Abstract
The reported incidence of post--dural puncture headache (PDPH) after neuraxial anaesthesia varies widely, depending on patient and procedural risk factors. Most previous studies have had small sample sizes and focused on obstetric patients. This study aimed to investigate the incidence of PDPH and factors associated with PDPH in non-obstetric and obstetric patients after neuraxial anaesthesia. We identified patients who underwent surgery with neuraxial anaesthesia between July 2010 and December 2017 from a Japanese nationwide inpatient administrative claims and discharge database. Factors associated with PDPH (body mass index (BMI), depression, spinal abnormalities, academic hospital and location of epidural anaesthesia) were examined using multivariable logistic analyses. The incidence of PDPH in non-obstetric patients after spinal anaesthesia, epidural anaesthesia and combined spinal epidural anaesthesia was 0.16%, 0.13% and 0.23% and in obstetric patients was 1.16%, 0.99% and 1.05%, respectively. Higher BMI was associated with decreased incidence of PDPH in non-obstetric patients receiving spinal anaesthesia and obstetric patients receiving epidural anaesthesia. In female patients receiving spinal anaesthesia, a history of depression was associated with increased incidence of PDPH. Being in an academic hospital was associated with decreased incidence of PDPH in male patients receiving spinal anaesthesia and female patients receiving spinal or epidural anaesthesia, but increased incidence of PDPH in male patients receiving epidural anaesthesia. Lumbar epidural anaesthesia was associated with increased incidence of PDPH in male patients, but decreased incidence of PDPH in obstetric patients compared with thoracic epidural anaesthesia. The present study identified several potential new risk factors for PDPH, and revealed that the incidence of PDPH in non-obstetric patients after neuraxial anaesthesia was lower than in obstetric patients.
Introduction
Post--dural puncture headache (PDPH) is an uncommon complication associated with neuraxial anaesthesia and is caused by cerebrospinal fluid leakage. The typical symptoms of PDPH are positional headache that is worsened by sitting or standing, often accompanied by nausea, neck stiffness, photophobia, tinnitus or hearing loss. PDPH disturbs early postoperative ambulation because the majority of cases occur within the first 24–48 h after dural puncture. 1
There have been several small studies on the incidence of PDPH after different types of neuraxial anaesthesia. In a single-centre retrospective study using chart reviews, the incidence of accidental dural puncture in obstetric patients was 0.5%, and approximately 73% of these patients developed PDPH. 2 Meanwhile, the incidence of PDPH after combined spinal epidural anaesthesia (CSEA) was similar to that after epidural anaesthesia (EA) according to studies on obstetric patients. 3 , 4 However, these previous studies on PDPH were limited by their small sample sizes. Furthermore, the studies focused mainly on obstetric patients with accidental dural puncture after EA, and thus there is a lack of studies investigating the incidence of PDPH in non-obstetric patients.
Some studies have identified risk factors associated with PDPH, including demographic factors (age, female sex, pregnancy, history of headache) and procedural factors (needle size, needle tip type, bevel orientation of cutting needles). However, these previous studies also had relatively small sample sizes.5–8
Therefore, the present study was conducted to examine the incidence of PDPH and potential factors associated with PDPH in non-obstetric and obstetric patients undergoing surgery with neuraxial anaesthesia, using a national inpatient database in Japan.
Materials and methods
Data source
Inpatient data were extracted from the Japanese Diagnosis Procedure Combination database, details of which have been described previously. 9 Briefly, the database includes administrative claims data and the following detailed patient data: age; sex; body mass index (BMI); diagnoses, comorbidities at admission and complications after admission recorded with text data in the Japanese language and encoded by International Classification of Diseases and Related Health Problems, 10th Revision (ICD-10) codes; medical procedures encoded by Japanese original codes; medications prescribed during hospitalisation; length of stay; and discharge status.
This article conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. Ethical approval was obtained from the Institutional Review Board (IRB) at The University of Tokyo (IRB number: 3501). The requirement for informed consent was waived by the IRB because the study was based on a secondary analysis of anonymous administrative data.
