Abstract
Transpulmonary thermodilution devices have been widely shown to be accurate in septic shock patients in assessing fluid responsiveness. We conducted a systematic review to assess the relationship between fluid therapy protocols guided by transpulmonary thermodilution devices on fluid balance and the amount of intravenous fluid used in septic shock. We searched MEDLINE, Embase and The Cochrane Library. Studies were eligible for inclusion if they were prospective, parallel trials that were conducted in an intensive care setting in patients with septic shock. The comparator group was either central venous pressure, early goal-directed therapy or pulmonary artery occlusion pressure. Studies assessing only the accuracy of fluid responsiveness prediction by transpulmonary thermodilution devices were excluded. Two reviewers independently performed the search, extracted data and assessed the bias of each study. In total 27 full-text articles were identified for eligibility; of these, nine studies were identified for inclusion in the systematic review. Three of these trials used dynamic parameters derived from transpulmonary thermodilution devices and six used primarily static parameters to guide fluid therapy. There was evidence for a significant reduction in positive fluid balance in four out of the nine studies. From the available studies, the results suggest the benefit of transpulmonary thermodilution monitoring in the septic shock population with regard to reducing positive fluid balance is seen when the devices are utilised for at least 72 hours. Both dynamic and static parameters derived from transpulmonary thermodilution devices appear to lead to a reduction in positive fluid balance in septic shock patients compared to measurements of central venous pressure and early goal-directed therapy.
Keywords
Introduction
Optimal fluid management in septic shock patients is unclear and consensus on the best approach has shifted greatly in the past 20 years.1–3 Multiple trials have demonstrated that fluid therapy in sepsis is highly complex and needs to be tailored to the individual patient rather than relying on static pressure–based goal therapy.4–6 To this end, advanced haemodynamic monitoring and dynamic assessment of how to best determine if a patient is fluid responsive has been well studied.7–9 Common dynamic parameters are the change in cardiac output/index following a fluid challenge or passive leg raise (PLR) and stroke volume variation. Static parameters include pressure-based measurements including central venous pressure (CVP) and pulmonary capillary wedge pressure (PCWP), and volumetric measurements including extravascular lung water index (EVLWI), intrathoracic blood volume index (ITBVI) and global end-diastolic index (GEDI). The benefit of advanced haemodynamic monitoring over CVP or other pressure-based measurements has been well shown for reducing mortality, duration of length of stay (LOS) and duration of mechanical ventilation (MV) in mixed intensive care unit (ICU) populations.10,11 The main problem with extrapolating these results to septic shock patients is these reviews have included non-calibrated devices and evidence suggests these devices are not able to accurately track the rapidly changing haemodynamics of septic patients when compared to either a pulmonary artery catheter (PAC) or transpulmonary thermodilution (TPTD) devices.12,13 Additionally, most studies regarding advanced haemodynamic monitoring and septic shock are concerned with the accuracy of a device’s ability to judge if a patient is fluid responsive and do not address whether the ability to determine a patient’s fluid responsiveness leads to any meaningful change in decisions around volume of intravenous (IV) fluid used in the resuscitation and maintenance of the septic patient.
The underlying assumption of these studies comparing the accuracy of advanced haemodynamic to pressure-based measurements is that if dynamic monitoring is more accurate in judging fluid responsiveness, meaningful outcomes with regard to a reduction in positive fluid balance should be seen when these devices are used. To this end, this systematic review was conducted to compare IV volume therapy and clinical outcomes when dynamic and/or static TPTD parameters are used in comparison to clinical examination and pressure-based measurements.
Methods
Protocol and eligibility criteria
This systematic review was prospectively registered with PROSPERO (CRD42018106744). Studies eligible for inclusion were conducted in ICU patients with a diagnosis of septic shock, prospective in nature and included a study arm with allocation to fluid therapy guided by a TPTD device requiring intermittent calibration (either the Pulse index Contour Cardiac Output (PiCCO) device or EV1000 with VolumeView). The comparator group could be any other modality to assess fluid responsiveness including early goal-directed therapy (EGDT), CVP or other haemodynamic monitoring tool. Only studies that evaluated the effect of TPTD devices on total IV fluid use or clinical outcomes were included; studies only assessing the ability of the devices to predict fluid responsiveness were excluded. The primary outcome assessed could include IV fluid use or fluid balance, with no limitation on follow-up time. Secondary outcomes included duration of MV, LOS and mortality. Only peer-reviewed published studies were eligible for inclusion and there was no language restriction. Retrospective studies, reviews, abstracts and case reports were excluded.
Search and study selection
Two authors (TS and YH) searched MEDLINE, Embase and The Cochrane Library from the respective inception dates of the databases to May 2019. A selection of the keywords used in the search strategy were: ‘Fluid responsiveness’, ‘Fluid balance’ or ‘Fluid replacement’, ‘Septic shock’, ‘Sepsis’, ‘PiCCO’, ‘EV1000’, ‘Pulse pressure variation’, ‘Stroke volume’ and ‘Passive leg raise’. A full search strategy for each database can be found in Supplemental data 1. The bibliographies of included papers and relevant review articles were also scanned for additional articles. A two-step process for identifying studies was employed; initially TS and YH identified all titles/abstracts that potentially fulfilled the inclusion criteria. Full texts were then assessed for eligibility; if there was disagreement between the two assessors, a third assessor (SO) made the final decision.
