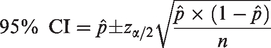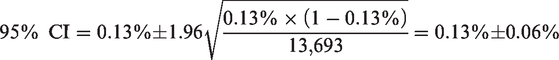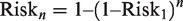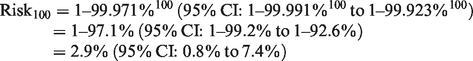Abstract

What is the probability that an anaesthetist will contract the SARS-CoV-2 virus from a patient who is under their care? This is an obvious question that is conspicuously absent from many guidelines and recommendations on personal protective equipment (PPE) during the COVID-19 pandemic. There is currently no way of quantifying the answer in any scientific way; however, I would like to propose the following model as a way of conceptualising the risk. The exact numbers are not as important as the underlying methodology, and the numbers are likely to change depending on geographical location and factors influencing community transmission; therefore, the result should be thought of as a ‘ballpark estimate’ rather than a scientifically accurate measurement of probability.
Two independent criteria need to be met before an anaesthetist can contract SARS-CoV-2 from a patient under their care:
The patient must be a carrier for the SARS-CoV-2 virus; The virus must be transmitted from the patient to the anaesthetist.
The probability that an anaesthetist will acquire COVID-19 from a patient can therefore be conceptualised by the following equation:
Probability of patient carrying SARS-CoV-2
In a confirmed case of COVID-19, the probability that the patient is a carrier of the SARS-CoV-2 virus is (by definition) 100%. In this context, it makes sense to minimise the risk of transmission by adopting very stringent infection control processes—including the use of airborne precautions. This argument could be extended to patients suspected of carrying the virus (e.g. close contact of a confirmed case) until their individual carrier status can be elucidated by testing. But what about patients from the general population with no indication for testing?
Stratifying the risk based on symptomatology seems logical, but the absence of fever and/or respiratory symptoms (e.g. cough, sore throat, shortness of breath) is not necessarily reassuring because the SARS-CoV-2 virus can be transmitted from asymptomatic or presymptomatic patients. 1 Likewise, the presence of symptoms is unhelpful because they may have an alternate non-COVID-19 explanation (e.g. fever in a patient with appendicitis).
A better way to assess the probability would be to focus less on symptomatology and instead ascertain the incidence of SARS-CoV-2 in the population who reside in your specific geographical location. For example, based on information publicly available at the time of writing
2
:
There have been 18 new cases in my home state (Queensland) over the past week; During that time 13,693 tests were reportedly performed; Incidence =
This ‘ballpark estimate’ (sample proportion) can be converted into a ‘ballpark range’ for my state of Queensland (population proportion) by calculating the 95% confidence interval (CI):
Note that this is a biased estimate because the sample population is symptomatic patients presenting from the community for testing (i.e. it is not a random sample). Therefore, if I were to anaesthetise an asymptomatic patient at my hospital today, it would be reasonable to assume they had less than a 0.13% (95% CI: 0.07% to 0.19%) probability of being a carrier for the SARS-CoV-2 virus.
Probability of transmission
Standard precautions such as hand hygiene, gloves, eye protection and flat surgical masks reduce the risk of SARS-CoV-2 transmission during contact with patients. However, one could make an argument that a higher level of PPE (e.g. an N95 mask) is required during general anaesthesia due to the aerosol-generating nature of many of the airway procedures undertaken. 3
It is true that N95 masks provide better protection than flat surgical masks when tested in a laboratory for surrogate markers such as inward leakage of air. 4 However it is also true that N95 masks and flat surgical masks both reduced SARS-CoV-1 transmission (in the original ‘SARS outbreak’ of 2003) by the same amount—about 80%. 5 Clearly, a flat surgical mask provides some non-trivial amount of protection to healthcare workers even when they are exposed to aerosol-generating procedures. 6
In a meta-analysis of nine observational studies assessing the effectiveness of any respiratory PPE (versus no PPE) during the SARS-CoV-1 outbreak of 2003, the odds ratio (OR) for SARS infection amongst exposed healthcare workers was 0.22 (95% CI: 0.12–0.40). 5 Therefore, a ‘ballpark estimate’ for transmissibility would be 22%, with a corresponding ‘ballpark range’ of 12% to 40%.
Risk of acquiring COVID-19 from an asymptomatic patient
We can use these estimates of incidence and transmissibility to estimate the risk that an anaesthetist will acquire COVID-19 from an asymptomatic patient undergoing general anaesthesia:
In other words, approximately 3500 patients (or somewhere between 1300 and 11,700 patients) would need to be anaesthetised today at my hospital before I would expect an anaesthetist to acquire COVID-19 from a patient. It is the very low incidence of SARS-CoV-2 in the community that conveys most of the protection—not the degree of aerosolisation or the specific type of PPE worn. Any risk if using an N95 mask would be at least as low as when using a flat surgical mask, but the number needed to treat in order to potentially prevent one transmission would be several thousand.
What about the cumulative probability of anaesthetising multiple patients over the course of the pandemic? Let’s call this Risk
n
(where n is the number of patients anaesthetised):
If one were to anaesthetise 100 patients over the course of the pandemic (n = 100) and the data for incidence and transmissibility remain unchanged:
The estimated incidence will fluctuate over time, and indeed vary considerably depending on geographic region. If testing reveals a hotspot of community transmission in one location, airborne precautions may be warranted in that area but not for the rest of the country. The estimate for transmissibility may be refined over the course of the pandemic as more data become available from observational studies of SARS-CoV-2 infection amongst healthcare workers. We may also identify situations where there is unusually high transmissibility (e.g. prolonged aerosolisation of the virus during ear, nose and throat (ENT) surgery). If such data were to become available, it may be reasonable to use airborne precautions in these specific circumstances.
What should remain constant is the underlying methodology and calculations for estimating the cumulative risk of a rare and uncertain event. Professional bodies that provide recommendations and guidance on PPE during the pandemic should include an estimate of this risk in their decision-making and communication.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.