Abstract
There is no clear consensus in the current guidelines published by major international anaesthetic associations on what is the most appropriate time for a patient to stop chewing gum. This open-label balanced-group randomised controlled trial aimed to evaluate whether the chewing of sugar-free gum caused an increased volume or reduced pH of residual gastric fluid in fasting patients. For this study 212 patients undergoing elective gastroscopy were randomised into a control group who followed routine fasting instructions and an intervention group who were asked to chew gum while fasting. Residual gastric fluid was aspirated under direct vision via a gastroscope under anaesthesia. The primary outcome was the incidence of a gastric residual volume >50 ml in participants who chewed gum compared with a control group. Secondary outcomes were variability in the overall gastric volume distribution and gastric pH distribution between the two groups. Nine out of 110 (8.2%) in the chewing gum group and six out of 102 (5.9%) in the control group had a residual gastric fluid volume >50 ml: incidence rate ratio 1.39 (95% confidence intervals (CI) 0.51–3.77; P = 0.60). However, only one patient (in the control group) had a residual gastric volume >73 ml. There was no statistically significant difference in gastric volume distribution between groups, odds ratio 1.60 (95% CI 0.99–2.58; P = 0.054) or in the distribution of gastric pH measurement, odds ratio 0.90 (95% CI 0.57–1.44; P = 0.67). These results indicate that if there is an increase in the incidence of residual gastric volume >50 ml in patients who chew gum preoperatively, it is likely to be small. Moreover, the absence of any patients in our chewing gum group with a residual gastric volume >73 ml is reassuring.
Keywords
Introduction
Preoperative fasting is a ubiquitous element of patient preparation for any elective procedure requiring anaesthesia. The small residual gastric fluid volumes commonly present after a typical fast (6 h for food, 2 h for clear fluids) 1 are associated with a very low rate of pulmonary aspiration, in the order of 1 in 4000 anaesthetics. 2 However, it is not known if chewing gum while fasting prior to elective surgery poses an increased risk of pulmonary aspiration if anaesthesia proceeds. 3 To compound this problem, there is no standard approach to the use of chewing gum in the fasting guidelines published by anaesthesia professional associations, including the Australian and New Zealand College of Anaesthetists (ANZCA), 4 European Society of Anaesthesiology (ESA), 5 Association of Anaesthetists of Great Britain and Ireland (AAGBI) 6 and American Society of Anesthesiologists (ASA). 7 This creates a situation where anaesthetists must use their own experiential judgement when deciding if a patient who has been chewing gum is adequately fasted for anaesthesia.
Reports from previous trials10–13 using blind suction of gastric fluid via a gastric tube have recorded mixed results, with chewing gum causing no increase in gastric fluid volumes or small statistically significant increases in volumes with doubtful clinical significance. A recent chewing gum study of 67 fasting patients 8 used the more accurate method of suctioning gastric fluid under direct vision. 14 This randomised controlled trial evaluated the effect of chewing gum on residual gastric fluid volume and demonstrated a small statistically significant increase in median gastric volumes (0.345 ml/kg versus 0.112 ml/kg for the control group), 8 equating to a clinically insignificant 19 ml difference for a 70 kg subject.
The residual gastric volume in patients who are chewing gum while otherwise fasting is a balance between increased saliva production and vagally stimulated gastric secretion production versus increased intestinal motility, also under vagal control. 8 , 9 , 15 However, the range of measured gastric fluid volumes with gum chewing in previous trials8–13 indicates there is inter-individual variability in the balance of these mechanisms. Thus, some patients who chew gum may potentially have a higher residual gastric volume than normally considered safe to proceed with elective general anaesthesia.
Historically a gastric volume >0.4 ml/kg (>25 ml) 16 has been used as the standard for increased aspiration risk; however, more recent primate studies 17 , 18 have demonstrated 0.8 ml/kg (50 ml for a 70 kg subject) gastric aspirate was required to induce a syndrome of aspiration pneumonitis. Indeed, some authors have stated volumes above 1.5 ml/kg (approximately 100 ml in a 70 kg patient) are not uncommon and therefore clinically acceptable. 2 To further complicate the issue, aspiration risk is influenced by individual patient factors such as intragastric pressure, gastric sphincter function and patient positioning under anaesthesia, which affects the fraction of gastric fluid that may be regurgitated and subsequently aspirated into the lungs. 2 ,19–22
In the absence of a widely agreed definition for the residual gastric volume predisposing to an unacceptable risk of pulmonary aspiration, we reviewed the control group data of previous chewing gum trials 8 ,10–13 and identified a consistently low incidence of subjects measuring gastric volumes >50 ml in the order of approximately 2%. Therefore, for this study a gastric volume of 50 ml was chosen as a volume for which we could reasonably expect a low incidence in fasting patients. This study aimed to compare the incidence of fasted patients who chewed sugar-free gum having a residual gastric fluid volume >50 ml, versus control patients who did not. In addition, we aimed to measure and compare the overall gastric volume distribution of the two groups and also compare the relative pH values of this gastric fluid.
