Abstract

‘There are numerous toxic states in which the eliminating organs of the body, most especially the kidneys, are incapable of removing…the natural or unnatural substances whose accumulation is detrimental to life. In the hope of providing a substitute in such emergencies, which might tide over a dangerous crisis…a method has been devised by which the blood of a living animal may be submitted to dialysis outside the body, and again returned to the natural circulation.’ John J. Abel, Leonard G. Rowntree and Benjamin B. Turner, May 1913.
1
Patient undergoing haemodialysis using the Portable/Wearable Artificial Kidney (WAK) developed in the Division of Artificial Organs, University of Utah, c. 1975. Image courtesy of the Spencer S. Eccles Health Sciences Library, University of Utah.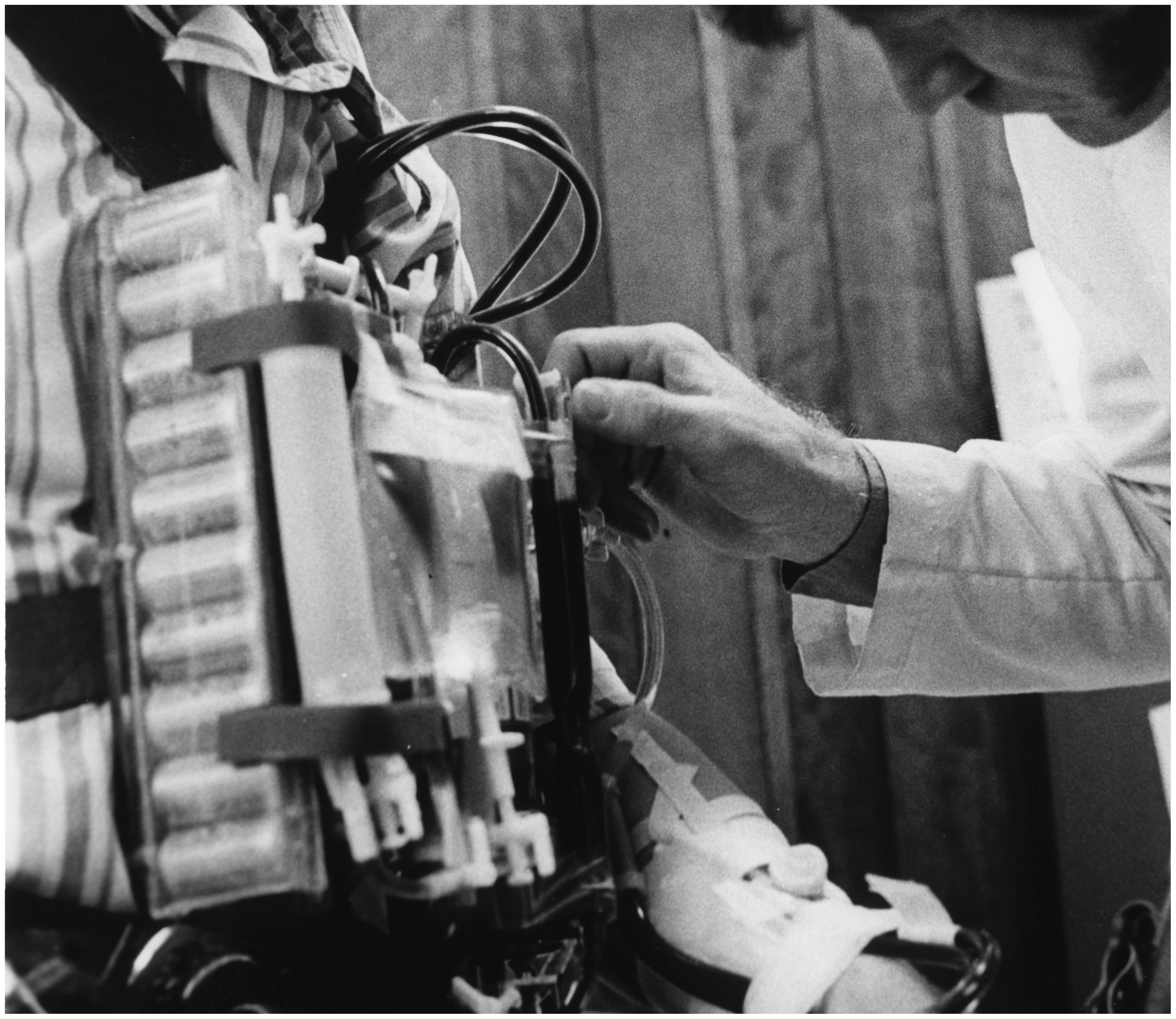
In 1912, John Abel and colleagues from the Pharmacological Laboratory, Johns Hopkins Medical School commenced animal experiments with a novel ‘vividiffusion’ apparatus.1,4 This comprised a series of semi-permeable tubes made from colloidion (cellulose nitrate), contained within a glass jacket filled with saline dialysate. Hirudinated blood entered the colloidion tubes from a cannula in the animal’s femoral artery and was returned to the femoral vein. Then one year later, Abel demonstrated the device at the 17th International Medical Congress in London, where it ‘excited the liveliest interest’. 5 The Times newspaper reported ‘Professor Abel has constructed what is practically an artificial kidney … it is possible that the principle may ultimately be adopted in the treatment of disease’. 6 Despite this early promise, Abel’s group confined their endeavours to quantitative analysis of substances appearing in the dialysate and never attempted the use of vividiffusion as a means of renal replacement therapy.
