Abstract
The aim of the study was to investigate an ultrasound-guided posterior antebrachial cutaneous nerve block based on visualising the nerve within the fat-filled flat tunnel and describe the area of cutaneous sensory loss. A total of 12 healthy volunteers were included in the study. The posterior antebrachial cutaneous nerve was identified within the fat-filled flat tunnel in the upper arm using high-frequency ultrasound. The nerve was blocked using an in-plane needle guidance technique with 1 ml 2% lidocaine. Sensory loss to pinprick was evaluated 15 minutes after performing the block and the cutaneous sensory block area mapped. Ultrasound visualisation of the posterior antebrachial cutaneous nerve in the fat-filled flat tunnel was possible in all volunteers. The median distance of the posterior antebrachial cutaneous nerve to the lateral epicondyle of the elbow was 67.5 (range 54–105) mm. Loss of sharpness sensation to pinprick extended from the posterior aspect of the distal upper arm and posterior forearm to the wrist. The median cutaneous sensory block area was 103 (range 61–341) cm2. Two volunteers had a sensory block over the anterior forearm of 29 and 10 cm2 respectively. This amounted to 11% and 4.5% of the total cutaneous sensory block area. One volunteer had a sensory block over the dorsum of the hand of 39 cm2 (15% of the total cutaneous sensory block area). The results of this study indicate that the fat-filled flat tunnel can be a useful sono-anatomical landmark in identifying the posterior antebrachial cutaneous nerve and may serve as a target for injection. Although sensory block is predominantly confined to the posterior distal arm and forearm, inter-individual variability in the area and distribution was observed. Sensory block in the anterior forearm and dorsum of the hand can occur. The study was prospectively registered at the Australian New Zealand Clinical Trials Registry, identifier ACTRN12618000891224.
Introduction
The skin and subcutaneous tissues of the forearm receive their innervation from three nerves: the medial, lateral and posterior antebrachial cutaneous nerves of the forearm. For the most part, knowledge of the distribution of cutaneous sensory nerves has been based on anatomical cadaver dissection. Due to variations in cutaneous nerve dispersal, anatomical textbooks present similar but not identical ‘standard’ diagrams of sensory cutaneous borders.1–2 Mapping of sensory borders following ultrasound-guided local anaesthetic block of the medial and lateral antebrachial cutaneous nerves (MABCN and LABCN) of the forearm have been reported.3–4 Blanco et al. suggest locating the posterior antebrachial cutaneous nerve (PABCN) with ultrasound in a ‘fat pocket above the long head of triceps’. 5 Finneran and Sandhu recently described an ultrasound-guided block of the PABCN ‘within the muscle belly of the lateral head of the triceps muscle’. 6
The aim of this study was to investigate the relative merits of visualisation and block the PABCN as it exits the hiatus of the deep fascia of the upper arm. At this location the nerve can be identified with high-frequency ultrasound as an echogenic oval structure travelling within an anechoic fat-filled flat tunnel (FFFT). Block of the PABCN is useful for supplementation of an incomplete brachial plexus block, analgesia or anaesthesia for cutaneous surgical incisions and coverage of forearm tourniquet pain when used in conjunction with LABCN and MABCN blocks. 7
Methods
The Sydney Local Health District Human Research Ethics Committee approved the study protocol (HREC/18/CRGH/94). Clinical Trial Notification was submitted to the Therapeutic Goods Administration (CT-2018-CTN-03469-1). The study was prospectively registered at the Australian New Zealand Clinical Trials Registry (ACTRN12618000891224). Written informed consent was obtained from 12 healthy volunteers recruited from the hospital staff. The inclusion criteria were male or female, aged 18–65 years old and a body mass index (BMI) of 18–35 kg/m2. Specific exclusion criteria were known allergy to local anaesthetic drugs, injury, deformity or previous surgery to the right arm or PABCN, pain or pre-existing neurological deficit of the right arm, and identifying features on the arm such as tattoos, moles, scars or birthmarks.
All blocks were performed in an operating theatre or the post-anaesthesia care unit. The right arm was used in all volunteers to maintain consistency. Volunteers were supine with the upper arm adducted and internally rotated and the elbow flexed over the anterior chest. Scanning was performed using a 15–6 MHz linear ultrasound probe and a transportable ultrasound machine (X-Porte, FUGIFILM SonoSite, Inc. Bothell, WA, USA). The probe was placed transversely on the upper arm at the level of the spiral groove to provide a short axis view of the radial nerve (RN). At this level the RN has an ovoid flattened profile and distinctive fascicular echotexture due to its close relationship with bone (Figure 1). The RN was then followed distally to identify where it gives rise posteriorly to the PABCN just before penetrating the lateral intermuscular septum (LIMS). The PABCN was then tracked as it traverses the fascial planes between the long head of triceps and brachialis muscles to exit via the hiatus in the deep fascia. Here, the PABCN enters the FFFT and becomes recognisable as an oval hyperechoic structure surrounded by anechoic fatty tissue (Figure 2).
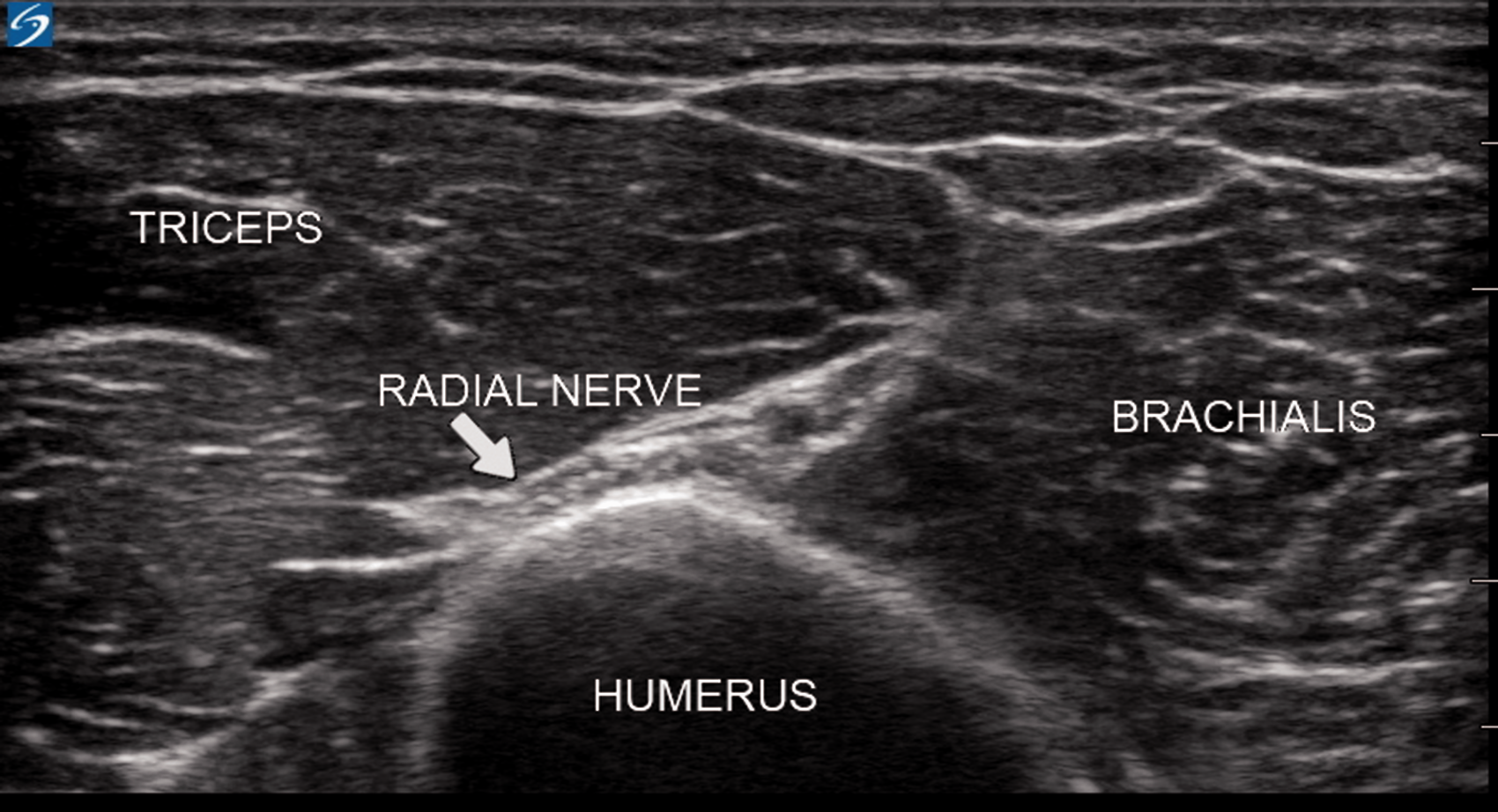
Short axis view of the radial nerve (RN) at the level of the spiral groove. At this level the RN has an ovoid flattened profile and distinctive fascicular echotexture due to its close relationship with bone.
