Abstract
Surgery and obesity are known risk factors for thromboembolic events due to the presence of a hypercoagulable state. Rotational thromboelastometry is a viscoelastic assay that can provide a measure of hypercoagulability via a comprehensive assessment of the coagulation process. This prospective study investigates haemostatic changes over time, presence of hypercoagulability and the association between body mass index and thromboelastometry results in patients undergoing major orthopaedic surgery. Fifty adult patients undergoing total hip or knee replacement surgery had serial thromboelastometry measures performed prior to and following surgery, and on postoperative days 1 and 3. A hypercoagulable state, defined by an ExTEM maximum clot firmness G score ≥11 dyne/cm2, was present in 28% of the patients at baseline. The mean ExTEM maximum clot firmness G score increased by an average of three units from 10 (95% confidence interval (CI) 9–11) dyne/cm2 at baseline to 13 (95% CI 13–14) dyne/cm2 on postoperative day 3, with 85% of patients having a G score ≥11 dyne/cm2. A decrease in ExTEM and InTEM clot formation time and an increase in ExTEM, InTEM and FibTEM clot amplitude at 10 minutes, alpha angle and maximum clot firmness were observed by postoperative day 3 (P < 0.001). There was no significant difference in the mean thromboelastometry values between patients with a body mass index <35 kg/m2or ≥35 kg/m2. Although a modest association between body mass index and the ExTEM maximum clot firmness G score was observed with exploratory data analysis, further study is required in a large cohort to test the effects of confounders, validate these findings, and determine their clinical importance.
Introduction
Changes in haemostatic and fibrinolytic pathways during surgery can contribute to hypercoagulability.1,2 This hypercoagulable state can extend up to four to six weeks following surgery3–5 and has been reported to be associated with thrombotic complications. 6 Obesity is a heterogeneous disease involving a complex interplay between genetic, epigenetic and environmental factors affecting numerous biological pathways, some of which are prothrombotic. 7 Increased levels of fibrinogen, Factor VII, Factor VIII, von Willebrand Factor (vWF) and plasminogen activator inhibitor, characteristic of a prothrombotic state and impaired fibrinolysis, have been observed in obese compared to non-obese patients. Therefore, obese patients are considered to be at a higher risk of thromboembolic events,8,9 particularly in the postoperative period.10,11
Current practice using standard coagulation tests to detect hypercoagulability has limitations. 5 Functional assays such as the viscoelastic tests thrombelastogram (TEG®, (Haemonetics Australia, Macquarie Park, New South Wales)) and rotational thromboelastometry (ROTEM) provide comprehensive, dynamic and timely assessment of clot initiation, propagation, clot quality and stability in whole blood. ROTEM® delta (Pentapharm GmbH, Munich, Germany) is a point-of-care (POC) testing device measuring the viscoelastic properties of multiple aspects of coagulation in a sample of citrated whole blood. POC haemostatic testing has been used in hypercoagulability screening and assessment of thrombotic risk, particularly in trauma, 12 following surgery13,14 and in cancer patients.15–17 According to previous studies, G score, a derived ROTEM parameter indicative of total clot strength, is a validated marker of thromboembolic risk,18,19 with every dyne/cm2 increase in G resulting in 25% increased odds of developing thromboembolic complications. 18 Studies done in the past were on non-cardiac surgery patients6,20,21 with a small sample of orthopaedic surgical patients, or were based on a single pre- or postoperative assay6,20 or on patients undergoing bariatric surgery.19,22 Other studies were TEG based,6,18,21,22 and absolute values from TEG and ROTEM assays may not be interchangeable in clinical settings. 23 While a hypercoagulable profile has been reported in morbidly obese patients compared to controls,21,24 there is a lack of evidence for the changes in ROTEM parameters during and after major elective orthopaedic surgery and their association with obesity. Obesity has been recognised as a chronic progressive disease which is best measured as a continuum rather than being categorised based on arbitrary boundaries. 25 This preliminary study was designed to investigate the association between body mass index (BMI) and (a) preoperative hypercoagulability and (b) coagulation changes measured using ROTEM assays over time in adult patients undergoing total hip replacement (THR) or total knee replacement (TKR).
Materials and methods
Following institutional ethics approval (HREC/15/QPCH/123) and written informed consent, a convenience sample of 50 adult patients undergoing THR or TKR at The Prince Charles Hospital (Brisbane, Australia) were included in this study. This project was a part of a larger study registered with the Australian New Zealand Clinical Trials Registry (ACTRN12615000825550). Patients with (a) a known history of thrombophilia, (b) other possible causes of hypercoagulability (active malignancy, prolonged bed rest, current use of oral contraceptives or hormone replacement therapy, previous history of venous thromboembolism (VTE), nephrotic syndrome) and (c) other pre-existing conditions requiring postoperative anticoagulation or patients needing long term anticoagulation (e.g. prosthetic valves) were excluded from this study.
