Abstract
Microbiota—defined as a collection of microbial organisms colonising different parts of the human body—is now recognised as a pivotal element of human health, and explains a large part of the variance in the phenotypic expression of many diseases. A reduction in microbiota diversity, and replacement of normal microbes with non-commensal, pathogenic or more virulent microbes in the gastrointestinal tract—also known as gut dysbiosis—is now considered to play a causal role in the pathogenesis of many acute and chronic diseases. Results from animal and human studies suggest that dysbiosis is linked to cardiovascular and metabolic disease through changes to microbiota-derived metabolites, including trimethylamine-N-oxide and short-chain fatty acids. Dysbiosis can occur within hours of surgery or the onset of critical illness, even without the administration of antibiotics. These pathological changes in microbiota may contribute to important clinical outcomes, including surgical infection, bowel anastomotic leaks, acute kidney injury, respiratory failure and brain injury. As a strategy to reduce dysbiosis, the use of probiotics (live bacterial cultures that confer health benefits) or synbiotics (probiotic in combination with food that encourages the growth of gut commensal bacteria) in surgical and critically ill patients has been increasingly reported to confer important clinical benefits, including a reduction in ventilator-associated pneumonia, bacteraemia and length of hospital stay, in small randomised controlled trials. However, the best strategy to modulate dysbiosis or counteract its potential harms remains uncertain and requires investigation by a well-designed, adequately powered, randomised controlled trial.
Introduction
The intricate relationship between a eukaryote and bacterium—likely Rickettsia prowazekii—started approximately 0.6–1.5 billion years ago when this aerobic bacterium was integrated into a eukaryote to become a mitochondrion, drastically shaping all advanced life forms, including humans, on our planet ever since.1,2 A dynamic and complex relationship between diverse communities of microbes and host organisms, including humans, continues today. 3
A microbiota is defined as a collection of microbial organisms colonising a habitat. The collective genome of these organisms is the microbiome. Due to availability of 16S rRNA sequencing, the microbiota is now recognised as a pivotal element of human health, and explains a large part of the variance in the phenotypic expression of many diseases.3–5 A reduction in microbiota diversity, and replacement of normal microbes with non-commensal, pathogenic or more virulent microbes both in and on different parts of the human body—in particular the gastrointestinal tract (also known as gut dysbiosis)—is now considered to play an important part in the pathogenesis of many acute and chronic illnesses, including obesity, diabetes mellitus, inflammatory bowel disease, non-alcoholic fatty liver disease, autoimmune diseases, cancers and even mental health conditions.5–24
The key words ‘probiotics’, ‘surgery’, ‘trauma’, ‘critical illness’, ‘burns’, and ‘microbiota’ were used to search for relevant studies published in the PubMed database between January 2014 and July 2019 to provide the most up-to-date information on the importance of gut microbiota in surgical and critically ill patients.
Factors affecting the gut microbiota
Food, antibiotics, other common medications and the effects of surgery and critical illness may all affect the gut microbiota. The human gastrointestinal tract is quickly colonised with 500–1000 species of microbes after birth from interactions with the environment. 25 A healthy individual’s gastrointestinal tract is predominantly colonised by Firmicutes (including Lactobacillus) and Bacteroides (>98% in total), with a smaller proportion constituted by Proteobacteria, Actinobacteria, Verrucobacteria and Fusobacteria. 26 Many factors can affect the diversity and relative proportion of different bacterial species in the gut. Food is the energy source for the gut microbes. Changes in the type of food ingested, including the quantity of micronutrients such as polyphenols, can modulate the gut ecosystem within one or two days (Table 1).25,27–33
Factors affecting gut microbiota and its possible effects on the body.

TMA: trimethylamine; TMAO: trimethylamine-N-oxide; SCFAs: short-chain fatty acids.