Data collection and items
The following data were extracted by TS and YH to a pre-designed form: number of patients, country, inclusion/exclusion criteria, comparator protocol, and protocol for TPTD, age, gender and illness severity scores including Sequential Organ Failure Assessment and Acute Physiology and Chronic Health Evaluation scores. The primary outcome of fluid balance and IV fluid expansion volumes were recorded for each available timepoint reported by the individual study. Secondary outcomes were outcomes related to MV, LOS and mortality, either in the ICU or hospital.
Risk of bias in individual studies
The risk of bias for the included randomised controlled trials (RCTs) was evaluated by the Cochrane Risk of Bias tool and prospective cohort trials were assessed by ROBINS-1.
Data analysis
A meta-analysis was deemed inappropriate due to the different devices used in the comparator group, outcomes measured in each study and different endpoints with regard to time frame.
As TPTD devices can either rely on volumetric or dynamic fluid assessments, included studies were split into two groups for primary outcome analysis: those that primarily used dynamic methods and those that primarily used static TPTD parameters. Studies were defined as dynamic method studies if the fluid protocol focused on IV therapy guided by judging fluid responsiveness by dynamic assessment whereas static method studies were those that guided fluid therapy based on volumetric endpoints.
To reduce the heterogeneity of the fluid outcomes, where available 72-hour fluid balances have been reported. When 72-hour fluid balances were not available, the closest fluid balance in timing was reported. For individual study outcome data, including primary and secondary outcomes, either the mean (standard deviation) or median (interquartile range) has been used in this systematic review based on the original study’s reporting method.
