgery; MI: myocardial infarction; OR: odds ratio; ACC/AHA: American College of Cardiology/American Heart Association; NA: not analysed; NS: not specified; OR: odds ratio. PROMUS™ and ION™ (Boston Scientific, Maple Grove, MN, USA); Xience (Abbott, Santa Clara, CA, USA); Endeavor® and Endeavor Resolute® (Medtronic, Santa Rosa, CA, USA); Biomatrix™ (Biosensors, Newport Beach, CA, USA); Nobori® (Terumo Europe, Leuven, Belgium); Optima (Sorin Biomedica, Milan, Lombardy, Italy); Cre8™ (CID-Alvimedica, Saluggia, Vercelli, Italy).
Smith et al. reported a retrospective cohort study that was exclusively limited to patients with second generation DESs (of which 95% were Xience V everolimus stents) undergoing elective and emergency surgery. 25 The authors reported a significantly high risk of MACEs (17.1%) in the first 90 days following PCI. Statistical significance was lost when emergent surgeries were excluded. 25 Saia et al., in a prospective observational study, found that there was a significantly higher risk of cardiac death or MI in the first 60 days following PCI among patients with newer generation DESs (odds ratio (OR) 2.58; 95% confidence intervals (CI) 1.15 to 4.47). 22 This was the only study that included patients with third and fourth generation DESs. When compared to newer generation DESs, patients with first generation DESs had a significantly higher risk of cardiac death or MI post-surgery for up to 12 months after PCI, and even beyond this time frame. 22 Only 6.4% of recruited patients received newer generation DESs.
In the other four studies identified, subgroup analyses were performed according to the time interval from PCI, but they were not exclusively limited to patients with newer generation DESs. Egholm et al. performed a retrospective matched cohort study based on Danish National Patient Registry data. Thirty-nine percent of patients had second generation DESs, the majority of which were implanted for ACS. 9 The risk of MI, cardiac death and all-cause mortality was only significantly increased within the first month after stent implantation. The authors did not perform a subgroup analysis according to stent generation type on the risk of MACEs in relation to the time interval from PCI to NCS. There was no significant difference between first and second generation DESs in MI, cardiac death or all-cause mortality, although the incidence of these complications was lower among those with first generation DESs. 9 The main finding of Egholm et al. was replicated by a retrospective study by Park et al., which reported that the incidence of MACEs was only significantly elevated following NCS after the first 30 days following DES implantation (9.2%; OR 2.21). 24 A second generation DES was implanted in 60.4% of patients. However, no subgroup analysis on the type of DES was performed. There was no difference between the first and second generation DESs in terms of the risk of MACEs. 24 Thim and colleagues subsequently performed another retrospective analysis on data from the Danish National Patient Registry and reported that first generation DESs were associated with a statistically significant reduction (adjusted OR 0.66) in all-cause death (but not MI) compared with second generation DESs. 26 The final relevant study was a retrospective cohort by Lo et al., in which 81.4% of patients had second generation DESs. 23 The incidence of MACEs was highest (9.1%) when surgery was performed within 45 days of stent implantation but this did not reach statistical significance. This could be explained by an insufficient sample size and a low absolute number of postoperative cardiac events. There was no significant difference between first and second generation DESs for any postoperative complication, including 30-day MACEs. 23
Perioperative antiplatelet therapy
Seven studies that analysed the incidence of perioperative MACEs according to antiplatelet therapy in patients with second generation DESs were identified.21,23–25,27–29 None of the studies included patients with third or fourth generation DES. These studies failed to show a protective effect of antiplatelet therapy on MACEs when the majority of subjects had second generation DESs; nor did they show an association between antiplatelet therapy and major bleeding. However, the studies were underpowered as a result of clinical heterogeneity and insufficient sample sizes.