Data extraction
From the database, we obtained the records of patients who underwent surgery with neuraxial anaesthesia (spinal anaesthesia (SA), EA and CSEA) between 1 July 2010 and 31 December 2017 during six types of procedures: (a) general surgery; (b) pulmonary surgery; (c) obstetric surgery; (d) gynaecological surgery; (e) urological surgery and (f) orthopaedic and plastic surgery. General surgery included oesophageal, gastrocolic, hepatobiliary, pancreatic and breast surgery. Pulmonary surgery included lung, tracheal, pleural and diaphragmatic surgery. Obstetric surgery included caesarean section and other surgeries related to pregnancy (abortion, peripartum hysterectomy, maternal-foetal surgery, cervical cerclage). Gynaecological surgery included hysterectomy and other pelvic surgeries. Urological surgery included kidney, adrenal, urinary tract, bladder, prostate, and genital organ surgeries. Orthopaedic and plastic surgery included musculoskeletal and skin surgeries for lower extremities. We excluded patients aged <20 years and those receiving multiple neuraxial blocks for different surgeries during the same admission.
We extracted information on patient characteristics including age, sex, BMI, spinal abnormalities, history of depression, type of surgery with neuraxial anaesthesia, type of EA (thoracic or lumbar) and type of hospital (academic or non-academic).
Study patients
We included patients with PDPH after admission using (a) ICD-10 codes G960 (cerebrospinal fluid leak) and G970 (cerebrospinal fluid leak from spinal puncture) and (b) ICD-10 code T885 (other complications of anaesthesia) together with text ‘cerebrospinal fluid leak’ or ‘headache after spinal anaesthesia’. We also included patients with recorded diagnosis of ICD-10 code R51 (headache) along with text ‘headache’ after admission, but excluded those at admission.
We identified patients with depression at admission using ICD-10 codes F32 (depressive episodes, depressive conduct disorder) and F33 (depressive disorder). Patients with spinal abnormalities at admission were identified by ICD-10 codes M40–43 (deforming dorsopathies), M45–49 (spondylopathies) and M50–53 (other dorsopathies).
Age was categorised into three groups: 20–49, 50–69, and ≥70 years. BMI (in kg/m2) was categorised into five groups: <18.5 (underweight), 18.5–24.99 (low normal weight), 25–29.99 (high normal weight), 30–34.99 (overweight) and ≥35 (obesity).
Statistical analysis
The incidence of PDPH was calculated for each type of neuraxial anaesthesia (SA, EA and CSEA) in non-obstetric patients and obstetric patients. Univariable and multivariable logistic regression analyses were performed to examine the associations of PDPH with patient and hospital characteristics. Among non-obstetric patients in the SA group, we selected type of hospital, history of depression and spinal abnormalities as well as age, which was reported to be a risk factor for PDPH, to identify significant factors associated with PDPH. Among non-obstetric patients receiving EA, we selected puncture location for EA (thoracic or lumbar) as a factor associated with PDPH in addition to age, BMI, type of hospital, history of depression and spinal abnormalities. We did not use age among obstetric patients who received SA, EA and CSEA as a factor associated with PDPH. Male, non-obstetric female and obstetric patients were analysed separately.
We used a significance level of P <0.05 for the statistical analyses. All analyses were conducted using Stata/MP 15.0 software (Stata Corp., College Station, TX, USA).
Results
During the study period, a total of 3,455,010 patients who underwent neuraxial anaesthesia were identified. We excluded patients aged <20 years (n = 113,010) and those who underwent multiple neuraxial blocks for different surgeries during the same admission (n = 396,083). Thus, 2,945,917 patients were eligible for the study. The estimated incidence of non-obstetric PDPH patients after SA, EA and CSEA was 0.16%, 0.13% and 0.23%, respectively. The incidence of obstetric PDPH patients after SA, EA and CSEA was 1.16%, 0.99%, and 1.05%, respectively.
