Abstract
Muscle wasting in the intensive care unit (ICU) is common and may impair functional recovery. Ultrasonography (US) presents a modern solution to quantify skeletal muscle size and monitor muscle wasting. However, no standardised methodology for the conduct of ultrasound-derived quadriceps muscle layer thickness or cross-sectional area in this population exists. The aim of this study was to compare methodologies reported for the measurement of quadriceps muscle layer thickness (MLT) and cross-sectional area (CSA) using US in critically ill patients. Databases PubMed, Ovid, Embase, and CINAHL were searched for original research publications that reported US-derived quadriceps MLT and/or CSA conducted in critically ill adult patients. Data were extracted from eligible studies on parameters relating to US measurement including anatomical location, patient positioning, operator technique and image analysis. It was identified that there was a clear lack of reported detail and substantial differences in the reported methodology used for all parameters. A standardised protocol and minimum reporting standards for US-derived measurement of quadriceps muscle size in ICU is required to allow for consistent measurement techniques and hence interpretation of results.
Keywords
Introduction
Critically ill patients can experience extensive muscle wasting that occurs rapidly from intensive care unit (ICU) admission. 1 Muscle wasting may contribute to the condition known as ICU-acquired weakness (ICU-AW) with consequent effects on strength and functional outcomes. Quantifying muscle size may therefore identify patients at risk of ICU-AW and poor functional recovery, and help guide interventions to combat atrophy and weakness. 2
The most direct methods to measure musculature are computed tomography (CT) and magnetic resonance imagining (MRI), however their use in critical illness is complicated by a lack of deployability, repeated radiation exposure and incompatible monitoring equipment. 3 In addition, CT scans of the third lumbar region to measure muscularity have been reported more recently in the literature, 4 however this remains a research appropriate rather than clinically relevant technique, due to radiation exposure preventing serial measurements. Accordingly, ultrasonography (US) has gained momentum as a non-invasive, bedside technique for the quantification of muscle size in these patient populations. 5
One of the barriers of using US to measure musculature is that differences in technique methodology can affect the results obtained. In order to maintain reliability of musculature measurements, a defined, standardised approach is required. Consistency in technique to prevent measurement error is required including type of probe, probe positioning, patient positioning, landmarking, choice of equipment, technical settings such as gain, pressure application and distortion minimisation. To our knowledge, there is currently no internationally accepted validated guideline or framework for the measurement of musculature in critical illness using ultrasound. While there are tutorial guides available in other populations,6,7 it should be recognised that these are not validated protocols and there is a clear lack of consensus in reporting US techniques and study methodologies. Furthermore, critically ill patients are a unique population with factors, such as fluid shifts, that influence the translation of evidence from other populations. 8
The quadriceps is the most extensively studied weight-bearing muscle group, with strong correlations to muscle mass and strength in a number of patient groups. 9 Muscle size of the quadriceps can be measured by either cross-sectional area (CSA) and/or muscle layer thickness (MLT). Measurement of CSA involves calculating the area of a target structure from a frozen ultrasound image. 10 MLT utilises on-screen callipers or programs to calculate the thickness of structure(s) of interest: either total quadriceps thickness or thicknesses of specific muscle groups. Within the literature, there appears to be no consensus on which method is more representative of true muscle size. Although Puthucheary et al. suggest that MLT underestimates muscle wasting and strength, 11 studies rarely explore benefit of one over the other.
As standardisation of US imaging and reporting is essential to the future validity and application of US musculature assessment, the aim of this systematic review is to compare the similarities and differences in published methodologies for US-derived measurement of quadriceps musculature in critically ill patients.
Methods
Electronic databases were searched on 31 January 2019 by a single reviewer (LW) using a systematic and reproducible approach as follows. Potential studies were identified using pre-existing literature databases and predefined search criteria (Table 1). Studies were then screened to identify eligible studies for review (LW). This involved the removal of duplicate publications and assessment of eligibility against the inclusion and exclusion criteria from title and abstract.