Methods
The study was an investigator-initiated, open-label, randomised controlled trial using parallel groups with a 1:1 allocation ratio. Ethics approval was provided by the Tasmanian Human Research Ethics Committee (HREC ref no H0016137) and the Calvary Health Care Tasmania Clinical and Research Ethics Committee (CREC project no 011216). The trial was registered with the Australian New Zealand Clinical Trials Registry (ACTRN12617000032358).
Recruitment spanned the period from February 2017 to August 2018. Included subjects were adult patients aged 18 years and over who were admitted for elective gastroscopy at a single day-stay endoscopy unit. All patients booked for a gastroscopy were emailed a participant information sheet and then telephoned a few days later by a research nurse for the purposes of eligibility screening. Exclusion criteria were a past history of upper gastrointestinal surgery, past history of gastrointestinal disease with disordered swallowing or gastroparesis, use of drugs known to decrease gastric motility, known allergy or intolerance to chewing gum, and patients receiving bowel preparation for a colonoscopy on the same day. Computer randomisation of study identification (ID) numbers for both groups was followed by the placement of printed ID number slips into numerically ordered envelopes matching the computer randomisation file. After gaining informed consent to participate, the research nurse opened the next envelope in the numerical sequence to allocate the patient to their group and give instructions as per the study protocol.
All patients were booked onto a regular endoscopy list and given their general fasting instructions by the gastroenterologists’ secretarial staff as part of their endoscopy admission paperwork. The control group was instructed to follow a standard minimum 6 h fast for solids and 2 h fast for clear fluids. The chewing gum group were advised to follow the same fasting guidelines but were also instructed, at the time of randomisation by the research nurse, to start chewing gum any time after ceasing oral intake of solids on the day of their gastroscopy. These patients were provided with a packet of chewing gum (Wrigley’s Extra® Spearmint sugar-free chewing gum, The Wrigley Company, Asquith, New South Wales, Australia). On the day of the gastroscopy, compliance with gum chewing was checked by the endoscopy unit nursing staff at the time of admission, and all patients in the intervention group were instructed to chew gum from this time until just before their procedure began. Prior to entering the endoscopy procedure room, nursing or anaesthesia staff would request any gum, if present, to be expectorated. Only the gastroenterologist taking the sample was blinded to the patients’ group allocation. Sedation was induced in the left lateral decubitus position and the gastroenterologist passed the gastroscope into the stomach, immediately suctioning all gastric fluid visualised into a suction trap to be analysed. There were no sprays, gargles or irrigation allowed before the stomach had been emptied. The volume and pH of the collected gastric fluid was measured and recorded (pH measured with Testo 206-pH1 instrument, Testo SE & Co. KGaA, Lenzkirch, Germany). Research staff also made a record of demographic data, use of proton pump inhibitors (PPIs) or H2 receptor antagonists (H2RAs), any swallowed chewing gum and any adverse events during the gastroscopy.
The expected proportion of patients with a volume of greater than 50 ml in the control group was estimated to be 2%. The primary outcome measure was to detect an increase of 10% (i.e. an overall rate of 12%) in the proportion of patients in the chewing gum group who measured a residual gastric volume greater than 50 ml compared with the control group. The study’s secondary outcomes included any difference between the groups’ residual gastric volume distribution, and any difference in the two groups’ pH measurement distributions. A sample size of 102 patients per treatment group was calculated to be sufficient to detect the primary outcome of a 10% absolute difference in the proportion of patients with an aspirated gastric fluid volume over 50 ml (alpha 0.05 and power 80%). Statistical analyses were performed on an intention-to-treat basis, with additional per-protocol reporting of the results. Initial patient characteristics were compared by using ordered logistic regression for continuous variables and Fisher’s exact test for binary variables. Primary and secondary outcome comparison between treatment groups was performed by estimation of incidence rate ratios (IRR; 95% confidence intervals (CI); P-values) using general linear negative binomial modelling. The distributions of gastric volumes and pH levels were compared using ordered logistic regression, a rank-ordered equivalent of analysis of variance (ANOVA) type multivariate regression. All analyses were performed using Stata MP2 version 14.2 (StataCorp, College Station, TX, USA).