Table 1 shows the patient and hospital characteristics after neuraxial anaesthesia. The numbers (proportions) of patients who underwent SA, EA and CSEA were 1,049,969 (35.6%), 1,807,200 (61.3%) and 88,748 (3.0%), respectively. The proportion of patients aged ≥50 years was higher than that of patients aged 20–49 years in the SA and EA groups, whereas the proportion of patients aged 20–49 years was highest in the CSEA group. The low normal weight group (18.50–24.99 kg/m2) was the largest group of patients in each group. The highest proportions of patients by type of surgery in the SA, EA and CSEA groups were orthopaedic and plastic surgery (34.6%), general surgery (46.8%) and obstetric surgery (81.1%), respectively.
Patient and hospital characteristics after neuraxial anaesthesia.
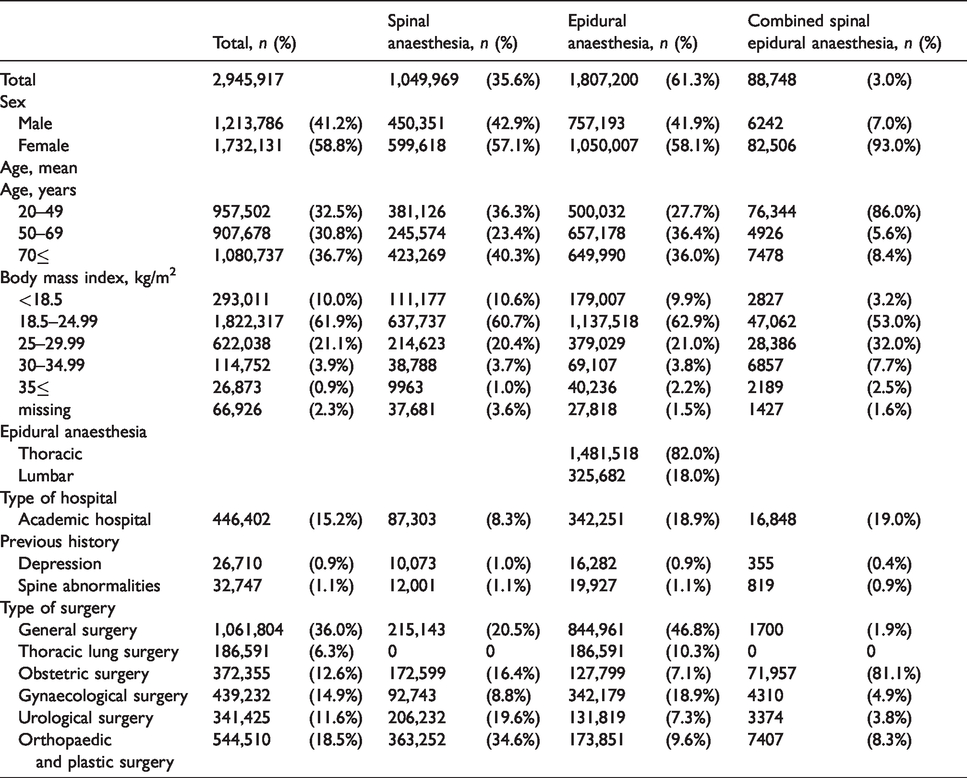
Table 2 shows the results of the univariable and multivariable logistic regression analyses for PDPH in male patients. In the multivariable analyses, high normal weight (odds ratio (OR): 0.57; 95% confidence interval (CI): 0.41–0.79; P = 0.001) and overweight (OR: 0.49; 95% CI: 0.29–0.82; P = 0.007) were significantly associated with lower incidence of PDPH compared with underweight in the male SA group. Being in an academic hospital was significantly associated with lower incidence of PDPH in the male SA group (OR: 0.39; 95% CI: 0.25–0.62; P <0.001), but significantly associated with higher incidence of PDPH in the EA group (OR: 1.36; 95% CI: 1.08–1.72; P = 0.009). Spinal abnormalities were significantly associated with higher incidence of PDPH in the male EA group (OR: 2.57; 95% CI: 1.32–4.99; P = 0.005). Lumbar EA was significantly associated with higher incidence of PDPH (OR: 1.31; 95% CI: 1.00–1.72; P = 0.048) compared with thoracic EA.
Univariable and multivariable logistic regression for PDPH in male patients.
BMI: body mass index; PDPH: post--dural puncture headache; Ref: reference; CI: confidence intervals.