Smith et al. reported the only study that investigated the risk of MACEs in 282 patients who had NCS after PCI with second generation DESs. 25 The incidence of MACEs was 6.8% in patients who were on DAPT within the seven days preceding NCS; 2.8% in patients who were on aspirin within seven days preceding NCS and 6.7% in those who did not receive antiplatelet medications within this time period (Fisher’s exact test, P = 0.315). This indicated that there was no significant difference in the incidence of MACEs in the three groups of patients. However, the lack of standardised surveillance protocols to detect postoperative MI and subsequent MACEs could have resulted in an underdiagnosis and underestimation of MACEs. The incidence of bleeding was lowest in the group that did not receive antiplatelet therapy (3%) compared to the groups that received single (6%) or DAPT (8%). Antiplatelet therapy was not associated with excessive surgical bleeding. The small number of patients (n = 30) who did not receive antiplatelet drugs prior to surgery could have contributed to a type 2 error.
Six other studies21,23,24,27–29 included patients with second generation DESs undergoing NCS, but no specific subgroup analyses on these patients were performed (Table 2). Of the six, two studies reported a significant association between antiplatelet therapy and MACEs.21,27 Using data from a Korean registry, Hong et al. retrospectively analysed 2179 patients, and 56% of these patients had second generation DESs. 27 Antiplatelet therapy was ceased in 43% of patients before NCS. There was no increased risk of net adverse clinical events (NACEs), which included all-cause death, MI, stent thrombosis and major bleeding postoperatively at 30 days in patients who discontinued antiplatelet therapy (hazard ratio 0.60; 95% confidence intervals (CI) 0.28 to 1.27). This was irrespective of whether surgery was performed one to 12 months post PCI or greater than 12 months post PCI. However, discontinuation of antiplatelet drugs for more than seven days was associated with a significantly higher risk of NACEs (adjusted hazard ratio 6.93) compared to discontinuation for less than seven days. 27
Perioperative antiplatelet therapy, MACEs and major bleeding.
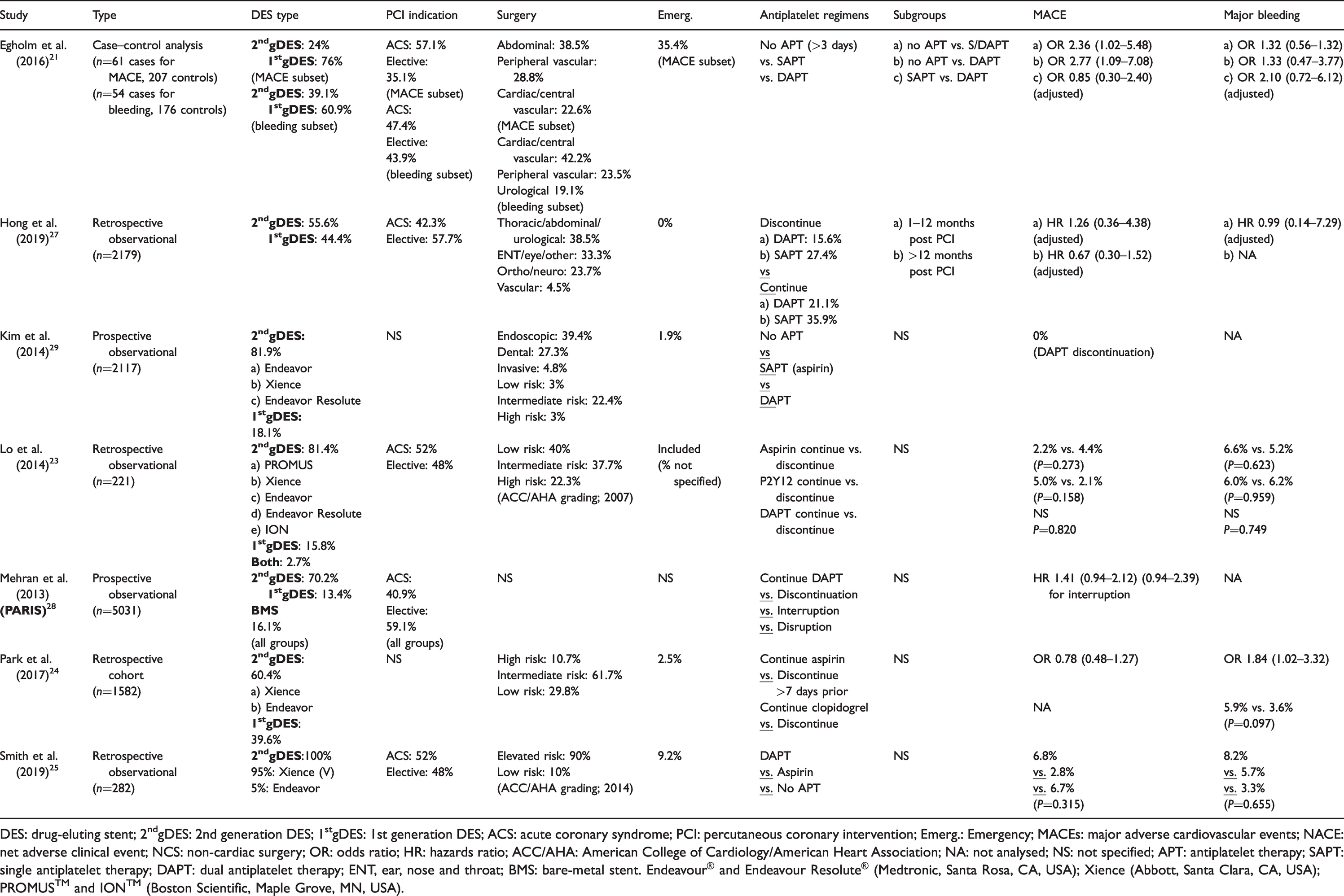
DES: drug-eluting stent; 2ndgDES: 2nd generation DES; 1stgDES: 1st generation DES; ACS: acute coronary syndrome; PCI: percutaneous coronary intervention; Emerg.: Emergency; MACEs: major adverse cardiovascular events; NACE: net adverse clinical event; NCS: non-cardiac surgery; OR: odds ratio; HR: hazards ratio; ACC/AHA: American College of Cardiology/American Heart Association; NA: not analysed; NS: not specified; APT: antiplatelet therapy; SAPT: single antiplatelet therapy; DAPT: dual antiplatelet therapy; ENT, ear, nose and throat; BMS: bare-metal stent. Endeavour® and Endeavour Resolute® (Medtronic, Santa Rosa, CA, USA); Xience (Abbott, Santa Clara, CA, USA); PROMUS™ and ION™ (Boston Scientific, Maple Grove, MN, USA).
