Abstract
Lung gas exchange efficiency deteriorates during general anaesthesia due to ventilation–perfusion (V/Q) scatter. Propofol total intravenous anaesthesia (TIVA) may preserve V/Q matching better than inhalational agents. We compared V/Q matching in patients randomized to either TIVA or sevoflurane anaesthesia, using deadspace and shunt measurements and the MIGET (Multiple Inert Gas Elimination Technique). Baseline arterial blood and mixed expired gas sampling was done before induction and repeated after one to two hours of relaxant general anaesthesia in 20 patients, supine with controlled ventilation at an FiO2 of 0.3 and a target end-tidal PCO2 of 30–35 mmHg. Blood samples for MIGET were processed after headspace equilibration by gas chromatography. The primary endpoint was a comparison of the two groups in the change from baseline of absolute difference between log standard deviation of ventilation and blood flow distributions (∂(σV−σQ)). Deadspace fraction increased and PaO2/FiO2 ratio decreased across both groups overall with anaesthesia, but change in deadspace was not different between groups (mean (standard deviation, SD) sevoflurane 21.8% (11.7%) versus TIVA 20.5% (10.6%), P = 0.601). Change in PaO2/FiO2 ratio was also similar between groups (mean (SD) sevoflurane −51.9 (69.1) mmHg versus TIVA −78.3 (76.9) mmHg, P = 0.43), as was change in shunt fraction (δQs/Qt mean (SD) sevoflurane −5.1% (12.6%) versus TIVA 0.4% (7.7%), P = 0.174). The primary endpoint ∂(σV−σQ) was not different between sevoflurane and propofol TIVA groups (mean (SD) 0.17 (0.81) versus 0.17 (0.29), P = 0.94). TIVA did not better preserve V/Q matching in patients undergoing anaesthesia with controlled ventilation compared with sevoflurane.
Keywords
Introduction
The efficiency of lung gas exchange deteriorates after induction of general anaesthesia due to a number of factors which contribute to impaired ventilation–perfusion (V/Q) matching with increased V/Q inhomogeneity or “scatter” in the lung. 1 Blood gas data from previous studies mainly in the setting of one-lung anaesthesia have suggested that propofol total intravenous anaesthesia (TIVA) may preserve V/Q matching better than inhalational agents, but with mixed results.2–5 There are limited data comparing V/Q matching with propofol TIVA and inhalational anaesthesia in routine two-lung ventilation. 2 A difference if confirmed might have implications for the choice of anaesthesia in patients with impaired lung gas exchange or lung disease undergoing surgery. These previous studies have used only routine respiratory blood gas–derived indices to estimate any treatment effect.
The Multiple Inert Gas Elimination Technique (MIGET) is a well-established method for more precise characterization of V/Q distributions in the lung, which can be adapted to use during general anaesthesia.6–14 The MIGET uses measured partial pressures, in arterial blood and expired breath, of six volatile gases with widely differing blood solubilities which are delivered intravenously at trace concentrations, to construct distributions of ventilation and blood flow ratios in the lung from retention and excretion curves calculated from the Fick equation describing mass balance for each gas in 50 lung compartments across a wide range of V/Q ratios.6–8 We compared the change between baseline (pre-anaesthesia induction) and after one to two hours of anaesthesia, in V/Q matching in patients receiving relaxant general anaesthesia, randomized to either propofol TIVA or sevoflurane maintenance anaesthesia. This was done using both standard indices derived from blood gas measurements (deadspace and shunt fraction measurements) and the MIGET.
Materials and methods
Ethics committee approval was obtained (Austin Health H2012/04591 May 2012) and trial registration lodged with the Australian and New Zealand Clinical Trials Registry (ACTRN12614000226606, 4 March 2014). Clinical Trials Notification was lodged with the Therapeutic Goods Administration in Australia for conduct of the MIGET.