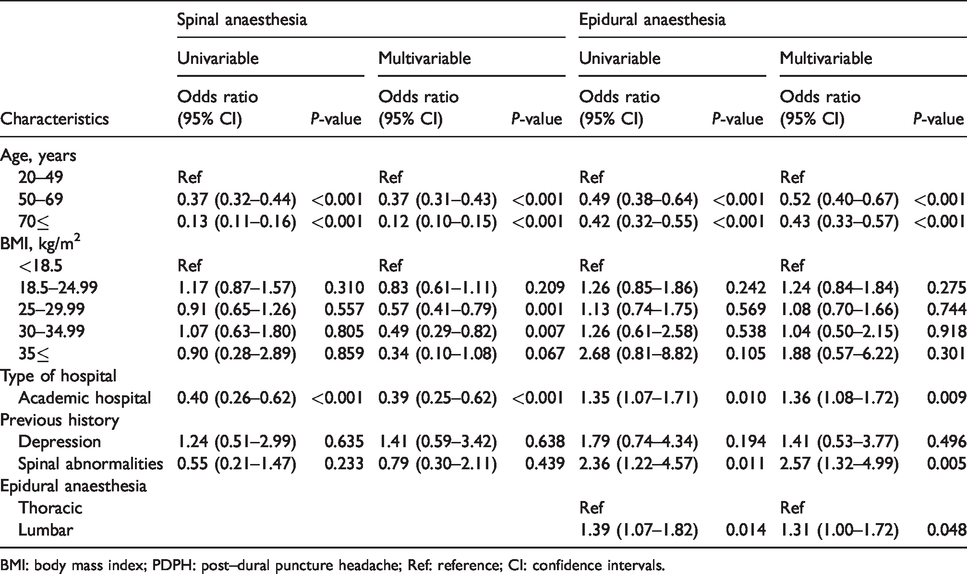
Table 3 shows the results of the univariable and multivariable logistic regression analyses in non-obstetric female patients after neuraxial anaesthesia. In the multivariable analyses, BMI categories including low normal weight (OR: 0.85; 95% CI: 0.74–0.98; P = 0.026), high normal weight (OR: 0.56; 95% CI: 0.45–0.70; P <0.001), overweight (OR: 0.57; 95% CI: 0.40–0.82; P = 0.002) and obesity (OR: 0.11; 95% CI: 0.03–0.45; P = 0.002) were significantly associated with lower incidence of PDPH compared with underweight in the non-obstetric female SA group. Low normal weight (OR: 1.17; 95% CI: 1.00–1.35; P = 0.045) in the EA group was significantly associated with a higher incidence of PDPH compared with underweight in the EA group. Depression was significantly associated with higher incidence of PDPH in the SA group (OR: 1.67; 95% CI: 1.05–2.68; P = 0.031). Being in an academic hospital was significantly associated with lower incidence of PDPH in the non-obstetric female SA (OR: 0.45; 95% CI: 0.35–0.57; P <0.001) and EA (OR: 0.78; 95% CI: 0.69–0.89; P <0.001) groups.
Univariable and multivariable logistic regression for PDPH in non-obstetric female patients.
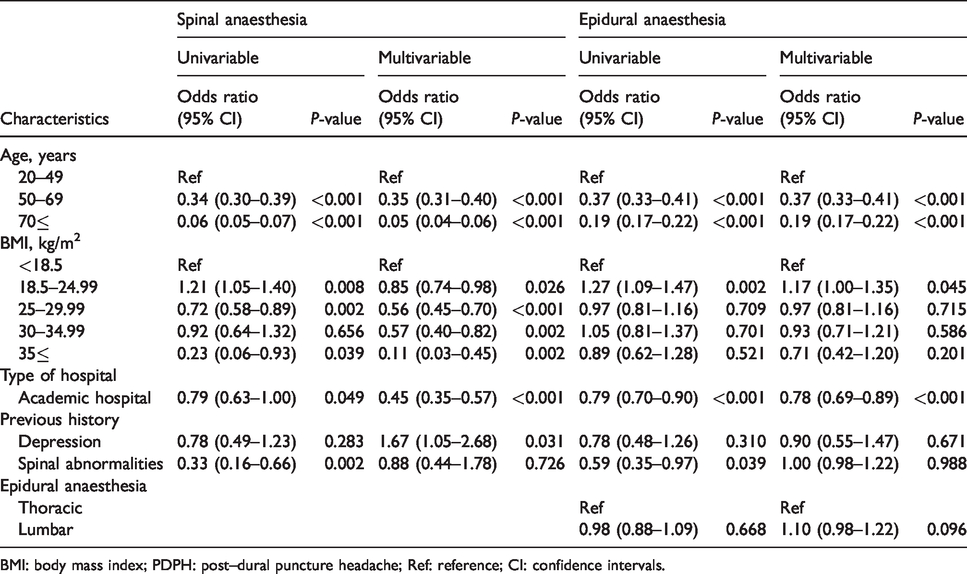
BMI: body mass index; PDPH: post--dural puncture headache; Ref: reference; CI: confidence intervals.
