Abstract

Deep vein thrombosis (DVT) and pulmonary embolism (PE) are common after major trauma. The major pathogenic mechanisms of DVT and PE include a hypercoagulable state, 1 venous stasis2,3 and vascular injury—collectively also called Virchow’s triad. Although anticoagulant prophylaxis remains the cornerstone in reducing thromboembolism in patients after major trauma, it is often considered contraindicated in many major trauma patients who are at risk of or have active bleeding, including those with multiple cerebral contusions with intracranial hypertension. Reducing venous stasis and vascular injury in these patients is pivotal as part of a multimodal approach to reducing the burden of thromboembolism. We hypothesised that femoral venous catheterisation and omitting bilateral lower limb pneumatic compression were significant, and potentially modifiable, mechanical risk factors for developing venous thromboembolism in patients with severe trauma. In this substudy of the da Vinci trial,4,5 we report the associations between the use of femoral venous catheterisation or lower limb pneumatic compression and venous thromboembolism.
In brief, the da Vinci trial recruited and randomly allocated 240 trauma patients, who had an injury severity score greater than 15, with clinical contraindications to anticoagulant prophylaxis within 72 hours of admission to a trauma centre, to receive either a prophylactic vena cava filter or standard care alone. Although there was no significant difference in the primary composite outcome of 90-day mortality or symptomatic PE between the two groups, as a priori secondary outcome, vena cava filters were effective in reducing symptomatic PE among those who could not be anticoagulated within seven days of injury (N = 80; 0% in the filter group versus 14.7% including one fatal PE in the control group; P = 0.01). The number of patients with thrombus entrapped in the vena cava filters (in the filter group) was identical to the number of patients with symptomatic PE in the control group, suggesting that filters were achieving the desired effect in preventing embolisation of lower limb thrombi to the pulmonary circulation. 6
In this substudy, we used univariable and multivariable logistic regression to assess the associations between the use of femoral venous catheterisation (for central vascular access or continuous dialysis), without bilateral lower limb pneumatic compression and venous thromboembolic events. Lower limb fractures, pelvic fractures and the use of prophylactic anticoagulant within seven days of injury were also assessed, and used as covariates to adjust for the independent effect of femoral venous catheterisation and a lack of using bilateral lower limb pneumatic compression as mechanical risk factors for venous thromboembolism.
Of the 240 patients included in the trial, femoral venous catheters were used in 33 patients (13.8%) and two femoral catheters were used in one of these patients within 90 days of enrolment. Six femoral catheters (18.2%) were used for continuous dialysis and the rest were used for central vascular access. The duration of the femoral catheters left in situ was not recorded in the trial, but most would have been used for longer than 24 hours given that none of the study centres had a unit protocol to remove femoral venous catheters within 24 hours of placement. In the univariable analyses, femoral venous catheterisation was associated with an increased risk of symptomatic PE (odds ratio (OR) 7.00, 95% confidence interval (CI) 1.66 to 29.53, P = 0.008), including one fatal PE (3.0% with femoral venous catheter versus 0% without femoral venous catheter; P = 0.012).
Of the 43 patients (17.9%) who were not treated with bilateral lower limb pneumatic compression initially, 32 (74.4%) had lower limb fractures; only seven of these 32 patients (21.9%) were treated with bilateral pneumatic compression after fixation of the fractures. Eleven of those 43 patients (4.6% of the total cohort) were never treated with any lower limb pneumatic compression despite the absence of lower limb injuries. Not having bilateral lower limb pneumatic compression during the immediate phase after severe trauma was associated with an increased risk of lower limb DVT (OR 2.80, 95% CI 1.15 to 6.81; P = 0.023), as well as the composite outcome of lower limb DVT, PE or thrombus entrapped in the filter (OR 2.79, 95% CI 1.27 to 6.16; P = 0.011).
The adverse associations between the two mechanical factors and thromboembolic events remained unchanged after adjusting for lower limb and pelvic fractures and a delay in initiating anticoagulant prophylaxis (Table 1). These findings suggest that femoral venous catheterisation should be avoided or removed as soon as possible (e.g. <24 hours), and bilateral lower limb pneumatic compression should also be used for all major trauma patients to reduce their risk of thromboembolism, especially if they have contraindications to prophylactic anticoagulants. A comprehensive multimodality approach is needed to reduce the burden of venous thromboembolism in patients with severe trauma.
Mechanical predictors of lower limb deep vein thrombosis, pulmonary embolism and thrombus entrapped in the vena cava filter in patients enrolled in the da Vinci trial (N = 240).