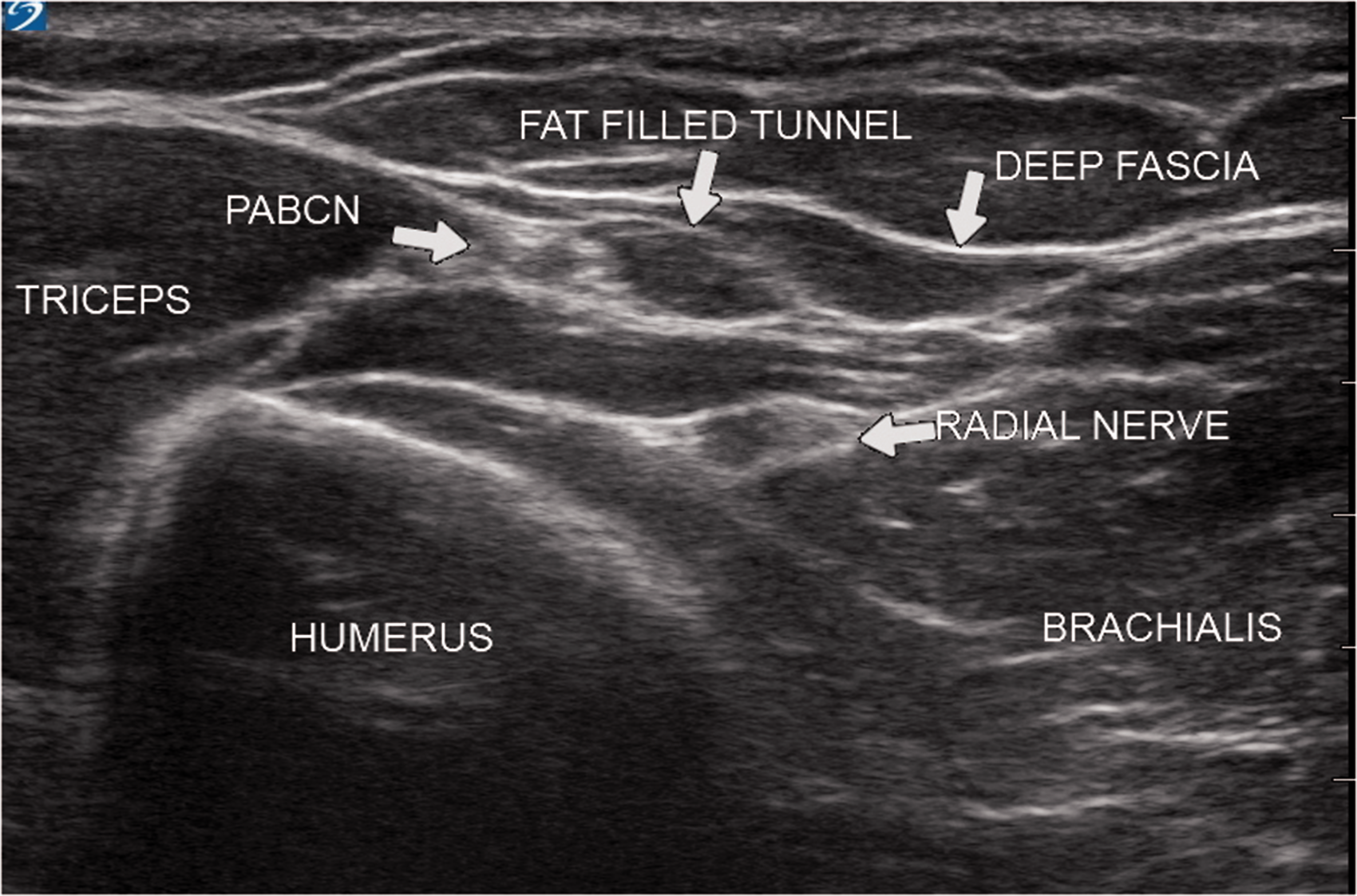
The posterior antebrachial cutaneous nerve (PABCN) seen entering the fat-filled flat tunnel (FFFT) recognisable as an oval hyperechoic structure surrounded by anechoic fatty tissue.
The position of the nerve at this site was marked on the skin and the distance to the lateral epicondyle recorded. A still image of the nerve in the short axis was saved to the hard drive of the ultrasound machine and the circumferential area of the nerve measured using proprietary software (Figure 3). The nerve was then tracked distally for 2–3 cm until its division and the number of branches noted. A 60-second video loop of the whole scan was recorded to permit later review.
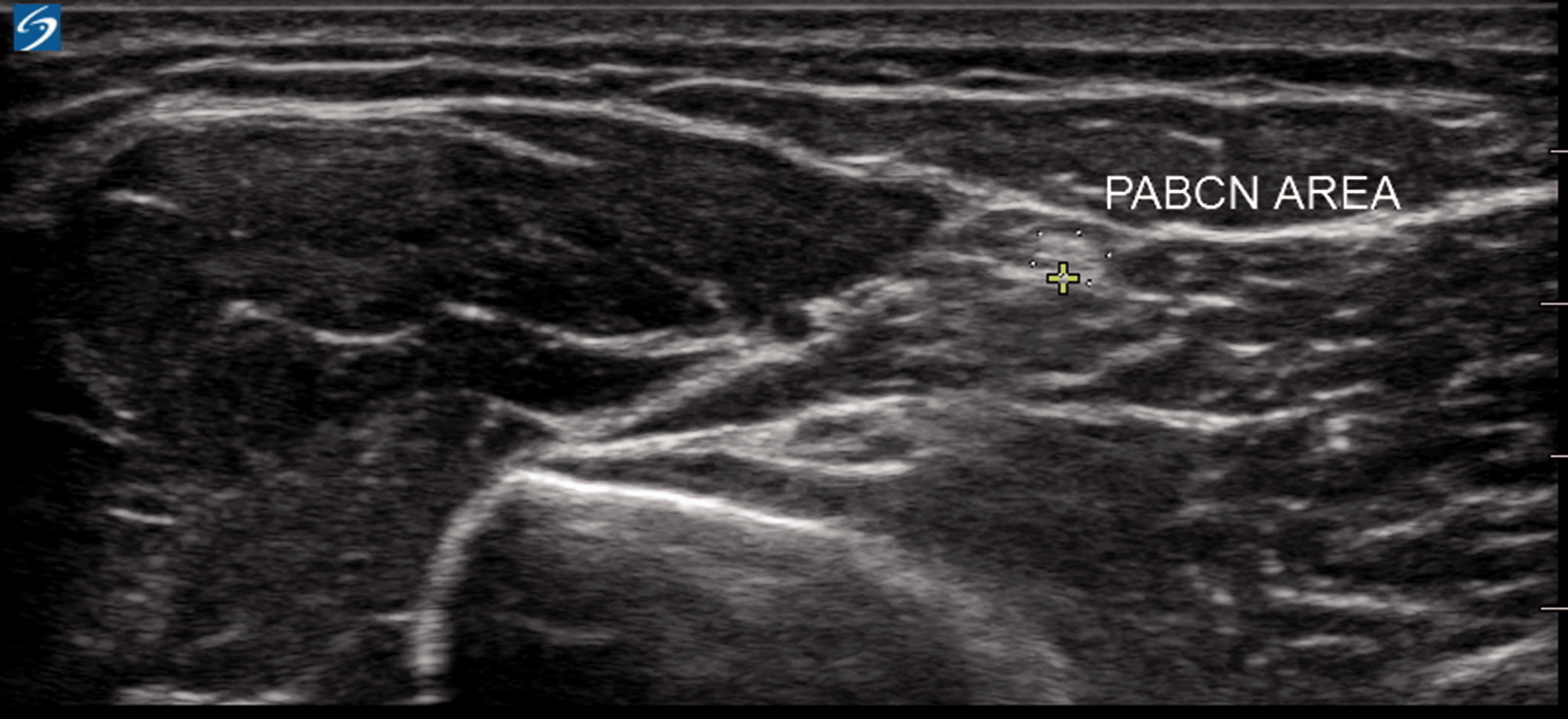
Measurement of the circumferential area of the posterior antebrachial cutaneous nerve (PABCN).
A sterile cover was placed over the probe and sterile gel applied. Following disinfection of the skin an insulin syringe (BD Ultra-Fine, Becton, Dickson and Company, Franklin Lakes, NJ, USA) was used to deliver a subcutaneous injection of 0.5 ml 2% lidocaine prior to the insertion of a 22 G×50 mm echogenic needle (SonoPlex STIM, PAJUNK® GmbH, Geisingen/Germany). The echogenic needle was connected via an extension line to a 3 ml syringe containing 1 ml 2% lidocaine. Using an in-plane technique the needle was then advanced in a lateral to medial direction into the FFFT (Figure 4). Following negative aspiration, an assistant injected 1 ml 2% lidocaine (20 mg) to achieve the circumferential spread of the local anaesthetic around the PABCN.
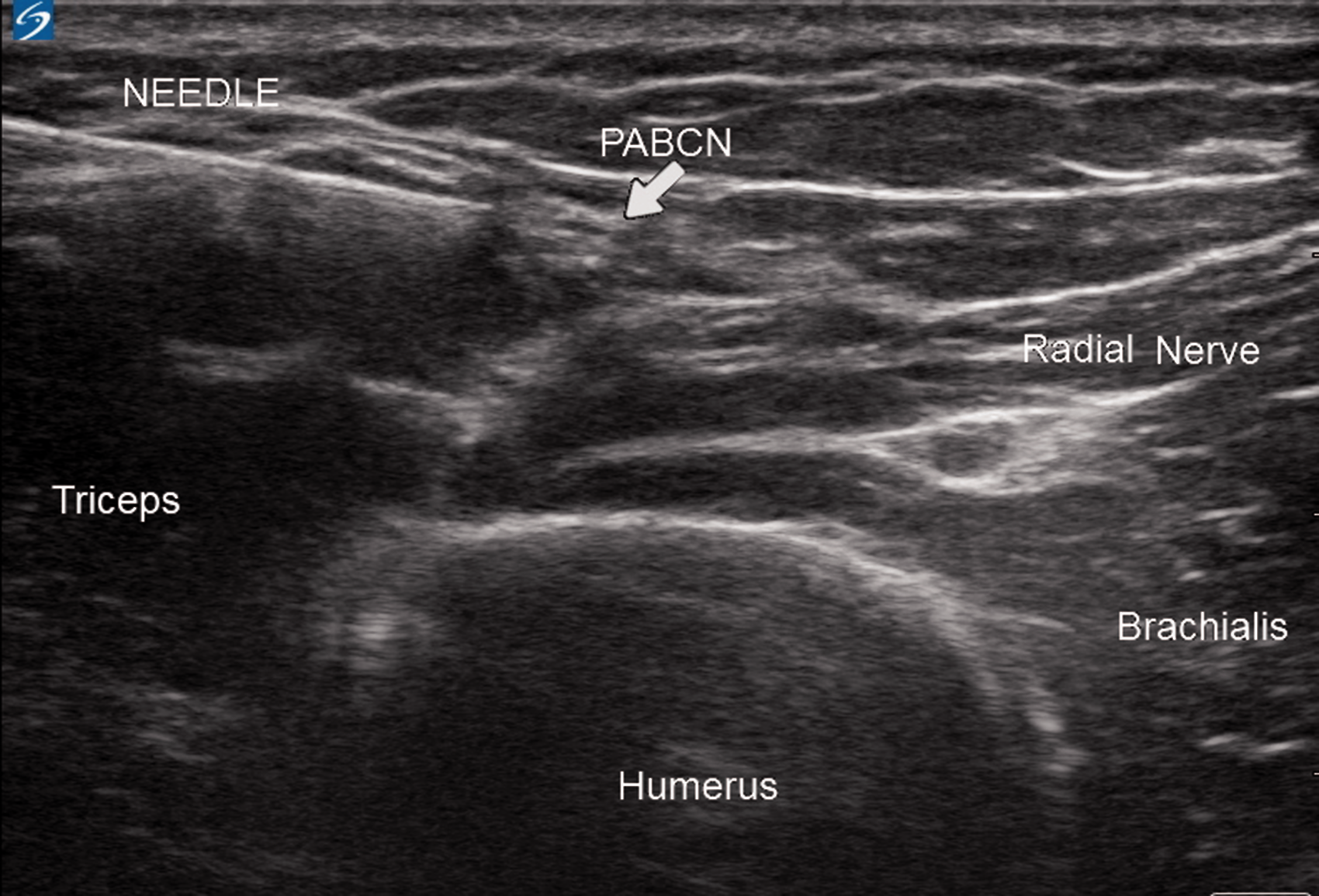
Using an in-plane technique the needle is advanced into the anechoic fat-filled flat tunnel surrounding the posterior antebrachial cutaneous nerve (PABCN).
