Abstract
A telephone survey of cardiac anaesthetists and perfusionists at the 29 public hospitals providing adult cardiac surgical services in Australia and New Zealand was carried out between December 2019 and January 2020. The aim was to investigate current practice with regard to selected contentious elements of anaesthetic and perfusion management during cardiopulmonary bypass; primarily relating to bypass circuit priming, blood conservation methods and point-of-care coagulation testing. There was a 100% response rate. The average number of adult public cardiopulmonary bypass cases per hospital was 508 (160–1400). For cardiopulmonary bypass cases, ten hospitals (34%) routinely used a cell saver and the remainder used a cell saver selectively. Residual blood remaining in the cardiopulmonary bypass circuit was processed using a cell saver routinely in four hospitals (14%) and selectively in 23 (79%). Acute normovolaemic haemodilution was rarely used. Retrograde autologous priming was used routinely in seven hospitals (24%) and selectively in 16 (55%). All hospitals had access to point-of-care coagulation testing. The majority of hospitals targeted an activated clotting time of 480 s (range 400–500 s) prior to commencing cardiopulmonary bypass. There was marked geographic variation in access to fibrinogen concentrate. The cardiopulmonary bypass circuit prime solution was primarily a balanced crystalloid in most hospitals; however, there was significant variation regarding the addition of human albumin, mannitol, sodium bicarbonate and other medications. Many of the interventions examined were used on a case-by-case basis. These findings support the need for further research to define more evidence-based practice of these interventions.
Keywords
Introduction
Several key professional bodies that represent anaesthetists, perfusionists and cardiothoracic surgeons provide evidence-based guidelines addressing perioperative blood management and the conduct of adult cardiac surgery involving cardiopulmonary bypass (CPB) (hereafter ‘on-pump surgery’).1–5 However, there is little high-level evidence to support the guidelines over a range of decisions and interventions available to clinicians managing patients on CPB.
There are 29 public hospitals that provide adult cardiac surgical services across Australia and New Zealand. These hospitals differ in case load, case-mix and provision of surgical and perfusion services. Established registries capture a wealth of data relating to cardiac surgery and perfusion practice, but not from all public hospitals in Australia and New Zealand that perform cardiac surgery. 6 , 7 The number of hospitals using a medical perfusionist only versus a clinical perfusionist only versus a mixed-perfusion staffing model (both medical and clinical perfusionist) is also unclear.
The primary aim of this survey was to investigate the range of current practice in Australian and New Zealand public hospitals in relation to selected contentious elements of anaesthetic and perfusion management during CPB, primarily relating to bypass circuit priming, blood conservation methods and point-of-care (POC) coagulation testing. A secondary aim was to document the approximate annual CPB caseload and to categorise differences in the perfusion staffing model across hospitals in Australia and New Zealand.
Materials and methods
The practices explored in this survey included acute normovolaemic haemodilution (ANH), retrograde autologous priming (RAP), bypass circuit prime volume and constituents, the type of pump used as the primary arterial pump, activated clotting time (ACT) targets pre CPB and the use of POC coagulation testing. We also explored cell salvage (CS) use both as an alternative to cardiotomy suction and to process all or a portion of the blood remaining in the CPB circuit after separation from CPB (hereafter ‘pump blood’). For the purpose of this paper the term RAP will be used to encompass both retrograde arterial and/or venous antegrade priming of CPB circuits.
This telephone survey of the 29 public hospitals in Australia and New Zealand that undertake adult cardiac surgery was prospectively approved by the Gold Coast Health Human Ethics Research Committee (GCHHS HREC Reference number LNR/2019/QGC/58653; date of approval 8 November 2019). The survey items are provided in Appendix 1. The lead author (SP) telephoned both a cardiac anaesthetist (a Fellow of the Australian and New Zealand College of Anaesthetists practising in the subspecialty of cardiac anaesthesia) and a perfusionist (clinical or medical) from each hospital. Calls were made during working hours through each hospital’s switchboard to either the department of anaesthesia or perfusion services. Arrangements were then made to speak with either an on-duty or on-call cardiac anaesthetist and a perfusionist. If the cardiac anaesthetist was also a practising medical perfusionist at the same hospital, a separate perfusionist was not contacted unless this was suggested by the anaesthetist. In each call, the lead author introduced himself and explained the survey and the approximate time required for participation. If the practitioner was unable to participate at that time, they were invited to reschedule a more convenient time or nominate a suitable available colleague. The anaesthetists and perfusionists were asked a standard and identical set of questions. Questions were clarified by the interviewer as needed and respondents were provided time to check details or confer with colleagues if required.