Although a formal meta-analysis was not possible due to heterogeneity, simple pooled analysis of primary and secondary outcomes has been performed along with a narrative summary of the individual studies. The Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach was applied to the dynamic TPTD compared to static measurements and the static TPTD compared to static measurement subgroups to provide an overall assessment of the evidence relating to both the primary and secondary outcomes. The summary of findings table was generated using the GRADEpro software. 14 Supplementary data file 2 contains justifications for assessment of the evidence ratings.
Results
Characteristics of included studies
As shown in Figure 1, from a total of 8162 citations retrieved, nine studies were selected for inclusion.15–23 The nine studies were all prospective, with eight being RCTs and one being a prospective cohort. Six of the studies were published in English and three in Chinese. All trials had septic shock patients as their primary population of interest with three exceptions: Yu et al. (2017) limited their study population to chronic obstructive pulmonary disease (COPD) patients in septic shock, Trof et al. included septic shock and all other forms of shock and Zhang et al. included both acute respiratory distress syndrome (ARDS) and septic shock patients in their trial. There were three trials that used a dynamic TPTD protocol and six trials that used a protocol based on static TPTD measurements. Five of the comparator groups used CVP pressures, three groups used EGDT based on CVP measurements and one group used PCWP to guide fluid therapy.
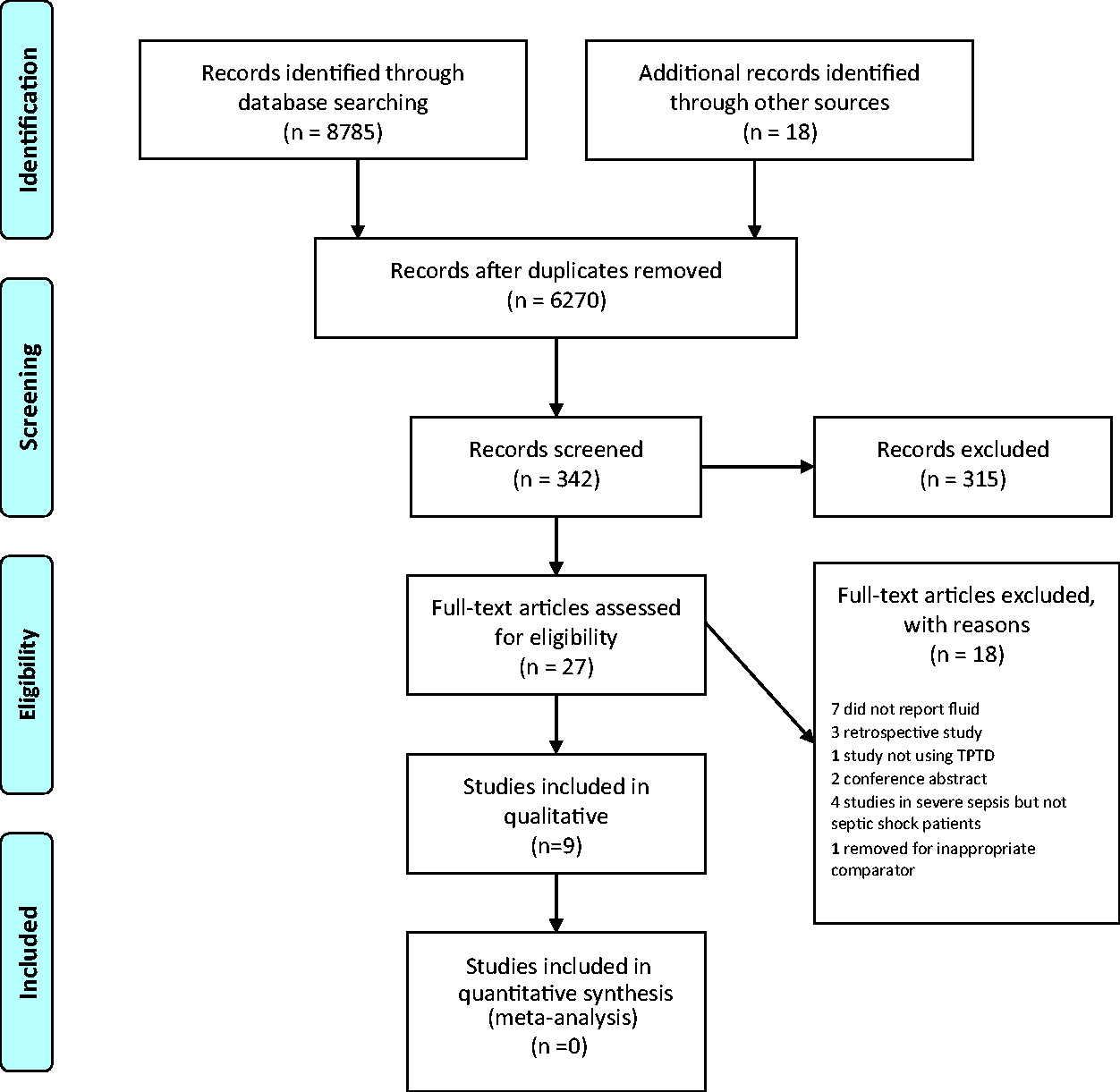
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-analyses) flow chart of study inclusion.
A total of 813 septic shock patients were evaluated from the nine included studies. All nine studies used the PiCCO device as the TPTD device. The characteristics of the included studies are shown in Table 1.
Characteristics of included studies.
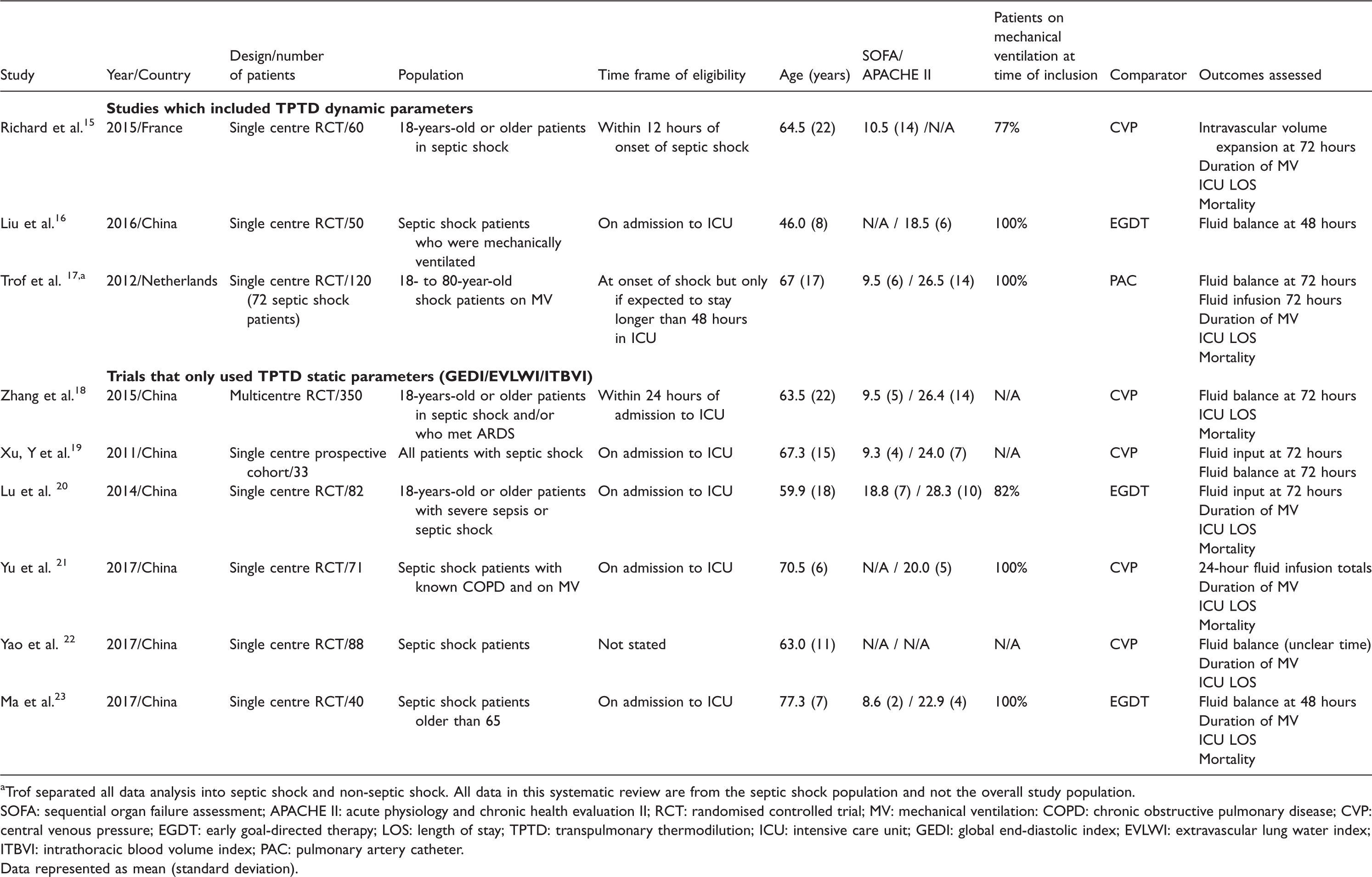
aTrof separated all data analysis into septic shock and non-septic shock. All data in this systematic review are from the septic shock population and not the overall study population.