Sensory loss to sharpness over the upper and lower arm was assessed 15 minutes later using a commercially available tool widely used for testing peripheral neuropathy in diabetic patients (Neuropen with 40 g Neurotips, Owen Mumford Ltd, Oxford, United Kingdom). This tool assesses sharpness or pain in small nerve fibres via a sterile single-use semi-sharp monofilament that exerts a force of 40 g when pressed into the skin.
The unblocked forearm was tested first to ensure the volunteer understood what sensation to expect. Volunteers were asked to state ‘sharp’ or ‘not sharp’ when the monofilament was pressed onto the skin. Testing of the blocked forearm commenced in an area of normal sensation and was repeated in approximately 1 cm incremental movements until loss of sensation (‘not sharp’) was reported. This area was outlined on the skin with a surgical marker pen and copied on to a paper transparency. The total cutaneous sensory block area (CSBA) in centimetres squared was then calculated from photographs of the transparency using SketchAndCalc (© 2008–2018 SketchAndCalc). 8
The anterior surface of the forearm was then defined with a marker pen using the following borders. A line was drawn joining the lateral epicondyle to the radial styloid and another from the medial epicondyle to the ulnar styloid. This delineated the lateral and medial borders. The anterior inter-epicondylar line at the elbow and anterior distal skin crease at the wrist were used to define the proximal and distal borders.
Any sensory block in the anterior surface of the forearm was deemed to have encroached into the territory of the LABCN and MABCN. This area was calculated as a percentage of the total CSBA using SketchAndCalc. 8
Any sensory block on the dorsum of the hand (distal to the wrist skin crease) was deemed to have encroached into the territory of the ulnar or RNs and calculated as a percentage of the CSBA using SketchAndCalc. 8
All volunteers received verbal instructions regarding the need to protect the area of skin from inadvertent injury (hot surface, sharps) until normal sensation had returned (2–4 hours).
All volunteers were reviewed the following day. The skin over the area of local anaesthetic injection was inspected for local signs of infection. Sensation and movement of the forearm was assessed.
Results
In total, 12 volunteers were included in the study, six male and six female. Median (range) age, weight, height and BMI were: 37 (23–62) years, 74.5 (48–88) kg, 163 (152–186) cm and 24.18 (18.07–33.33) kg/m2, respectively.
Ultrasound visualisation of the PABCN was possible in all cases. The median distance of the PABCN to the lateral epicondyle of the elbow was 67.5 (range 54–105) mm. Branching of the nerve distal to the FFFT was seen in all volunteers. There were four branches in two, three branches in four and two branches in six volunteers respectively. The median circumferential area of the PABCN was 0.085 (range 0.03–0.16) cm2.
Sensory loss was detected in all volunteers. The median CSBA was 103 (range 61–341) cm2. Two volunteers had sensory block over the anterolateral forearm of 29 and 10 cm2 respectively. This amounted to 11% and 4.5% of the total CSBA. One volunteer had a sensory block over the dorsum of the hand of 39 cm2 (15% of the total CSBA).
Photographs of the sensory block distribution of all volunteers were evaluated (Figures 5–16). Although the sensory block varied between volunteers the majority had a block that involved the posterior aspect of the distal upper arm and forearm extending to the wrist. Sensory block over the lateral epicondyle was present in three volunteers. Two volunteers had sensory block extending over the anterior forearm and one had sensory block extending over the dorsum of the hand (Figure 10). One volunteer had a sensory block in the upper distal arm but not in the forearm (Figure 14).
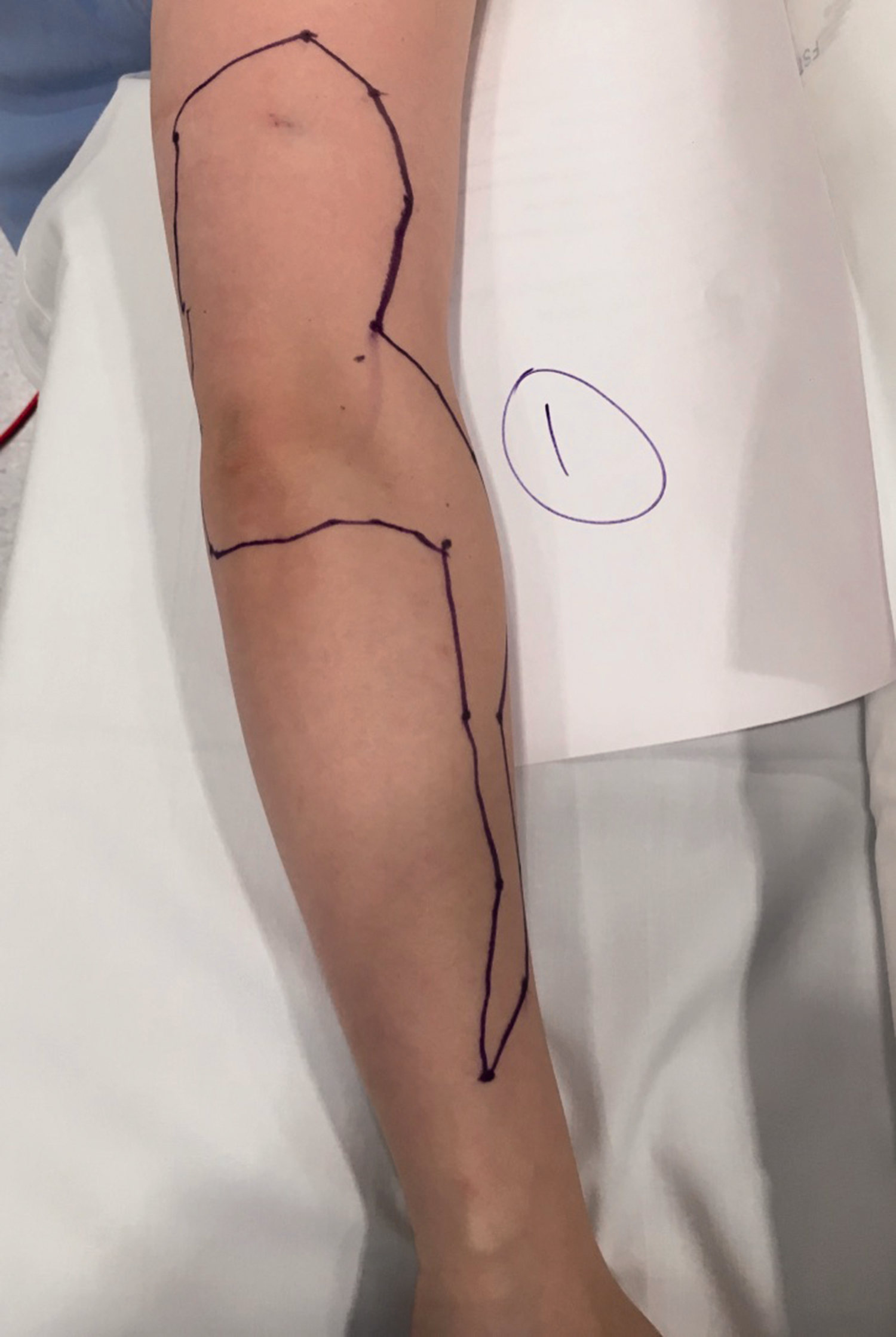
Sensory map of subject 1.