Antibiotics have rapid and potentially long-lasting effects on the microbiota. Even a couple of doses of perioperative antibiotics can affect the microbiota of the gut, respiratory tract and breastmilk,34–36 and the latter could affect an infant’s gut microbiota for up to three months. 37 The effect of using antimicrobials to decontaminate the digestive tract selectively in the critically ill is also a concern because recolonisation of the gut by antibiotic-resistant bacteria may occur upon intensive care unit (ICU) discharge after cessation of selective decontamination. 38
A number of non-antibiotic drugs often used in critically ill patients, such as atypical antipsychotics and proton pump inhibitors, can have substantial direct antimicrobial activity 39 which would affect microbial diversity in the gut.40–42 In addition to direct antimicrobial activity, non-antibiotic drugs such as opioids and non-steroidal anti-inflammatory drugs can also affect gut microbiota indirectly by their cytotoxic effect and by altering gut motility and biliary acid metabolism.43–47 Although it is not easy to apply to urgent surgical and critically ill patients, physical activity, as part of prehabilitation, 48 may have beneficial effects on gut microbiota diversity, which can, reciprocally, improve patients’ muscle strength.49–52
Finally, just the stress of surgery, injury, haemorrhage and critical illness itself, even without the use of antibiotics or therapeutic interventions, may also reduce the gut microbiota diversity within hours.22,53,54 Conversely, use of probiotics (i.e. live bacterial cultures that confer health benefits), prebiotics (i.e. food that encourages the growth of gut commensal bacteria) or a combination of prebiotics and probiotics (which is also called synbiotics) may improve microbiota diversity. 55
Links between gut dysbiosis and organ dysfunction
The relationship between the normal commensals inhabiting the human gut and their healthy human host is known as mutualism. The host provides a conducive environment, and the microbiota provides nutrients and energy to the host from the ingested food that the host enzymes are unable to break down, as well as producing metabolic bioactive signalling molecules that can affect multiple organ systems, including the neural,56–58 hepatic, 59 renal,60,61 cardiovascular,62,63 respiratory, 64 immune 65 and coagulation systems (Figure 1).66–68 A number of biologically active gut microbiota–derived metabolites, including trimethylamine-N-oxide (TMAO), uraemic toxins, short-chain fatty acids (SCFAs), phytoestrogens, anthocyanins, bile acids and lipopolysaccharide, have been shown to have substantial effects on different organ systems. 69 In this review, we will focus on two of the most important and well-established microbiota-derived metabolites: TMAO and SCFAs.
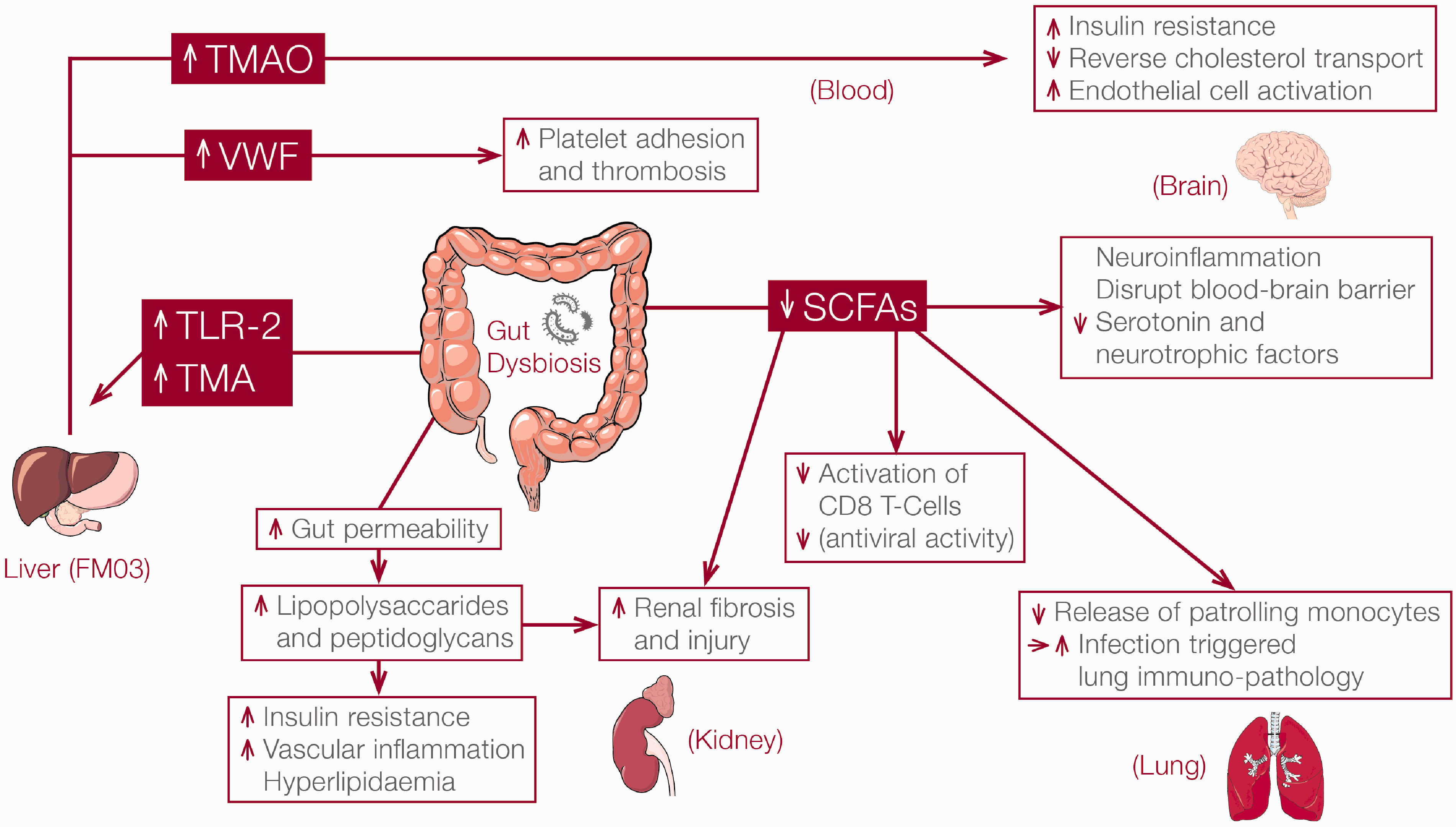
Links between dysbiosis and organ dysfunction. TMA: trimethylamine; TMAO: trimethylamine-N-oxide; SCFAs: short-chain fatty acids; vWF: von Willebrand factor; TLR-2: Toll-like receptor-2; FMO3: flavin-containing mono-oxygenase-3.
TMAO
Choline is water-soluble nutrient from food sources such as eggs, red meat, liver, soybeans and pork. It is a precursor for phospholipids (including phosphatidylcholine) which are essential components of membranes and of the neurotransmitter acetylcholine. Many species of gut bacteria, though not the major normal commensal Bacteroides, can metabolise choline into trimethylamine (TMA), which is then rapidly absorbed into the splanchnic circulation before further transformation to TMAO by the flavin-containing mono-oxygenase-3 (FMO3) enzyme in the liver. A reduction in Bacteroides and an increase in TMA-forming microbes, such as Firmicutes, in the gut microbiota will increase the production of TMA and hence also plasma TMAO. Choline itself, however, does not impact on the abundance of TMA-producing bacteria. Of the five functional FMO enzymes in the liver, FMO3 is the only one which effectively catalyses the conversion of TMA to TMAO.70–72 In addition to choline,