Egholm et al. performed two nested case–control studies, and investigated the relationship between antiplatelet therapy, MACEs and major bleeding. 21 Of note, second generation DESs constituted a minority of the stents in these analyses (24% and 39%, respectively). Overall, the absence of antiplatelet therapy was associated with a significantly elevated risk of 30-day MACEs compared to either single or DAPT (OR 2.36, 95% CI 1.02 to 5.48). There was no relationship between antiplatelet therapy and reoperation for bleeding. 21 Based on these results, the authors concluded that patients undergoing moderate- to high-risk surgery should continue at least one antiplatelet agent. The remaining four studies23,24,28,29 found no association between antiplatelet therapy and MACEs. Lo et al., in a retrospective observational study, reported that there was no difference in the incidence of MACEs in patients who continued aspirin (2.2% versus 4.4%, P = 0.273), P2Y12 inhibitors (5.0% versus 2.1%, P = 0.158) or DAPT (P = 0.820) compared to patients who discontinued antiplatelet therapy. 23 The incidence of major bleeding was not associated with antiplatelet therapy. 23 In contrast, a retrospective cohort study by Park et al. reported that the continuation of aspirin until the time of surgery was associated with a significant increase in major bleeding (7.1% versus 4.0%, P = 0.042) and did not reduce the incidence of MACEs (6.9% versus 8.7%, P = 0.319). 24
The Patterns of Non-Adherence to Anti-platelet Regimens in Stented Patients (PARIS) study was a large prospective observational study of over 5000 patients, in which subgroups of patients were predefined as physician-recommended discontinuation, brief interruption for surgery, or due to disruption related to bleeding or non-compliance. In that study, 70.2% of patients had second generation DESs. 28 Compared to patients who were on DAPT, the adjusted hazard ratio for MACEs due to interruption of DAPT was 1.41 (95% CI 0.94 to 2.12; P = 0.10) and disruption was 1.5 (95% CI 1.14 to 1.97; P = 0.0042), suggesting that the risk of MACEs was not increased in the two groups of patients. Finally, Kim et al. reported no association of MACEs with DAPT discontinuation among patients with unexpected requests for NCS. 29 Although 81.9% of patients in that study had second generation DESs, the results must be interpreted with caution because of the small sample size (only 111 patients actually discontinued antiplatelet therapy).