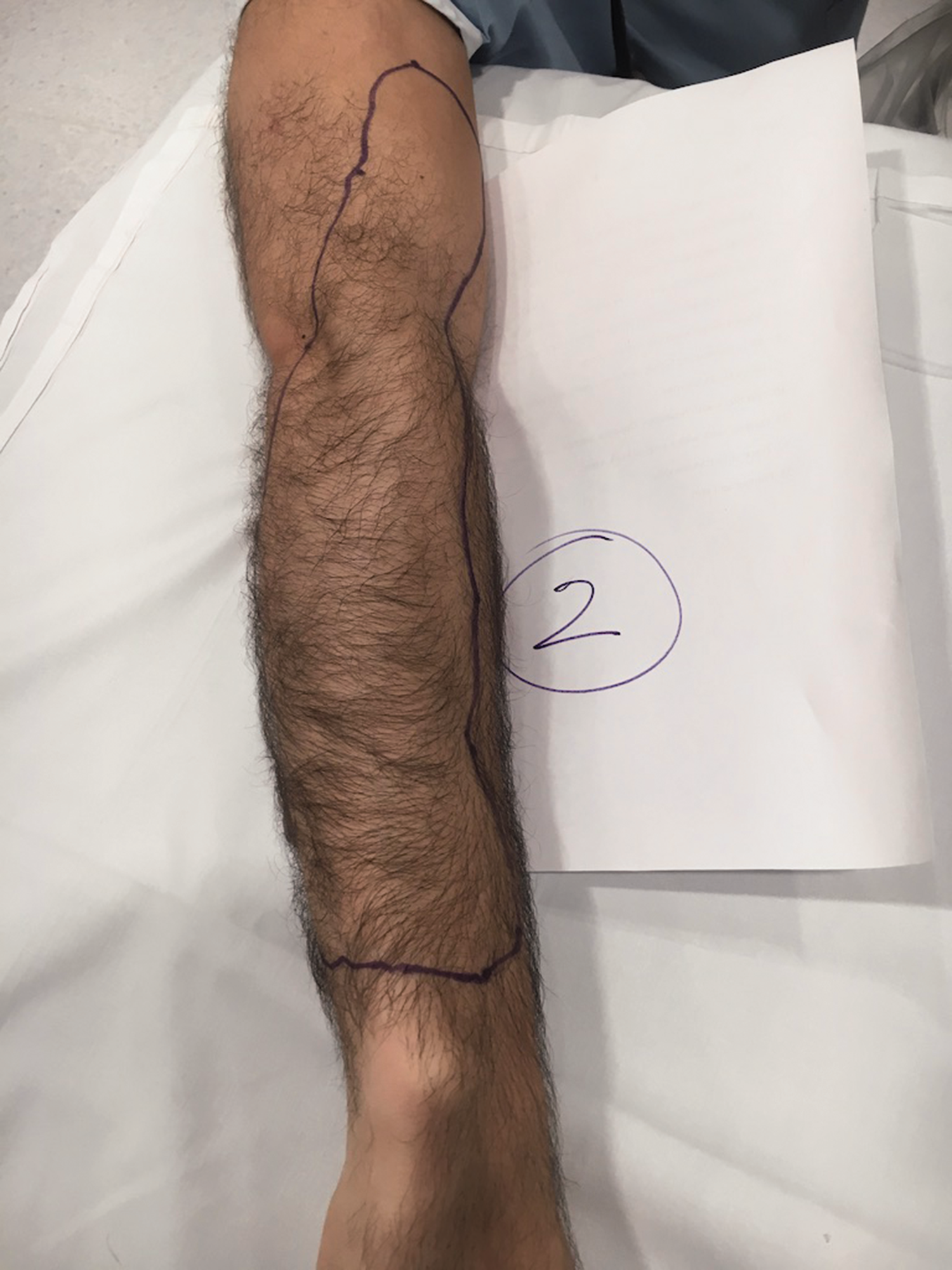
Sensory map of subject 2.
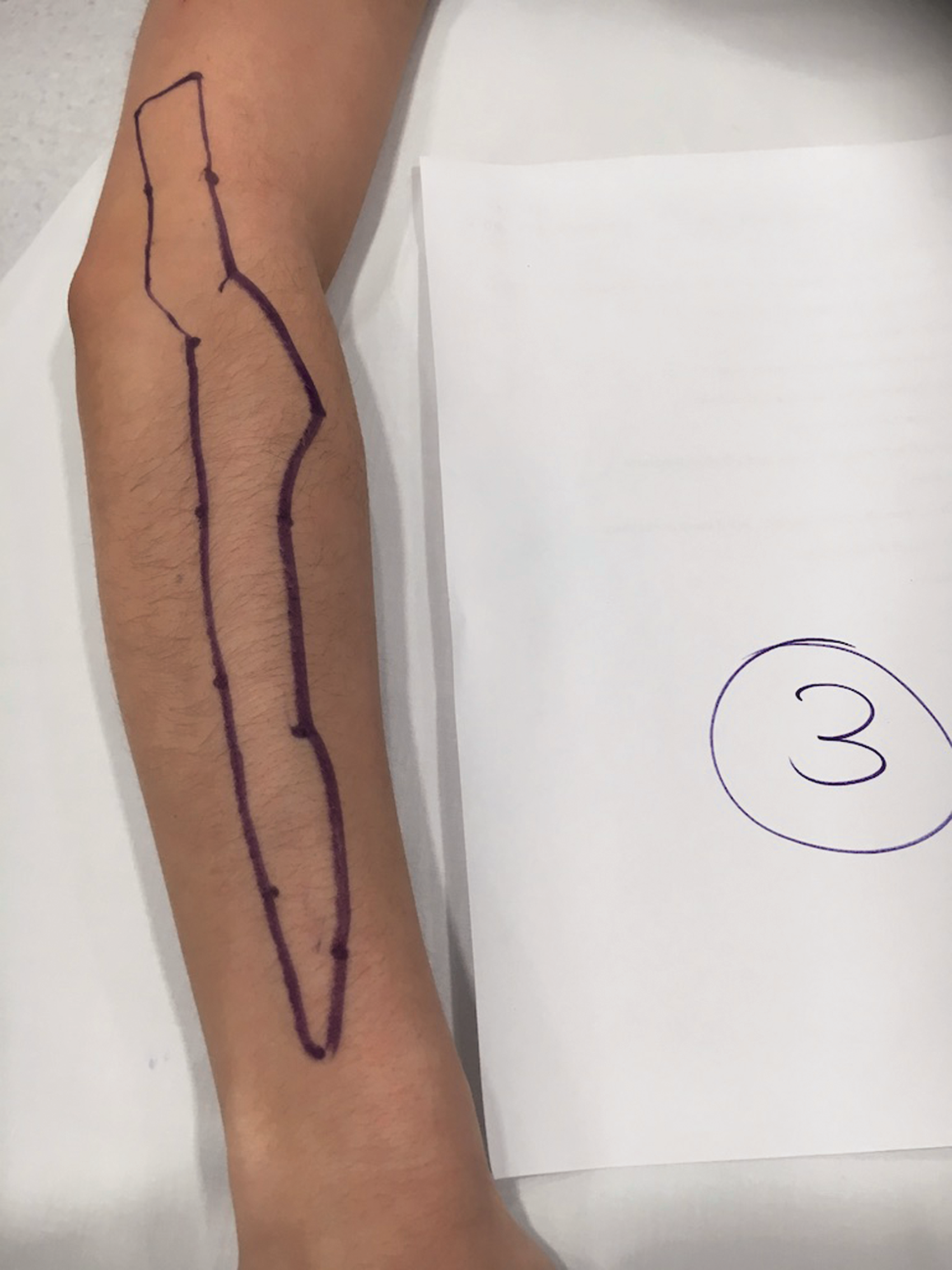
Sensory map of subject 3.
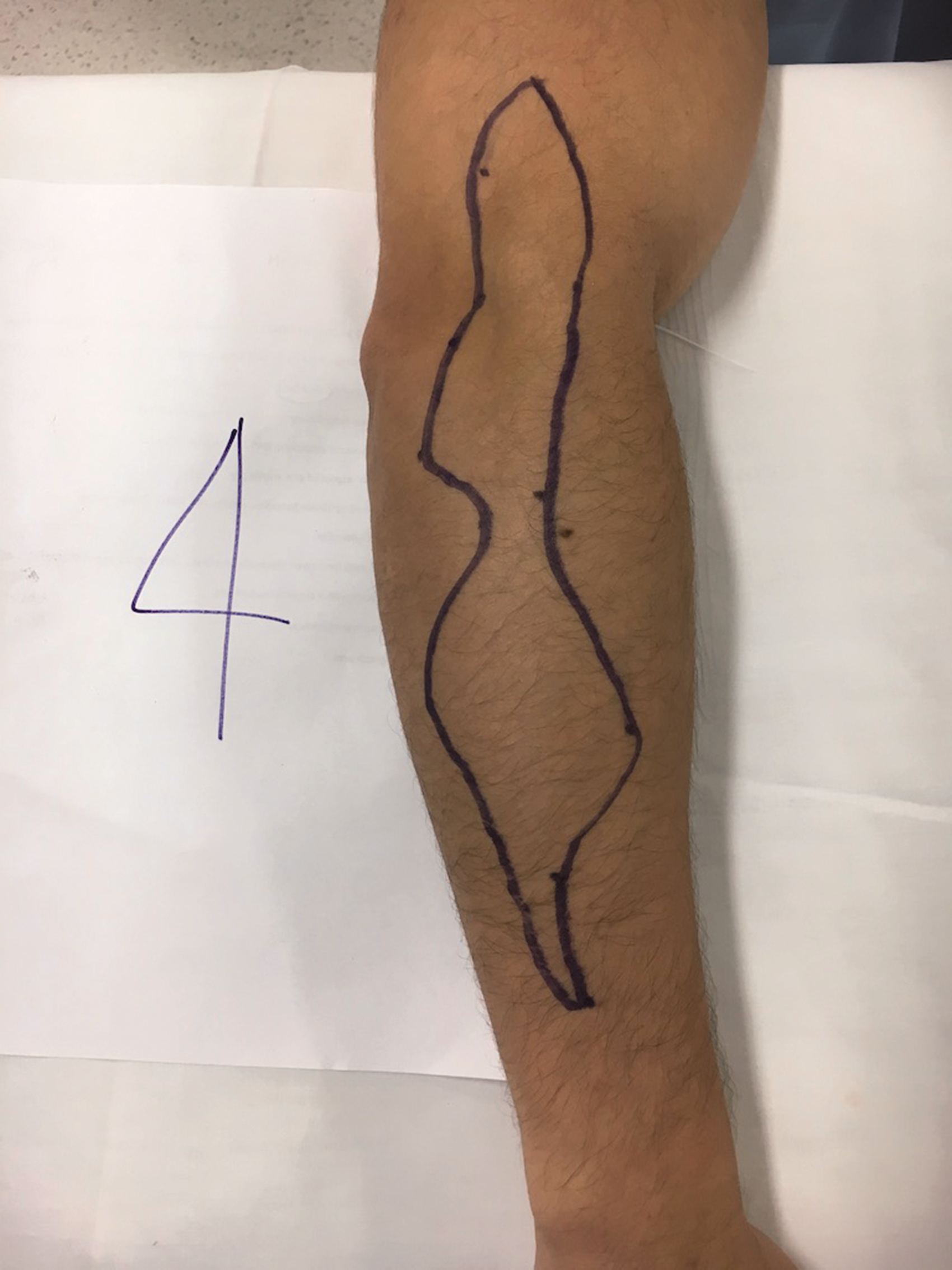
Sensory map of subject 4.