Baseline BMI was determined from height and weight measured during the preoperative assessment. Potential confounders of interest included age, sex, smoking, presence of metabolic syndrome, 26 preoperative aspirin intake, comorbidities, American Society of Anesthesiologists physical status, type of procedure and use of a tourniquet during surgery. Anaesthetic and surgical management of the study participants were at the discretion of the treating specialists. Tranexamic acid (1–2 g) was administered to patients as an intravenous bolus, with or without another 1 g applied topically, as a part of standard practice in our hospital. Routine blood tests performed and data relevant to this study were recorded.
Blood samples for ROTEM assay were collected from participating patients prior to surgery, immediately post surgery and on postoperative days (POD) 1 and 3. POD 3 was chosen as the last timepoint because of differences in the discharge time between THR and TKR patients. Fresh venous blood samples were collected in citrated vacutainer tubes (2.7 mL containing 3.2% sodium citrate). Assays were performed using ROTEM® delta in accordance with the manufacturer’s instructions. InTEM and ExTEM clotting times (CT), clot formation time (CFT), alpha angle (α), clot amplitude at 10 minutes (A10), maximum clot firmness (MCF), and FibTEM A10 and MCF, were analysed. The G score for ExTEM MCF at each timepoint was derived using the formula (5000 × MCF)/100–MCF. 19
The study participants received routine thromboprophylactic measures post surgery that involved graduated compression stockings and either aspirin 300 mg/day for six weeks from POD 1 (in the majority) or other agents such as enoxaparin or direct-acting oral anticoagulants according to the discretion of the surgical team. Intermittent sequential compression devices were placed on the non-operative limb before the procedure.
The primary outcome measure was the ExTEM MCF G score, measured on a continuous scale. Hypercoagulability was defined as a G score of ≥11 dyne/cm2. 19 Distributions of ROTEM and clotting function measures were examined by inspecting histograms and boxplots; associations with BMI were explored using scatterplots. Baseline variables were compared across the categories of hypercoagulability and BMI using Student’s t-test or Wilcoxon’s rank sum tests for continuous variables or Fisher’s exact test for categorical variables. Associations between BMI and ROTEM measures were assessed by fitting mixed-effects linear regression models. To assist in the interpretation of results, BMI was centred at a value of 30 kg/m2 and rescaled so the unit of measurement was 5 kg/m2. CFT had a skewed distribution and so was log transformed for use in modelling. The predictive marginal mean values for each variable at each timepoint were obtained from these models. For the primary outcome, confounders of interest were tested in multivariable models. Analyses were performed using Stata v15 (StatCorp, College Station, TX).
Results
Fifty patients (60% female) were recruited between June 2016 and February 2018 with a mean age of 67 (standard deviation (SD) nine) years. Baseline characteristics by hypercoagulability and BMI category are shown in Table 1. Hypercoagulability was present in 14 (28%) patients at baseline, comprising 5/31 (16%) patients with a BMI <35 kg/m2 and 9/19 (47%) patients with a BMI ≥35 kg/m2 (P = 0.03). BMI ranged from 22 to 56 kg/m2 (median 31 kg/m2; interquartile range (IQR) 28–38 kg/m2). Scatter plots of observed ExTEM MCF G score by BMI at baseline and POD 3 are presented in Figure 1. Baseline clotting function measures did not differ by hypercoagulability status; mean fibrinogen levels were 4.1 g/L in those with baseline hypercoagulability compared to 3.7 g/L in those without. Summary statistics for baseline and postoperative ROTEM measures and clotting function tests by BMI category are shown in Table 2. Mean ROTEM values did not differ significantly between people with a BMI <35 kg/m2 compared to those with a BMI ≥35 kg/m2. At baseline, the mean ExTEM MCF G score was 9.6 dyne/cm2 (95% confidence interval (CI) 8.9–10.2) in those with a BMI <35 kg/m2 compared to 10.6 dyne/cm2 (95% CI 9.5–11.8; P = 0.08) in those with a BMI ≥35 kg/m2. The ExTEM MCF G score increased from baseline in both groups, with the highest value (mean 14.2 dyne/cm2; SD 2.9) observed on POD 3 in patients with a BMI ≥35 kg/m2. A hypercoagulable state was present in 85% of the patients on POD 3.