Following informed consent, adult patients aged 18–85 years were recruited to the study who were undergoing elective non-laparoscopic surgery under relaxant general anaesthesia planned to take at least one hour and in whom placement of an arterial blood pressure monitoring line was considered appropriate for clinical purposes for cardiovascular monitoring by the attending anaesthetist. All subjects were in the supine position, and had normal lung function on history and examination, or spirometry (forced expiratory volume in one second, (FEV1) or forced vital capacity, (FVC) > 70% predicted) and body mass index (BMI) < 35 kg/m2. Participating consented patients were randomized using sealed envelopes shuffled prior to sequential numbering.
After arrival in the operating room, intravenous (IV) access appropriate for anaesthesia and surgery was established and an intravenous (IV) infusion commenced at 3 mL/minute of a pre-prepared 1 L sterile bag of normal saline containing a dissolved mixture of trace concentrations of six inert gases of widely ranging blood gas partition coefficients (λ) (sulphur hexafluoride (SF6 λ = 0.007), ethane (λ = 0.11), cyclopropane (λ = 0.47), halothane (λ = 2.5), diethyl ether (λ = 13.9) and acetone (λ = 417)) used for purposes of the MIGET according to standard methodology as described by Roca and Wagner. 8 The infusion was run for a minimum of 30 minutes prior to collection of samples for the baseline MIGET and continued throughout the experiment, while an arterial blood pressure monitoring line was placed under mild midazolam sedation and local anaesthesia, and the patient connected to standard clinical anaesthesia monitors, which included electrocardiograph (ECG), pulse oximetry, Vigeleo/FloTrac continuous pulse waveform cardiac output monitor (Edwards Lifesciences, Irvine, CA, USA), bispectral index monitoring of anaesthetic depth (Aspect Medical, Norwood, MA, USA), and tidal capnography, oxigraphy and inhaled agent monitoring (Zeus®, Draeger Medical, Lubeck, Germany).
Sampling for baseline (pre-anaesthesia) MIGET measurements was then performed. With an occlusive nose peg applied, the patient quietly breathed room air into a custom-designed, low resistance, gas-tight length of mixing tubing for two minutes, at which point a mixed expired gas sample was collected into a gas-tight 50 mL glass syringe. Minute ventilation was measured using a Wright spirometer and cardiac output measured using the Vigeleo/FloTrac.
Three arterial blood samples (total approximately 30 mL) were simultaneously collected into gas-tight heparinised glass syringes for subsequent blood gas measurement using a radiometer ABL 625 blood gas analyser, and for analysis of MIGET gas partial pressures. This was done by addition of an equal 10 mL of high-purity nitrogen headspace gas to each glass syringe and subsequent equilibration in an agitated warm water bath at 36°C for a minimum of 45 minutes, prior to gas-tight transfer of the headspace gas into a 10 mL glass syringe for measurement.
Partial pressures of the six MIGET gases in both headspace gas from arterial blood samples and in mixed expired gas samples were measured by an Agilent 7890A gas chromatograph fitted with a flame ionization detector and electron capture detector. Gas separation was achieved using a 30 m length, 0.32 mm internal diameter PLOT capillary column (JW Technologies, Denver, CO, USA), with oven temperature ramped up from 150 to 250°C over eight minutes. A high degree of linearity (r2≥ 0.99) for all gases was confirmed using serial dilution across a 250-fold concentration range, which encompassed the concentrations encountered in the patient sample under investigation. Clear separation of chromatographic peaks for cyclopropane and halothane from that for sevoflurane were achieved.
Mixed expired and arterial CO2 partial pressures and arterial oxygen partial pressure were measured to allow shunt fraction (venous admixture) to be estimated by the MIGET program and to calculate physiological deadspace using the Bohr–Enghoff equation.
