Abstract
Reducing unnecessary routine diagnostic testing has been identified as a strategy to curb wasteful healthcare. However, the safety and efficacy of targeted diagnostic testing strategies are uncertain. The aim of this study was to systematically review interventions designed to reduce pathology and chest radiograph testing in patients admitted to the intensive care unit (ICU). A predetermined protocol and search strategy included OVID MEDLINE, OVID EMBASE and the Cochrane Central Register of Controlled Trials from inception until 20 November 2019. Eligible publications included interventional studies of patients admitted to an ICU. There were no language restrictions. The primary outcomes were in-hospital mortality and test reduction. Key secondary outcomes included ICU mortality, length of stay, costs and adverse events. This systematic review analysed 26 studies (with more than 44,00 patients) reporting an intervention to reduce one or more diagnostic tests. No studies were at low risk of bias. In-hospital mortality, reported in seven studies, was not significantly different in the post-implementation group (829 of 9815 patients, 8.4%) compared with the pre-intervention group (1007 of 9848 patients, 10.2%), (relative risk 0.89, 95% confidence intervals 0.79 to 1.01, P = 0.06, I2 39%). Of the 18 studies reporting a difference in testing rates, all reported a decrease associated with targeted testing (range 6%–72%), with 14 (82%) studies reporting >20% reduction in one or more tests. Studies of ICU targeted test interventions are generally of low quality. The majority report substantial decreases in testing without evidence of a significant difference in hospital mortality.
Introduction
Ordering routine, daily diagnostic tests, including pathology tests and chest radiographs (CXRs), is common in the intensive care unit (ICU). This occurs despite evidence that such testing may lead to harmful over-investigation, may contribute to iatrogenic anaemia, and is costly; and that a substantial proportion of these tests are normal or unchanged on repeat testing.1–4 Targeted diagnostic testing, in which clinicians order tests in response to specific clinical questions, may reduce unnecessary diagnostic test ordering by up to 50% and has been identified as a priority area to decrease waste by both clinicians and consumers.5,6 However, concern has been raised that targeted testing may increase the risk of missed diagnoses, and high-quality evidence of the safety and effectiveness of targeted compared with routine diagnostic testing is lacking.7,8 The primary aim of this systematic review was to evaluate interventional studies reporting on the implementation of a targeted diagnostic testing strategy in adult patients admitted to the ICU.
Methods
The study was conducted according to a predetermined protocol (PROSPERO registration number: CRD42018115218) and is reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines (Supplementary Appendix eFigure 1).9
Search strategy
The search strategy included OVID MEDLINE, OVID EMBASE and the Cochrane Central Register of Controlled Trials from inception until 20 November 2019. We searched for interventional studies describing the implementation of an intervention designed to reduce routine pathology and/or CXR use in patients admitted to the ICU. The search strategies included the terms “critical care”, “investigation”, “pathology test” and “chest radiograph”. A full list of the search terms is provided in the supplementary appendix. There were no date or language limitations. Conference proceedings were not searched. Two authors (EL and KH) independently searched, screened and determined study eligibility, with any disagreement resolved by consensus. Study quality was assessed using the Cochrane Collaboration’s Risk-of-Bias in Non-Randomized Studies of Interventions and the revised Cochrane Risk-of-Bias Tool for Randomized Trials.10,11
Eligibility criteria
Publications were eligible for inclusion if they were an interventional study (pre- and post-implementation or randomised clinical trial (RCT)), included an adult patient population admitted to an ICU and reported the numbers of at least one routine diagnostic test in the pre- and post-implementation periods. Studies that were presented only in abstract and publications that did not report any outcomes of interest were excluded.
Data collection
A case report form was piloted and refined by the study authors. Study data were then extracted from eligible studies onto a final pre-specified case report form independently, in duplicate, by two authors (EL and KH). Disagreement was resolved by consensus. Where relevant, requests were made to the corresponding authors for data not reported in the primary manuscripts.