Discussion
To the best of our knowledge, this is the first review that specifically examined the risk of MACEs according to the time interval from PCI and the role of antiplatelet therapy in patients with newer generation stents undergoing NCS.
PCI to surgery interval
Overall, the current evidence suggests that the risk of perioperative MACEs is only significantly increased in the first three months after stent implantation in patients with newer generation DESs undergoing NCS. 30 While the risk of MACEs appears no longer significant after three months, the possibility that there is an increased risk cannot be reliably excluded as most of the studies identified were inadequately powered. These findings primarily apply to second generation DESs as five out of six studies identified did not include patients with third or fourth generation DESs. In the non-surgical setting, there is limited evidence that third generation DESs with biodegradable polymers confer a lower risk of stent thrombosis, cardiac death or target vessel vascularisation compared to second generation DESs. 31 Polymer-free fourth generation DESs (with one month of DAPT) were associated with a lower risk (risk difference –3.6%) of stent thrombosis, cardiac death or MI compared to BMSs in high bleeding risk patients (LEADERS FREE trial). 32 However, in the ONYX ONE study, no significant difference was reported between these fourth generation DESs and polymer-based zotarolimus-eluting DESs in high-risk bleeding patients who received DAPT for one month. 33 Patients with polymer-free stents may be less prone to MACEs in the perioperative period and urgent NCS could be considered one month after PCI. The European Society of Cardiology (ESC) 2017 guidelines shortened the time interval between stent implantation and NCS to one month, irrespective of the type of stent provided that aspirin therapy could be continued and access to a cardiac catheterisation laboratory was available 24 hours. 