Literature search criteria.

ICU: intensive care unit
Study inclusion criteria
Studies were eligible for review if they:
were a published peer-reviewed original research article with a quantitative study design; studied critically ill adult participants admitted to an ICU; included ultrasound measurement of the quadriceps muscle for MLT and/or CSA; and were written in the English language
No restrictions were placed on studies with or without a control arm or on year of publication.
Study exclusion criteria
Studies that solely reported other ultrasound-derived measures such as echogenicity (muscle quality assessment) or pennation angle (force contractility), or measurement of muscle groups other than the quadriceps were excluded.
Where further clarification was required to assess relevance, the full-text was obtained (LW). Data extraction was then carried out (LW and LC) using predefined characteristics (Table 2). Any discrepancies in data extraction were settled by a third data extraction (MS). Figure 1 shows the review process.
Data extraction parameters.

aAny method that makes direct reference to ‘elimination of distortion’ with an associated reduction method.
bIndividual(s) responsible for image acquisition only.
cIndividual(s) not involved in image acquisition and only in MLT/CSA measures.
US: ultrasonography; MLT: muscle layer thickness; CSA: cross-sectional area.
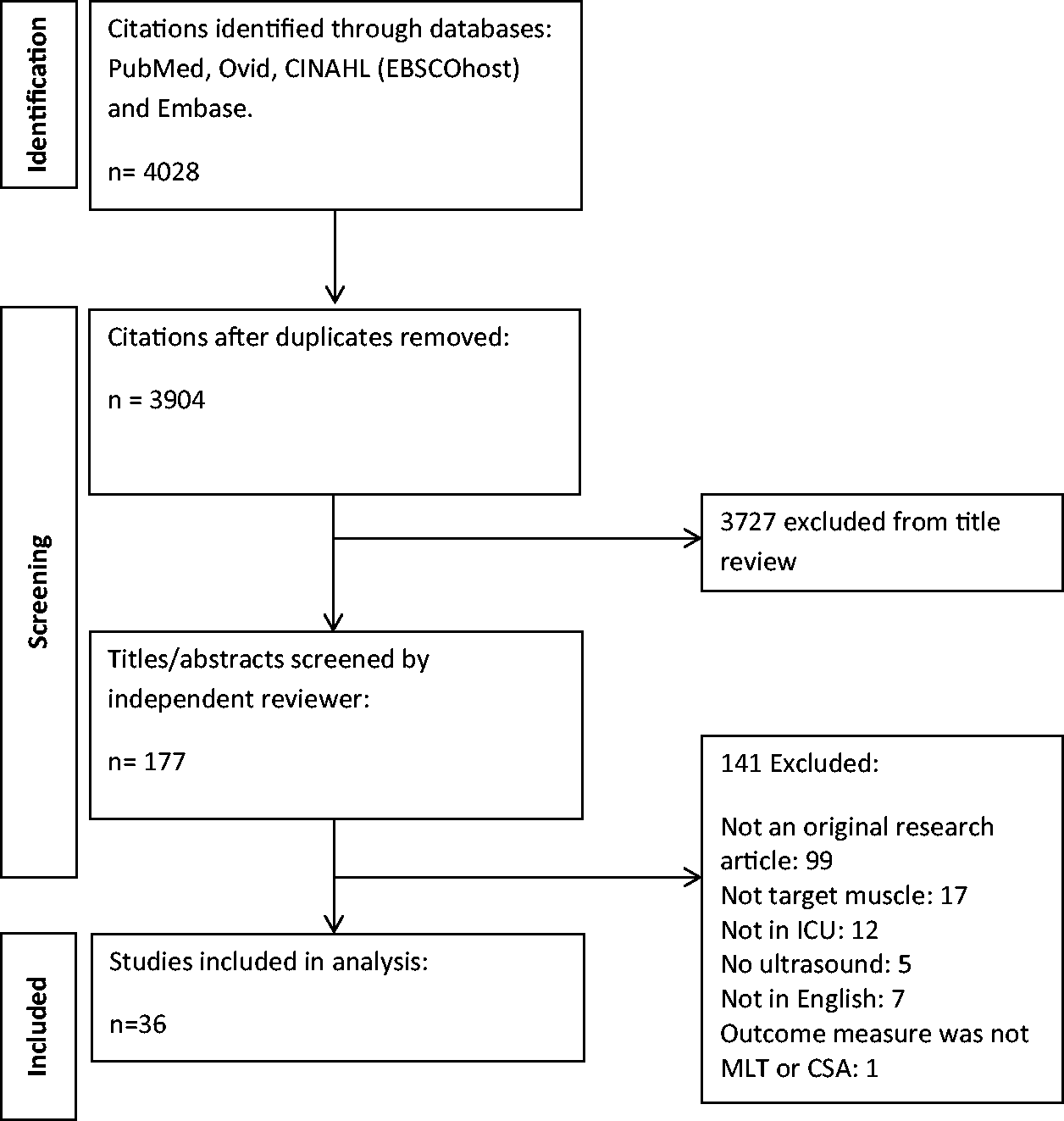
Literature search results following initial search, screening and number of studies included in analysis. ICU: intensive care unit; MLT: muscle layer thickness; CSA: cross-sectional area.
Results