Statistical analysis
Eligible studies were pooled for meta-analysis using a random effects model. The primary safety outcome was hospital mortality. The primary efficacy outcome was aggregated tests per patient bed-day. Key secondary outcomes included ICU and hospital length of stay (LOS), duration of mechanical ventilation and individual test numbers. For categorical data, the number of participants with an event was compared with the total number of participants with and without an event. For continuous data, the participant number, mean and standard deviation (SD) were used. Where outcome data were only reported as number, median and range, and where author contact failed to provide means and SD, these were imputed for inclusion in the meta-analysis using validated methods.12 Weighted mean difference (WMD) and relative risk (RR) with 95% confidence intervals (CI) were calculated for continuous and categorical data respectively. A P <0.05 was considered significant. Heterogeneity was measured using the I2 statistic with an I2>40% considered significant heterogeneity. A pre-specified sensitivity analysis was conducted excluding those studies with a high risk of bias in any domain. Publication bias was measured with a funnel plot with the odds ratio (OR) for the primary outcome plotted against the standard error of the log OR. To provide comparative estimates of cost savings, the reported currency was adjusted for inflation using the gross domestic product deflator from the International Monetary Fund: World Economic Outlook Database and then exchanged to US dollars.13,14 The statistical analysis was conducted using Stata (Intercooled Version 11.2, StataCorp, College Station, TX, USA).
Results
The search identified 94 potentially relevant studies. Of these, 26 studies with more than 44,000 patients were included in the systematic review.15–40 There were three studies that did not report the total number of patients.21,32,33 The process of identifying eligible studies is provided in Figure 1.
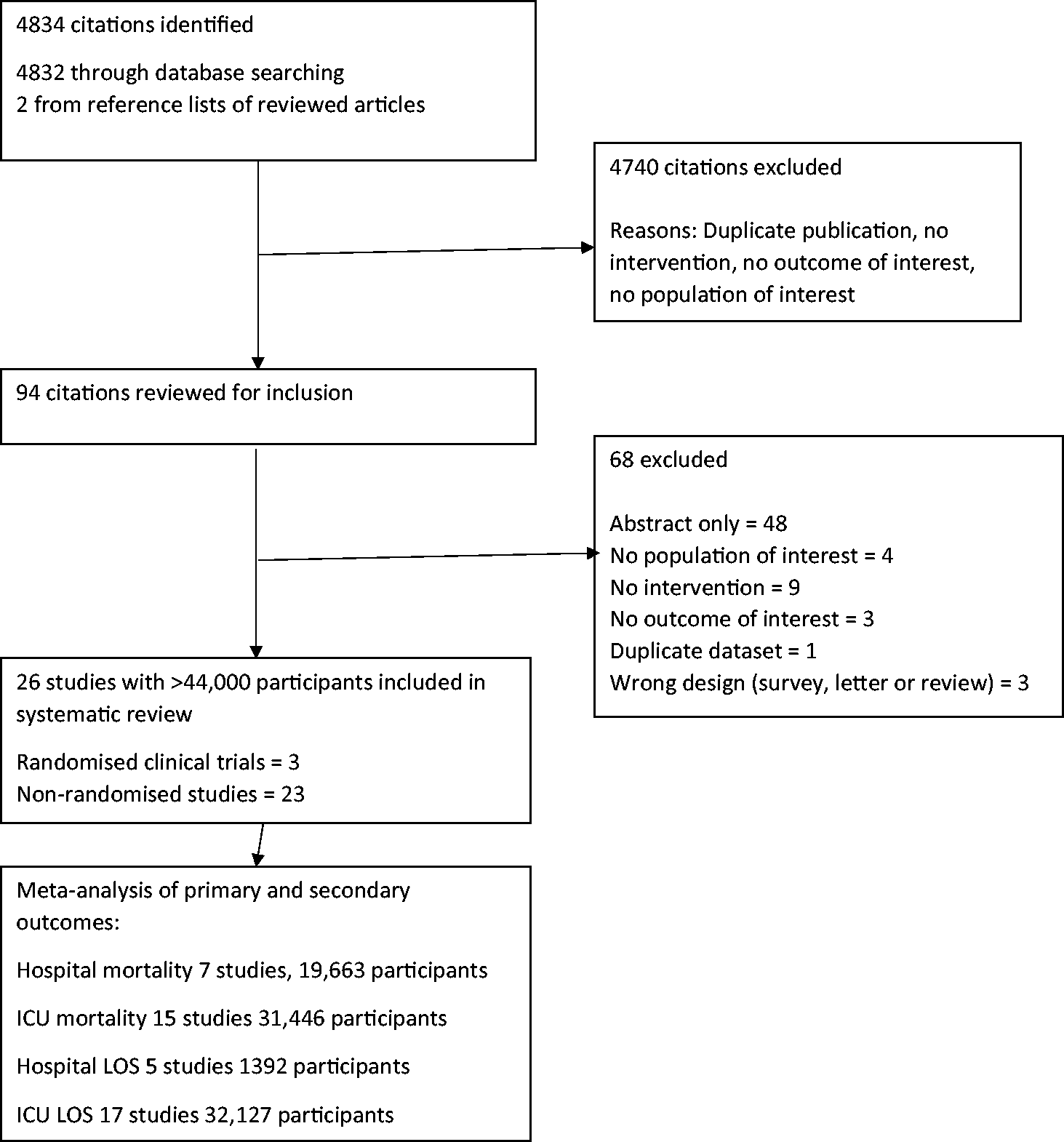
Preferred Reporting Items for Systematic Reviews and Meta-analyses study screening, inclusion and meta-analysis.