Anaesthesia protocol
Following preoxygenation, anaesthesia was induced with propofol 1–2 mg/kg and neuromuscular blockade and an opioid at doses according to the anaesthetist’s judgement. After insertion of an endotracheal tube and confirmation of correct position, the patient was connected to the circle breathing system of the anaesthesia machine (Zeus®, Draeger Medical, Germany), and volume-controlled ventilation of the lungs was commenced at a rate of 10–12 breaths/minute and tidal volume of 7–10 mL/kg with no positive end-expiratory pressure, so as to maintain an end-tidal PCO2 of 30–35 mmHg as measured by capnography. The fresh gas flow rate was set to a rate equal to minute ventilation to prevent expired gas recirculation, with an FiO2 of 30% or as near as possible to this for maintenance of an SpO2 of not less than 96%. Body temperature monitoring and maintenance with a forced-air warming device was started.
Maintenance of anaesthesia post-induction and intubation was according to randomization. The Inhalational group (Group Sevo) had maintenance of anaesthesia with sevoflurane in oxygen/air. The TIVA group (Group TIVA) had maintenance of anaesthesia with propofol intravenously. In both groups the delivered concentration or infusion rate was adjusted to maintain a bispectral index (BIS) of 40–60.
After one hour of surgery, the mixing tubing was connected into the expiratory limb of the breathing system. After establishment of steady haemodynamics and ventilation for a minimum of ten minutes, a second set of blood and mixed expired gas samples was taken and other variables recorded in similar fashion to those taken at baseline. Minute ventilation was measured from the spirometer in the breathing system.
Statistical analysis and sample size
The log standard deviation (σ) of each distribution of ventilation and blood flow, equivalent to the second moment of the distribution plotted on a logarithmic scale, was calculated by the MIGET program. We sought a single metric of V/Q scatter itself, rather than separate measures of distribution of V and Q, which may be harder to interpret in combination. The absolute difference between σ of ventilation and blood flow was used as the primary measure of V/Q scatter for each set of measurements (σV−σQ), as described by Colburn et al. 15 Where the distributions of either or both V and Q widen along a horizontal axis of V/Q ratio, the degree of mismatch between them will also be expected to increase. For any patient, MIGET data was accepted for inclusion in pooled analysis only if the residual sum of squares (RSS) of both baseline and intraoperative retention and excretion curves calculated by the MIGET program were less than 10. The RSS is a statistical measure of goodness of fit of measured curves to predicted smooth retention and excretion curves across the range of solubilities of the MIGET gases, and is an index of raw data quality and reliability in MIGET measurements. The presence of outliers, due to artefact or measurement error for one or more MIGET gas species, will produce an excessive RSS. V/Q distributions generated with an excessive RSS should be viewed sceptically, and we prospectively rejected them from the final analysis. The threshold of RSS of ten we chose is arbitrary but consistent with previously published experience and guidelines. 8
The primary endpoint for the trial was the change in (σV−σQ) from baseline to intraoperative measurement (∂(σV−σQ)) for each patient. A relative difference between the groups in ∂(σV−σQ) of 20% or more was considered significant.
From previously published studies using older anaesthetic agents such as halothane or enflurane,9–14 a change in σ of 50% above awake baseline values is typical after establishment of anaesthesia, with a range of around 25% either side of this. Using a t-test for unpaired data, at an alpha of 0.05 and a beta of 0.9, a minimum of 16 patients was required. To allow for more modest changes with modern anaesthetic agents, and to allow for some data attrition, recruitment of 24 patients was stipulated.
Secondary endpoints were change from baseline in physiologic deadspace, shunt fraction calculated from V/Q distributions generated by the MIGET program, and PaO2/FiO2 ratio calculated from blood gas measurements.
Analysis of data consistent with a normal distribution on the Shapiro–Wilk test was done using a two-tailed t-test for unpaired data, and non-normal data were compared using the Kruskal–Wallis test, in STATA 12.