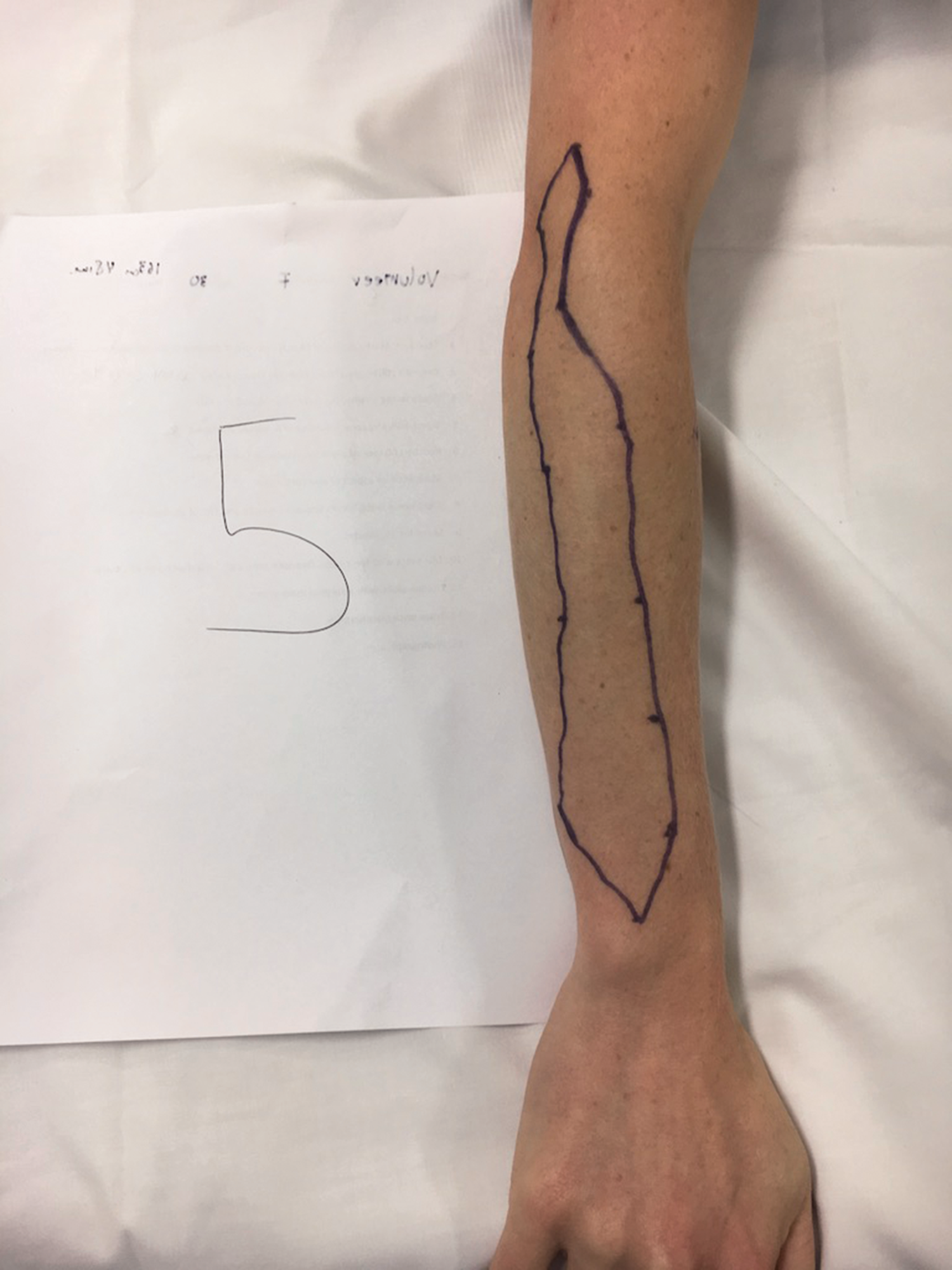
Sensory map of subject 5.
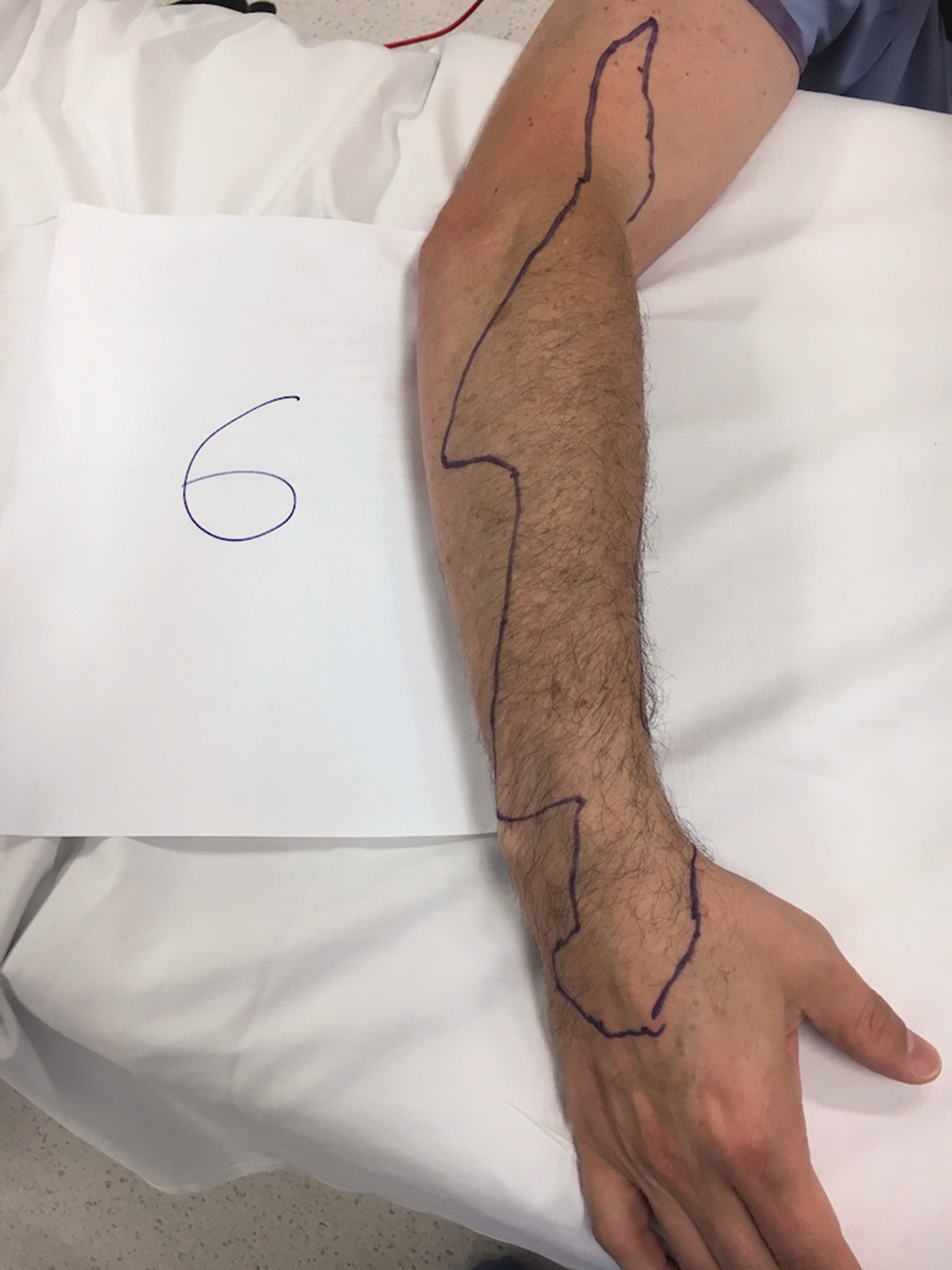
Sensory block of subject 6 extending beyond the wrist to the dorsum of the hand.