Characteristics of the included trials
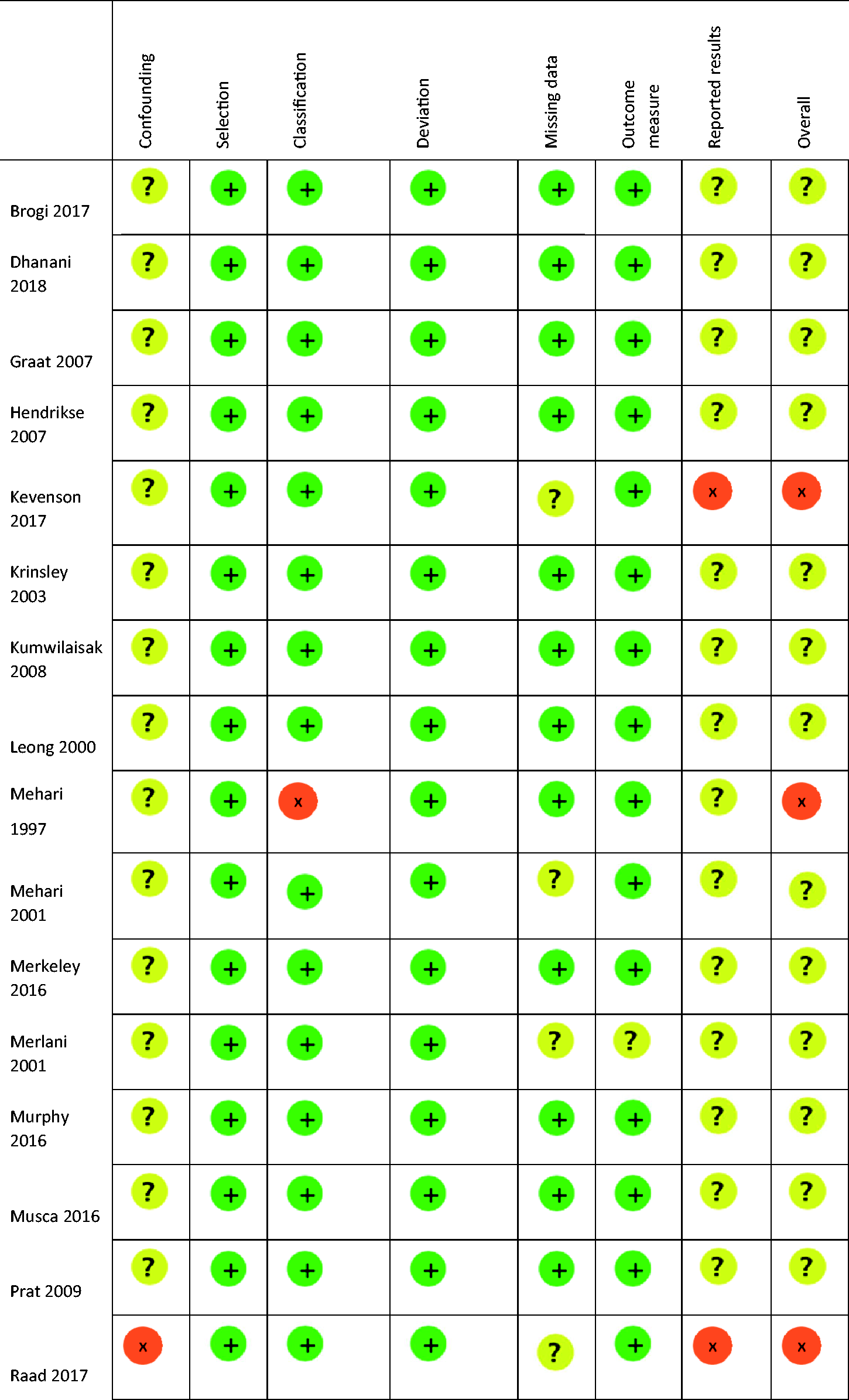
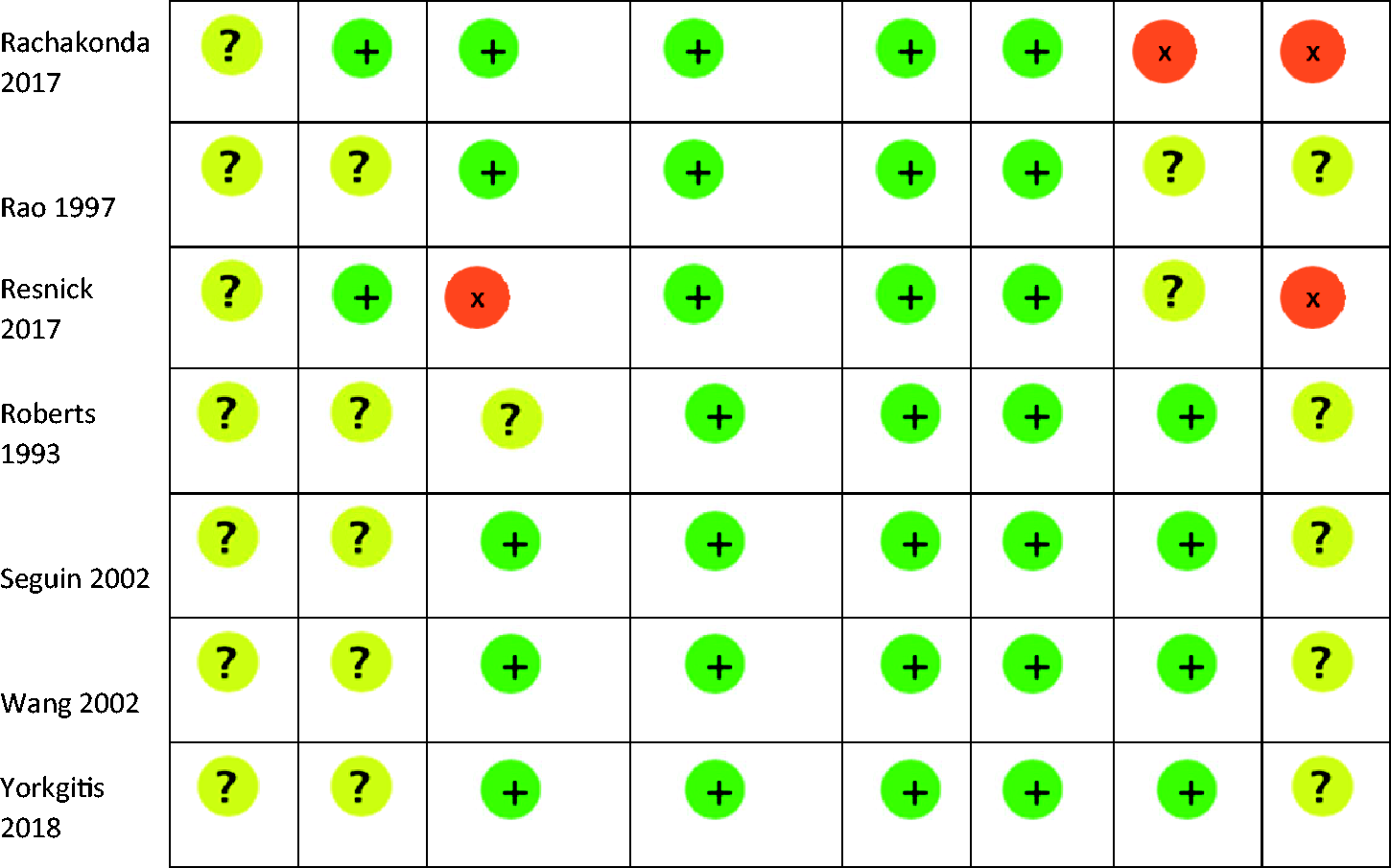
The included trials were published between 1993 and 2018. They were conducted in Europe, North America and Australasia. Additional data for the primary outcome were received from two authors.20,30 There was substantial variation in the intervention design and tests targeted for reduction (Table 1). Overall, the quality of the included studies was low, with all included studies having at least one domain of greater than low risk of bias (Table 2). A description of the rationale for each bias assessment is provided in the supplementary appendix eTable 1.
Study characteristics.40

N: number of participants; ICU: intensive care unit; LOS: length of stay; CXR: chest radiograph; C: control; I: intervention; PI: post-intervention; MV: mechanical ventilation; NR: not reported; ABG: arterial blood gas; CLABSI: central line–associated bloodstream infection; CCU: coronary care unit; F/U: follow-up; #: duration of study defined from date of control group data to date of intervention or post-intervention data. A period of intervention or absence of data collection between these two points is represented when overall duration is greater than stated period of control, intervention and post-intervention.*Group A and C included from reported study. Group A was pre-intervention, Group C was post intervention post-intervention, Group B was an intermediate group that occurred temporarily between A and C and so was excluded.