SOFA: sequential organ failure assessment; APACHE II: acute physiology and chronic health evaluation II; RCT: randomised controlled trial; MV: mechanical ventilation: COPD: chronic obstructive pulmonary disease; CVP: central venous pressure; EGDT: early goal-directed therapy; LOS: length of stay; TPTD: transpulmonary thermodilution; ICU: intensive care unit; GEDI: global end-diastolic index; EVLWI: extravascular lung water index; ITBVI: intrathoracic blood volume index; PAC: pulmonary artery catheter.
Data represented as mean (standard deviation).
With respect to bias, only one trial was assessed to have a low risk of bias, all other trials had a moderate, high/serious or unclear risk of bias with the most common risk of bias due to lack of blinding (Table 2).
Assessment of individual trial bias.
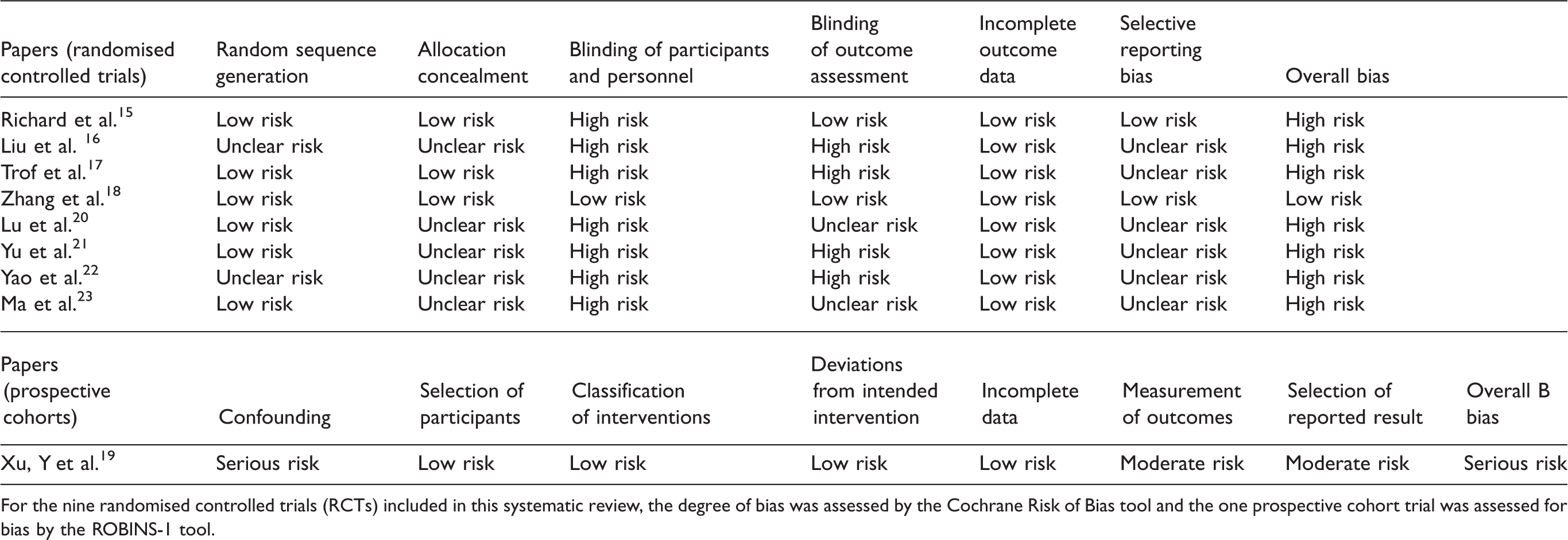
For the nine randomised controlled trials (RCTs) included in this systematic review, the degree of bias was assessed by the Cochrane Risk of Bias tool and the one prospective cohort trial was assessed for bias by the ROBINS-1 tool.
Effect of TPTD on fluid therapy and clinical outcomes
The individual results of fluid-based outcomes for the three trials that used dynamic parameters measured by the TPTD are demonstrated in Table 3. The individual results of fluid-based outcomes for the six trials that used static parameters measured by the TPTD are demonstrated in Table 4. Overall, the effect of using TPTD monitoring in comparison to pressure-based measurements on fluid balance in septic shock is mixed and appears dependent on the intervention that TPTD is compared to and the time frame of the study more than the TPTD protocol used. Studies that reported at 6 or 24 hours either found no difference in fluid balance or infusion totals between TPTD and pressure measurements 23 or found an increase in fluid use in the TPTD group.20,21 In contrast, studies that reported at 48 and 72 hours either found no significant difference15,17,19 or a reduction in positive fluid balance for the TPTD group.16,23 Finally, only one trial reported to seven days and found a less positive fluid balance on day seven for the TPTD group. 18 In the seven trials comparing TPTD to CVP or EGDT based primarily on CVP, four demonstrated there was a significant reduction in the positive fluid balance when TPTD was used. In comparison, in the one trial that compared TPTD to PCWP, there was no difference in fluid balance. 17 Assessing the effect of TPTD protocols based on dynamic compared to static measures, when dynamic TPTD studies were pooled for fluid balance, there was a moderate level of evidence to show a less positive fluid balance compared to pressure-based measurements. When static TPTD studies were pooled for fluid balance, there was a low level of evidence to show a reduction in positive fluid balance compared to pressure-based measurements.