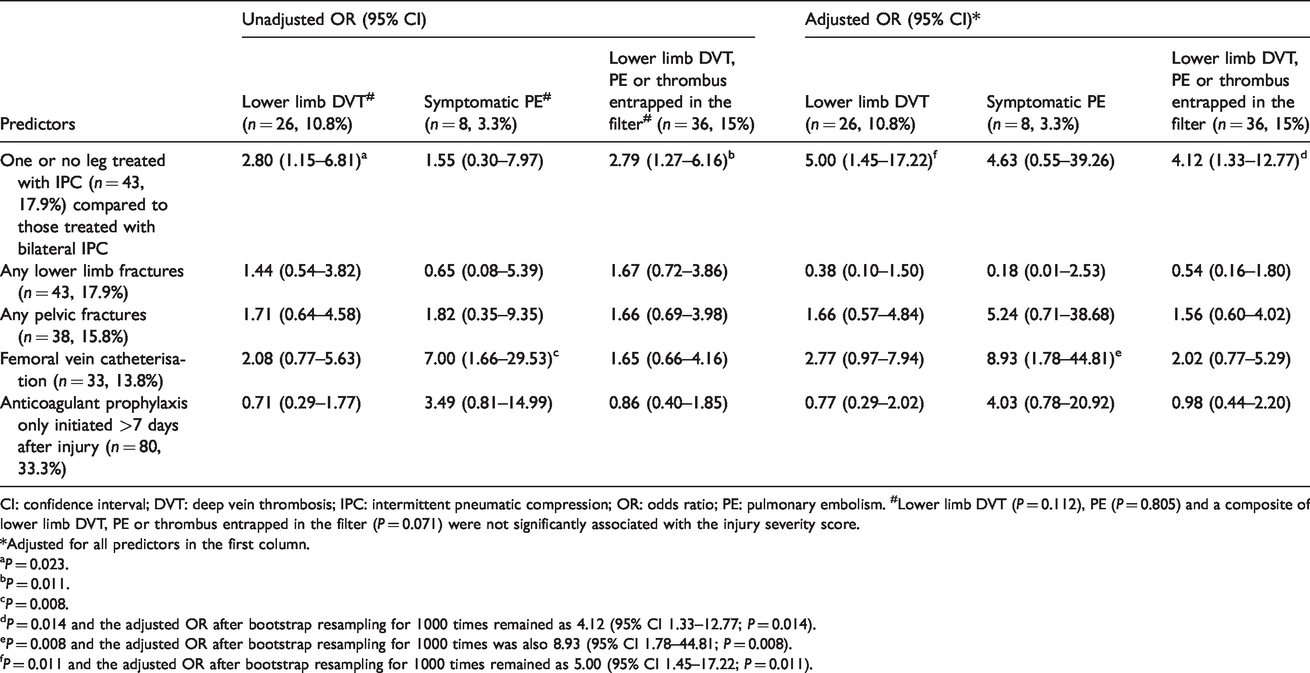
CI: confidence interval; DVT: deep vein thrombosis; IPC: intermittent pneumatic compression; OR: odds ratio; PE: pulmonary embolism. #Lower limb DVT (P = 0.112), PE (P = 0.805) and a composite of lower limb DVT, PE or thrombus entrapped in the filter (P = 0.071) were not significantly associated with the injury severity score.
*Adjusted for all predictors in the first column.
aP = 0.023.
bP = 0.011.
cP = 0.008.
dP = 0.014 and the adjusted OR after bootstrap resampling for 1000 times remained as 4.12 (95% CI 1.33–12.77; P = 0.014).
eP = 0.008 and the adjusted OR after bootstrap resampling for 1000 times was also 8.93 (95% CI 1.78–44.81; P = 0.008).
fP = 0.011 and the adjusted OR after bootstrap resampling for 1000 times remained as 5.00 (95% CI 1.45–17.22; P = 0.011).
Footnotes
Author contribution(s)
Data availability
De-identified data of the trial will be available to reasonable requests after all the substudies have been completed.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KM Ho was funded by the Raine Medical Research Foundation to initiate the da Vinci trial and he has received an honorarium from Cardinal Health for delivering an educational activity related to venous thromboembolism in 2019.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The da Vinci trial was jointly funded by the Raine Medical Research Foundation, WA Health and the Medical Research Foundation of Royal Perth Hospital. The funders had no involvement in the design of the trial, data analysis or the decision to submit this manuscript for publication.