There were 4028 papers identified following the literature search. Following the screening process 36 papers were included in the final data extraction and analysis (Figure 1, Table 3).
Ultrasound protocol data extraction for measuring the quadriceps musculature.
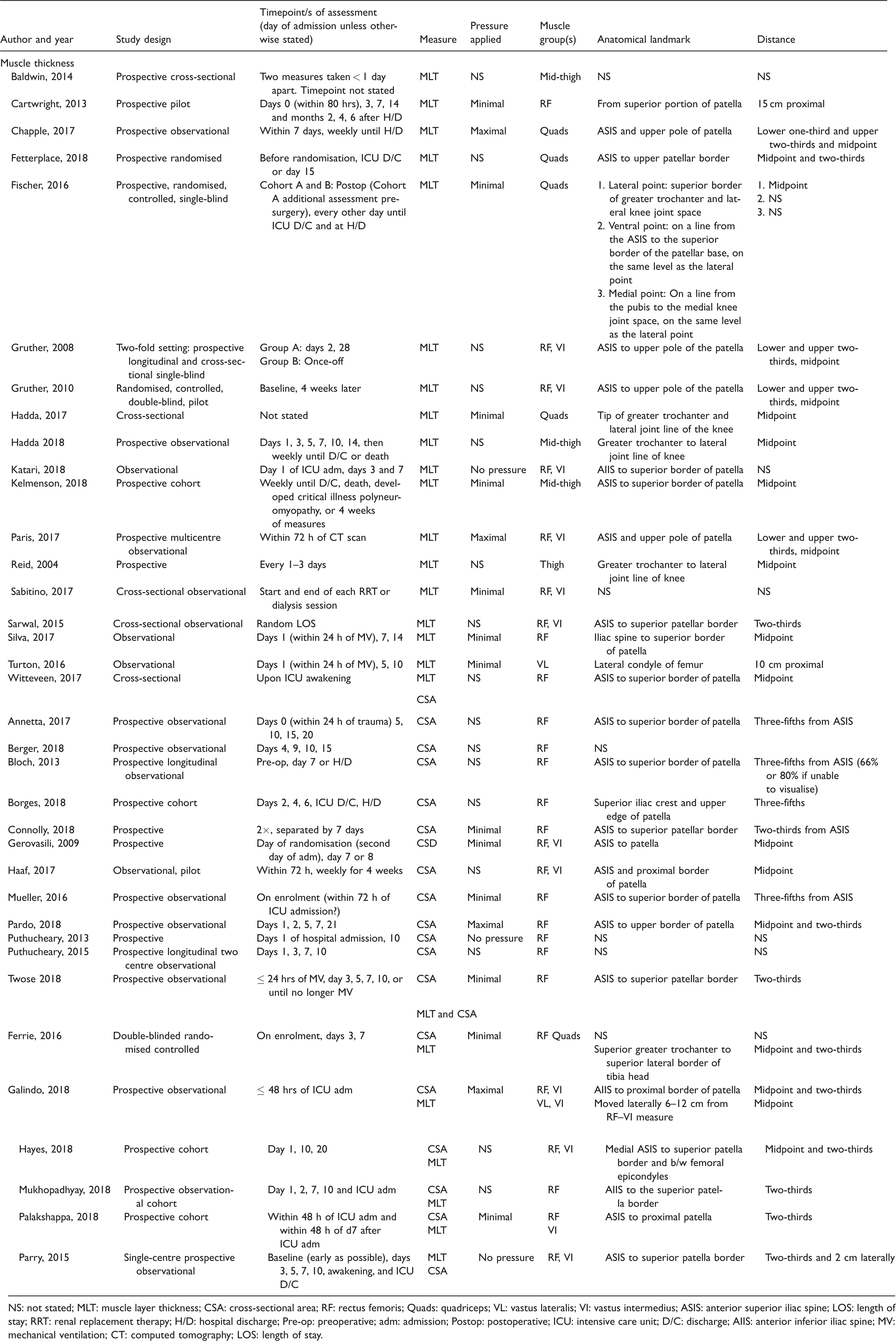
NS: not stated; MLT: muscle layer thickness; CSA: cross-sectional area; RF: rectus femoris; Quads: quadriceps; VL: vastus lateralis; VI: vastus intermedius; ASIS: anterior superior iliac spine; LOS: length of stay; RRT: renal replacement therapy; H/D: hospital discharge; Pre-op: preoperative; adm: admission; Postop: postoperative; ICU: intensive care unit; D/C: discharge; AIIS: anterior inferior iliac spine; MV: mechanical ventilation; CT: computed tomography; LOS: length of stay.
Patient population and study design
Inclusion/exclusion criteria
Four papers did not exclude patients with pre-existing conditions specifically noted to have a detrimental effect on peripheral muscle size.5,12–14
Timepoint of assessments
All but seven8,14–19 authors reported serial US measurements. Of these, only six explicitly reported being conducted after ICU discharge5,20–24 (Table 3). The large majority of studies were observational with only six having a randomised study design.12,13,22,25–27
Patient related
Positioning, contraction/relaxation and rotation
The majority of studies reported supine positioning, with eight not referencing any patient positioning.17,21,22,27–31 Mueller et al. and Turton et al. referenced the head of bed at a 30° and 45° elevation, respectively,16,32 while one paper reported a 30°–45° head of bed elevation, 19 and another referenced semi-supine positioning. 