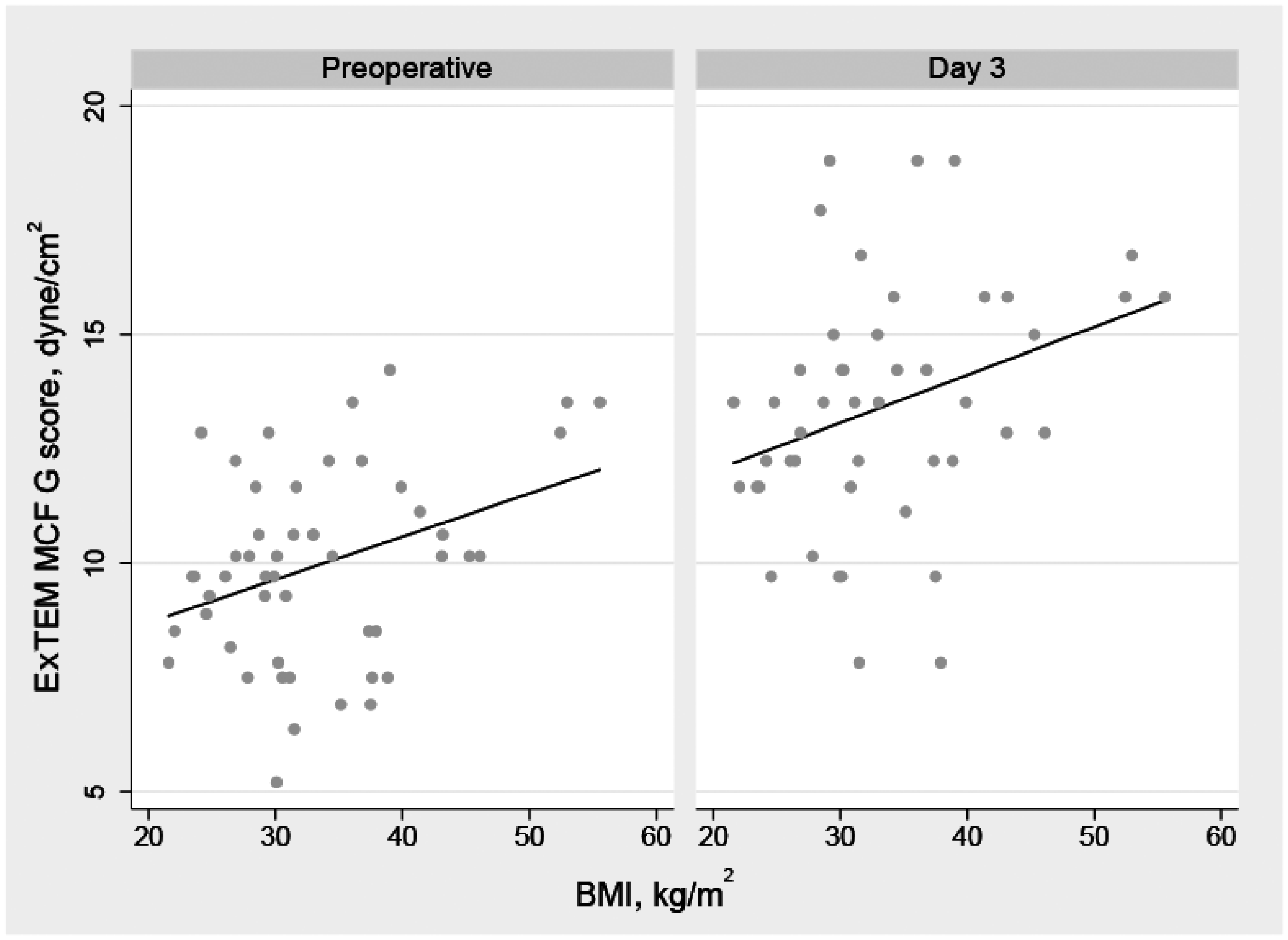
Scatter plots of ExTEM MCF G score by BMI, with fitted linear regression lines at baseline and POD 3. MCF: maximum clot firmness; BMI: body mass index; POD: postoperative day.
Distribution of baseline and intraoperative patient characteristics by elevated baseline ExTEM maximum clot firmness G score (≥11 dyne/cm2) and classification of obesity (body mass index ≥35 kg/m2).