30 This recommendation was based on the results of the Danish registry study discussed above. 9 The ESC guidelines incorrectly cited a matched cohort study by Holcomb et al. as further justification for their recommendation to reduce the minimum duration of DAPT. 34 Holcomb et al. compared patients undergoing NCS within 24 months of stenting with a matched group of non-stented patients. Only 46% of patients had DESs implanted. Contrary to the ESC guidelines, this study did not support the class IIa recommendation by ESC that a P2Y12 inhibitor could be discontinued one month after stent implantation for elective NCS. The authors, in fact, reported that the stented cohort had an increased risk (2%) of 30-day MACEs, postoperative MI and revascularisation for the first 12 months following coronary stenting compared to non-stented patients. 34
Antiplatelet therapy
There is no consistent evidence that discontinuation of antiplatelet therapy for less than seven days increases the risk of MACEs, but the risks increase if it is ceased for more than seven days. Most studies reported no difference in the incidence of major bleeding with antiplatelet therapy.21,23,25 There were no studies that included patients with third or fourth generation DESs. The Surgery After Stenting (SAS) guideline, a European consensus guideline developed by Rossini et al. in 2014, standardised perioperative antiplatelet therapy by balancing the ‘combined ischaemic risk’ of the patient, based on clinical and angiographic features, against the predicted risk of surgical bleeding. 35 Further developments in stent technology, antithrombotic therapy and surgical procedures resulted in the publication of the SAS2 guidelines in 2018. 