33 The term ‘limb extension’ was reported by 21 studies.5,8,12–16,19,23–26,33–41 One paper referenced ‘legs flat’ and was not included as referencing extension. 20 Relaxation of limbs was reported by 11 authors,5,13,14,16,20,26,35,36,38,39,41 with one using a pillow under the knees for muscular relaxation. 22 Four studies reported foot positioning; toes facing the roof.8,19,36 Neutral rotation/positioning of the lower limbs was reported by five authors12,15,23,33,37 and external rotation of the lower limb by one. 32
Ultrasound procedures
MLT was reported by 18 articles, 12 reported CSA, and six reported both CSA and MLT (Table 3). For MLT four papers measured the rectus femoris (RF), one the vastus lateralis (VL), one the vastus intermedius (VI), one measured VL and VI, and five measured total quadriceps MLT. Four studies reported the ‘mid-thigh’ or ‘thigh’ with no further detail of anatomical site of measurement (Table 3). For CSA 13 papers measured RF and five measured RF and VI. Landmarking of anatomical locations and thickness measures were reported by the majority of authors, however the location descriptors were variable (Table 3). The side that image acquisition was carried out was generally bilateral5,13,14,21–23,26,31,34–36,38,39,41,42 or on the right.8,12,15–20,32,33,43,44 No studies analysed solely the left side aside from Hayes et al. (2018) 37 taking measures from the non-cannulated leg with no further detail of the side used. The majority of studies did not report how landmarking of anatomical sites was performed. Out of the 29 studies that conducted serial measures, only half of these reported the use of a marker to ensure accuracy for consequent acquisitions.