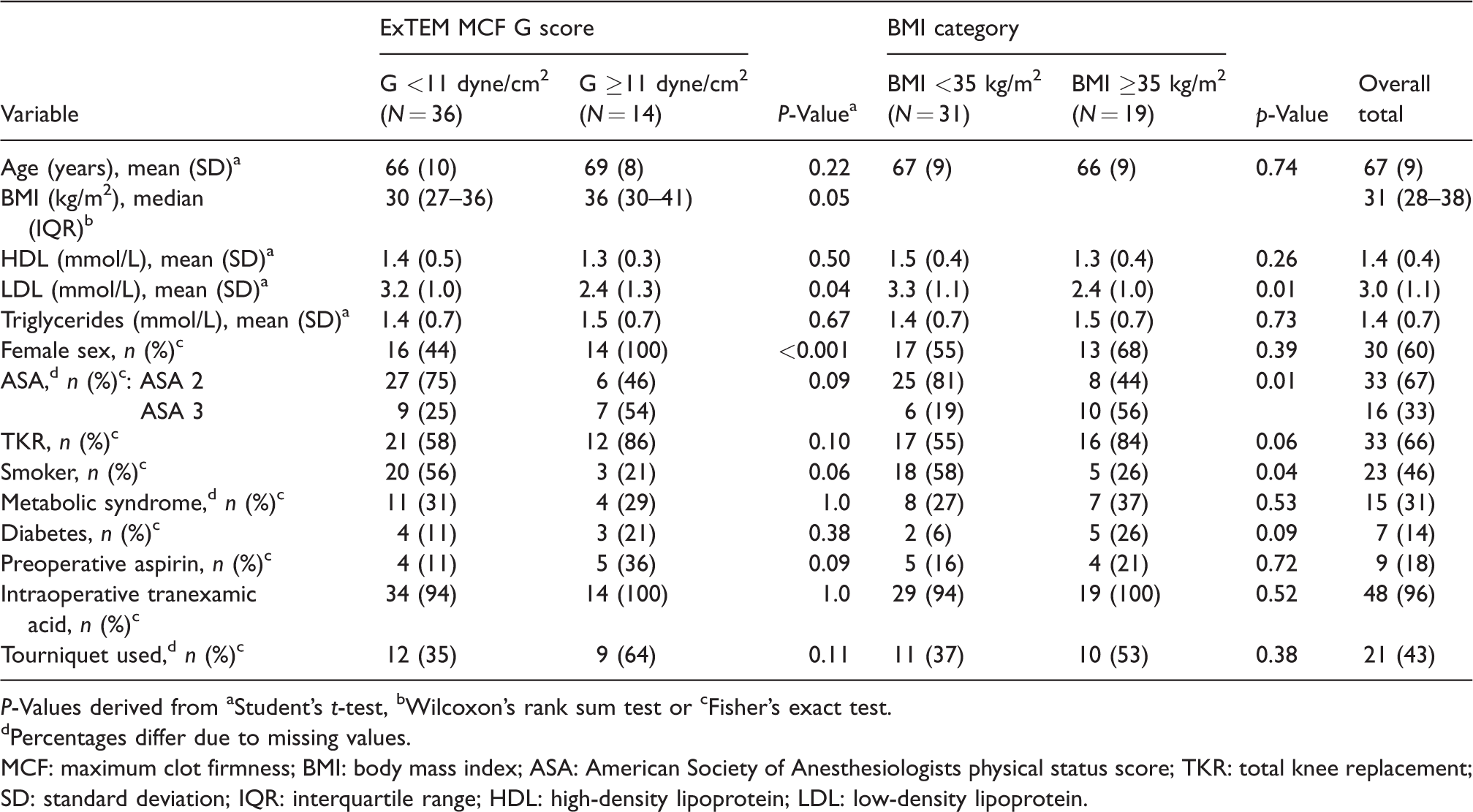
P-Values derived from aStudent’s t-test, bWilcoxon’s rank sum test or cFisher’s exact test.
Percentages differ due to missing values.
MCF: maximum clot firmness; BMI: body mass index; ASA: American Society of Anesthesiologists physical status score; TKR: total knee replacement; SD: standard deviation; IQR: interquartile range; HDL: high-density lipoprotein; LDL: low-density lipoprotein.
Distribution of rotational thromboelastometry and clotting function test results by body mass index category at baseline and postoperatively.
