Abstract
There is a recognition of the contribution to global warming from emissions of anaesthetic gases into the atmosphere. We audited sevoflurane use to help guide future initiatives to reduce consumption. We observed sevoflurane use during paediatric anaesthesia in a single operating theatre over eight weeks. We recorded demographics, timing of induction and maintenance of anaesthesia, type of circuit used and amount of liquid sevoflurane used (in mL). Ninety-four cases were available for analysis. Of these, 65 had gas inductions and 29 had intravenous (IV) inductions. The median sevoflurane use was 19 mL (interquartile range, IQR 13–24 mL). The median duration of cases was 50.5 min (IQR 35–78 min). The median sevoflurane consumption for cases with a gas induction was 22 mL (IQR 16–26 mL) and for those with an IV induction was 11 mL (IQR 7–17 mL; P < 0.00001). The duration of cases for the gas and IV induction cohorts were similar. During maintenance of anaesthesia, there was no difference between the IV and gas induction cohorts. There was little difference in sevoflurane use between the T-piece and circle system groups. Cases performed with gas inductions consumed twice the sevoflurane as those with IV inductions. Future interventions to reduce sevoflurane consumption should focus on this period.
Introduction
Climate change will likely have a substantial impact on the environment, economy, infrastructure and even global health. 1 There is an increasing awareness that the healthcare sector contributes to global warming.2,3 For anaesthetists, our contribution is largely from emissions of anaesthetic gases into the atmosphere. 4
During paediatric anaesthesia, sevoflurane is commonly used for both induction and maintenance of anaesthesia. Sevoflurane is a greenhouse gas with a global warming potential (GWP) 440 times that of CO2. 5 To reduce sevoflurane emissions during paediatric practice, we need first to improve our understanding of how sevoflurane is used during everyday delivery of anaesthesia.
Sevoflurane consumption is determined by the fresh gas flow (FGF) and the setting on the vaporiser dial. Many anaesthetists will employ higher FGFs during paediatric anaesthesia than in adult anaesthesia. High FGF is perceived as especially useful during the inhalational (gas) induction of anaesthesia because a high FGF will increase the speed of rise of sevoflurane concentration in the circuit to more closely match that on the vaporiser dial. Furthermore, during both induction and maintenance of anaesthesia, high FGFs will help overcome any leak or loss of volume from the circuit occurring during facemask ventilation, or around a supraglottic airway or uncuffed endotracheal tube.
This paper describes an observational audit investigating sevoflurane use in our department to help guide future initiatives and research to reduce consumption.
Methods
This audit was approved by the University of Otago, the Canterbury District Health Board, and Te Komite Whakarite, our hospital’s committee for consultation with Mãori.
Over an eight-week period during December 2017 and January 2018, we observed sevoflurane use in a single operating theatre and its associated induction room at Christchurch Hospital. This theatre is mostly used for paediatrics and is the only one equipped for anaesthetic room induction in our hospital. All cases during normal working hours, involving children 16 years of age or younger, were included. Cases performed include those from paediatric surgery, otorhinolaryngology, orthopaedics and plastic surgery. Anaesthetics were delivered by a consultant anaesthetist or a supervised trainee.
We recorded basic demographics (age and weight), timing of induction and maintenance of anaesthesia, type of circuit used, type of airway and amount of liquid sevoflurane used (in mL) during induction and maintenance. Liquid sevoflurane consumption data were taken from the logs of a GE Avance (GE Healthcare, Madison, WI, USA) anaesthesia machine in the induction room and a GE Aisys CS2 in the operating theatre. These machine logs have been shown to be accurate within 10% of the weighed amount of the volatile agent. 6 The GE Aisys CS2 machines were equipped with Et Control providing automated target control for the volatile agent. Breathing systems used were Limb-O™ circuits (CareFusion, San Diego, CA, USA) and Ayres T-pieces (Intersurgical Ltd, Wokingham, UK).
Children receiving a propofol infusion to maintain anaesthesia were excluded from the study.
We defined the induction period as the time from the commencement of anaesthesia until the patient was ready for positioning for surgery. Maintenance was defined as the time from the completion of induction until the patient was ready to leave the operating theatre.
Subjects’ ages and weights, and sevoflurane usage, were compared between the intravenous (IV) and inhalational induction groups using the two-tailed Mann–Whitney U test.
Results
We collected data from 112 anaesthetic episodes.
Eighteen cases were excluded. Cases were excluded because of lost or incomplete data (four cases), propofol infusions during the procedure (five cases) and those in which the GE Avance anaesthetic machine did not measure the amount of sevoflurane use during T-piece use via the auxiliary outlet (nine cases).