Measurement procedures
The amount of pressure applied to the US probe for image acquisition was largely reported as minimal (15/36 articles) (Table 2). Elimination of distortion was referred to in six studies; five stated the use of excess gel,14,25,28,34,36 while the remaining study referenced gentle contraction, relaxation techniques and passive mobilisation to help visualise structures. 33 US probe positioning was largely uniform with perpendicular positioning being the most predominant (Table 3). The plane of acquisition was not referenced in 19 studies.
US technical settings
The depth to which the US image was taken was reported by 12 authors. Of these, three reported a specific depth—5, 15 1.9–6.0 41 and 4.9 cm 37 —with the others referring to either a standard/constant setting (e.g. always at 6 cm depth)21,34 or alteration to visualise the whole structure(s) of interest.14,17,28,32,33,38,39 Other settings (gain, focus and contrast) were made reference to in ten studies13,15,17,21,23,28,33,34,37,38 with only two providing more information than standardisation of or constant settings.21,33
Type of US transducer used
The type of US transducer used was reported by 30 studies (Table 4). Sound wave frequency (MHz) and the array of the transducer footprint used varied. Linear array probes were reported in half of the studies, with three reporting a curvilinear probe and one the use of a sector array when the thigh cross section could not be visualised completely.
Ultrasound protocol data extraction for ultrasound transducer procedures and equipment.
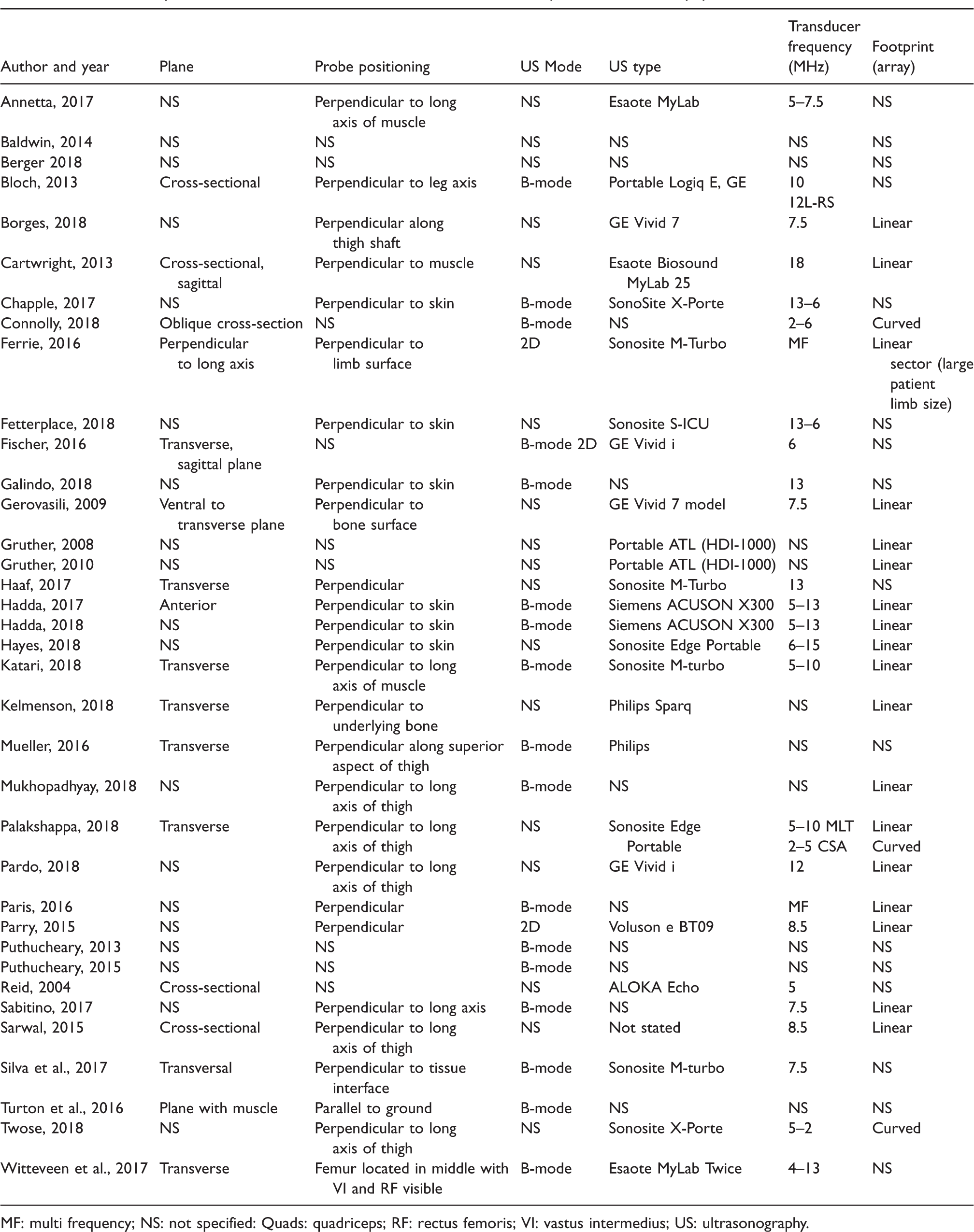