Questions 1–5 addressed basic demographics, case numbers and organisation of perfusion services. In hospitals where both public and private services were offered, public case numbers were estimated by the respondents. Questions 6–7 sought to address ANH prior to CPB. Responses were categorised as never, selectively, or always. If a hospital selectively used ANH, respondents were asked it the practice was ‘rare’ or ‘common’. Questions 8–11 related to CS use and management of pump blood after aortic decannulation. Where selective processing of pump blood was undertaken, additional information regarding hospital guidelines or indications was sought. Questions 12–17 were around perfusion practice. Coagulation management during and post CPB was explored in questions 18–22.
Results
The survey was conducted between December 2019 and January 2020. Respondents were all practising clinicians working regularly in the cardiac theatres of the hospital surveyed. All cardiac anaesthetists were Fellows of the Australian and New Zealand College of Anaesthetists (FANZCA). The demographic data obtained from FANZCA and clinical perfusion respondents are presented in Table 1.
Respondent demographics and hospital data.

FANZCA: Fellow of the Australian and New Zealand College of Anaesthetists.
Clinical perfusionist total includes one practitioner from a mixed perfusion staffing model; ‘on-pump’ refers to cardiac surgical cases involving cardiopulmonary bypass.
The extrapolated number of CPB cases performed in Australian and New Zealand public hospitals per year was 14,739, with 2560 (17.4%) performed in New Zealand. The extrapolated number of CPB cases performed by each hospital per year ranged from 160 to 1400, with an average of 508; a further breakdown is provided in Table 1.
Perfusion services were provided entirely by clinical perfusionists in 73% of the public hospitals. Public hospitals with an exclusively medical perfusion service were limited to Queensland and New South Wales. Western Australian hospitals had mixed models. All medical perfusionists (seven) surveyed were also practising cardiac anaesthetists (the authors accept this may not always be the case).
The majority of hospitals (62%) never performed ANH prior to cardiac surgery, and the procedure was rarely performed at ten of the remaining 11 hospitals. One hospital used ANH for approximately 40% of CPB cases.
All hospitals surveyed had CS capability. In total, ten (34%) used CS routinely for all CPB cases and 19 (66%) used CS selectively based on a risk assessment of patient and surgical factors or surgeon preference. Surgical indications for CS included one or more of the following: redo-sternotomies, double valve surgery or combined valve surgery plus coronary artery bypass grafting (CABG), emergency cases, cardiac transplant surgery, and ventricular assist device implant or explant procedures. Heparin was used as the anticoagulant for the CS circuit in all hospitals. Several respondents from hospitals that selectively used CS reported that routinely using CS for all CPB cases did not appear to be cost-effective, as the volume of blood collected was often insufficient for processing.
Use of CS to process pump blood after separation from CPB varied (Table 2). Practice at the majority (79%) of surveyed hospitals was to consider CS to process pump blood selectively. Many respondents reported that the greatest limitation of this practice was the time required to process pump blood with CS, given their need to have blood available for volume replacement in the early post CPB period. Other factors considered at hospitals selectively processing pump blood with CS included consideration of pathophysiological factors individual to the patient, total pump blood volume and surgeon preference. One respondent described how the duration of CPB was a factor considered at their hospital, where the perception was that after relatively long CPB times CS may improve the ‘quality’ of pump blood through the removal of inflammatory mediators. Of the four hospitals (13%) where pump blood was routinely processed, respondents from two hospitals described an approach where a portion of pump blood (e.g. 400–1000 ml) is routinely collected for immediate use by the anaesthetist and the remaining pump blood is processed using CS. A method to maximise the collection of pump blood for CS processing was described by one respondent, which involved infusing a crystalloid solution through the bypass circuit after its connection to the cell saver.
Hospital use patterns of specific anaesthetic and perfusion interventions.
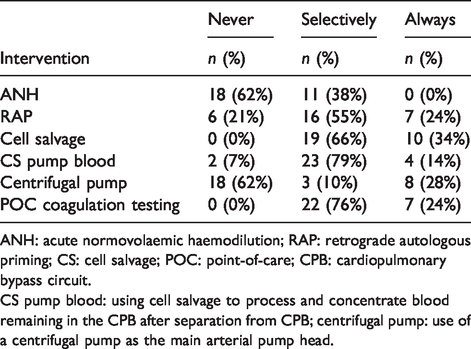
ANH: acute normovolaemic haemodilution; RAP: retrograde autologous priming; CS: cell salvage; POC: point-of-care; CPB: cardiopulmonary bypass circuit.