Fluid-based outcomes in studies using dynamic transpulmonary thermodilution measures.
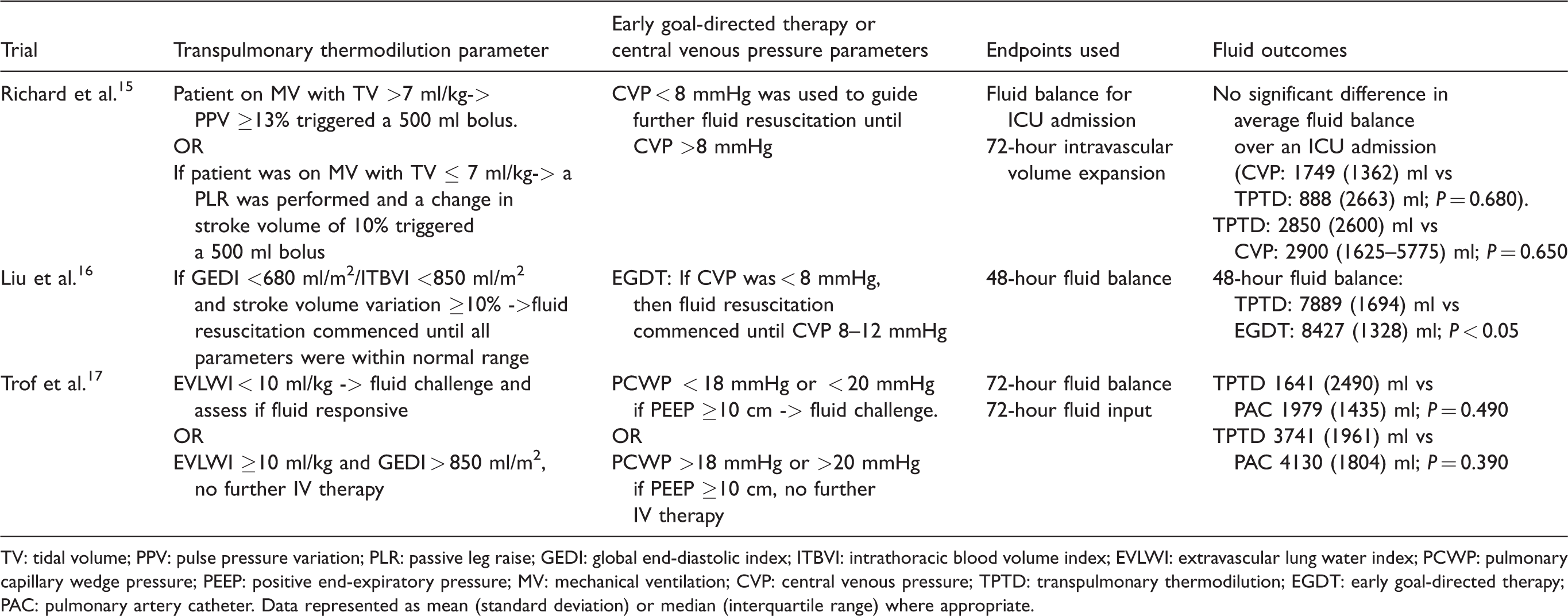
TV: tidal volume; PPV: pulse pressure variation; PLR: passive leg raise; GEDI: global end-diastolic index; ITBVI: intrathoracic blood volume index; EVLWI: extravascular lung water index; PCWP: pulmonary capillary wedge pressure; PEEP: positive end-expiratory pressure; MV: mechanical ventilation; CVP: central venous pressure; TPTD: transpulmonary thermodilution; EGDT: early goal-directed therapy; PAC: pulmonary artery catheter. Data represented as mean (standard deviation) or median (interquartile range) where appropriate.
Fluid-based outcomes in studies utilising static transpulmonary thermodilution measures.