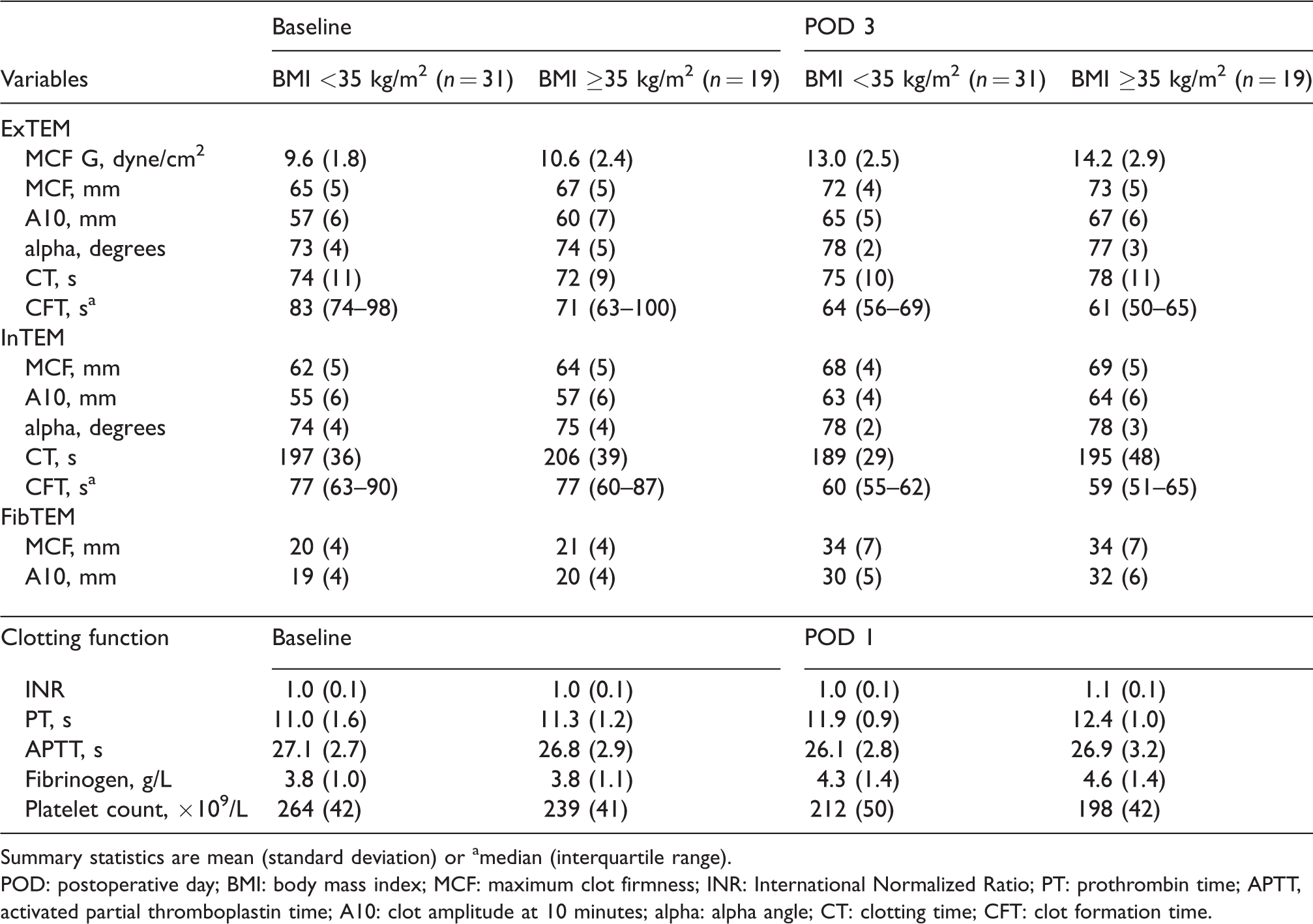
Summary statistics are mean (standard deviation) or amedian (interquartile range).
POD: postoperative day; BMI: body mass index; MCF: maximum clot firmness; INR: International Normalized Ratio; PT: prothrombin time; APTT, activated partial thromboplastin time; A10: clot amplitude at 10 minutes; alpha: alpha angle; CT: clotting time; CFT: clot formation time.
Significant changes over time were observed for all measures of clotting function, both standard coagulation laboratory tests and ROTEM assays in all patients (Table 3). Mean prothrombin time, International Normalized Ratio and fibrinogen increased between baseline and POD 1 (P < 0.001) while activated partial thromboplastin time and platelet count decreased between baseline and POD 1 (P = 0.005 and P < 0.001, respectively; Table 3). Compared to baseline values, a significant shortening in CFT (ExTEM and InTEM) as well as a significant increase in A10, α and MCF (ExTEM, InTEM and FibTEM; P < 0.001) were observed on POD 3.
Postoperative changes and effects of body mass index on rotational thromboelastometry parameters in 50 surgical patients.