CS pump blood: using cell salvage to process and concentrate blood remaining in the CPB after separation from CPB; centrifugal pump: use of a centrifugal pump as the main arterial pump head.
During CPB all hospitals used cardiotomy suction to collect and return shed blood to the CPB circuit, and no hospitals reported using CS exclusively as an alternative to cardiotomy suction. A small number of respondents described the use of CS in addition to cardiotomy suction during CPB.
Roller pumps (RP) were used routinely as the primary arterial pump head in 21 hospitals (74%) with centrifugal pumps (CP) used in the remaining eight (26%). Of the hospitals using RP as the primary arterial pump, 15% reported selective use of CP.
The mean and median pump prime volume, prior to cutting out any filters or RAP, was 1500 ml. The highest prime volume was 2000 ml. If respondents reported more than one prime volume due to possible variations in circuit configuration, the volume corresponding to the most commonly used adult circuit configuration was used. Data regarding pump prime volume are presented in Table 3. Of the cardiac anaesthetist respondents, 22 (76%) were not aware of the exact prime volume or its constituents or additives.
Cardiopulmonary bypass circuit prime composition and volume.
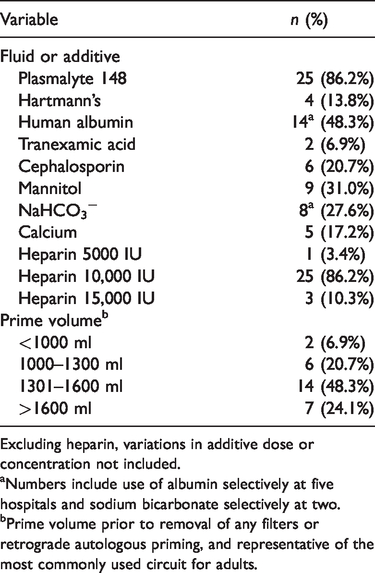
Excluding heparin, variations in additive dose or concentration not included.
aNumbers include use of albumin selectively at five hospitals and sodium bicarbonate selectively at two.
bPrime volume prior to removal of any filters or retrograde autologous priming, and representative of the most commonly used circuit for adults.
Plasmalyte 148 (Baxter International Inc. Sydney, New South Wales) was used as the main priming fluid in 25 hospitals (86%). The remaining hospitals used Hartmann’s solution (Baxter International Inc. Sydney, New South Wales). None of the hospitals used synthetic colloids in their CPB prime.
Additions to the prime solution varied. All hospitals added unfractionated heparin to the CPB circuit prime, with most hospitals adding 10,000 IU (Table 3). Albumin was added to the prime in 50% of hospitals (to make up the total prime volume); either as a 20% or a 4% solution (Table 3).
Overall, 23 hospitals (79%) performed RAP, seven (24%) did so for all CPB cases and 16 (55%) on a selective basis (Table 3). The primary reason stated for both routinely and selectively undertaking RAP was the preservation of blood. Several factors were described that were seen to limit the utility of performing RAP such as the neutral or slightly negative fluid balance of patients pre CPB. Some perfusionists reported that the cardiac anaesthetists at their hospitals administered minimal intravenous fluid prior to CPB, and if RAP was performed invariably the displaced prime would need to be returned to the reservoir during CPB. Others felt that performing RAP was beneficial despite this, because the initial ‘crystalloid load’ received by the cerebral circulation was lower when CPB was initiated and the nadir haematocrit was higher. Some perfusionists alluded to a lack of support from other perioperative team members as an issue preventing RAP; for example, one reported that ‘our surgeon is very fast and we don’t have time to RAP safely’.
POC coagulation testing was available at all hospitals, with 53% using rotational thromboelastometry (ROTEM; ROTEM Sigma or Delta analyser, Instrumentation Laboratory, Bedford, MA, USA) and 47% using thrombelastography (TEG; TEG 5000 or 6s; Haemonetics Corporation, Braintree, MA, USA).
One hospital used both technologies. For CPB cases seven (24%) hospitals used POC coagulation testing always and 22 (76%) used it selectively. Less than half of surveyed hospitals had a specific local algorithm for blood product administration based on POC viscoelastic coagulation assay results.