Table 4 shows the results of the univariable and multivariable logistic regression analyses in obstetric patients after neuraxial anaesthesia. In the multivariable analyses, BMI categories including low normal weight (OR: 0.70; 95% CI: 0.52–0.95; P = 0.023), high normal weight (OR: 0.62; 95% CI: 0.45–0.85; P = 0.003) and overweight (OR: 0.52; 95% CI: 0.36–0.76; P = 0.001) were significantly associated with lower incidence of PDPH compared with underweight in the obstetric EA group. A history of depression in the SA group (OR: 1.89; 95% CI: 1.01–3.54; P = 0.047) was significantly associated with increased incidence of PDPH. Being in an academic hospital was significantly associated with lower incidence of PDPH in the obstetric SA (OR: 0.45; 95% CI: 0.39–0.53; P = 0.001) and EA (OR: 0.54; 95% CI: 0.46–0.64; P <0.001) groups. Lumbar EA was significantly associated with lower occurrence of PDPH in the obstetric EA group (OR: 0.72; 95% CI: 0.61–0.85; P <0.001).
Univariable and multivariable logistic regression for PDPH in obstetric patients.
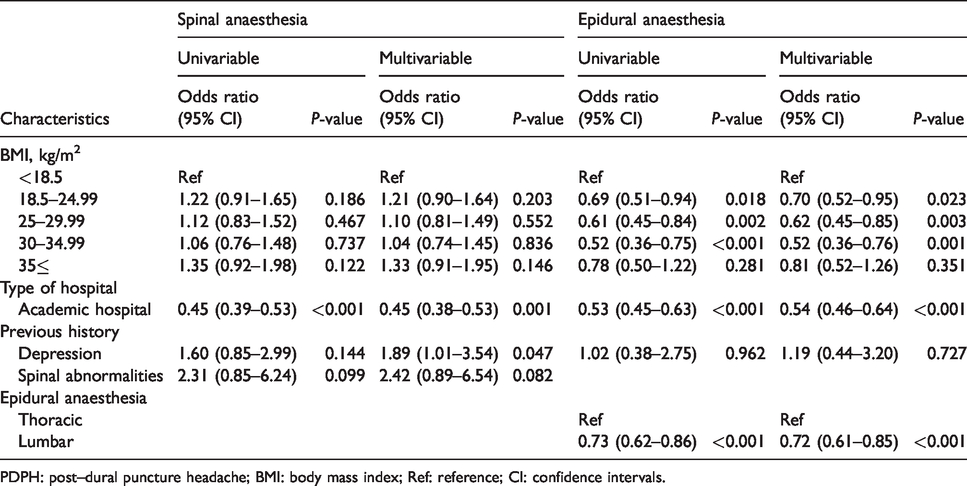
PDPH: post--dural puncture headache; BMI: body mass index; Ref: reference; CI: confidence intervals.