Results
A total of 24 patients (ten male and 14 female) were recruited. All patients were able to complete the baseline data collection and subsequent intraoperative data sampling was collected from all patients. However, in two patients in each group, data was excluded due to excessive RSS in retention/excretion curves generated by the MIGET program in one or both of the baseline and intraoperative samples, leaving a total of 20 patients (ten in each group) for analysis.
Table 1 shows demographic data from the included patients. There were no significant differences between the two groups in baseline demographics. Table 2 shows the primary and secondary outcome data by group.
Demographic data for patients.
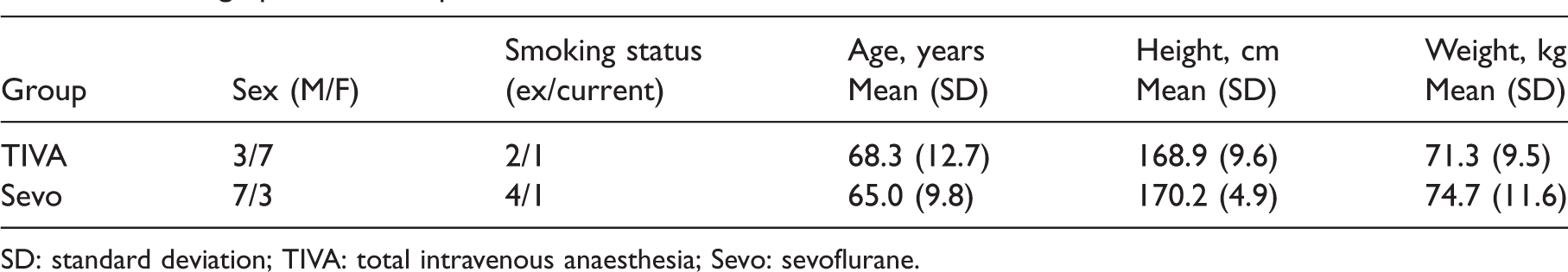
SD: standard deviation; TIVA: total intravenous anaesthesia; Sevo: sevoflurane.
Primary and secondary outcome data by group.
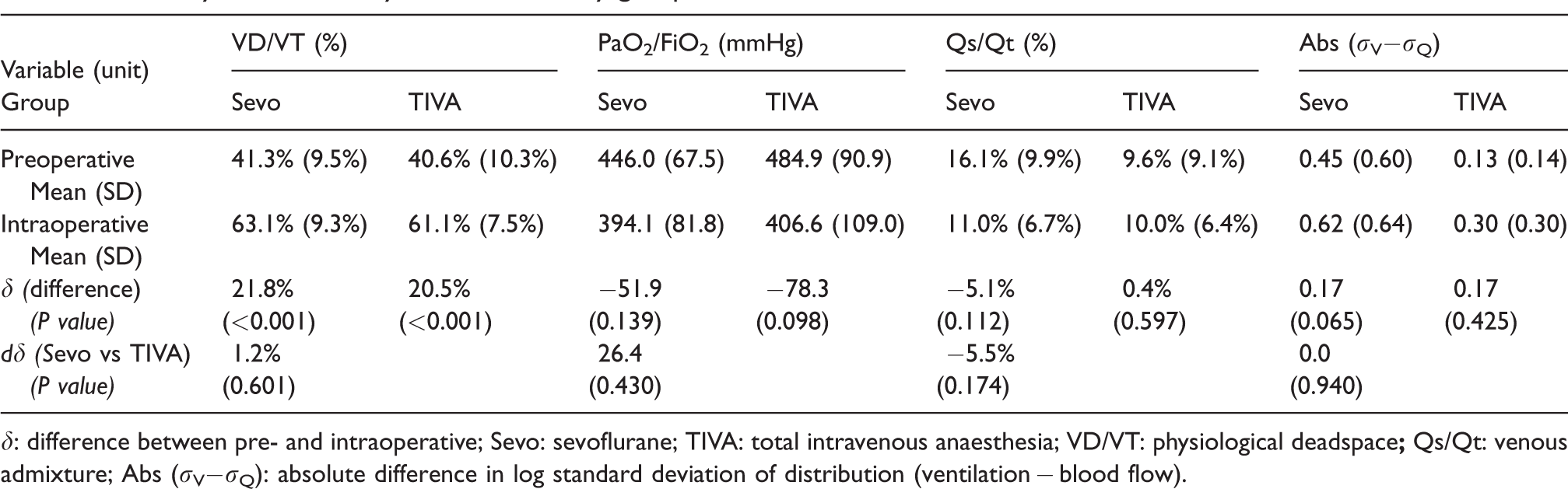
δ: difference between pre- and intraoperative; Sevo: sevoflurane; TIVA: total intravenous anaesthesia; VD/VT: physiological deadspace
Changes from baseline to intraoperative
Physiological deadspace fraction (VD/VT) increased (see Table 2) with anaesthesia from baseline in both groups and overall (mean (standard deviation, SD) 40.9% (9.7%) to 62.1% (8.3%), P < 0.0001). The PaO2/FiO2 ratio decreased (465.4 (80.5) to 400.3 (94.0) mmHg across both groups overall, P = 0.024), but shunt fraction was not significantly changed in either group or overall (12.8% (9.8%) to 10.5% (6.4%), P = 0.58).
Comparison of changes between groups
Change in physiological deadspace fraction (δVD/VT) was not significantly different between groups (mean (SD) sevo 21.8% (11.7%) versus TIVA 20.5% (10.6%), P = 0.601). The change in PaO2/FiO2 ratio was also similar between groups (mean (SD) sevo −51.9 (69.1)mmHg versus TIVA −78.3 (76.9) mmHg, P = 0.43), as was change in shunt fraction (δQs/Qt mean (SD) sevo −5.1% (12.6%) versus TIVA 0.4% (7.7%), P = 0.174). The primary endpoint ∂(σV−σQ) was not different between sevoflurane and propofol TIVA groups (mean (SD) 0.17 (0.81) versus 0.17 (0.29), P = 0.94).