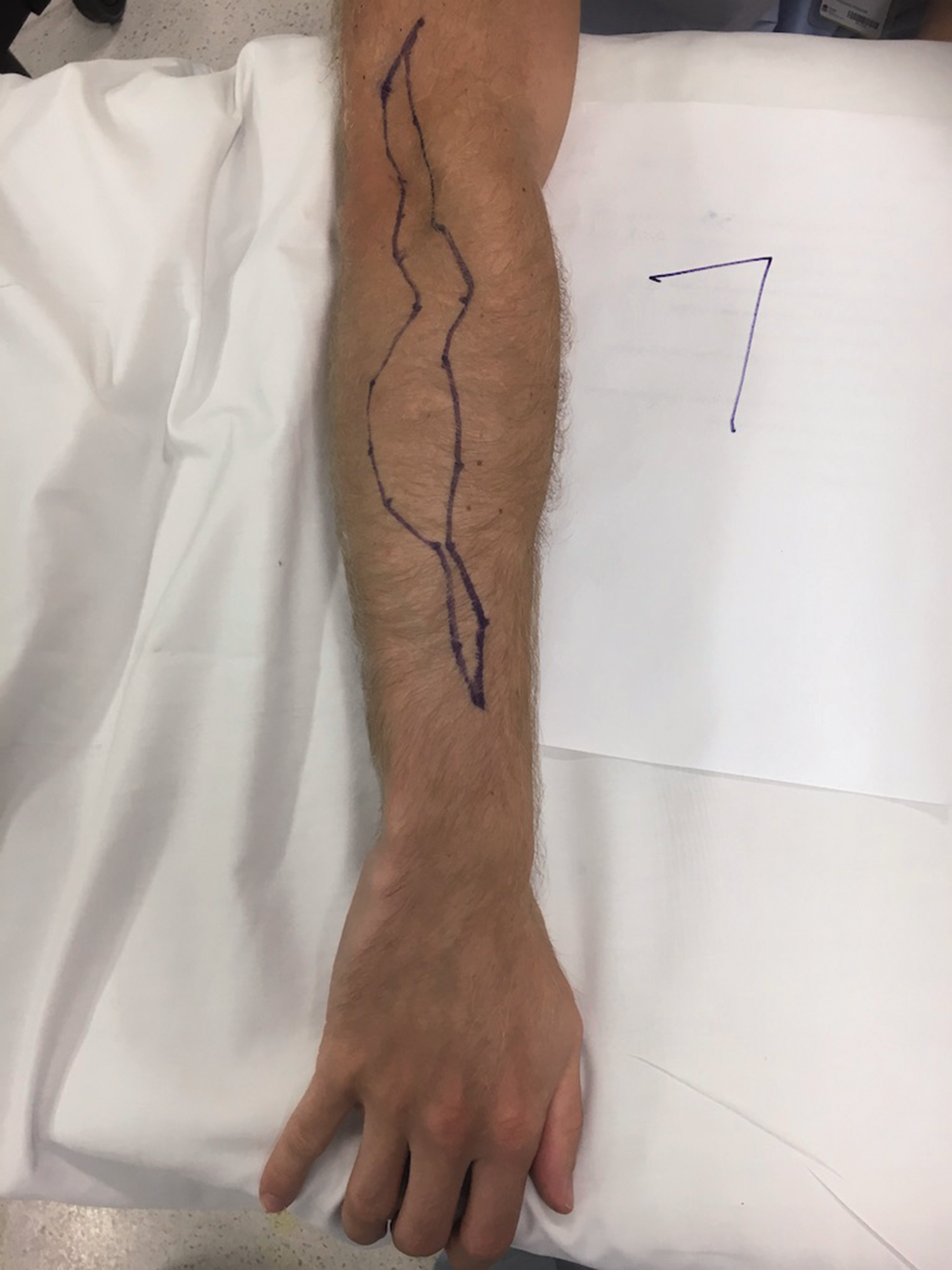
Sensory map of subject 7.
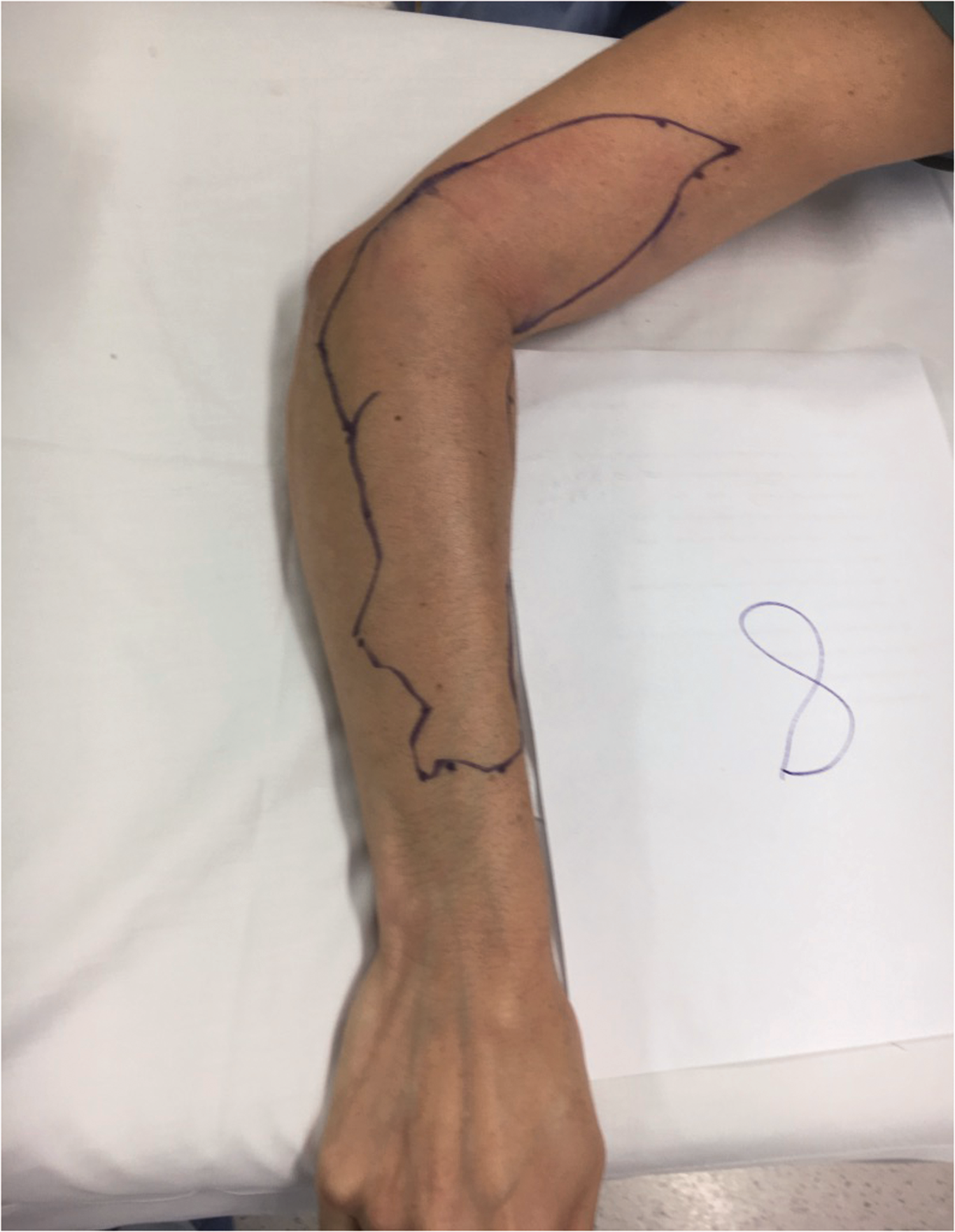
Sensory map of subject 8.
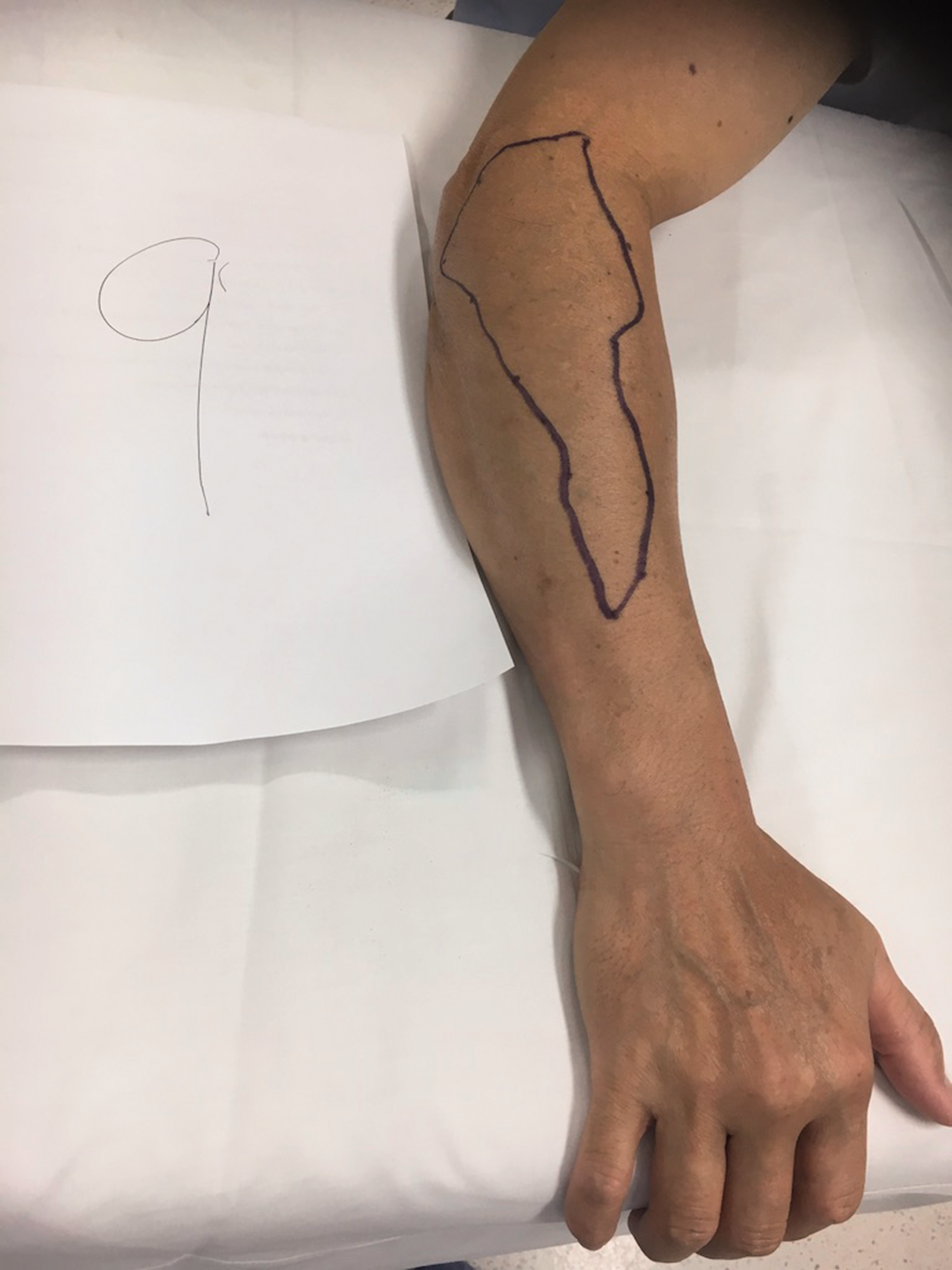
Sensory map of subject 9.