MF: multi frequency; NS: not specified: Quads: quadriceps; RF: rectus femoris; VI: vastus intermedius; US: ultrasonography.
Operators
The operator was defined as an individual(s) conducting image acquisition only at the bedside, while an assessor solely conducted image analysis. For those studies that included more than one operator (n = 28), three articles clearly stated that the operator was not blinded16,22,45 and eight that operators were blinded.8,12,15,25,26,35,36,42 The majority of studies only included one operator (19/37; 51%) with six having two, 8,17,24,25,36,42 one having three 32 and the remaining not reporting this parameter.14,15,18,20,22,27–30,40 Experienced15,21,23,33,39 or trained5,8,12,13,16,22,30,32,34,36–38,41,42 operators were referenced in 19 papers.
Image analysis
Number of images at each anatomical site
Thirteen authors did not state the number of US images obtained8,15,16,18,21,25,27,29–31,38,42,43. Four studies acquired measures in duplicate.13,14,34,36 Triplicate measures were recorded by 13 authors8,12,17,20,22–24,28,33,37,40,41,45 while three took measures in triplicate only if there was a difference of >10%5,39 or 0.1 cm. 19 The remaining authors reported taking one, 26 six, 32 or ‘the best three images’. 30
Measurement tools
There was great variation in the measurement tool used to analyse the images. Nine articles used inbuilt callipers on the US machine,8,13,14,16,19,24,34,41,45 nine used Image-J,15,20,21,23,30,32,33,37,40 one used Photoshop Elements version 12 28 , one used EchoPAC (General Electric Healthcare) 22 , and the remaining 16 did not state the method used.