Discussion
In the present study, we examined the incidence of PDPH and factors associated with PDPH in non-obstetric patients and obstetric patients after neuraxial anaesthesia (SA, EA and CSEA) using a nationwide inpatient database in Japan. In male patients, the occurrence of PDPH was associated with age, BMI, being in an academic hospital, spinal abnormalities and location of EA: high normal weight (OR: 0.57) and overweight (OR: 0.49) were associated with decreased PDPH after SA, whereas spinal abnormalities (OR: 2.57) and lumbar EA (OR: 1.31) were associated with increased PDPH after EA (Table 2). In non-obstetric female patients, the incidence of PDPH was associated with age, BMI, being in an academic hospital and a history of depression: low normal weight (OR: 0.85), high normal weight (OR: 0.56), overweight (OR: 0.57), and obesity (OR: 0.11) were associated with decreased PDPH after SA, whereas depression (OR: 1.67) was associated with increased PDPH after SA (Table 3). In obstetric patients, the incidence of PDPH was associated with BMI, being in an academic hospital, a history of depression and location of EA: low normal weight (OR: 0.70), high normal weight (OR: 0.62), overweight (OR: 0.52) and lumbar EA (OR: 0.72) were associated with decreased PDPH after EA, whereas depression (OR: 1.89) was associated with increased PDPH after SA (Table 4). Being in an academic hospital showed opposite effects for sex and type of neuraxial anaesthesia (OR: 0.39 in male SA group, OR: 1.36 in male EA group, OR: 0.45 in both non-obstetric female and obstetric SA group, OR: 0.78 in non-obstetric female EA group and OR: 0.54 in obstetric EA group).
The incidences of PDPH after SA in the present study were 0.16% in non-obstetric patients and 1.16% in obstetric patients. These incidences were lower than those in a previous study in which the incidences of PDPH in obstetric patients after SA ranged from 3.1% to 8.7% depending on the type and size of the needle. 5 Another study showed that the overall PDPH incidence was 2.0% in ambulatory surgical patients aged 15–45 years. 10
Our study showed that the incidence of PDPH after EA and CSEA in non-obstetric patients was 0.13% and 0.23%, respectively. The incidence of PDPH after EA was similar to that in a previous study, in which the incidence of PDPH was 0.14% in non-obstetric patients who underwent surgery with EA. 11
In our study, the incidence of PDPH after EA and CSEA in obstetric patients was 0.99% and 1.05%, respectively. A previous study found that the incidence of accidental dural puncture during EA in obstetric patients was 0.4%, and that 60.5% of these patients developed PDPH. 12 However, this previous study focused mainly on the incidence of PDPH following accidental dural puncture after EA, and therefore the present findings cannot be directly compared with this previous finding.
Our findings may have been affected by coding inconsistencies and ascertainment problems, although a validation study on the database used in this study showed that the diagnosis records for several common diseases (malignant tumours, cardiac diseases, stroke, and renal diseases) had moderate sensitivity and high specificity. 9 Nevertheless, our use of a large-scale dataset allowed us to determine the incidence of PDPH in not only obstetric patients, but also non-obstetric patients.
We identified several potential new risk factors for PDPH after neuraxial anaesthesia. Although the actual aetiology of PDPH remains unclear, two pathophysiologic mechanisms have been proposed: (a) leakage of cerebrospinal fluid after dural puncture may lead to intracranial hypotension and compensatory venous dilatation that result in headache and cranial nerve palsies, and (b) intracranial hypotension caused by cerebrospinal fluid leakage may lead to sagging of intracranial structures, which stretches sensory intracranial nerves and results in headache. 13 , 14 There may also be several other possible causes of PDPH.
The present study showed that high BMI was associated with reduced incidence of PDPH in non-obstetric patients undergoing SA and obstetric patients undergoing EA. In male patients receiving SA, the ORs were lower (0.83 to 0.34) as the BMI categories increased (Table 2). In non-obstetric female patients receiving SA, the ORs were also lower (0.85 to 0.11) as the BMI categories increased (Table 3). In obstetric patients receiving EA, low (OR: 0.70) and high (OR: 0.62) normal weight and overweight (OR: 0.52) were significantly associated with decreased incidence of PDPH, compared with underweight (Table 4). Meanwhile, in non-obstetric female patients receiving EA, low normal weight (OR: 1.17) was significantly associated with increased occurrence of PDPH, compared with underweight (Table 3). A previous study found that a lower BMI group (≤25 kg/m2) had a greater likelihood of PDPH among research volunteers who underwent a diagnostic lumbar puncture, compared with a higher BMI group (>25 kg/m2). 15 In contrast, other studies showed no relationship between incidence of PDPH and BMI. 16 , 17 The reason for a potential association between high BMI and low incidence of PDPH may be inactive behaviour and lack of movement of obese patients to the upright position during the early postoperative period. Furthermore, studies showed that obesity with high intra-abdominal pressure produced less cerebrospinal fluid volume in the lower spine and resulted in intracranial hypertension. 18 , 19 Additional studies on the pathophysiologic mechanisms utilising clinical assessments are warranted.