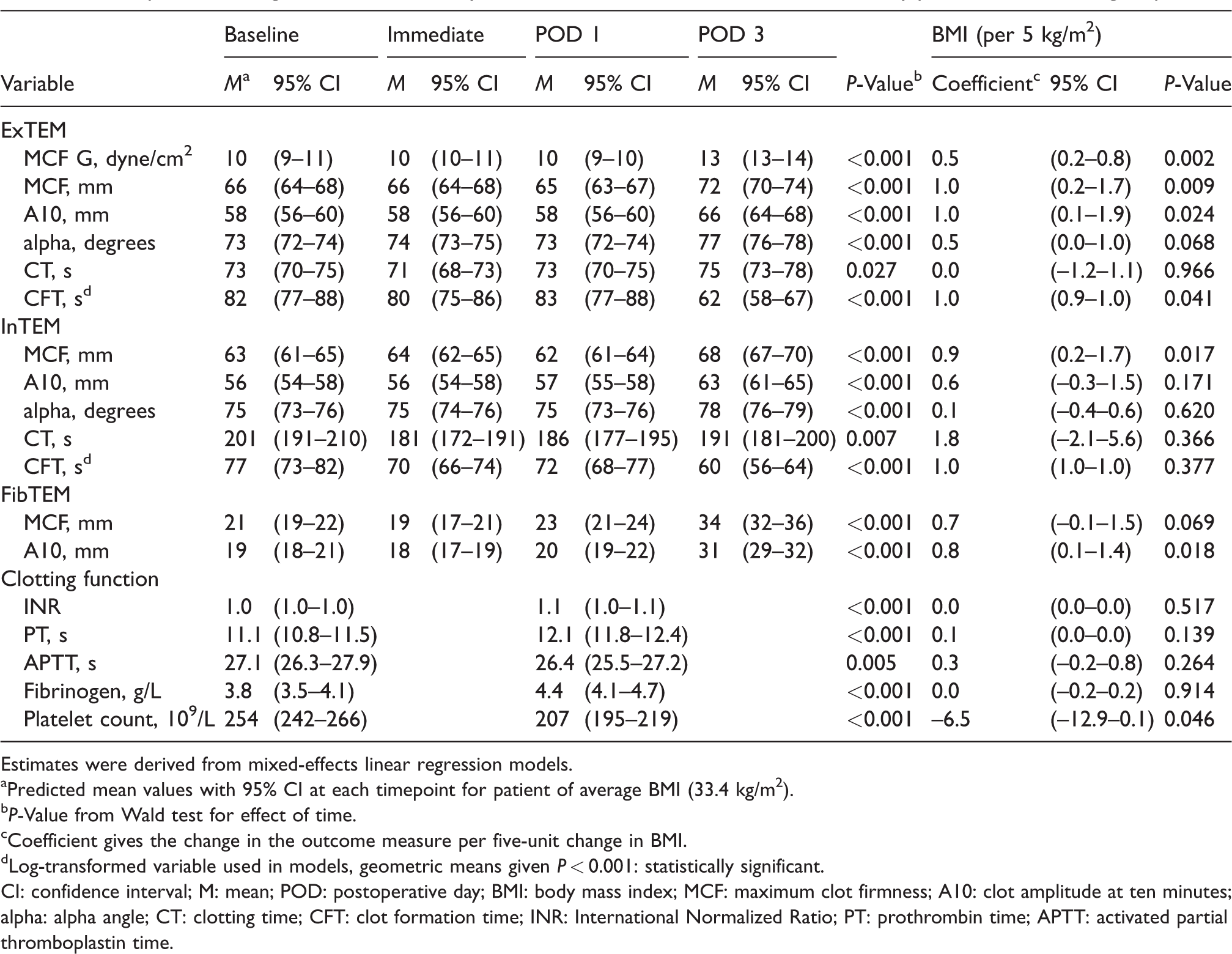
Estimates were derived from mixed-effects linear regression models.
Predicted mean values with 95% CI at each timepoint for patient of average BMI (33.4 kg/m2).
P-Value from Wald test for effect of time.
Coefficient gives the change in the outcome measure per five-unit change in BMI.
Log-transformed variable used in models, geometric means given P < 0.001: statistically significant.
CI: confidence interval; M: mean; POD: postoperative day; BMI: body mass index; MCF: maximum clot firmness; A10: clot amplitude at ten minutes; alpha: alpha angle; CT: clotting time; CFT: clot formation time; INR: International Normalized Ratio; PT: prothrombin time; APTT: activated partial thromboplastin time.
A significant association was observed between BMI and the ExTEM MCF G score, with the mean G score increasing by 0.5 dyne/cm2 (95% CI 0.2–0.8; P = 0.002) for each 5 kg/m2 increase in BMI. Based on this model, the predicted marginal mean values of the ExTEM MCF G scores with 95% CI by BMI at baseline and POD 3 are presented graphically in Figure 2. The horizontal line at a G score of 11 dyne/cm2 represents the threshold used to define hypercoagulability. 19 The association between BMI and G score persisted after testing potential confounders in the model.