Discussion
We found no evidence in patients with normal baseline lung function that propofol TIVA resulted in better preservation of V/Q matching in the lung than anaesthesia with sevoflurane. This finding was consistent across all our measures, including MIGET derived variables and traditional indices of V/Q matching based on both CO2 elimination and oxygenation.
Both groups had significant increases in alveolar deadspace following induction of anaesthesia and controlled ventilation, as expected from previous studies. 1 PaO2/FiO2 ratio decreased overall as well, although shunt fraction estimated by MIGET was highly variable and did not change significantly in either group or overall. The greater variability of oxygenation-based indices of V/Q mismatching than those based on carbon dioxide partial pressure measurements may explain why we found no statistically significant effect of anaesthesia on oxygenation in each treatment group in our study, which would mainly reflect effects on low V/Q lung regions and shunt calculations.
Our results contrast with findings of previous workers investigating a similar population. Praetel et al. reported a comparison of propofol TIVA and isoflurane anaesthesia in 40 ventilated patients undergoing elective surgery in the supine position, using a randomized crossover design with patients acting as their own controls. 2 They showed a greater decrease from pre-anaesthesia baseline values in PaO2/FiO2 ratio and increase in physiological deadspace calculated from capnographic waveform analysis, with isoflurane. It is unclear whether our contrasting findings reflect a difference in the pharmacology of sevoflurane compared to the older inhalational agent, or some other factor. This possibility is not supported by data from a study in a pig model of low V/Q induced by pneumoperitoneum, where pulmonary shunt fraction was increased with sevoflurane but not isoflurane relative to propofol TIVA. 16 However, these comparisons are made difficult by heterogeneity in the mechanisms for inducing V/Q mismatch between different studies and populations.