Female patients with history of depression had an increased incidence of PDPH after SA in the present study. This finding was comparable with a previous study. 20 Specifically, the previous study examined the incidence, duration and risk factors for PDPH and the risk of getting a migraine attack in migraine sufferers and non--migraine sufferers, and showed that history of depression was associated with longer duration of PDPH, but not an increased incidence of PDPH. 20
To our knowledge, other factors such as being in an academic hospital, spinal abnormalities, and location of EA were not examined in previous studies. Being in an academic hospital was associated with an increased incidence of PDPH in male patients after EA (OR: 1.36), but decreased incidence of PDPH in male patients after SA (OR: 0.39) and female patients after SA (OR: 0.45 in both non-obstetric female and obstetric patients) or EA (OR: 0.78 in non-obstetric female patients and OR: 0.54 in obstetric patients). We expected that being in an academic hospital would be a factor associated with increased incidence of PDPH, because academic hospitals employ many junior anaesthesiologists, and thus patients may experience multiple SA attempts and accidental dural punctures during EA may occur more frequently than in non-academic hospitals. The reason why academic hospitals showed opposite results for sex and neuraxial anaesthesia remains unknown, although the incidence of PDPH after SA may be influenced by differences in the needle size or needle tip type used in individual hospitals.
Our study showed different results between male and female patients in terms of the associations between spinal abnormalities and PDPH. Because of the small number of patients, we could not confirm or exclude an association between spinal abnormalities and PDPH.
Lumbar EA was a potential factor associated with decreased PDPH in obstetric patients (OR: 0.72). Meanwhile, male patients who received lumbar EA had an increased incidence of PDPH (OR: 1.31). We speculate that accidental epidural puncture after lumbar EA occurs more frequently than that after thoracic EA because the distance from skin to epidural space in lumbar EA is shorter than that in thoracic EA.
There are several limitations to the present study. First, it was a retrospective study using an administrative claims database, and thus the recorded diagnoses of PDPH may have lower sensitivity than those in chart reviews. In addition, we could not investigate other possible risk factors for PDPH such as certain demographic factors (history of headache, history of PDPH) and procedural factors (needle size, needle type, bevel direction, patient cooperation, anaesthesiologist skill). Second, our findings have internal validity when applied to Japanese patients, but may not be generalisable to patients in other countries. Third, the patients identified using ICD-10 code R51 and text ‘headache’ may include those who suffered from headache from causes unrelated to PDPH. Thus, we may have overestimated the incidence of PDPH. Finally, mild PDPH patients may not have been recorded and the incidence of PDPH may have been underestimated.
In conclusion, the present study determined the incidence and potential factors associated with PDPH after neuraxial anaesthesia, using a large-scale national database. Although we could not adjust for unmeasured confounders, high BMI in patients undergoing SA or EA was associated with a decreased incidence of PDPH, while history of depression in female patients undergoing SA was associated with an increased incidence of PDPH. Being in an academic hospital showed different effects for sex and type of neuraxial anaesthesia. Lumbar EA was associated with an increased incidence of PDPH in male patients, but decreased incidence of PDPH in obstetric patients compared with thoracic EA.
Footnotes
Authors' note
K Makito contributed to this article in her professional capacity. The views expressed in the submitted article are her own and do not necessarily represent the views of the institution or funder.
Author contribution(s)
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Ministry of Health, Labour and Welfare, Japan (19AA2007 and H30-Policy-Designated-004) and the Ministry of Education, Culture, Sports, Science and Technology, Japan (17H04141).