There was a marked geographical variation in access to fibrinogen concentrate (FC). Only two hospitals (7%) reported unrestricted access, whereas seven reported restricted access (24%) and in the remaining 20 hospitals (69%) it was unavailable. In hospitals with restricted access, criteria for access included permission from a haematology consultant, agreement between two consultants, and criteria related to TEG or ROTEM results. Hospitals in New South Wales, the Australian Capital Territory and Victoria all reported no access to FC. Several respondents felt that the cost of FC was the main barrier to its use in their hospitals.
Prior to commencing CPB most hospitals targeted an ACT of 480 s (Table 3). Other hospitals targeted 400–500 s (Table 3).
Discussion
From this survey of 29 hospitals, the estimated total number of cardiac surgeries on CPB performed in Australian and New Zealand public hospitals was about 14,739 per year, with New Zealand contributing about 2560 cases. For comparison, data collected by The Australian and New Zealand Society of Cardiac and Thoracic Surgeons Surgery Database Program (ANZSCTS) in 2018, reported 13,810 on-pump cardiac surgeries across 37 public and private Australian hospitals. 7 The case numbers in the current survey complement the ANZSCTS registry data, as at the time of publication only one New Zealand hospital was a member of the registry and several public hospitals in Australia do not contribute data to the ANZSCTS registry.
The results indicate that CS would be used routinely for around 31% of on-pump surgeries undertaken at public hospitals in Australia and New Zealand. This is comparable to 2014–2015 data collected from nine hospitals by The Australian and New Zealand Collaborative Perfusion Register (ANZCPR), which showed CS use for 30% of on-pump CABG surgeries. 8 The use of CS for cardiac surgery in Australia and New Zealand is lower than in North America, where CS use has been reported for about 99.5% of CABG surgeries. 8 The higher rate of CS in North American data may reflect the grade I level recommendation made in 2011 by The Society of Thoracic Surgeons and the Society of Cardiovascular Anesthesiologists that CS be used during cardiac surgery as a method of blood conservation. 9
This survey has demonstrated that within Australian and New Zealand public hospitals there is significant practice variation regarding how CS is used for on-pump cardiac surgery cases. According to the National Blood Authority (NBA) conceptual framework of patient blood management, CS is a component of the second ‘pillar’ of blood management, ‘minimising blood loss’. 10 The NBA provides a grade 3 level recommendation that CS be used for adult patients undergoing surgery where substantial blood loss is anticipated. 10 The document ‘Guidance for the Provision of Intraoperative Cell Salvage’ published by the NBA in 2014 does not contain specific advice regarding how CS be utilised during cardiac surgery. 11 Neither of these recommendations relates specifically to on-pump cardiac surgery, where salvage of shed blood directly into the CPB circuit is possible.
Recommendations specific to CS use in cardiac surgery are available in more recent guidelines published by the Association of Anaesthetists of Great Britain and Ireland (AAGBI) and jointly by the European Associations for Cardio-Thoracic Surgery and Anaesthesiology (EACTS). 1 , 3 The AAGBI recommend the routine use of CS during all cardiac surgery (at least in a collect only mode, discussed below) as an effective means of reducing allogenic transfusions, whereas EACTS guidelines recommend its consideration (IIa evidence). 1 , 3 The substitution of CS in lieu of cardiotomy suction during CPB is not recommended in either document. Moreover, despite evidence that CS removes inflammatory mediators present in cardiotomy suction blood that impair coagulation and platelet function ex vivo, the use of CS as an alternative to cardiotomy suction results in greater transfusion requirements due to the loss of platelets and clotting factors. 3 ,12–15 In regard to the processing of pump blood in a CS unit, the AAGBI guidelines suggest this may reduce allogenic transfusion requirements, but neither the AAGBI nor the EACTS recommend this as routine practice. 1 , 3 Of relevance to this treatment option, the EACTS guidelines caution that the ability of CS to reduce transfusion requirements is balanced by the effects of platelet and coagulation factor loss and that the reinfusion of >1000 ml of CS blood may impair coagulation. 1
Some survey respondents from hospitals selectively using CS mentioned that their service questioned the cost-effectiveness of using CS for all cases, as during ‘low-risk’ cases the volume of blood collected within the CS unit may be insufficient for processing. This survey did not explore the cost implications of a ‘collect only’ CS mode, where only the consumables required to collect anticoagulated blood are used, unless a threshold amount of blood is collected. The AAGBI recommends the use of a ‘collect only’ CS for all cardiac surgical cases and suggests that if more than 500 ml of blood is collected, it is processed. 3