Studies assessed using ROBINS-I: Risk of Bias in Non-randomized Studies of Interventions assessment.

Hospital mortality
In-hospital mortality, reported in seven studies (n = 19,663), occurred in 828/9815 patients (8.4%) in the targeted testing group and 1007/9848 patients (10.2%) in the comparator group, (relative risk (RR) 0.89, 95% CI 0.79 to 1.01, P = 0.006, I2 38.5%) (Figure 2). There was no evidence of publication bias on funnel plot (eFigure 2).
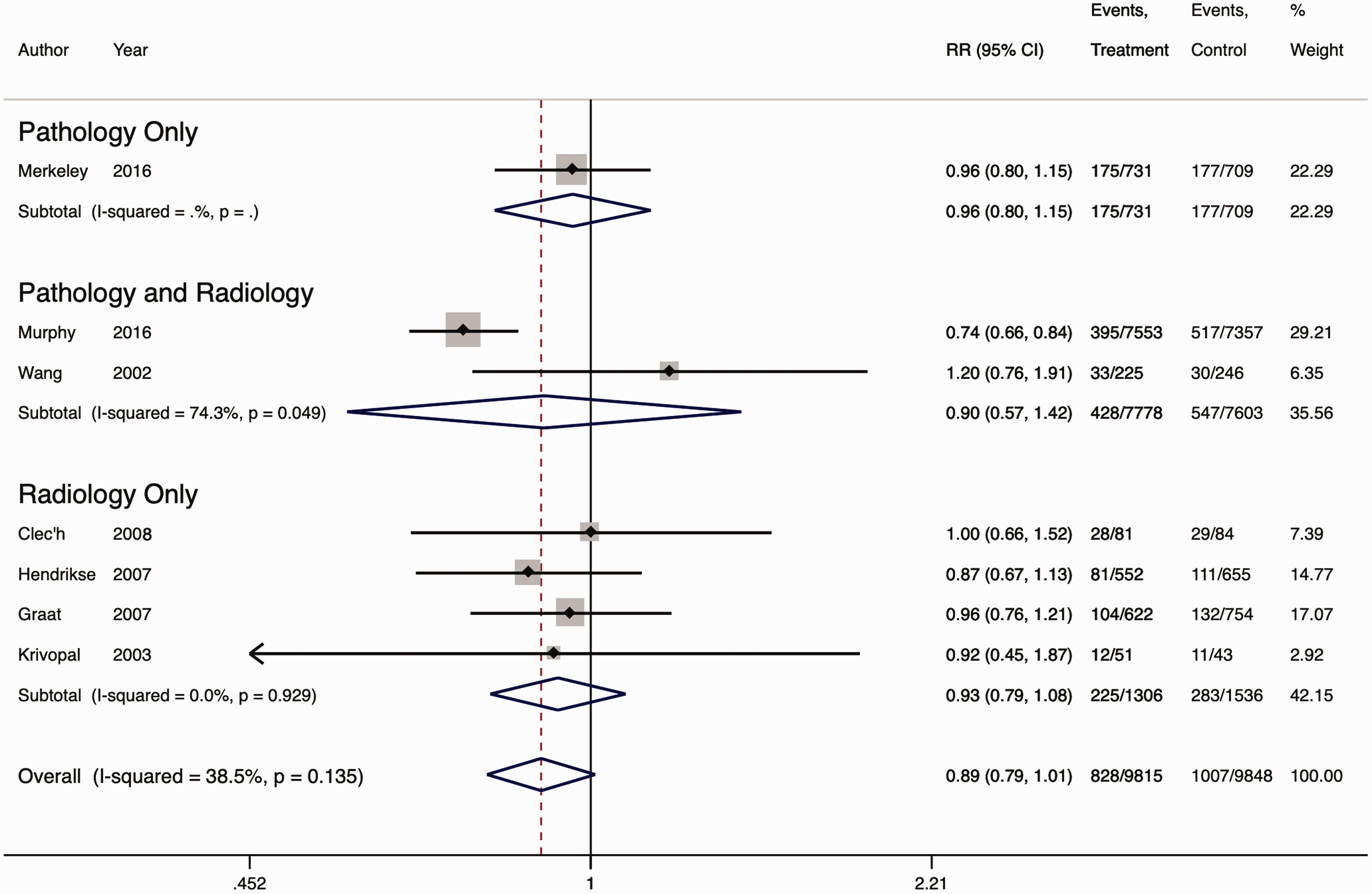
Forest plot for the association between targeted test reduction interventions and hospital mortality.