36 The thrombotic and haemorrhagic risks are individually graded as low, intermediate or high, and recommendations for antiplatelet therapy are tailored according to these risks and the type of surgery (general, cardiac, vascular, urological). 36 The combined ischaemic risk is primarily determined by four factors: PCI to surgery interval, stent type, clinical and angiographic characteristics and premature DAPT cessation (Table 3). Among patients with second generation DESs without clinical or angiographic at-risk features, the combined ischaemic risk is high in the first month following PCI, intermediate at one to three months, low/intermediate from four to six months and low from six months onwards. In patients with clinical or angiographic at-risk features, the combined ischaemic risk remains high for the first three to six months following PCI and only becomes low after 12 months. The SAS2 guidelines suggest that elective surgery should be delayed six months to one year if the combined ischaemic risk is intermediate or high (Table 4). 36 The haemorrhagic risk is based on anticipated blood loss as well as expected difficulties in achieving surgical haemostasis (Table 5).
Surgery After Stenting 2 guideline; 36 combined ischaemic risk.
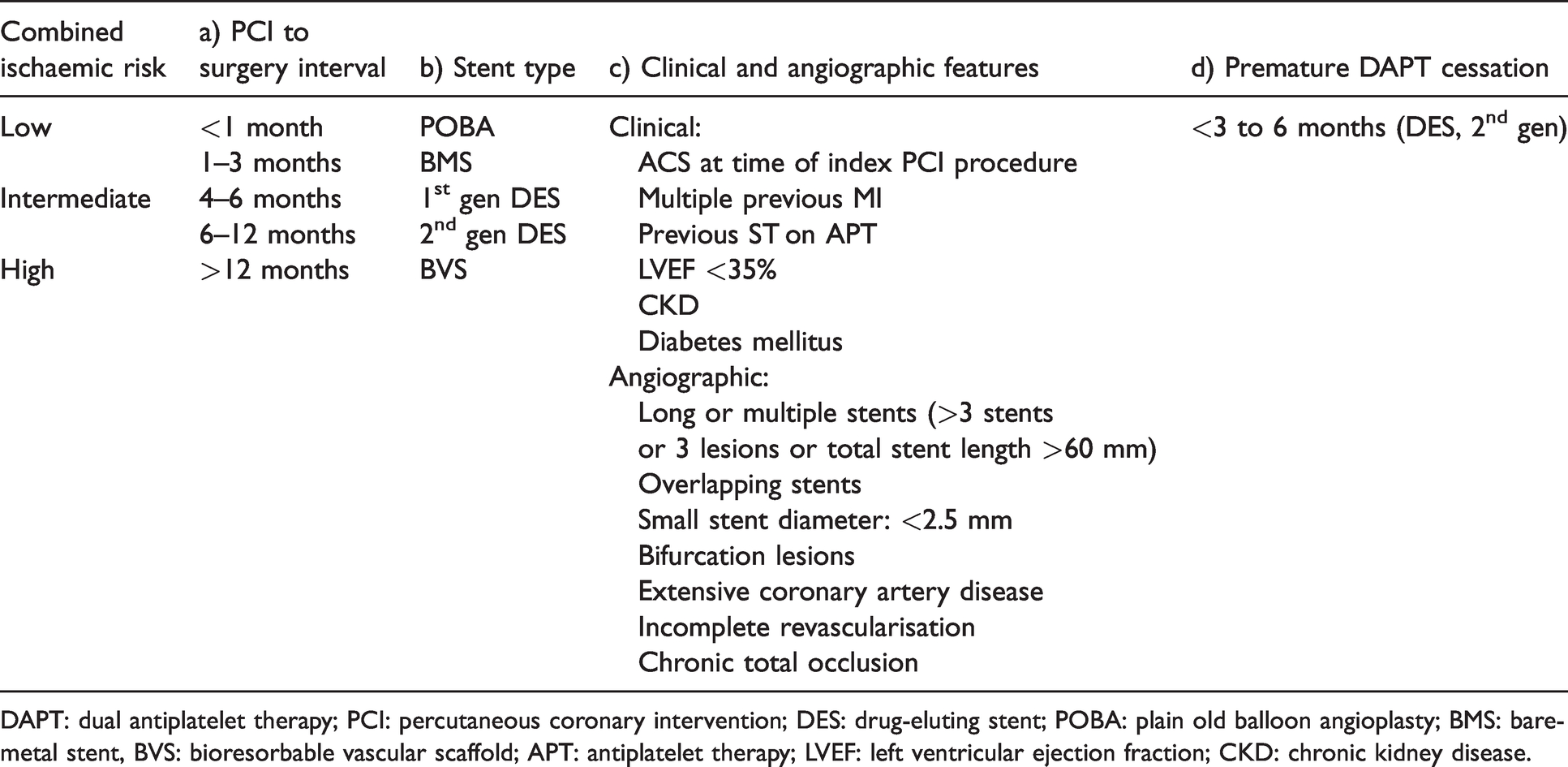
DAPT: dual antiplatelet therapy; PCI: percutaneous coronary intervention; DES: drug-eluting stent; POBA: plain old balloon angioplasty; BMS: bare-metal stent, BVS: bioresorbable vascular scaffold; APT: antiplatelet therapy; LVEF: left ventricular ejection fraction; CKD: chronic kidney disease.
Surgery After Stenting 2 guideline; 36 combined ischaemic risk in patients with second generation DES.

DES: drug-eluting stent; PCI: percutaneous coronary intervention.
Surgery After Stenting 2 guideline; 36 haemorrhagic risk.
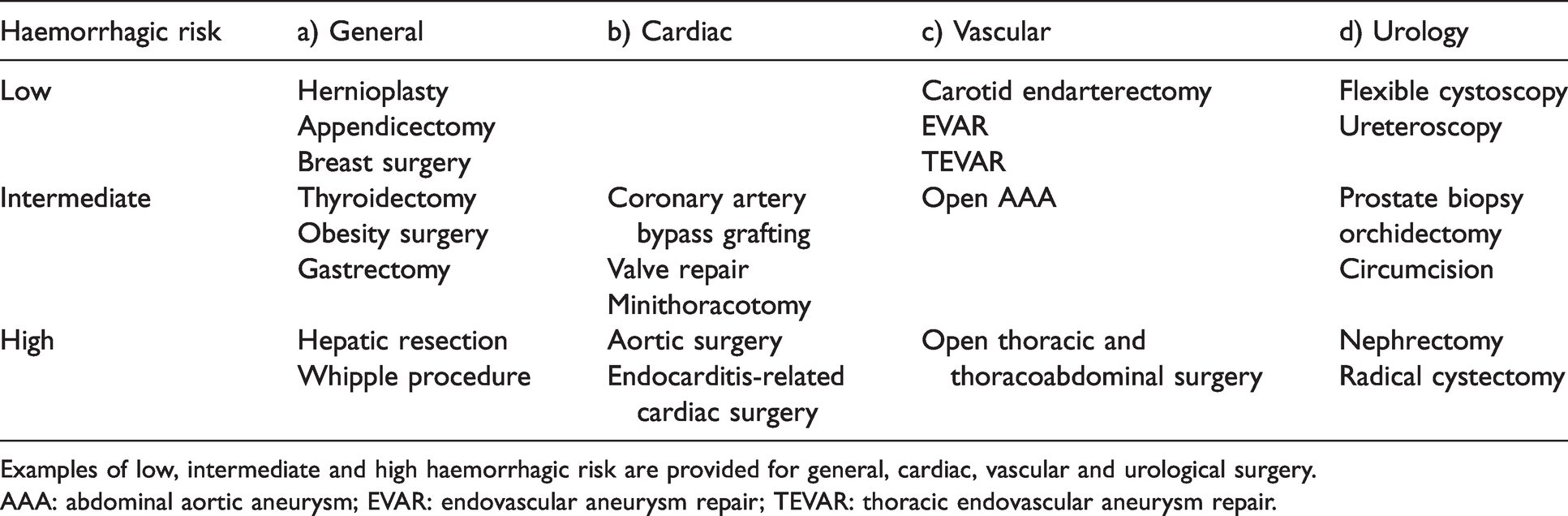
Examples of low, intermediate and high haemorrhagic risk are provided for general, cardiac, vascular and urological surgery.