Results
A total of 925 patients were assessed for inclusion into the study, of whom 237 were recruited. Twenty-five subjects were withdrawn after randomisation; the reasons for these withdrawals are detailed in Figure 1. Patients’ demographic and clinical characteristics are detailed in Table 1. All patients in the intervention group confirmed they chewed gum while fasting from clear fluids, with 64% of patients chewing “some of the time”, 18% chewing “most of the time” and 18% chewing “all of the time”.
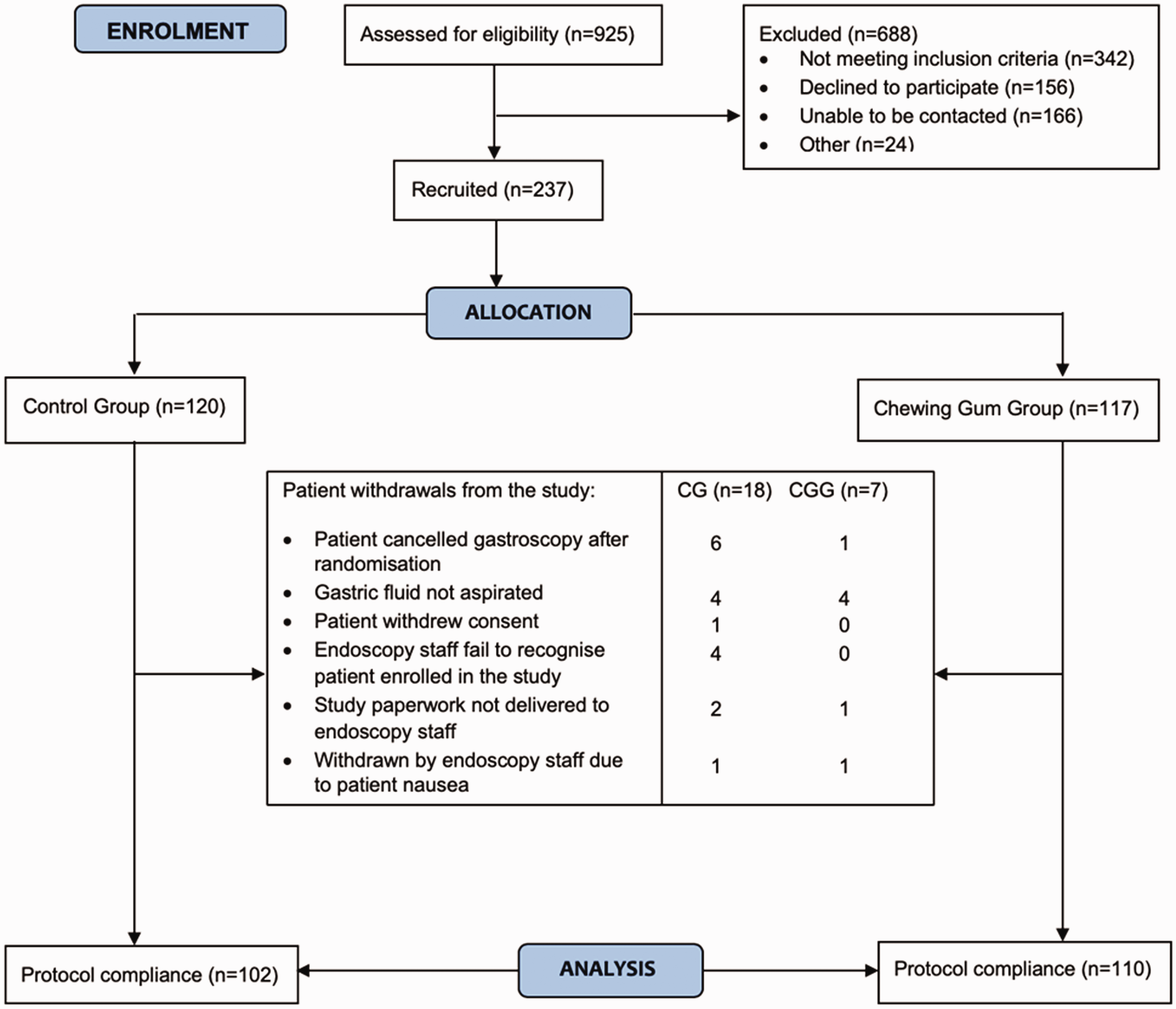
Study participant flow diagram.CG: control group; CGG: chewing gum group.