This survey indicates that ANH is rarely performed for cardiac surgery involving CPB in Australian and New Zealand public hospitals. The efficacy of ANH appears to be associated with the volume of blood harvested, with the greatest benefits seen when >800 ml of blood is collected, which may not be tolerated in many patients. 16
Our results show a median total prime volume of 1500 ml and that RAP was used routinely by 24% and selectively by 55% of hospitals. This is similar to ANZCPR registry data from nine hospitals collected between 2014 and 2015, showing a total RAP use rate of 20.3% and median total prime volume of 1500 ml. 8 The net volume of prime solution remaining after RAP or circuit modification is directly related to the degree of haemodilution occurring when CPB is commenced and is associated with the need for exogenous blood transfusion. 17 A systematic review published in 2020 examined all adult studies comparing RAP to conventional practice published within the last ten years. In keeping with contemporary evidence-based guidelines, this review concluded that overall, the available evidence supports the use of RAP as a blood-conservation technique. 1 , 18 The authors of the review also reported that all studies concluding that RAP did not reduce the need for allogenic blood transfusion used markedly greater net prime volumes following RAP than the positive trials. 17 Patients who are anaemic or have a smaller body surface area are likely to benefit the most from efforts to minimise haemodilution. 1 , 19 There is a considerable body of evidence supporting the practice of reducing net prime volume (and haemodilution) through circuit componentry selection and use of RAP; 17 , 18 however, the scale and design of existing studies has generally limited the ability to control the full range of confounding variables affecting volume status. 1 , 4
In comparison to a survey of perfusion equipment used in Australian and New Zealand hospitals in 2003 (which included private hospitals), the use of CPs as the main arterial pump appears to have increased slightly (28% in current survey versus 13.6% in the 2003 survey). 20 A systematic review and meta-analysis of studies comparing RPs and CPs as the main arterial pump have not demonstrated significant differences in patient-derived outcomes or transfusion requirements. 4
Although balanced crystalloids were used as the main CPB circuit priming fluid at all hospitals, there was variation both between and within hospitals regarding what medications were added to the prime solution (Table 3). Recent guidelines report no consensus regarding the ideal prime solution and offer no recommendations for the addition of medications. 4 , 21 Our results are similar to those of a recent and larger survey of bypass pump prime composition in Australian hospitals (both public and private), which showed that Plasmalyte 148 was the most common prime solution and sodium bicarbonate, mannitol and human albumin are common additives in varying doses. 22 In our survey over 48% of hospitals added human albumin to their prime solution. Results of an international survey published in 2017 demonstrated the addition of albumin to CPB prime is significantly more popular in Australia and New Zealand than in North America, Europe or South America. 23 The addition of heparin to pump prime was universal in our survey; however, this practice is less common internationally, being added by less than 40% of perfusionists responding to a similar survey in North America and Europe. 23
By asking perfusionists and cardiac anaesthetists identical questions, our survey provides an additional insight that pump prime composition is often not a shared knowledge, as most cardiac anaesthetists were not aware of the exact composition of the prime solution used in their hospitals. This survey was not designed or intended to explore medication safety and governance issues, or compare medications administered in the prime solution to medications administered by the anaesthetic team. Consideration of these issues may be useful on a local level.
This survey delivery method was chosen to maximise the response rate, with consideration given to the relatively small sample size and the risk of non-response bias. Further, given the survey explores the approach taken to some contentious areas of practice, the authors felt the telephone survey format would allow for ease of clarification and facilitate the collection of additional qualitative data offered by respondents that may provide valuable context to the results. As a survey of hospital practice, the responses of both anaesthetists and perfusionists were used to assist the capture of relevant data and the flexibility of the survey method was intended to limit recall bias. The authors attempted to limit observer bias by using a single interviewer who consistently asked a structured series of questions.