Much of the change from baseline in indices of V/Q matching we measured relate to the direct effect of controlled positive pressure ventilation (PPV). A typical example of change in V/Q distributions measured by the MIGET in our study is shown in Figure 1, where introduction of PPV produced a secondary peak, representing the development of high V/Q lung units not present in the preoperative spontaneous breathing situation. This may explain why we showed no significant differences in V/Q scatter of our MIGET-calculated primary endpoint (∂(σV−σQ)) between groups, or in either group between pre- and post-induction measurements, despite clear changes in alveolar deadspace. Our sample size calculations allowed for a 20% difference between groups in the primary endpoint to be detected, but assumed a much smaller variability of change from baseline of log standard deviation of ventilation (but not blood flow) than we observed, which was more than twice the 50% range we had assumed, due to the common occurrence of bifid peaks such as shown in Figure 1b. Bifid V/Q distributions depart from the theoretical distributions that underlie our nominated primary endpoint and make it hard to interpret. Nevertheless, our alveolar deadspace and oxygenation-based measurements make it clear that no difference between treatment groups in V/Q scatter was present in our study.
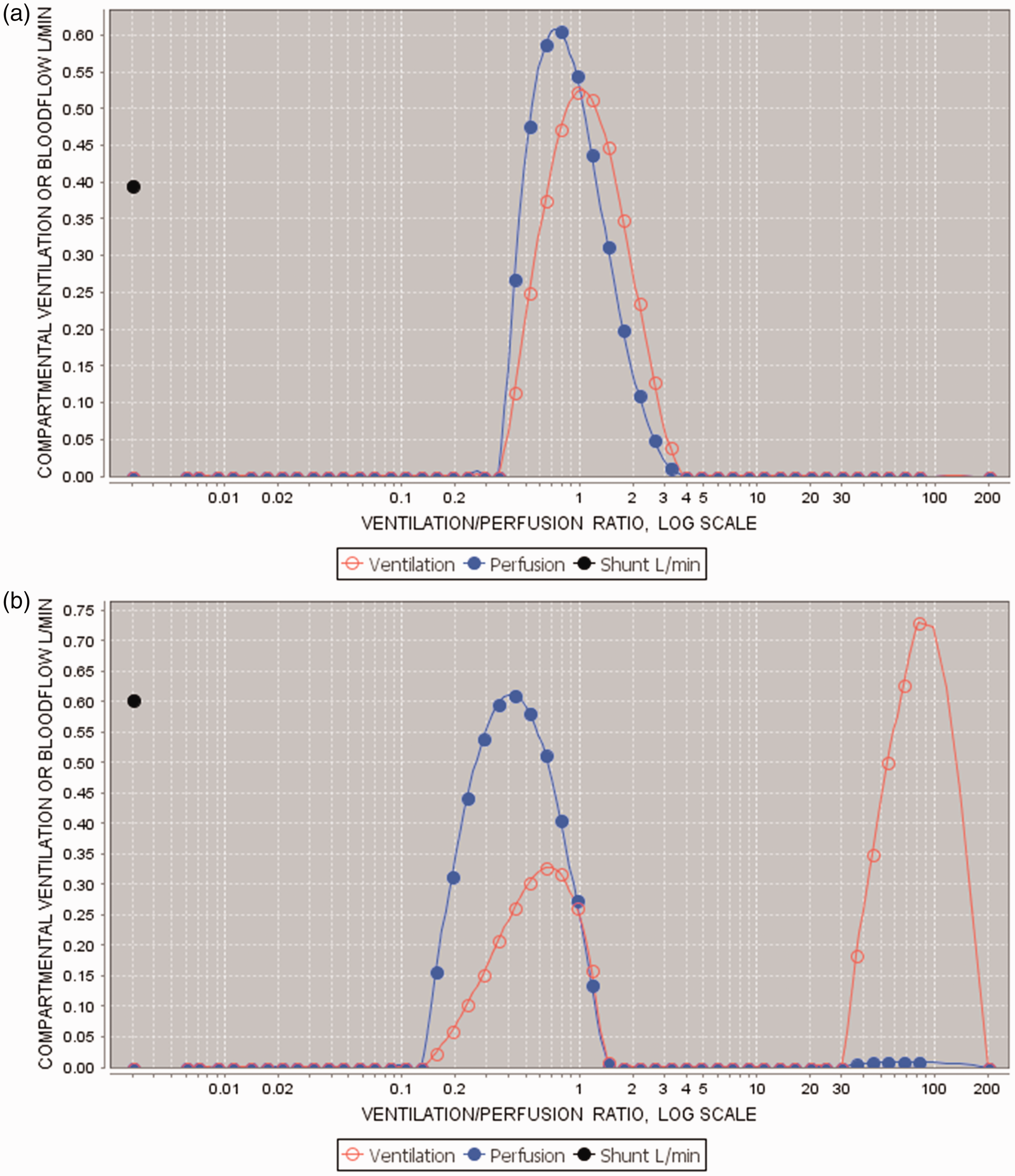
(a) Baseline data from one patient: σV = 0.47, σQ = 0.45, σV−σQ = 0.02. (b) Intraoperative data from same patient: σV = 2.39, σQ = 0.69, σV−σQ = 1.70, ∂(σV−σQ) = 1.68.
Interestingly, Nyren et al. found in healthy volunteers undergoing spontaneous ventilation inhalational anaesthesia with sevoflurane that the distribution of blood flow but not of ventilation, measured using electrical impedance tomography, was altered from pre-anaesthesia baseline, suggesting that anaesthesia affects mechanisms such as hypoxic pulmonary vasoconstriction, leading to less favourable V/Q matching. 17 While, consistent with this concept, oxygenation efficiency appears to have deteriorated overall with anaesthesia in our patient sample who were ventilated, we could show no difference between treatment groups.