Demographic and clinical characteristics.
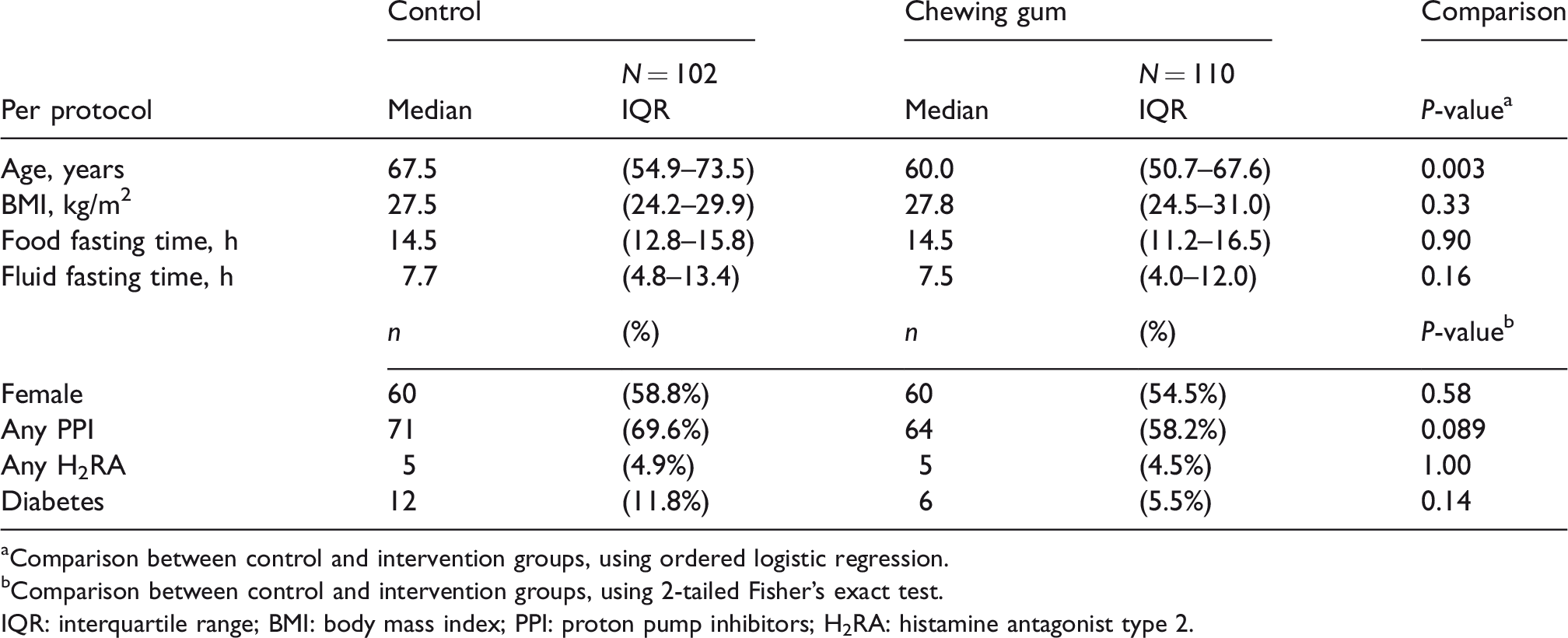
aComparison between control and intervention groups, using ordered logistic regression.
bComparison between control and intervention groups, using 2-tailed Fisher’s exact test.
IQR: interquartile range; BMI: body mass index; PPI: proton pump inhibitors; H2RA: histamine antagonist type 2.