TMAO is normally excreted by the kidneys, but it can induce adverse systemic effects, including platelet hyper-reactivity, endothelial cell and macrophage foam cell activation, modulation of cholesterol and sterol and/or bile acid metabolism, and vascular inflammation.62,70,74–76 Compared to a control group, mice receiving a diet containing choline (1.2%) or TMAO (0.12%) for 12 weeks developed more pulmonary oedema, cardiac enlargement and myocardial fibrosis, and reduced left ventricular ejection fraction. 77 Similarly, patients with heart failure have elevated TMAO concentrations that correlate with plasma B-type natriuretic peptide (BNP) levels, and are predictive of five-year mortality in a dose–response fashion. 78 A large epidemiological study of 4007 patients undergoing elective coronary angiography also found a concentration-dependent relationship between elevated plasma TMAO and major adverse cardiac events of death, myocardial infarction and stroke over a three-year period, independent of traditional cardiovascular risk factors. 79 Furthermore, elevated TMAO levels were also shown to be associated with the concentrations of pro-inflammatory intermediate monocytes (CD14++CD16+; correlation = 0.7) 80 and atherosclerotic burden on coronary angiography in patients with coronary artery disease. 81 Similarly, microbiota-related metabolites of the TMAO pathway were reported to be associated with two important complications after heart transplantation: rejection and total atheroma volume which is a precursor of cardiac allograft vasculopathy. 82 Interestingly, statin use is associated with a reduced level of TMAO in patients with coronary artery disease, suggesting that statins may work beyond reducing the plasma cholesterol levels by interacting with the microbiota–heart axis.83,84
Beside inducing TMAO, dysbiosis can also generate Toll-like receptor-2 signalling which will increase von Willebrand factor synthesis by the hepatic endothelial cell, resulting in acceleration of platelet deposition and thrombus growth after vascular endothelial injury. 66 In addition to cardiovascular adverse effects, TMAO is also believed to play a role in the relationship between dysbiosis and conditions including progressive renal fibrosis, 85 insulin resistance 86 and non-alcoholic fatty liver disease. 87
SCFAs
SCFAs are now considered as one of the most important beneficial microbiota-related metabolites. SCFAs, including acetate, propionate and butyrate, are derived from the bacterial fermentation of dietary fibres, and can act as either substrates for metabolism or signalling molecules. SCFAs are rapidly absorbed (>90%) by the colonic cells into the portal circulation and then mostly used by the hepatocytes to synthesise glucose, cholesterol and fatty acids, with only small amounts of SCFAs reaching the systemic circulation. SCFAs have multiple important physiological roles in the gastrointestinal tract, including maintenance of mucosal barrier integrity by regulating the expression of tight junction proteins to reduce bacterial translocation, improving mucous production and mobility, and anti-inflammatory and immunomodulatory properties. 58 SCFAs are multi-target messengers and can stimulate three G-protein coupled receptors on many types of cells beyond those in the gastrointestinal tract, including tissue-resident (or localised) immune cells in the peripheral blood, bone marrow, lung, small intestine, adipose tissue and brain.88–91