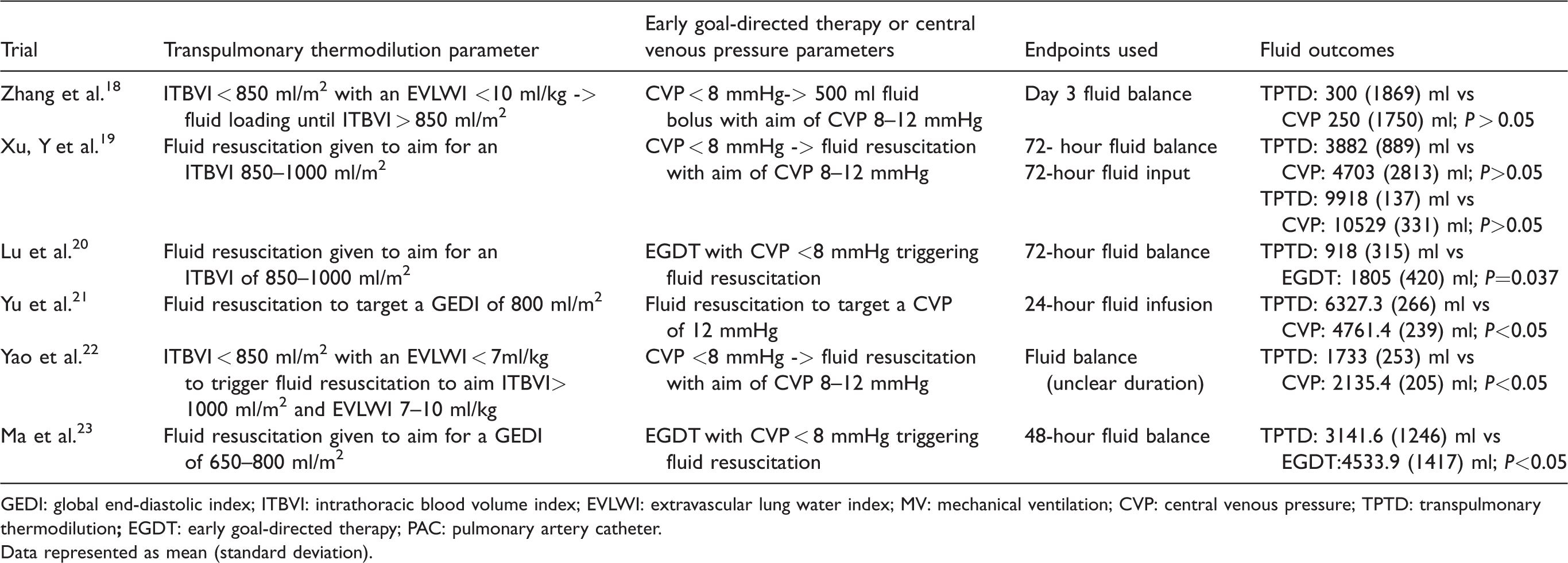
GEDI: global end-diastolic index; ITBVI: intrathoracic blood volume index; EVLWI: extravascular lung water index; MV: mechanical ventilation; CVP: central venous pressure; TPTD: transpulmonary thermodilution
Data represented as mean (standard deviation).
Studies that assessed the fluid infusion totals demonstrated that total IV fluid use was either the same15,17,19 or increased 21 in the TPTD group compared to the pressure-based comparator groups. When dynamic TPTD studies were pooled for fluid infusion totals, there was a low level of evidence to show TPTD had no effect on infusion totals. When static TPTD studies were pooled for fluid infusion totals, there was a low level of evidence to show that static TPTD monitoring increased the total volume of IV fluid infusion use compared to pressure-based measurements.
Finally, the individual outcomes of each trial on effect of LOS and duration of MV (seven trials), as well as the mortality outcomes (six trials) are shown in Table 5. There was moderate evidence that TPTD monitoring lowered the duration of MV compared to pressure-based measurements in septic shock. The GRADE system has been used to generate a summary of evidence table (Table 6), presenting the quality of evidence for all the above outcomes.
Clinical outcomes for transpulmonary thermodilution in septic shock.
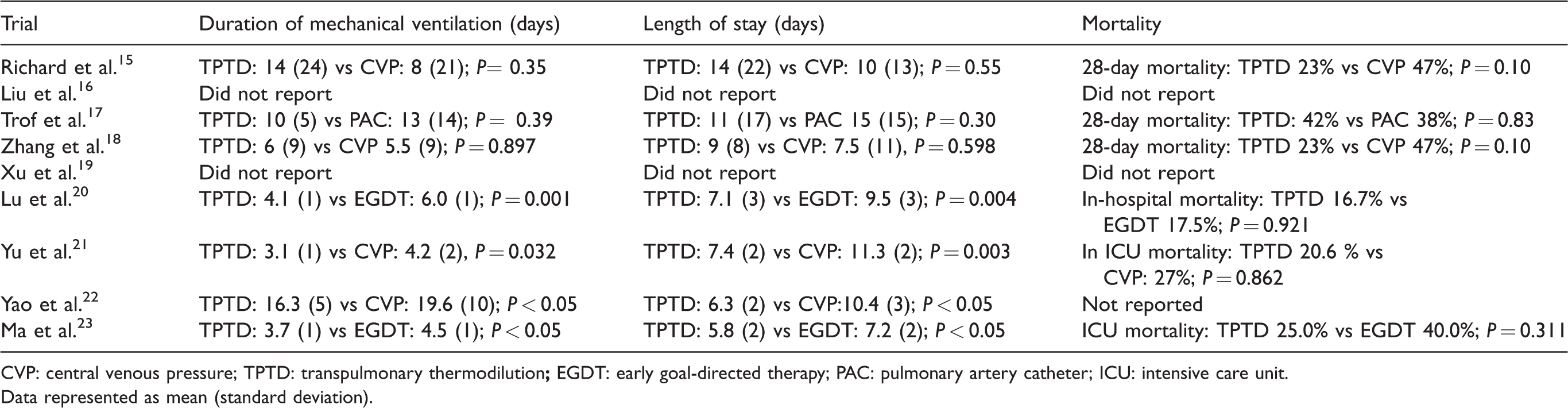
CVP: central venous pressure; TPTD: transpulmonary thermodilution
Data represented as mean (standard deviation).