Of the 212 patients who had their gastric fluid aspirated, 15 patients had a residual gastric fluid volume of greater than 50 ml, six out of 102 (5.9%) in the control group and nine out of 110 (8.2%) in the chewing gum group with an IRR of 1.39 (95% CI 0.51–3.77; P = 0.60). Only one patient (in the control group) had a volume in excess of 100 ml (140 ml). The maximum volume in the chewing gum group was 72 ml, and overall for the 212 patients, 99.5% of patients had a residual gastric volume <73 ml.
Results for the secondary outcomes are outlined as follows. For the gastric volume distributions, the control group had a median (interquartile range (IQR)) volume of 8 (4–19) ml compared with 12 (5–27) ml for the chewing gum group. Figure 2 provides a graphical representation of the relative proportions of patients in both groups with their measured gastric volumes across an arbitrarily selected range of 10 ml increments. This graph is markedly skewed in favour of lower values with a far greater number of patients in both groups in the volume ranges of 0–10 ml and 11–20 ml, and decreasing proportions of patients measuring volumes in the higher range values. The comparison of these gastric volume distributions showed an odds ratio (OR) 1.60 (95% CI 0.99–2.58; P = 0.054) in favour of a higher volume in the chewing gum group. With regard to the pH measurements, the pH for the control group was a median (IQR) of 3.0 (1.8–6.2) compared with 2.5 (2.1–4.5) for the chewing gum group, with an OR 0.90 (95% CI 0.57–1.44; P = 0.67).
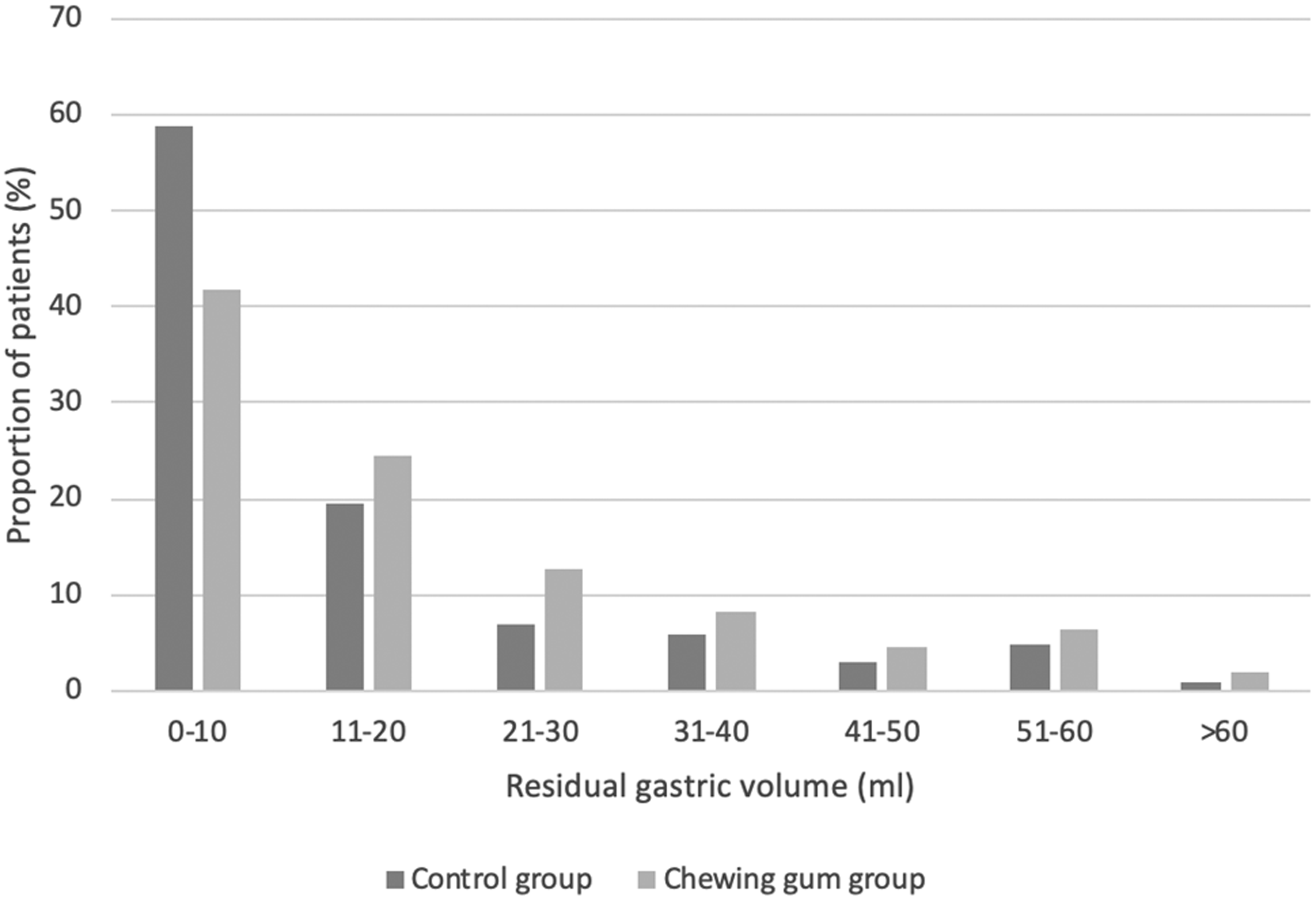
A comparison of the relative proportions of patients with gastric volumes at increasing 10 ml increments.