The actions of SCFAs on the heart have received much attention recently. In a murine model of chronic left anterior descending artery ligation, gut microbiota suppression by an oral antibiotic mixture (vancomycin, neomycin, metronidazole and ampicillin) markedly increased the rates of post myocardial infarction ventricular rupture and death in a dose-dependent fashion. Crucially, this mortality outcome could be substantially attenuated by faecal transplantation (from untreated mouse donors) before myocardial infarction, suggesting that gut microbiota are involved in the early phase of myocardial remodelling after myocardial infarction. 92 This interesting study showed that there was a dramatic reduction in monocytic cell infiltration into the tissue affected by myocardial infarction in animals with microbiota suppressed by antibiotics compared to animals untreated with antibiotics prior to the infarction. Administering enteral SCFAs or intravenous infusion of the monocytic cell line in addition to antibiotics one day after infarction not only restored the normal monocytic infiltration into the tissue of the study animals but also improved survival of these animals with less myocardial rupture. Furthermore, administration of SCFA-producing probiotics (Lactobacillus acidophilus, Bifidobacterium bifidum, Lactobacillus casei, Lactobacillus paracasei and Lactobacillus rhamnosus) was associated with both improved ejection fraction and levels of monocytic infiltration in the infarcted myocardium, partially overcoming the adverse effects of microbiota suppression. This study strongly suggests that SCFAs are the key mediators through which the gut microbiota can modulate myocardial tissue repair (or left ventricular remodelling) by the tissue-resident immune cells after injury. Statins are widely used in patients with coronary artery disease, and in an animal setting, statins do not appear to affect the production of SCFAs, and the gut bacteria are not sensitive to physiological concentrations of statins. 93
In addition to the cardiovascular system, a lack of SCFAs due to dysbiosis is also believed to induce a wide range of adverse effects on other organ systems through a number of different receptors in the brain, sympathetic ganglia, vagal nerves, peripheral nerves, gut endocrine cells and renal juxtaglomerular apparatus, inducing neuroinflammation, disrupting the blood–brain barrier and causing a reduction in serotonin and neurotrophic factor biosynthesis. These pathological changes may, in part, contribute to the development of chronic renal, mental health and neurodegenerative diseases.58,94–97 The fact that supplementing SCFAs can attenuate the development of some of these diseases in different experimental settings substantially strengthens the causal role of SCFA deficiency in these diseases.92,97–101
Clinical trials of probiotics and synbiotics in surgical and critically ill patients