Grading of Recommendations Assessment, Development, and Evaluation summary of findings for the use of transpulmonary thermodilution in septic shock.

CVP: central venous pressure; TPTD: transpulmonary thermodilution
Data represented as mean (standard deviation).
Discussion
In our systematic review, we found the use of TPTD devices led to an overall reduction in positive fluid balance in septic shock compared to CVP and EGDT. All trials in this systematic review utilising EGDT as the comparator group were reliant on static measurements, most commonly CVP targets. Pressure-based targets in fluid management in septic shock patient have been shown to be detrimental to fluid balances and patient outcomes, likely because of the inherent pitfalls in interpreting volume status from pressure measurements.3,6,24
Interestingly, although both dynamic and static measures of fluid assessment with TPTD devices led to significantly less positive fluid balances, dynamic fluid assessments had no effect on the total IV fluid used and static measures resulted in an increase in total IV fluid administration. It is unclear why there is a trend towards a less positive fluid balance with these devices if they result in an increase in IV fluid therapy. This has been observed in other trials using non-calibrated devices in septic patients. 25 One possible reason is that the presence of advanced haemodynamic devices encourages the clinician to give fluid boluses to measure fluid responsiveness. 26 This may result in higher IV fluid infusions but changes the clinician’s decision-making in terms of strategies to limit the overall fluid balance.
The major limitation of this systematic review is the heterogeneity in the time frame of reporting on fluid outcomes. A prominent feature of trials linking fluid balance to mortality is that a prolonged large positive fluid balance at the end of day 2 to 4 following the onset of critical illness leads to the increase in mortality, not necessarily the volume given in the initial resuscitation period.27,28 Few sepsis trials appreciate the significance of the phases of sepsis and will usually recruit all septic patients, irrespective of duration of sepsis, or use a surrogate time for the onset of sepsis (i.e. admission to the ICU). Based on the results of this systematic review, it appears the time frame of fluid endpoints has a large influence on the outcome and makes interpretation of pooled results difficult due to high levels of heterogeneity. The impact of this potential biphasic effect of fluids in septic shock is that it makes trials with different time frames extremely difficult to interpret in a systematic review. What is likely underestimated is the dynamic nature of sepsis and how quickly the patient may shift from one phase to the next. It has been demonstrated that the later fluids are initiated, the less positive effect they seem to have. 29 As illustrated by Zhong and Xue (2015), 85.7% of patients in septic shock were judged fluid responsive at the time of enrolment to the study but at 72 hours, only 28.6% patients were fluid responsive. This effect of changing fluid responsiveness may explain the time-related effect on fluid outcomes within each trial. Ma found no difference at 6 or 24 hours in fluid balance between GEDI/EVLWI protocol and EGDT but there was a significantly lower positive fluid balance at 48 hours for the GEDI/EVLWI group. 23 Zhang found no difference in fluid balance at 72 hours but demonstrated a significant reduction in positive fluid balance on day 7 for the TPTD group. 18 Richard demonstrated no difference at 72 hours for fluid infusion between the two groups but when considering total fluids over the duration of admission with average LOS of 12 days, there was significantly less IV fluid use on a daily basis in the dynamic TPTD group compared to CVP. 15 Trof showed higher fluid use in the first 24 hours for the TPTD group but less at 72 hours. 17 The one trial, by Yu et al. (2017), which demonstrated increased fluid infusion totals using a TPTD device reported only the first 24 hours. 21
It should be noted that not all TPTD directed therapy is equal in its accuracy to guide fluid therapy. Previous trials have demonstrated that dynamic parameters are far more accurate than static parameters in judging fluid responsiveness and therefore, accurate fluid administration.30–32 Static volume-based measurements (GEDI/EVLWI) have numerous pitfalls in the septic shock population. In studies directly assessing the predictive ability of GEDI/ITBVI for fluid responsiveness, it has been shown to be no better than CVP.33–35 There are other changes in the septic state that confound many of the static parameters. Septic cardiomyopathy changes, including global cardiac dilatation and left ventricular systolic impairment, likely impair the GEDI ability to guide fluids in the septic population and a standard GEDI of 680–800 ml/m2 is almost certainly not appropriate for all septic patients.35,36 Easley et al. (2009) showed the impairment of hypoxic pulmonary vasoconstriction, seen in some forms of sepsis, can cause an increase in the EVLWI without an actual increase in the amount of pulmonary oedema. 37 Although these limitations should indicate that dynamic assessments have a greater impact on fluid therapy, the evidence from this review suggests static and dynamic measures have relatively similar outcomes on fluid therapy.
Two additional major limitations that should be addressed in future trials are the lack of blinded randomised controlled trials assessing effect of fluid responsiveness on fluid balances and the complexity of dynamic measures protocols leading to lack of compliance.