Targeted test reduction
Of the 18 studies reporting a difference in testing between the two groups, all reported a decrease associated with targeted testing (range 6%–72%), with 14 (82%) studies reporting >20% reduction in one or more tests and 11 (65%) studies reporting >30% reduction in one or more tests. Of the 23 studies reporting whether testing reduction was statistically significant, reduction was significant for 19 (83%) studies. Heterogeneity in reporting (e.g. reporting ICU admission number rather than bed days) and missing data (e.g. lack of estimated variance) precluded meta-analysis of the diagnostic tests per patient bed-day. Overall, there was a consistent reduction in proportion of tests conducted, ranging from 6% to 64%. A summary of the effect of targeted testing on testing frequency is provided in Table 3.
Targeted test reduction.
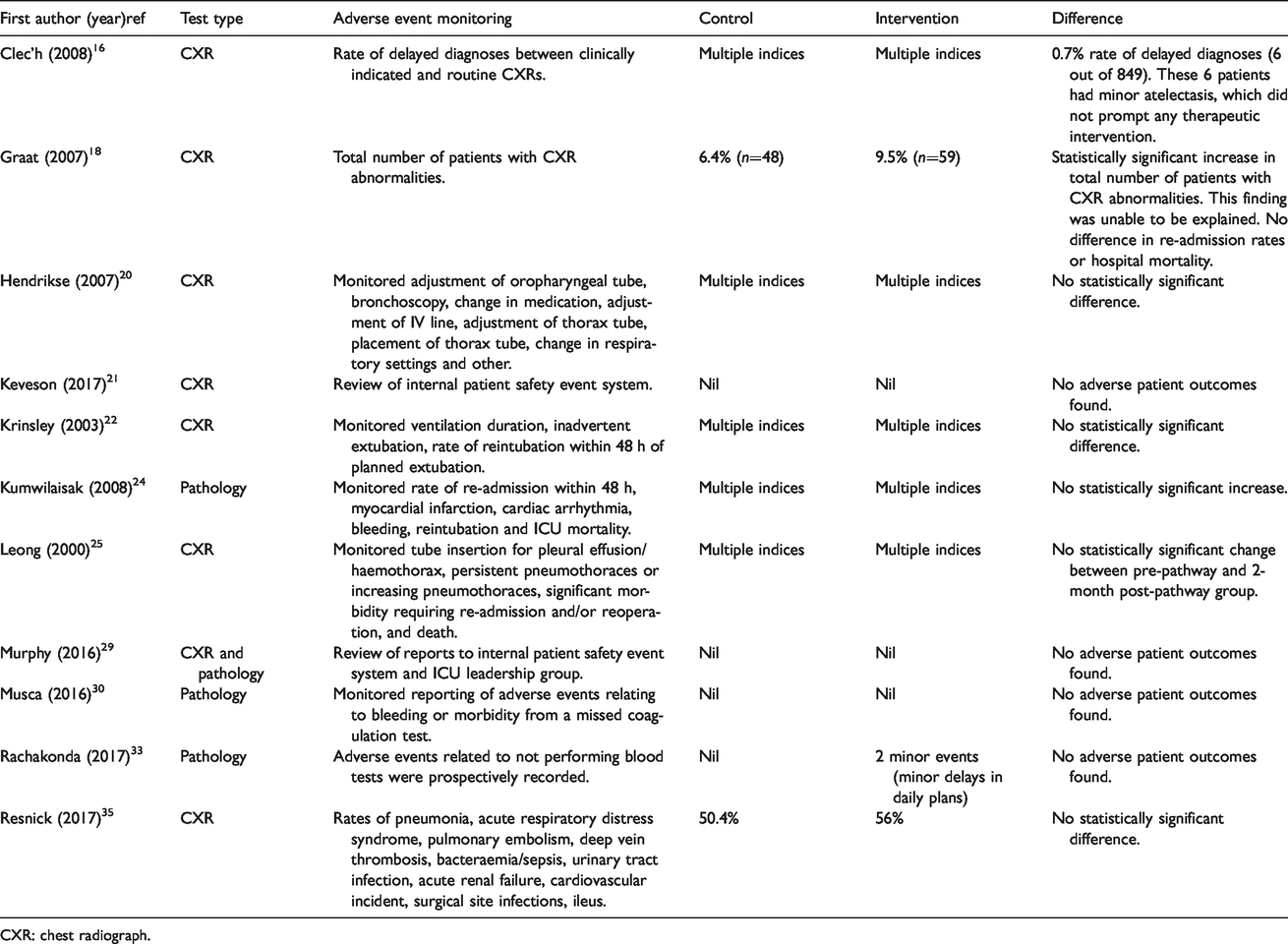
NR: not reported; N/A: not applicable; CXR: chest radiograph; ABG: arterial blood gas.