Scoring of images
An assessment of muscle wasting using a scoring method in order to allow for interpretation of muscle size was only reported by two authors.17,20 Reasons for exclusion of images included excessive oedema not allowing definition of the RF,16,20,25,37 images without standardised gain settings 28 and removal of images with the highest and lowest width. 32
Assessors
In seven studies the same person acquired the US image (operator) and measured MLT/CSA (assessor).13,16,19,33,39,42,45 In five studies, the operator was a different person to the assessor,12,15,22,26,35 and all assessors were blinded to the US image. Of the six interventional studies, four studies reported that the person conducting the ultrasound measurement was blinded to the intervention.12,22,25,35 Of the 30 studies that included serial US measures, only four reported that the investigator taking the US image was blinded to previous measures12,22,30,37 and two explicitly reported that no blinding to previous measures occurred.25,32
Inter- and intra-rater reliability
Inter-rater reliability was reported in 11 studies8,14,16,18,26,28,30,36,37,42,45 and intra-rater reliability was reported in seven studies.8,14,15,22,24,36,42 The most common method of reporting reliability was using intra-class coefficients.
Discussion
This systematic review identified 36 studies that analysed quadriceps muscle thickness or cross-sectional area in critically ill patients using US. Across these studies, it was observed that there was a clear lack of agreement in the methodological approach used, and the reporting of such, in regards to the US technique.
This review identified that the most common method of reporting musculature using US in the ICU literature is MLT, as opposed to CSA, although the authors generally provided little justification for the chosen methodology. CSA measurements have been shown to be a more reliable indicator of muscle wasting and strength in 19 patients up to day 10 of ICU admission. 11 However, MLT may have greater translation into a clinical setting when compared to CSA, with greater feasibility as results can be obtained quickly and in real time, as reported by Paris et al. 14 Further, MLT may be easier to identify than CSA in the setting of muscle wasting, where distinction between muscle groups to allow CSA measurements may not be feasible. The development of an ultrasound protocol for use in critical care requires consideration of both the feasibility of measurement, as well as quantification of the amount of muscle on admission, and degree of muscle loss during hospitalisation that has an impact on outcomes.
Of the included studies, while CSA was primarily of the RF muscle, there was great variation in the conduct of linear measures, with both individual muscle groups and total muscle layer being measured. Of even greater concern is the inconsistency in the reporting of anatomical sites of measurements. While the majority of studies measured between the anterior superior iliac spine (ASIS) and patella, this was not unanimous. Further, the distance at which the US measurement was taken between these two sites varied greatly. Measures were taken at one-third, two-thirds, one-half, three-fifths, 10 or 15 cm proximal to the patella, or at a combination of sites. With no consensus on the appropriate anatomical site to conduct the measurement either the most accessible, 46 or methods translated from non–critically ill populations, have been referenced by the included papers.18,34 This discrepancy in anatomical site of measurement is similarly reflected in tutorial guides: the most commonly proposed method is to measure muscle at two sites—at two thirds and the midpoint between the ASIS and the top of the patella6,46—yet the anterior inferior iliac spine has also been proposed. 47 Little justification behind the proposed sites is provided. It is likely that using more than one landmark provides a more global, and hence accurate, reflection of muscle size and subsequently a better representation of muscle atrophy. 48 A consensus on appropriate landmarking technique is required to ensure muscle atrophy is being captured optimally and that consequent studies are both relatable and comparable. This is particularly important as we move forward to demonstrate relationships between interventions and degree of muscle wasting.
Although the majority of studies reported applying minimal pressure on the muscle group measured, the question of the correct amount of pressure to apply during image acquisition remains unanswered, with alternating theories for each. Critically ill patients are susceptible to conditions that may complicate visualisation of muscle wasting such as oedema. 49 Correct pressure applied to a US probe may improve image quality and therefore muscle measurements in the presence of these complications. 50 It has been hypothesised that minimal pressure may be easier to standardise across operators and may be able to identify, and therefore quantify, oedema. 6 Surprisingly though, other reports state that maximal and not minimal compression is able to better allow for standardisation 51 and muscle quantification in the presence of oedema. 48 These factors have not yet been thoroughly investigated 6 and consideration of the ability to maintain consistent pressure in serial measurements is required. This dilemma provides a potential application for technology to standardise pressure application in the setting of muscle wasting.