There are few studies available to assess a TPTD’s effect on fluid therapy in septic shock; only nine studies were available for inclusion that focused on the overall fluid therapy and not the ability of the device to predict fluid responsiveness. Additionally, only one of the nine trials was blinded. Non-blinded study designs certainly lend themselves to a high degree of observer bias and Hawthorne effect, introducing a great degree of bias that weakens any result of these trials. A common issue with dynamic fluid responsiveness guided fluid management is that despite their superior evidence for accuracy, they are driven by complex protocols. These complex processes lead to a high rate of non-compliance/adherence to the dynamic fluid protocols, likely leading to erroneous outcomes. Richard et al. (2015) reported 43% of patients in the dynamic group had at least one protocol violation.
15
A possible reason for this non-adherence may be due to many clinicians being unfamiliar with dynamic fluid assessment techniques. As illustrated by Fischer et al. (2016), only 60% of senior ICU physicians could correctly apply pulse pressure variation (PPV); this fell to 17% when considering physicians who do not commonly use PPV.
38
This issue of non-compliance can be addressed and when education is provided, more profound effects of dynamic TPTD measurements are uncovered. In a trial conducted by Rameau et al. involving PLR and PiCCO monitoring in septic shock patients, once non-compliance was addressed, there was a significant fall in the average 48-hour fluid balance from 4.3 l to 3.1 l.
39
Based on the individual studies and GRADE assessment, there is a less clear picture for the effect of either dynamic or static TPTD protocols on LOS, duration of MV and mortality. Although the study protocols that relied on only volume-based measurements (GEDI and EVLWI) had weak evidence for improving the fluid balance compared to EGDT or CVP, they had similar effects on LOS and duration of MV as protocols that incorporated both dynamic and static parameters. This may indicate that although static parameters derived from TPTD should not be used to judge if a patient is fluid responsive, they are still highly useful in the management of the septic shock patient.40–45
A strength of this systematic review is the focus of analysis on septic shock patients, as a frequent criticism of many ICU trials is that the largely heterogeneous group of patients in any given ICU may weaken results.46,47 Sepsis is a diverse population; including other diseases to this systematic review would likely confound any meaningful conclusions to be drawn.
There are multiple limitations of this systematic review, which weaken any conclusions that can be derived about the effects of TPTD monitoring on septic shock patients. An inherent limitation of our paper due to its narrow focus is the limited number of trials available for inclusion in this systematic review and the overall small number of patients. There are multiple studies that demonstrate the effectiveness of advanced haemodynamic monitoring for improving fluid therapy in the ICU. However, these trials had to be excluded from this review, either because they focused on the non-septic population,48,49 were in the postoperative population or utilised non-calibrated haemodynamic monitoring. 50 Another potential weakness in this systematic review is the failure to include other novel techniques, including peripheral arterial velocities and tidal volume challenges.51,52 At the time of writing, these novel techniques do not yet have the level of evidence to determine whether they can be accurately applied in the septic shock population. However, these techniques are highly promising as they are less invasive than TPTD or PAC and easily applied, which therefore may reduce non-adherence to dynamic fluid protocols.
A final limitation of this review is not including PAC trials in this review article. Although both PACs and TPTDs have strong evidence supporting their accuracy in predicting fluid responsiveness in septic shock, TPTDs are preferred to PACs in many ICUs due to a perceived increased risk of PACs and assumed no added benefit over TPTDs.53–56 The decision to exclude PACs from this review was made based on the evidence of decreasing PAC use in ICUs. 56 However, there is an increase in the use of PACs with growing recognition of their importance in septic shock patients with cardiac dysfunction, particularly involving the right ventricle. Although a TPTD is as good as a PAC device for tracking real-time haemodynamic changes in hyperdynamic states such as sepsis, it has limitations when right ventricular dysfunction or severe pulmonary hypertension are present and may be worse than a PAC for management of ARDS.33,56,57 In the one trial that compared TPTD to PAC in this systematic review, there was no significant difference between fluid balance between the two groups in the septic shock population. 17 However, the most effective and accurate method will likely be when TPTD or PAC is used in conjunction with other assessment methods to deliver a more comprehensive answer to the patient’s fluid status. A study by Vignon et al. (2018) assessing agreement of TPTD and transthoracic echocardiography found that echocardiography was able to identify situations where TPTD measurements were likely to be inaccurate. 58 This underlines that the best form of haemodynamic monitoring for fluid therapy in complex patients will likely be a combination of modalities and not one single device or measurement.
Conclusion
The evidence for the effect of TPTD devices on septic shock fluid therapy suggests that both dynamic and static measurements result in a significant reduction in positive fluid balance in the septic shock patient compared to pressure-based fluid protocols. However, the effect is likely underestimated for multiple reasons including non-adherence to protocols and inadequate study designs. Higher-quality trials are required in this field to properly assess the effect of haemodynamic monitoring on septic shock.
Supplemental Material
AIC893703 Supplemental material - Supplemental material for The effects of static and dynamic measurements using transpulmonary thermodilution devices on fluid therapy in septic shock: A systematic review
Supplemental material, AIC893703 Supplemental material for The effects of static and dynamic measurements using transpulmonary thermodilution devices on fluid therapy in septic shock: A systematic review by Timothy G Scully, Yifan Huang, Stephen Huang, Anthony S McLean and Sam R Orde in Anaesthesia and Intensive Care
Footnotes
Availability of data and materials
Complete search data and search histories are available from the corresponding author on request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