Most hospitals targeted a pre CPB ACT value of 480 s, with responses ranging between 400 and 500 s, consistent with current international practice patterns. 23 Current North American guidelines suggest an ACT of 480 s continues to be a reasonable target, and report that 400 s is a reasonable target when using certain analysers that ‘maximally activate’ sample coagulation. 5 The extensive limitations of ACT measurement to guide heparin management during CPB are well described elsewhere. 5 This survey did not explore the emerging role of POC heparin concentration monitoring to guide anticoagulation during CPB, which current European guidelines suggest should be considered over ACT monitoring to reduce bleeding (level B evidence). 1
The use of POC viscoelastic coagulation assays and targeted algorithms to assist blood management decisions during cardiac surgery has resulted in modest reductions in allogenic blood transfusion and is supported by many current guidelines. 1 , 10 , 24 , 25 All surveyed hospitals had access to either TEG or ROTEM; however, hospitals varied in relation to their routine or selective use (i.e. when there were specific concerns regarding haemostasis). The routine use of ROTEM (including INTEM and HEPTEM analysis) after protamine administration has been shown in one series to have poor positive predictive value, but high negative predictive value for increased post-operative bleeding. 26 :
Targeted fibrinogen replacement as one of the first haemostatic measures in bleeding hypofibrinogenaemic patients is well described. 1 , 27 FC is an anhydrous human plasma-derived but purified alternative to cryoprecipitate for fibrinogen replacement. Suggested benefits compared with cryoprecipitate include more consistent fibrinogen concentrations, less pathogen load and ease of storage, availability and delivery. Recent studies suggest FC is not inferior to cryoprecipitate in the management of acquired hypofibrinogenaemia after cardiac surgery. 28 FC is already commonly used in Europe and considered the first line for treatment of acquired hypofibrinogenaemia in some European guidelines. 29 FC availability was explored in this survey to highlight geographical variation and promote discussion. Availability and access were variable with two-thirds of hospitals reporting having no access to FC with the majority of the remaining hospitals having restricted access. Cost was cited as the primary reason. The cost of FC is around A$800 per gram and the average dose for an adult patient is 2–4 g, which equates to 15–20 single donor units of cryoprecipitate. 30 Regional variations in how cryoprecipitate and FC are funded on a hospital and state level could possibly influence the uptake of FC use.
This survey has several limitations. Only one anaesthetist and one perfusionist were surveyed in each hospital and although questions were framed to address institutional and not personal practice, the responses may have been biased by the individual practice or knowledge of the respondents. The hospital caseload data obtained are subject to recall bias of respondents and per annum estimates required extrapolation. Similarly, the demographic data obtained may provide a useful snapshot, but the respondents represent only a small proportion of the population of cardiac anaesthetists and clinical perfusionists working in the region. This survey provides new information regarding how public hospitals are approaching some selected contentious areas of practice; however, the results cannot be used to form broader conclusions regarding the overall approach of the surveyed hospitals to blood conservation or efforts to minimise haemodilution while on CPB. For example, this survey did not address most elements of three pillars of perioperative blood conservation, which would include questions addressing the preoperative optimisation of red cell mass, a more exhaustive set of questions addressing blood loss minimisation and the management of anaemia. 10 Similarly a more complete survey of efforts to minimise haemodilution of CPB would address fluids given pre CPB, cardioplegia volume, use of filtration/ultrafiltration and diuretic use. Additionally, the use of biocompatible (including heparin bonded) coating of bypass circuits and the use of leukocyte depletion filters were not explored in this survey. Both have been addressed in recent guidelines. 1 , 3 Given the delivery method of this survey it was necessary to focus on a limited number of areas related to CPB.
Future research aiming to strengthen the level of evidence in each respective area will be of greater relevance to clinicians if informed by current practice patterns and this survey report may also be of use by stimulating dialogue and further investigation within individual hospitals and on a regional level.
Conclusions
This survey of cardiac anaesthetists and perfusionists from all public adult cardiac hospitals in Australian and New Zealand achieved a response rate of 100% and has given a snapshot of how public hospitals in the region approach certain practices in cardiac anaesthesia and perfusion where best practice is unclear. Most of the practices explored related to mechanical mechanisms of blood conservation, practices that limit haemodilution, and POC coagulation testing. It was found that in the majority of hospitals clinicians make ‘case-by-case’ decisions of when to use CS to collect shed blood or process residual pump blood, when to perform RAP and when to use POC coagulation testing. The addition of human albumin and hypertonic medications to the CPB prime solution varied considerably. The ongoing acceptance of locally formulated prime mixtures stands in sharp contrast to the rigorous scientific scrutiny and discourse surrounding intravenous fluids and their use in other contexts. These findings suggest there is an urgent need for adequately powered, well-designed prospective trials to identify which interventions provide the most benefit to patients and to inform best practice during CPB.
Supplemental Material
sj-pdf-1-aic-10.1177_0310057X20960730 - Supplemental material for Adult cardiopulmonary bypass in Australian and New Zealand public hospitals: A survey of practice
Supplemental material, sj-pdf-1-aic-10.1177_0310057X20960730 for Adult cardiopulmonary bypass in Australian and New Zealand public hospitals: A survey of practice by Simon J Pattullo and David M Samson in Anaesthesia and Intensive Care
Footnotes
Author contribution(s)
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