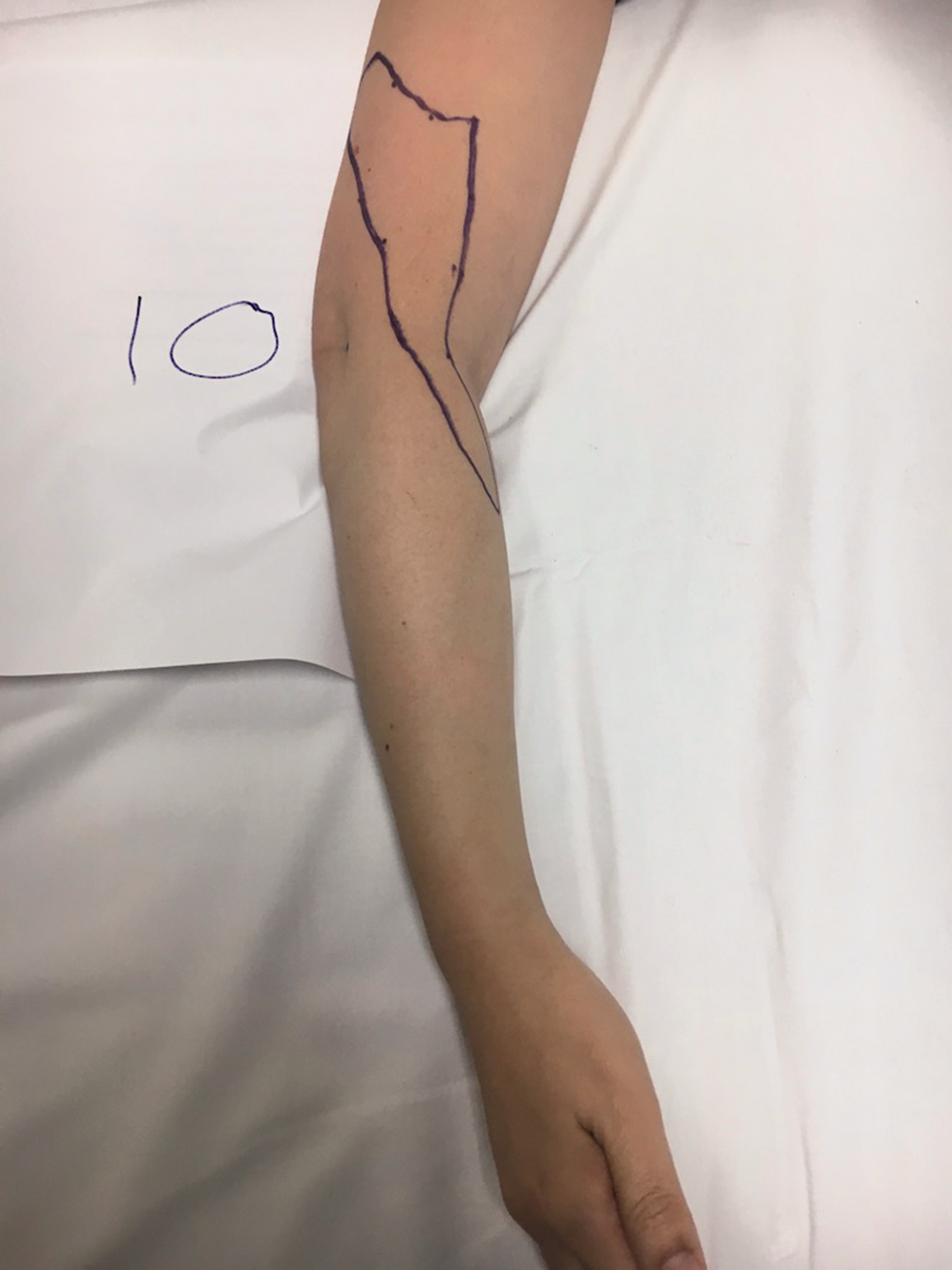
Sensory map of subject 10.
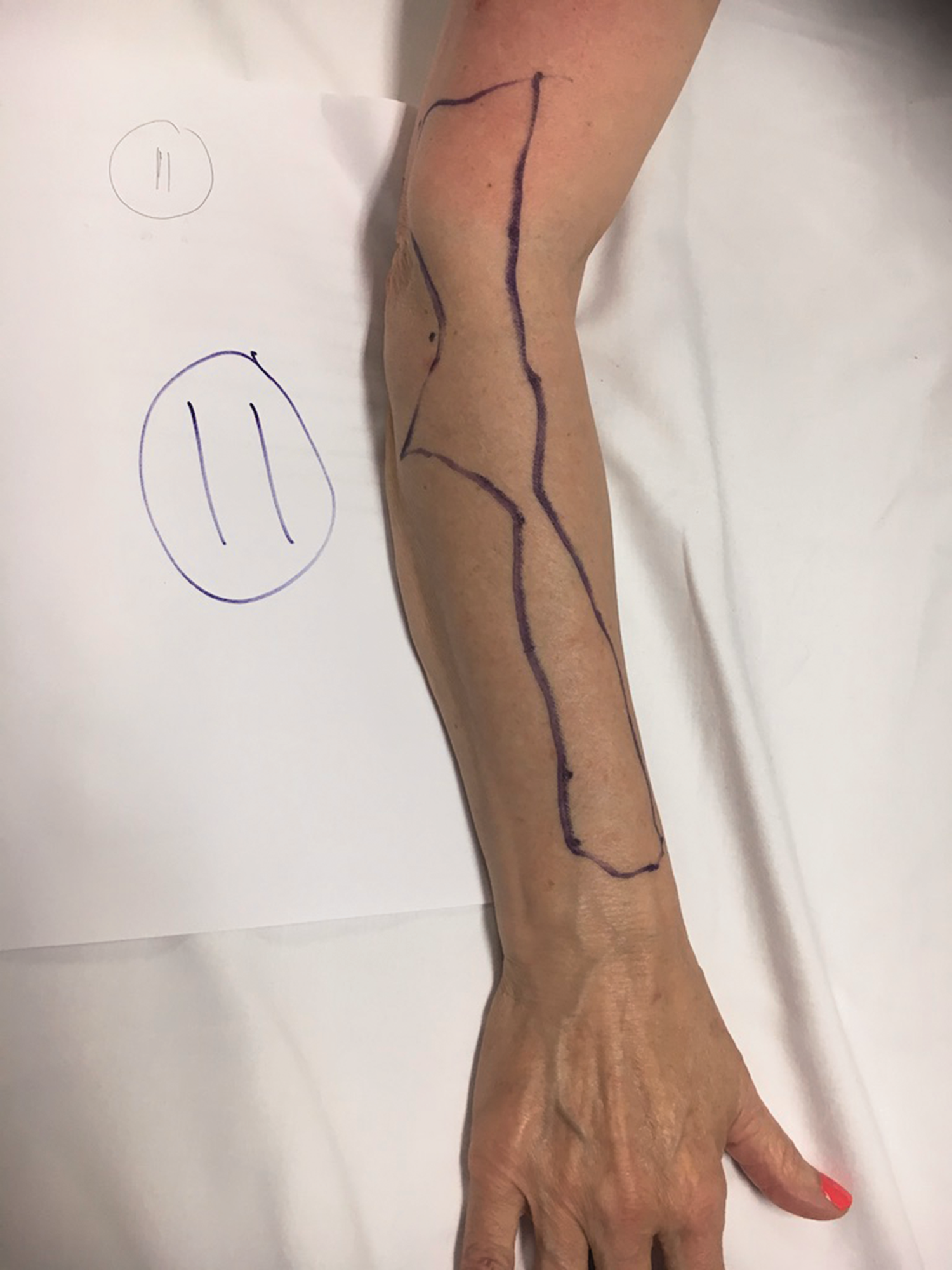
Sensory map of subject 11.