A great deal of data comparing propofol TIVA and inhalational anaesthesia comes from studies in the setting of one-lung ventilation (OLV) for thoracic surgery, with mixed findings. Kellow et al. found in 22 patients that shunt fraction increased substantially more in patients randomized to isoflurane than propofol TIVA after instigation of OLV for open lung resection. 3 Abe et al. in 20 patients undergoing oesophageal surgery found that shunt fraction increased when anaesthesia was changed from propofol to either isoflurane or sevoflurane during OLV. 4 However, Reid et al. could find no difference in oxygenation in 30 patients randomized with crossover design to propofol-alfentanil or isoflurane anaesthesia during OLV for thoracic surgery. 5 Other factors have been suggested to be potentially important to consider in choice of anaesthesia for lung surgery, including the effects of immunosuppression on rates of lung reperfusion injury or postoperative infection. In a recent multicentre trial of 460 patients, there was no evidence that choice of anaesthetic affected risk of adverse postoperative pulmonary outcomes or other complications after lung surgery involving OLV. 18
Arguably, the clinical implications of a difference in preservation of V/Q matching and lung gas exchange efficiency from anaesthetic choice would be greater for patients with impaired baseline lung gas exchange, such as patients with obstructive or restrictive pulmonary disease or systemic sepsis, than for patients with normal baseline lung function such as we studied. However, the mixed findings of these previous studies and ours in patients with normal baseline lung function and in thoracic surgery patients do not provide encouragement that a significant treatment effect of choice of anaesthesia is likely to be found in these higher risk patient populations.
In summary, we could find no benefit of propofol TIVA over sevoflurane anaesthesia in preservation of V/Q matching in the lung, and type of general anaesthesia has no clear influence on lung gas exchange efficiency.
Footnotes
Acknowledgements
Thanks to Chris Stuart-Andrews, Philip Zeglinsky and Andrew Ellis for their technical assistance.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Philip J Peyton: None; Harry Marsh: None; Bruce R Thompson: None.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a project grant from the Australian and New Zealand College of Anaesthetists.