AAA: abdominal aortic aneurysm; EVAR: endovascular aneurysm repair; TEVAR: thoracic endovascular aneurysm repair.
Summary of recommendations.

DES: drug-eluting stent.
Limitations
There are several limitations in this analysis. Only a few studies which specifically investigated the perioperative outcomes of patients with newer generation DESs undergoing NCS were identified. No randomised controlled trials were available. The studies were generally retrospective and observational in nature and were undertaken at a single centre. Most of the studies were not limited to patients with newer generation DESs and the proportion of patients with newer generation DESs was not recorded. Consequently, many of the studies were inadequately powered to detect significant differences in MACEs or major bleeding among patients with newer generation DESs. There was substantial clinical heterogeneity (type of DES, antiplatelet therapy, patient demographic characteristics) between the studies.
Implications
Although there is no appreciable increase in the risk of MACEs once three months has elapsed between DES implantation and surgery, it may be prudent to defer elective surgery for six months for those patients at higher risk due to previous infarction or complex angiographic features on coronary revascularisation. 30 However, analysis of data from several registries has shown that surgery can be undertaken one month after DES placement with an acceptable risk.9,24 Interventional cardiologists now have the option of using fourth generation DESs to shorten the delay for urgent surgery to one month and minimise the risk of perioperative MACEs in patients with acute coronary syndromes who require urgent NCS. 32
High-quality studies which compare the incidence of MACEs according to the type of antiplatelet therapy (dual, single or no antiplatelet therapy) in patients with newer generation DESs undergoing NCS are not available. There is no consistent evidence that the discontinuation of antiplatelet therapy for less than seven days increases the risk of MACEs, but the risks increase substantially when antiplatelet drug cessation exceeds seven days. Most surgeons consider continuation of DAPT as a relative contraindication to major NCS. Aspirin should be continued but P2Y12 inhibitors could be withheld for three to seven days. In elective surgery (such as intracranial procedures) in which excessive bleeding may be fatal, cessation of DAPT and bridging therapy with tirofiban or intravenous cangrelor (a reversible P2Y12 inhibitor) should be considered. 30 Bridging with low molecular weight heparin is of little benefit because heparin does not inhibit platelet aggregation, a major cause of perioperative stent thrombosis. 35
Patients with low functional capacity, lung disease, diabetes and renal failure have an increased risk of complications during NCS. Scoring systems such as the revised cardiac risk index are useful in evaluating the cardiovascular risk for complex patients presenting for high- and intermediate-risk NCS. Biomarkers such as troponin and brain natriuretic peptide (BNP) plasma levels measured pre- and postoperatively may have additional prognostic value for cardiac events and long-term mortality after major NCS. 33 In patients with acute coronary syndromes, a multidisciplinary approach with a risk–benefit evaluation must be undertaken to decide on the timing of surgery following DES implantation.
Large multicentre studies (or registries) that focus on biodegradable or polymer-free DESs are necessary to define further the optimal window for NCS following PCI with the newer generation DESs and the bleeding risk with continued DAPT.
Footnotes
Author contribution(s)
Declaration of conflicting interests
The author(s) have no conflicts of interest to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