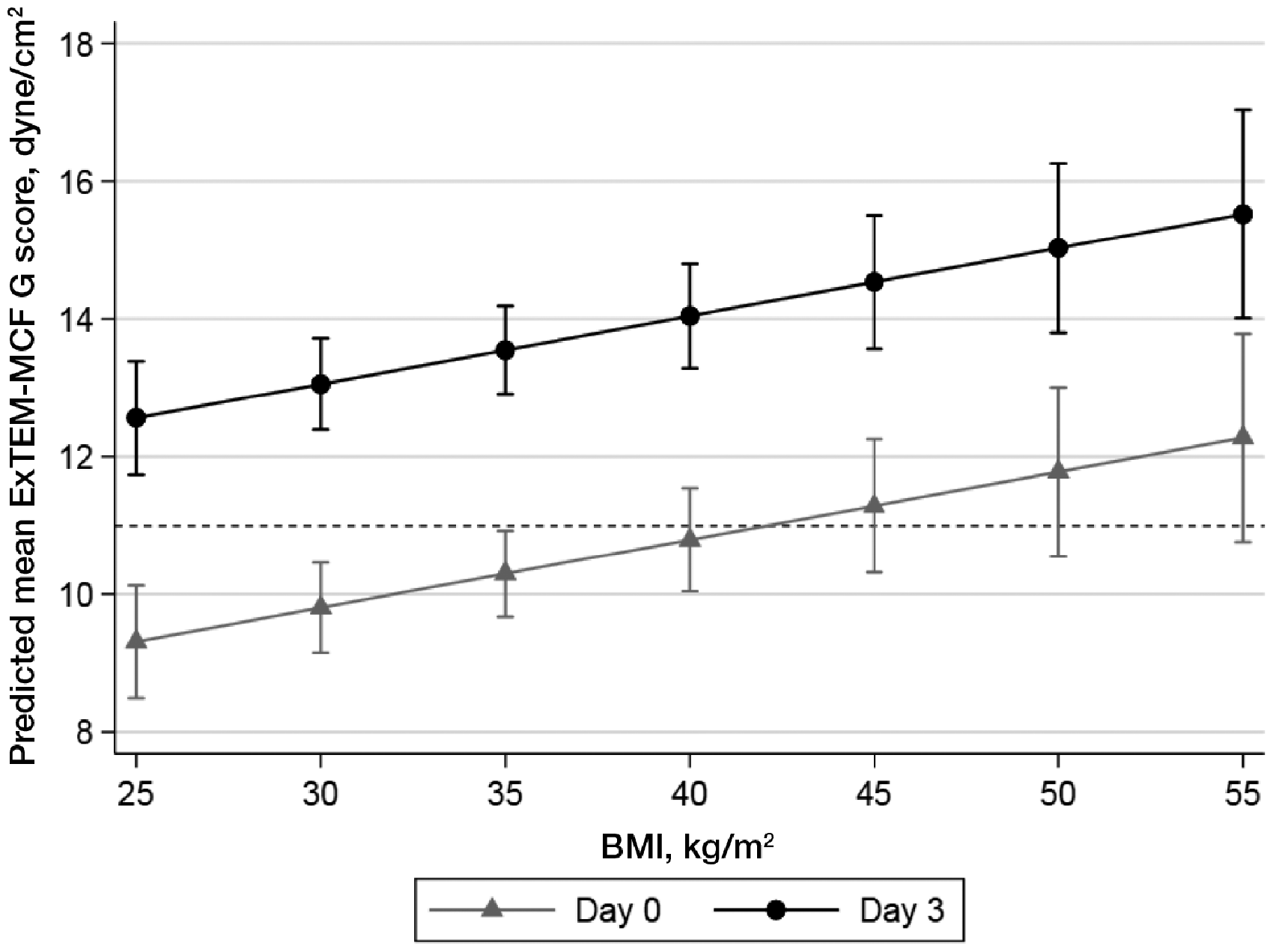
Predicted mean values of the ExTEM MCF G scores derived from a linear mixed-effects model by BMI at baseline and POD 3. The dashed horizontal line at a value of 11 dyne/cm2 represents the threshold used to define hypercoagulability.MCF: maximum clot firmness; BMI: body mass index; POD: postoperative day.
Discussion
Our study explored coagulation changes measured using ROTEM assays and their relationship with BMI on a cohort of 50 patients who underwent primary unilateral THR and TKR during the study period. We observed perioperative changes from baseline to POD 3 in all ROTEM measures, with an increase in clot firmness (elevated MCF) and alterations in clot kinetics (increase in A10, α and a decrease in CFT), which are consistent with alterations in haemostasis characteristic of hypercoagulability, as described in the literature. 27 Consequently, around 85% of patients were classified as hypercoagulable on POD 3, with a G score of ≥11 dyne/cm2, compared to 28% of patients at baseline. Notably, these changes occurred despite our standard institutional thromboprophylactic measures.
Significant changes from baseline ROTEM measures were not observed until POD 3, which is consistent with the findings from previous published studies. Taura et al. observed no modifications with clot strength until POD 2 in bariatric surgery patients. 19 In a study by Lison et al., of 51 patients who underwent major surgery, the increase in fibrinogen, Factor VIII and vWF, as well as increase in MCF, were noted from POD 2 to POD 6. 1 According to the literature, a hypercoagulable state can extend up to four to six weeks following surgery3–5 and has been reported to be associated with increased risk of thromboembolic events. 6 A study by Lapidus et al. reported that the majority of cases of deep venous thrombosis following THR (92%) and TKR (55%) were detected after hospital discharge, suggesting a prolonged postoperative hypercoagulable state and an increased risk of VTE complications. 28 Hyperfibrinogenaemia has been reported to confer an increased thrombotic risk. 29 In our study, patients with a G score ≥11 dyne/cm2 had higher mean fibrinogen levels compared to those with a G score <11 dyne/cm 2 , as observed in other studies,19,22 but the difference was not statistically significant.