A decade later, in Giessen, Germany, Georg Haas was the first to perform experimental haemodialysis (HD) in humans with severe uraemia. Beginning in October 1924, Haas utilised a dialyser constructed from colloidion tubes, which he painstakingly prepared and sterilised himself; hirudin (and later heparin) was used to prevent coagulation.7,8 Although he concluded that ‘blood purification by dialysis can be carried out in humans without exposing the patient to injury’, 7 his work received little recognition from colleagues and Haas abandoned experimentation in 1928.
Around the same time, the Chicago-based Visking Corporation began marketing NOJAX® synthetic sausage casings. 9 Made from regenerated cellulose (cellophane), these were supplied as long, thin tubes and used in the manufacture of skinless hot dogs (the casings were removed once the sausages had been cooked). Chemists quickly discovered that these served as effective membranes for laboratory dialysis and ultrafiltration, 10 and in 1937, William Thalhimer constructed an artificial kidney from 30 cm lengths of NOJAX® tubing, immersed in physiological saline. This was successfully utilised to dialyse nephrectomised dogs, using heparin as an anticoagulant. 11
During the early 1940s, apparatus for human HD was simultaneously and independently developed in the Netherlands, Canada and Sweden by Willem Kolff, 12 Gordon Murray, 13 and Nils Alwall 14 respectively. All three were influenced by Thalhimer’s work 15 and employed cellophane sausage casings in their designs. Kolff’s rotating drum dialyser was the first of these devices to emerge. Fabricated in the Enamel Works, Kampen, in late 1942, the prototype made use of aluminium salvaged from a downed Luftwaffe aircraft, a Ford Model T water pump and a sewing machine motor. 16
HD was initially trialled in patients with acute renal failure (ARF) due to a variety of causes, including sulphonamide crystalluria, 17 rhabdomyolysis 18 and mercury bichloride poisoning. 19 Patients had often been anuric for several days prior to the institution of therapy. By 1949, Murray reported survival of approximately 50% of patients managed with HD. Those who died ‘were found on the whole to have had chronic kidney disease, and even though purification of the blood was accomplished satisfactorily, they relapsed into a uraemic state’. 20 Despite these results, many remained sceptical of HD. An accompanying editorial in the British Medical Journal described the artificial kidney ‘as still only an ideal and not an accomplished fact…its translation into practice brings difficulties to the doctor and real hazards to the patient’. 21
The use of HD for ARF gained widespread acceptance during the 1950s, 22 spurred on in part by the excellent outcomes achieved in a United States Army Renal Insufficiency Centre, established during the Korean War. 23 In 1960, the introduction by Wayne Quinton and Belding Scribner of Teflon arteriovenous shunts 24 solved many of the problems associated with vascular access and facilitated the use of HD as a maintenance therapy for end-stage renal disease. 25 Long-term HD imposed significant limitations on patients’ lifestyles, and in the mid-1970s, a team at the University of Utah experimented with a Portable/Wearable Artificial Kidney. 26 This weighed 17 kg, excluding dialysate, and proved to be an idea ahead of its time.
In 1977, in Göttingen, Germany, Peter Kramer described the use of continuous arteriovenous haemofiltration (CAVH) as a means of managing diuretic-resistant fluid overload. 27 This employed a hollow fibre haemofilter (originally designed as an alternative to HD for chronic renal failure) 28 and produced 300–600 ml/hr of ultrafiltrate (plasma water and solutes up to a certain molecular weight) by convection. The simple, pumpless system made use of temporary dialysis catheters sited in the patient’s femoral artery and vein and could be rapidly established in critically ill patients.
Using an isotonic salt solution for fluid replacement, CAVH was soon extended to the management of ARF 29 and in 1982, Kramer presented his experience with its use in more than 150 intensive care patients at a meeting of the American Society for Artificial Internal Organs. 30 This served as ‘an epiphany’ for those in the audience, 31 and the technique was quickly adopted in many intensive care units. CAVH was superseded by continuous venovenous haemofiltration in the mid-1980s and several other forms of continuous renal replacement therapy subsequently emerged.