A total of 29 studies conducted serial measures. For studies that report change in muscle size over time it is vital that the landmarking technique used encompasses appropriate strategies to ensure serial measures are acquired from the same location and in the same manner, particularly with regard to patient positioning and the degree of pressure applied.
Of the 36 studies, 19 reported that the person conducting the ultrasound was experienced. This is a concern given taking accurate US measures are highly dependent on training and experience. 15 Although some studies have demonstrated reliability between muscle size acquisitions, these have often been in healthy populations 52 for which measurements are likely to be less challenging than for the critically ill, without barriers such as positioning, fluid retention and wounds. 15 Inter-rater reliability needs to be further demonstrated in populations that experience muscle wasting, and take into consideration barriers to limb positioning which adds complexity to the visualisation of anatomical structures.
This review also identified inconsistencies in patient positioning for ultrasound measures. While the majority of studies placed the patient in a supine position, only two referenced head of bed elevation. Head of bed elevation is often employed for reasons such as high intracranial pressure and reduction of ventilator-associated pneumonia risk. 53 Hacker et al. demonstrated significant differences (P < 0.001) in RF CSA across four different hip flexion positions (0°, 20°, 30° and 60°). 54 Therefore, alterations in elevation can give significantly different readings. In addition, limb extension was often reported, although relaxation and rotation were reported by few studies. As muscle structure length and angle are altered by contraction55,56 and rotation, alterations of these parameters will affect measures.
With the increasing use of US to measure muscle atrophy, determining what muscle size, or degree of change, relates to clinical outcomes and/or is considered to constitute muscle wasting in the critically ill population is essential. The development of ‘cut-offs’ that equate to clinically relevant muscle wasting, similar to that done by Mourtzakis et al. with CT in cancer patients, 57 is required in order for US of musculature to be appropriately interpreted and implemented into clinical practice. Only one study in this review provided discussion on the degree of muscle loss that may constitute clinically relevant muscle wasting: Bloch et al. classified those with greater than 9.24% muscle loss as having muscle wasting, determined by a Bland–Altman plot with the coefficient of variation. 20 There is currently no score to determine what constitutes muscle wasting in the critically ill population and further work is required in this area.
This systematic review has a number of strengths and limitations. The search criteria were limited to those articles that measured the quadriceps muscle, as justified earlier. However, it should be recognised that there have been recent reports on the association between protein delivery and change in ultrasound-derived forearm thickness 12 as well as other authors measuring muscle size in other muscle groups. 45 Further, the inclusion of other US-derived measurements such as echogenicity and pennation angle may provide additional information on muscularity in critical illness, however this was beyond the scope of this review. During data extraction, only data specifically reported by the authors was included in this review, and hence contacting the authors may have provided more completeness to the data obtained. Similarly, only methodology reported in the primary publication or online supplements was extracted. Some authors may have referenced techniques, however as it is difficult to ascertain which elements of the reference papers were included in the current publication, these details were not reported. The strengths of this review include the use of multiple databases and ranges of terminology to capture as many papers as possible, and a rigorous assessment of many aspects that may affect ultrasound measurement of musculature.
Conclusion
This review identified that there are substantial differences in the reported methodology used to measure CSA/MLT of the quadriceps muscle in critically ill patients. The reporting of US methodology was not comprehensive, often failing to provide a complete understanding of the specific methods used. Where methods were reported, the US methodology varied greatly between studies preventing consistency and comparison between studies. As a result, developing a standardised training ‘package’ in this area, that includes reliability testing and minimum reporting standards, would be of great benefit ensuring US users are not only adequately trained in measuring musculature but also to ensure that the way in which US methodology is reported allows for accurate data interpretation within and between studies and methodology replication.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