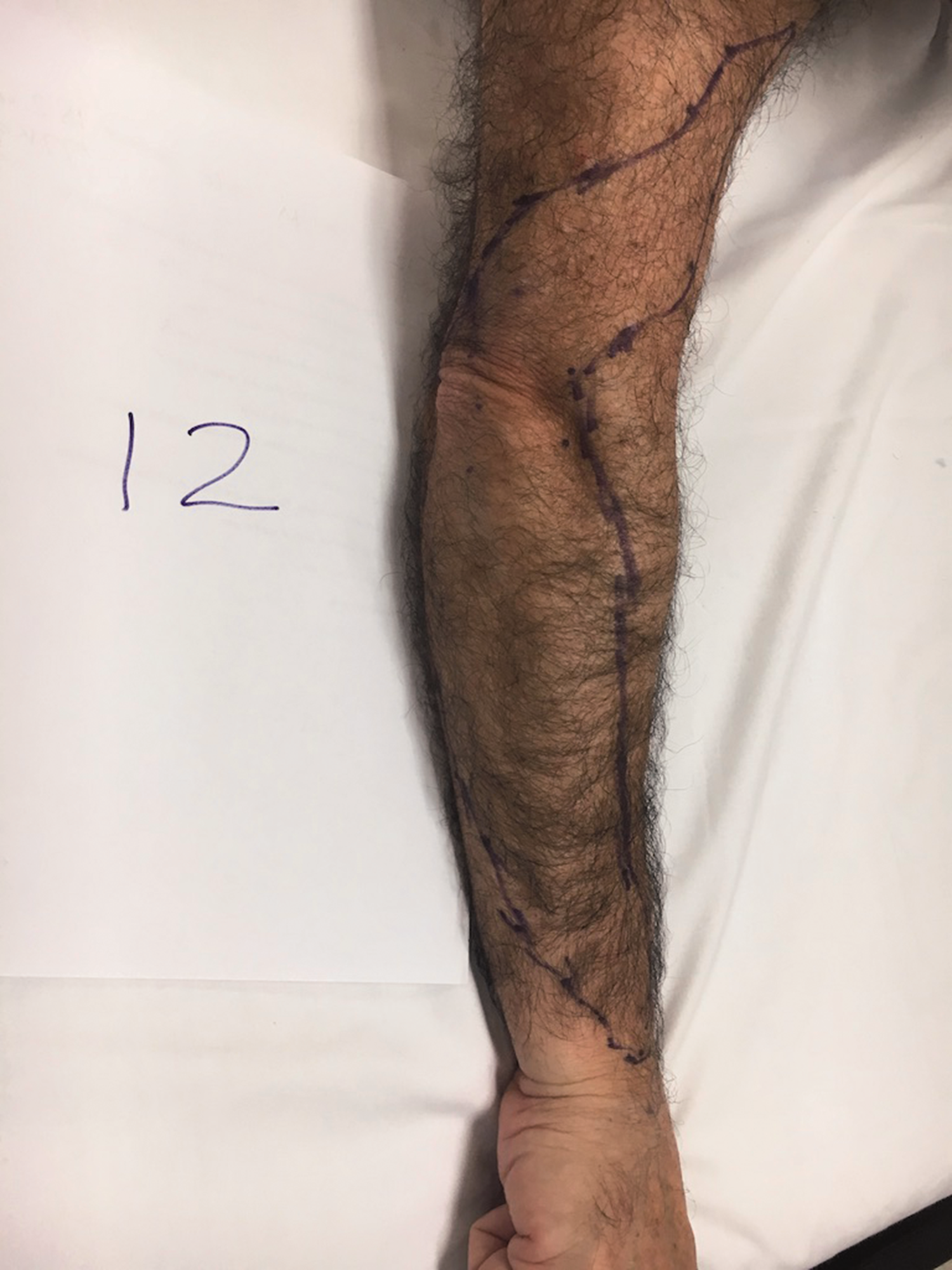
Sensory map of subject 12.
Volunteers were reviewed 24 hours after the performance of the PABCN block. No study-related adverse effects were detected.
Discussion
This volunteer study describes an ultrasound-guided PABCN within the FFFT and the resulting area of cutaneous sensory block. The nerve could be visualised in all of the volunteers as a discrete hyperechoic oval structure within an anechoic FFFT. Sensory block was achieved in all volunteers. In the majority, sensory loss extended from the distal posterior arm and forearm to the wrist. Sensory loss was found in the anterolateral forearm in two volunteers. In one volunteer sensory loss was detected over the dorsum of the hand.
The PABCN arises from the RN within the posterior compartment of the arm. The path of the PABCN follows the spiral groove initially, diverging as the RN pierces the LIMS. The PABCN emerges from the posterior compartment through a hiatus in the deep fascia between the lateral head of triceps and brachialis proximal to the lateral epicondyle. 9 At this location the nerve can be identified with high-frequency ultrasound as a hyperechoic oval structure travelling within the anechoic FFFT (Figure 2).
After entering the FFFT, the PABCN then travels distally posterior to the LIMS and bifurcates at a variable distance into anterior and posterior divisions. The anterior division descends subcutaneously anterior to the lateral epicondyle and then along the dorsum of the forearm to the wrist supplying the skin with sensation in its course and joining near its end with dorsal branches of the LABCN. The posterior division travels subcutaneously to supply sensation to the lateral epicondyle region.
Fascia can be divided into superficial and deep layers. The superficial layer contains an extensive amount of fat that forms distinctive channels known as FFFTs. The FFFTs function as conduits to allow unrestricted movement of nerves and vessels between the skin and underlying tissue. The ceiling of these tunnels is the deep layer of the stratum subcutaneum. The floor is composed of superficial fascia. 10 Both can be visualised with ultrasound as hyperechoic white lines. Fat within the tunnel is anechoic and appears black on ultrasound. Nerves characteristically have a hyperechoic outer structure of connective tissue that surround hypoechoic fascicles. The contrast of hyperechoic/hypoechoic nerves within an anechoic fat tunnel enhances ultrasound discernibility and provides a target for local anaesthetic injection. 11
In this study, the median distance of the PABCN in the FFFT to the lateral epicondyle of the elbow was 67.5 (range, 54–105) mm. This is consistent with two previous studies that found the PABCN emerged from the hiatus at 70 mm and 66 mm from the lateral epicondyle respectively.9,12
Previous authors have reported the ability of ultrasound to identify peripheral nerves.13–15 In the upper limb, high-frequency ultrasound has been used to identify and block the MABCN and LABCN.3–4 Ultrasound identification of the MABCN and LABCN is assisted by their close proximity to the basilic and cephalic veins in the cubital area. Unlike the MABCN and LABCN, the PABCN does not course in close proximity to a venous structure. This study advocates visualising the anechoic FFFT to facilitate identification of the hyperechoic PABCN and may serve as a target for injection.
Two studies have examined the use of ultrasound in the identification of the PABCN in an unembalmed cadaveric model.13,16 Maida et al. were able to sonographically identify the PABCN and several distal branches traversing over or directly adjacent to the lateral epicondyle. 16 Moritz et al. were able to identify the PABCN as it bifurcates in the LIMS. 13 Neither of these studies used the FFFT as a sono-anatomical landmark to identify the PABCN.
Blanco et al. have described the ultrasound appearance of the cutaneous nerves of the upper limb. 5 They located the PABCN in a ‘fat pocket above the long head of the triceps muscle in the posterior side of the arm, distal third’. An accompanying image depicts the PABCN exiting above the biceps muscle. This is anatomically incorrect as the PABCN emerges from the posterior compartment through a hiatus in the deep fascia between the lateral head of triceps and brachialis proximal to the lateral epicondyle. 9 During the preparation of this manuscript, Finneran and Sandhu published an ultrasound-guided block of the PABCN ‘within the muscle belly of the lateral head of the triceps muscle’. 6 Block distribution was tested by cutaneous sensory deficit to ice. The resulting sensory cutaneous block was not described although it is depicted in photographs.
Local anaesthetic block of the PABCN is useful for providing cutaneous anaesthesia for surgical procedures involving the posterior forearm. 7 Examples would be drainage of skin abscess, removal of retained foreign body, burn debridement and skin grafting. The distribution and area of sensory block achieved in this study varied between volunteers. In some, there was only a narrow strip of cutaneous anaesthesia over the posterior forearm. One explanation for this inter-individual variability is the existence of anastomoses between cutaneous sensory nerves. 17 This variability in cutaneous innervation would suggest that multiple sensory nerves may need to be blocked to ensure adequate anaesthesia and prevent block failure. Block of the PABCN alone would not cover tourniquet pain.
The posterior division of the PABCN is said to innervate the skin and subcutaneous tissues over the lateral epicondyle. 9 As such, sensory block of the PABCN may be beneficial in the evaluation of patients with refractory lateral elbow pain syndromes (e.g. tennis elbow).13,18 The present study found sensory loss in the vicinity of the lateral epicondyle was present only in three volunteers, implying innervation over this area may derive from other cutaneous nerves.
A cadaver study by Chodewaratham et al. examined branching of the PABCN. They found three to four branches emerged proximal or distal to the hiatus in half of the specimens. 12 Two specimens had a branch that passed dorsal to the lateral epicondyle. Interestingly, communicating branches between the PABCN and the LABCN were not uncommon. The present ultrasound study identified four branches in two, three branches in four and two branches in six volunteers respectively.
Although the sensory distribution area was predominantly confined to the posterior distal arm and forearm, there was significant inter-individual variation. In one volunteer sensory loss extended beyond the wrist to the dorsum of the hand (Figure 10). Although the hand classically derives its innervation from the median, radial and ulnar nerves, a recent study observed that large areas are not innervated by any of these three nerves. 17 The present study suggests the PABCN can innervate the dorsum of the hand. This may be the result of communicating branches with the LABCN or an anomalous distribution. 19 In two volunteers, sensory loss was detected in the anterolateral aspect of the forearm. This likely represents a shared of area of innervation between the PABCN and the LABCN. In one volunteer sensory loss was identified only in the inferodorsal aspect of the upper arm. This may be due to atypical innervation or failure to block all of the branches of the PABCN.
This study is subject to some limitations. It is a single investigator non-blinded study with potential for bias. The number of volunteers is small and the results may not be generalisable. The BMI of volunteers studied was normal. Therefore, the results may not apply to other patients. Volunteers were able to view the ultrasound display in the course of locating the PABCN in the FFFT and subsequent local anaesthetic injection. This may have produced performance bias. Sensory block was investigated using pinprick sensation. Other investigators have used a cold stimulus to assess sensory loss.4,6 Cold and pain perception differ in regard to sensory receptors, nerve conduction and cortical processing. Sensory testing with cold may have given rise to contradictory findings. Additionally, this study used a relatively small volume of local anaesthetic. Larger volumes may have yielded different results.
In conclusion, this study found that PABCN can be readily visualised by high-frequency ultrasound within a FFFT. The FFFT could serve as a sono-anatomical target for local anaesthetic deposition. Mapping of the CSBA revealed a high degree of inter-individual variability in the innervation of the arm and forearm. Sensory loss can extend to the dorsum of the hand. These findings could help explain unexpected cutaneous nerve block failure despite having been adequately executed.
Contributor statement
PJ Corke was responsible for the design, conduct and reporting of the study and performed all blocks. This study was submitted as the research component for an MSc in Regional Anaesthesia at the University of East Anglia, Norwich, United Kingdom.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