*: follow-up study demonstrating sustained reduction; #: % reduction/patient bed-day; ^: no significant reduction between pre- and two-month post-pathway group.
Secondary outcomes
ICU mortality was reported in 15 studies (n = 31,446) and occurred in 1153/16,052 patients (7.2%) in the targeted testing group and 1288/15,394 (8.4%) patients in the comparator group, (RR 0.91, 95% CI 0.81 to 1.02, P = 0.10, I2 45%), (Supplementary Appendix eFigure 3).
ICU LOS was reported in 17 studies (n = 32,127). Diagnostic test reduction was associated with a WMD in ICU LOS of –0.28 (95% CI –0.48 to –0.07, P = 0.01, I2 95.6%; see Supplementary Appendix eFigure 4). Hospital LOS was reported in five studies, all investigating CXR reduction only (n = 654). Diagnostic test reduction resulted in a WMD in hospital LOS of –0.14 (95% CI –0.51 to 0.23, P = 0.46, I2 9%; see Supplementary Appendix eFigure 5).
Adverse events
Of the 11 studies reporting adverse events, 9 (82%) found no adverse events or no statistically significant difference in adverse events (Table 4).
Adverse events.
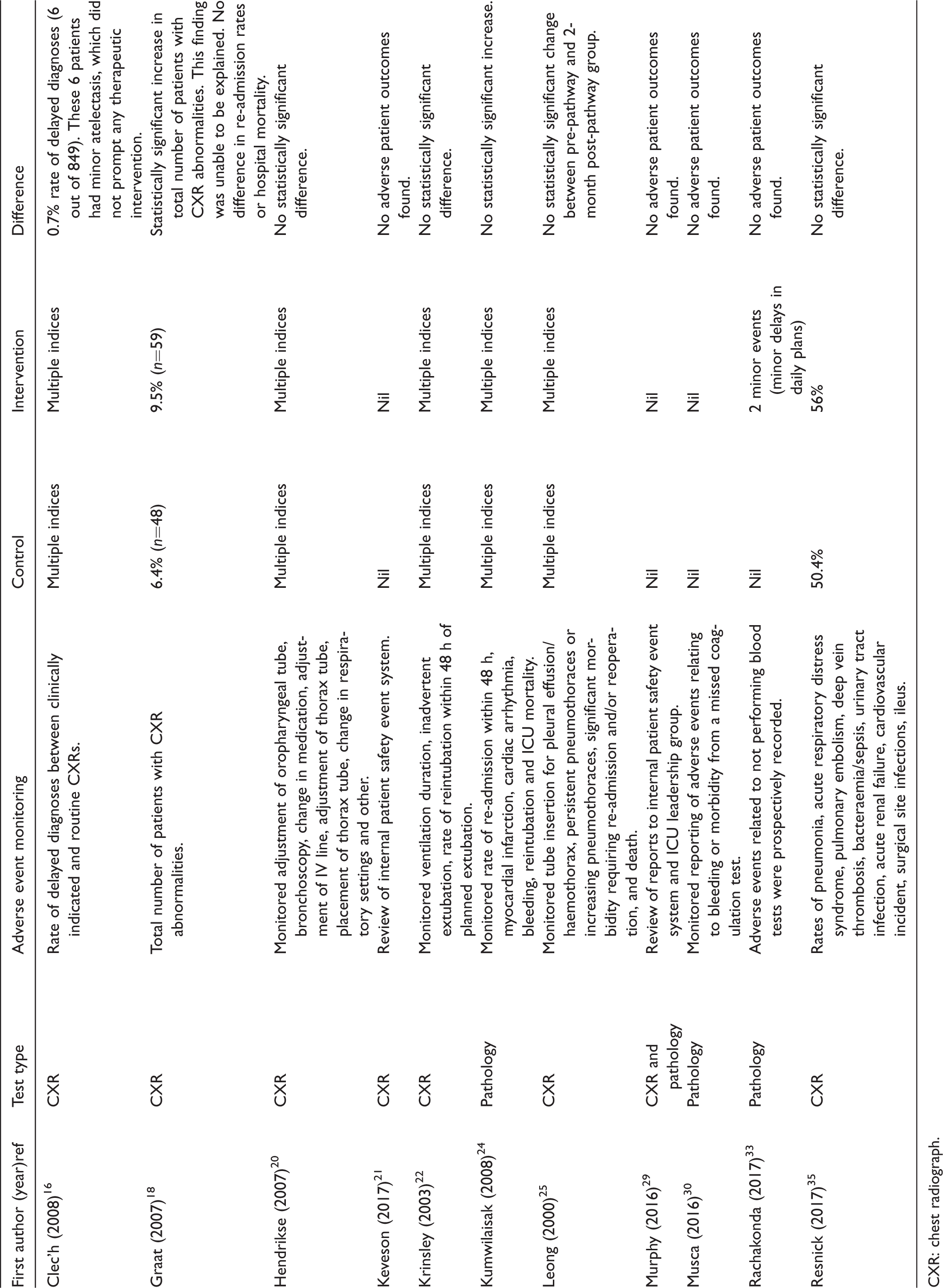
CXR: chest radiograph.