Although the evidence to support the causal link between dysbiosis and many chronic diseases is increasing, the relationship between dysbiosis and acute illnesses in surgical or critically ill patients is much more contentious. A number of clinical studies have shown that changes in gut microbial diversity, with a lower relative abundance of Firmicutes and Bacteroidetes and an increased relative abundance of Proteobacteria resulting in a reduction in SCFAs level, can occur within hours of an acute insult to the body.16,22,102,103 The effects of dysbiosis on a perioperative or critically ill patient are confounded further by medical interventions that can affect the microbiota directly, including blood transfusion, 104 parenteral nutrition105,106 and the use of antibiotic and non-antibiotic drugs, including antipsychotics, proton pump inhibitors, non-steroidal anti-inflammatory drugs, hypoglycaemic agents and chemotherapy.34–37,43,47,107–109
Clinical trials on surgical and critically ill patients
Ultimately, any causal link between dysbiosis and adverse outcomes after surgery or in critical illness would require confirmation by clinical trials showing a reduction in adverse outcomes after interventions to modulate dysbiosis. In patients undergoing gastrointestinal surgery, anastomotic leaks after bowel resection are a major concern because of the associated morbidity and mortality. Observational studies on patients with leaks showed that a lack of microbial diversity and abundance of mucin-degrading Lachnospiraceae are common in their microbiota,110,111 offering a plausible mechanistic reason for the causal link between dysbiosis and leaks after bowel resection. 112 There are animal and human controlled trials showing that applying topical antimicrobial agents to reduce non-commensal gut microbes in the areas of bowel anastomosis would improve healing and reduce leaks after bowel resection. 113 A number of small randomised controlled clinical trials, mostly published prior to 2014, also suggested that the use of pro/pre/synbiotics was associated with a reduced incidence of infection after a variety of different surgical procedures and a reduced incidence of anastomotic leaks after bowel resection.114,115 Unfortunately, a strong conclusion cannot be drawn from these older studies because the infection endpoint was not clearly defined. 114
More randomised controlled trials evaluating the benefits of probiotics/synbiotics involving surgical and critically ill patients in a variety of clinical settings have been reported in the past five years (Table 2).116–140 Study probiotics were administered enterally in most controlled trials, but in two trials, topical probiotics were administered to either the oral cavity or burn wound to assess their effects against bacterial colonisation compared to standard antiseptic solutions. The use of topical Lactobacillus plantarum instead of antiseptic for burn-wound dressing resulted in similar rates of wound healing and bacterial infection, and more importantly, Lactobacillus plantarum was not recovered from either peripheral blood or wound samples. 132 Of the 21 studies comparing enteral probiotics/synbiotics with placebo, 14 (67%), including five that were double-blinded,118,128–131,134,138 reported some form of clinical benefit after using probiotics/synbiotics compared to placebo: a reduction in ventilator-associated pneumonia, bacteraemia, length of mechanical ventilation and hospital stay. These studies were all relatively small (N ≤ 362), and none was powered to detect a mortality difference. 141 In terms of safety, none of these studies reported any episodes of bacteraemia or liver abscess related to the live bacteria contained in the probiotic/synbiotic treatment, although this potential complication has been reported in the literature, particularly in patients who are immunocompromised.142–145
Randomised controlled clinical trials assessing the effects of probiotics or synbiotics on outcomes of surgical and critically ill patients.