Obesity has been demonstrated to be associated with increased production of procoagulant microparticles and an increase in thrombin generation. 30 Campello et al. reported a significantly higher MCF in both InTEM and ExTEM in patients with a BMI ≥35 kg/m2 compared to age- and sex-matched healthy volunteers with a BMI <25 kg/m2. 30 In our study, we did not observe a significant difference in ExTEM G scores between patients with a BMI ≥35 kg/m2 versus a BMI <35 kg/m2. Hence, we are unable to support an effect of BMI on hypercoagulability based on the preliminary findings in our small sample. However, with our exploratory analysis, we observed a modest association between BMI and ROTEM parameters related to increased clot strength, especially the ExTEM MCF G score, consistent with some previous studies.24,30 Based on our model, the expected mean baseline ExTEM MCF G score for patients with a BMI ≥43 kg/m2 is above the threshold used to define hypercoagulability (Figure 2).
Interestingly, baseline hypercoagulability was observed in females but not in males. Sex-based differences in coagulability have also been demonstrated previously by Sucker et al. 31 In their study, the sex difference was noted in all age groups, even when females who were not on oral contraception and males were compared. Nevertheless, oral contraception per se did induce a significant change in the ROTEM parameters. 31 Another large prospective study had reported an increased risk of VTE in women on antidepressants. 32 We did not collect the relevant history from our participants and hence are not able to comment if the use of hormone replacement therapy or antidepressants could have contributed to our findings. Further work is required to explore sex differences in the association between BMI and hypercoagulability.
To our knowledge, this is the first study to explore ROTEM assays comprehensively to characterise perioperative haemostatic changes in patients undergoing lower limb joint replacement and explore the association between BMI and hypercoagulability before and after surgery. As the treating team were blinded to ROTEM results, the clinical management, including choice of thromboprophylaxis in the perioperative period, was not influenced by our results.
Our study has limitations. Although we observed changes in ROTEM parameters suggestive of hypercoagulability with increasing BMI, it is possible that these associations were due to confounding factors. We were unable to explore the effect of confounders adequately due to our limited sample size. However, our effect estimates were not altered when measured confounders (e.g. preoperative aspirin or female sex) were included in models. While multiple hypothesis tests may have increased the likelihood of type I error, the ROTEM measures are inter-related, and the results across MCF measures were consistent.
A standard definition of hypercoagulability status based on ROTEM measures is lacking and further work is required to develop clinically relevant thresholds applicable to surgical populations. While we chose to define hypercoagulability as a G score ≥11 dyne/cm2 based on previous relevant studies,19,22 there were studies with various other cut-off values and parameters predictive of thromboembolic complications.6,18,20 The reference ranges we applied were from a different population. 33 ROTEM assays were performed only until POD 3. Since the majority of the VTE events are diagnosed after hospital discharge following THR and TKR, 28 it is likely that these changes in ROTEM parameters may persist after discharge and have not been captured by our study. According to recent studies with conventional ROTEM analysis, a clinical dose of enoxaparin does not show any significant change in ROTEM parameters, 34 whereas significant changes are seen mainly in ExTEM CT in the presence of high levels of rivaroxaban with no impact on any MCF.34–36 With our data, we are unable to comment on the effects of postoperative VTE prophylaxis on ROTEM parameters, as only three of our patients received enoxaparin as bridging anticoagulation to rivaroxaban at discharge and the rest of our patients received aspirin as postoperative VTE prophylaxis.
In summary, in our preliminary study, we observed perioperative variations in ROTEM parameters between baseline and POD 3. Hypercoagulability was present in 28% of our patients at baseline, increasing to 85% of patients on POD 3. There was no significant difference in the mean ROTEM values when comparing patients with a BMI ≥35 kg/m2 to those with a BMI <35 kg/m2. The modest association observed between BMI and the ExTEM MCF G score with exploratory analysis, though showing a consistent relationship between BMI and hypercoagulability, could possibly be due to confounders. Further work in a larger cohort is needed to validate these findings.
Footnotes
Acknowledgements
We would like to thank the consultants in the anaesthesia and orthopaedic departments, The Prince Charles Hospital, Brisbane, Australia, for allowing us to recruit their patients and for assisting us with the data collection. We also appreciate the assistance from Pathology Queensland and from Dr Chris Clifton Chan, Resident Medical Officer, The Prince Charles Hospital, during the study period. We also acknowledge the contribution by Dr Nina Raju, haematologist, and Dr Scott Mckenzie, cardiologist, at The Prince Charles Hospital during the initial discussion for this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was partly funded by The Prince Charles Hospital Foundation New Investigator grant (ref. NI2015-126) and by Haemoview Diagnostics® (for the consumables).