Costs
Estimated annual ICU cost savings were reported in 15 studies. The annual saving per ICU bed in US dollars and adjusted for inflation to 2018 ranged from $1027 to $24,163 (Supplementary Appendix eTable2).
Discussion
Our systematic review and meta-analysis included 25 studies, which were generally of low to moderate quality, of interventions for reduction in ICU CXR, pathology or a combination of the two. Meta-analysis of seven studies suggested that targeted testing interventions were not associated with a significant difference in hospital mortality (RR 0.89, 95% CI 0.79 to 1.01, P = 0.006). These findings were consistent with a point estimate for ICU mortality and ICU and hospital LOS that favoured targeted testing but was not statistically significant. Although meta-analysis of overall test reduction per patient bed-day was not feasible due to trial reporting heterogeneity, included studies consistently achieved a substantial reduction in testing.
Our findings have several important implications. Previous systematic reviews have only quantitatively summarised CXR interventions.41,42 Our review demonstrated that several interventions include both pathology and radiology interventions, and that decrease in testing appears to be consistent across diagnostic testing type. Although no difference in hospital mortality associated with targeted testing is somewhat reassuring, this was reported in only a minority of included studies. Furthermore, the design and quality of these studies precluded definitive conclusions about the safety of targeted testing interventions. Future studies with robust designs, powered to exclude a clinically meaningful difference in mortality and conducted in multiple centres, are needed to provide high quality, generalisable evidence of safety.43 Such studies appear warranted given that stakeholders consider unnecessary testing in ICU to be a highly prevalent problem, ICU care is extremely costly with pathology and radiology costs representing 5%–25% and 4%–10% of direct costs respectively, and we consistently found in this study that targeted testing interventions were associated with decreased test ordering and costs.44–48
Our review found that adverse events were not increased in the majority of studies, and that in the two studies that did observe an increase in adverse events, these were minor and not associated with an increase in ICU LOS or mortality. These findings are reassuring given the concern that decreasing testing may increase the number of missed diagnoses.7,8 However, adverse events were only reported in 11 (44%) studies and generalisability remains uncertain.
A wide range of cost savings were reported in the included studies, but the majority range in the order of the salary of a junior medical officer to a staff specialist. Recouping, or being able to redirect this amount of funding to equipment or staffing, has the potential to increase efficiency and safety, in an era in which the global health sector is growing faster than the economy.49
There are several limitations to our systematic review. None of the included studies were of low risk of bias and the majority were before and after designs that are particularly vulnerable to bias resulting from secular trends. Although only seven studies contributed to the primary outcome, the direction of effect was consistent with the 15 studies included in the ICU mortality meta-analysis. Heterogeneity precluded an aggregate estimate of test reduction and few of the included studies described the process of developing the intervention or of assessing its sustainability.
Conclusion
Studies of ICU targeted test interventions are generally of low quality. The majority report a substantial decrease in testing and associated costs without evidence of significant differences in hospital mortality or adverse events.
Supplemental Material
sj-pdf-1-aic-10.1177_0310057X20962113 - Supplemental material for Safety and efficacy of routine diagnostic test reduction interventions in patients admitted to the intensive care unit: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-aic-10.1177_0310057X20962113 for Safety and efficacy of routine diagnostic test reduction interventions in patients admitted to the intensive care unit: A systematic review and meta-analysis by Katherine P Hooper, Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Writing original draft, Conceptualization, Formal analysis, Methodology, Supervision, Writing review editing, Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Writing review editing in Anaesthesia and Intensive Care
Footnotes
Author contribution(s)
Acknowledgements
The investigators would like to thank all the authors of the primary research material and in particular Dr Steven Musca and Dr Peter Spronk who provided additional data or clarification of their work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Edward Litton is supported by a National Health and Medical Research Foundation Early Career Fellowship.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.