RR: risk ratio; CI: confidence intervals; ICU: intensive care unit; VAP: ventilator-associated pneumonia.
Although the evidence to support the benefits of modulating dysbiosis in acute surgical and critically ill patients is strengthening, significant challenges as to how this can be translated into improved patient-centred outcomes remain. First, not all gut commensals are equally important, and some may offer more health benefits than others.30,39,40 Response to different types of probiotic treatment may vary and be specific to the strain of live bacteria used.24,146,147 Although not proven, the different types and doses of live bacteria and duration of probiotic therapy used in the studies may, at least in part, explain why the benefits of probiotics/synbiotics were not consistently reported (Table 2). Second, the emerging science is almost exclusively derived from 16S rRNA sequencing. This describes the quantity but not the function of bacteria, and does not look at viruses, fungi or protozoa. Unlike animal studies, important microbiota-related metabolites in the plasma, such as TMAO and SCFAs, were not measured in most, if not all, clinical trials. It is paramount to confirm that any live bacteria used in the probiotics/synbiotics can directly tackle the likely pathogenic pathways of dysbiosis by successfully colonising the colon with healthy microbiota that can generate SCFAs and reduce TMAO in the blood. 148
Finally, there are other interventions that can attenuate the potential harms of dysbiosis, including faecal microbiota transplantation, 149 SCFA supplementation,91,100,101 use of choline structural analogues to reduce TMA production by gut microbes,150,151 and reducing conversion of TMA to TMAO by inhibiting FMO3 in the liver. 152 In addition, early enteral nutrition, 153 a restrictive approach to using antimicrobials, 154 use of lactulose as a prebiotic155,156 and use of enteral digestive protease inhibitors such as tranexamic acid to reduce generation of TMAO may also be helpful. 157 Whether these alternative interventions are better than probiotics or if they can have additive benefits is not known, but this deserves further investigation by adequately powered randomised controlled trials.
Probiotics in Clostridium difficile–related diarrhoea and Helicobacter pylori infection
Clinical trials evaluating the efficacy of probiotics on specific bowel-related infections have been reported with mixed results.158–162 Although probiotics appear to be useful as an adjunct to therapeutic oral antibiotics to improve bowel symptoms in both Clostridium difficile–related diarrhoea and Helicobacter pylori infections, probiotics alone are certainly inadequate for eradicating these infections. 159
Conclusions
We begin to fully appreciate the importance of a healthy microbiota ecosystem in determining health and disease. In addition to antibiotics, commonly prescribed non-antibiotic drugs, acute illness and surgery all can have a substantial effect on microbiota diversity and risk of dysbiosis, which can occur within hours in surgical and critically ill patients.
Dysbiosis can affect different organ systems that are not anatomically in close proximity to the gastrointestinal tract through a number of different microbiota-derived metabolites, including TMAO and SCFAs. Results from animal and human studies are supportive of a causal role for dysbiosis in many chronic illnesses, especially cardiovascular and metabolic diseases. Using probiotics/synbiotics in surgical and critically ill patients to reduce or modulate dysbiosis has been increasingly reported to confer a number of important benefits, including a reduction in ventilator-associated pneumonia, bacteraemia and length of hospital stay. These trials are, however, small and have substantial limitations. As such, adequately powered randomised controlled trials assessing the effect of probiotics/synbiotics on mortality of surgical and critically ill patients are crucial before probiotics/synbiotics are used routinely in surgical and critically ill patients. From an ecology perspective, the most challenging—but also the best—approach to maintain our health, as a ‘supra-organism’, 163 is to achieve mutualism.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