No patient suffered a complication or came to harm during the study. However, one patient was noted to have gum in their mouth on induction of anaesthesia.
Discussion
Unlike the established preoperative fasting guideline for adults (6 h for solids and 2 h for clear fluids), 1 there is currently no consensus in fasting guidelines when gum is chewed while fasting.19–23 ANZCA states that gum must be discarded due to its risk as a foreign body, rather than for reasons of increased gastric content. 4 The ESA advocates that patients should not have their operation cancelled or delayed because of gum chewing. 5 The AAGBI advocates a 2 h fast after chewing gum, 6 while the Royal College of Nursing in the United Kingdom states that chewing gum should not be permitted on the day of surgery. 23 In North America, the ASA fails to mention chewing gum in their most recent position statement of preoperative fasting. 7 This lack of general consensus in the fasting guidelines forces individual anaesthetists to rely solely on their own clinical judgement in assessing the aspiration risk of a patient who has been chewing gum pre-operatively. Some anaesthetists may draw the conclusion that a patient who has been chewing gum is inadequately fasted for sedation or general anaesthesia, and subsequently delay or cancel that patient who is otherwise fully prepared for their procedure. 2 , 3 The rationale for conducting this study was to provide evidence to practising anaesthetists to improve their ability to make clinical decisions about patient safety when faced with this problem.
Our study detected an overall low incidence (7.1%) of fasting patients having a residual gastric volume >50 ml. All patients who withdrew from the study did so before a gastric fluid sample was collected, so only those patients who completed the study protocol have been included in the statistical analysis. On comparing the two groups in the study, there was a slightly higher incidence of gastric volume >50 ml in the chewing gum group (8.2%) compared with the control group (5.9%). This difference did not reach statistical significance; IRR 1.39 (95% CI 0.51--3.77; P = 0.60). This analysis was based on our study design to detect a 10% difference between the two groups, but given our sample size, alpha 0.05 and beta 0.20, we cannot exclude the possibility of a true difference of <10% in the incidence of gastric volume >50 ml. Detection of such a difference would require a study with a greater number of participants and an adjustment to the expected control group rate to 6% instead of the underestimated rate of 2% that we anticipated in our study design. The possibility of an increased gastric volume with chewing gum may be inferred from the comparison of the relative gastric volume distributions. Graphically there is an appearance of slightly higher volumes in the chewing gum group in Figure 2. The statistical comparison of the gastric volume distributions was calculated as OR 1.60 (95% CI 0.99–2.58; P = 0.054). This indicates higher volumes in the chewing gum group but narrowly fails to achieve statistical significance, with the lower 95% CI almost reaching 1.
Overall, the results of this study provide encouraging information on fasting gastric volumes, whether or not patients chew gum preoperatively. Importantly, 99.5% of all patients had a residual gastric volume less than 73 ml, a volume comfortably below van Putte’s 2 threshold for residual gastric volume of 1.5 ml/kg. Both groups of patients had a >90% incidence of low gastric fluid volume (<50 ml). Moreover, the effect of chewing gum on the incidence of higher residual gastric volumes was small and potentially clinically irrelevant, particularly as no patients who chewed gum had a residual gastric volume >73 ml.
There are some limitations to the generalisability of our results. The patients were all undergoing an identical procedure (elective gastroscopy) and though the fasting guidelines were the same for most other procedures requiring anaesthesia, specific patient or surgical factors may mean our results are not applicable in some clinical scenarios. Age was the only demographic factor where we detected a statistically significant difference between the groups with control median (IQR) age 67.5 (54.9–73.5) versus 60.0 (50.7–67.6) years for the chewing gum group, though the clinical significance of this difference is doubtful. However, our patients were mostly middle-aged or elderly, and as such, our results may not be applicable to the younger adult subset and are definitely not applicable to children, as patients aged less than 18 years were excluded from the study. Despite randomisation there were twice as many patients with diabetes in the control group: 12 (11.8%) versus 6 (5.5%), which was not statistically significant (P = 0.14). Notably, no patient with diabetes had a gastric volume >50 ml, indicating this imbalance may have been unlikely to affect the results for the primary outcome. Patients with gastroparesis were excluded from this study, so it is possible that patients with diabetic gastroparesis may have been correctly identified by our research staff. Given the increasing prevalence of diabetes globally, 24 and the variable effects of abnormal blood glucose levels on gastric emptying, 25 diabetes as a potential confounding factor would warrant consideration in any further study using gastric volume measurement.
With regard to gastric pH, the median measurements were in the relatively acidic range for both groups, with the interquartile range of pH measurements showing a broader range in the control group 3.0 (1.8–6.2) versus a slightly more acidic pH range of 2.5 (2.1–4.5) for the chewing gum group. When these pH distributions were compared there was no statistically significant difference, with OR 0.90 (95%CI 0.57–1.44; P = 0.67). However, this analysis has marked limitations. First, pH is a logarithmic scale, and as such there is a difference in the hydrogen ion concentration [H+] of two orders of magnitude between the 75th quartile measurements of pH 6.2 (controls) versus 4.5 (chewing gum). This raises the possibility that if [H+] had been used rather than pH for the secondary outcome, a higher OR may have been calculated. Second, a majority of patients in the study were taking regular gastric acid production -- modifying medication (PPIs and H2RAs) as noted in Table 1, with a higher number of patients in the control group taking PPIs (69.6%) than in the chewing gum group (58.2%). Though this difference was not statistically significant at the 5% level (P = 0.089), it does raise the question of PPI use being a potential confounding factor in the analysis of our pH results. These issues could be addressed in future studies by controlling for gastric acid--modifying drugs in the study design, and the measurement of [H+] values, which has a ratio scale, rather than measuring pH.
In addition, there were long fasting times endured by some patients. The median fasting times for solids (14.5 h, both groups) and clear fluids (7.7 h, controls and 7.5 h, chewing gum group) were substantially longer than the minimum fasting guideline of 6 h for solids and 2 h for clear fluids 1 set out in our study protocol. These prolonged times were most likely due to a large proportion of patients enduring overnight abstinence from food and fluid before a morning procedure. However, previous trials comparing prolonged overnight fasting with 2–3 h fasting from fluids 1 have shown no significant changes in gastric volumes caused by longer fasting times. We also acknowledge the longer fasting times were not ideal, but it is noteworthy that long fasting times do still occur elsewhere in the elective surgical population, 26 , 27 thus not ruling out the external validity of our results.
In conclusion, in this randomised controlled trial there was no statistically significant difference in residual gastric volumes >50 ml between adult patients who chewed gum preoperatively and those who did not. However, due to power and sample size concerns we cannot exclude a small difference. Nevertheless, none of the patients who chewed gum had a residual gastric volume >73 ml. These findings suggest that pre-operative gum chewing poses a low risk of residual gastric volumes >50 ml in adult patients scheduled for gastroscopy.
Footnotes
Data sharing statement
De-identified individual participant data that underly the results reported in this study are securely stored at the Clifford Craig Foundation, Launceston, Australia (as per HREC requirements). This data will be available to researchers providing a methodologically sound proposal to the authors, which can be directed to
Acknowledgements
For their assistance during the study we thank Dr M Veldhuis (Launceston General Hospital), J Niekamp, E Vardon, M Davey, H Melville and S Hutchins (Clifford Craig Foundation), the anaesthetists, nursing, and administrative staff of the endoscopy unit at Calvary Health Care Tasmania (St Vincent’s campus), and the clerical staff of the three gastroenterologists. Thanks to Dr Karl Gadd and Dr Bruce Newman with preparation of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Clifford Craig Foundation, Launceston General Hospital, Launceston, Tasmania who supplied funding for the study’s consumables and research staff (Project 159 – Chewing Gum study).