Ninety-four cases were available for analysis. Of these, 65 had gas inductions and 29 had IV inductions. The age and weight of patients involved are presented in Table 1. Those receiving IV induction tended to be slightly older and heavier than those receiving gas induction. Seventy cases were induced in the induction room and transferred to the operating theatre following induction. Twenty-four cases were induced in the operating theatre.
Patient age and weight of included cases.
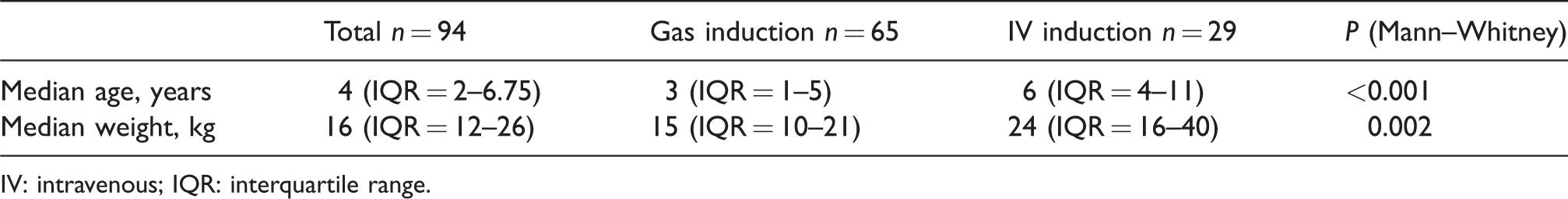
IV: intravenous; IQR: interquartile range.
Sevoflurane use during paediatric anaesthesia
The median amount of liquid sevoflurane used was 19 mL (interquartile range, IQR 13–24 mL). The median duration of the cases was 50.5 min (IQR 35–78 min) (Table 2).
Median liquid sevoflurane consumption in millilitres.

*Induction phase is defined as the time from the commencement of anaesthesia until the patient was ready for positioning for surgery. IV: intravenous; IQR: interquartile range.
The median sevoflurane consumption for cases with a gas induction was 22 mL (IQR 16–26 mL) and for those with an IV induction was 11 mL (IQR 7–17 min; P < 0.00001). The duration of cases for the gas induction cohort was similar (49 min; IQR 35–75 min) to those receiving IV induction (57 min, IQR 33–79 min; P = 0.79).
The total amount of sevoflurane used for the 70 patients induced in the induction room (median 19 mL, IQR 13–24 mL) was the same as for the 24 induced in the operating theatre (median 19 mL, IQR 13–26.5 mL).
Induction phase of anaesthesia
The median sevoflurane consumption was 13.5 mL (IQR 11–18 mL) during the gas induction phase and 4 mL (IQR 1–7 mL) for the IV induction phase (P ≤ 0.00001). Because the induction phase was defined as the time from the commencement of anaesthesia until the patient was ready for positioning for surgery, those cases induced with propofol also consumed some sevoflurane. Gas inductions took longer (median duration 10 min; IQR 8–13 min) compared with IV inductions (median 6 min; IQR 6–10.5 min; P = 0.002).
The maximum FGF seen was 6 L/min which is the default setting on our anaesthetic machines.
Of those 65 cases performed with an inhalational induction, 53 also used nitrous oxide. Only one case of the 29 IV inductions used nitrous oxide during the induction phase.
Maintenance of anaesthesia
Median sevoflurane use during the maintenance of anaesthesia was 7 mL (IQR 5–10 mL) with no difference between the IV induction and gas induction cohorts (median amount of sevoflurane consumed 8 and 7 mL respectively), P=0.53 (Table 2). The median duration of the maintenance phase was 43 min (IQR 27–71.5 min).
Data were collected on 77 of 94 cases on the use of Et Control during anaesthesia. Sixty-four of 77 cases (83%) used Et Control for all or part of the maintenance of anaesthesia.
No cases used nitrous oxide during maintenance of anaesthesia.
The effect of T-piece versus circle systems
There was little difference in sevoflurane use between the T-piece and circle system groups.
Of the gas induction cases, a T-piece was used during some part of the case in nine cases. A circle system was used for the entire case in 55. The median sevoflurane consumption for cases with a T-piece used during the case was 19 mL (IQR 15–24 mL) and for the circle system 22 mL (IQR 16–28 mL; P = 0.57).
Discussion
There is a strong desire to reduce the impact of healthcare on the environment. Anaesthesia is not excluded from this.2,3
Although anaesthesia accounts for a small percentage of overall global greenhouse gas emissions, the effect of anaesthetic gases is not insignificant.4,5 Desflurane has the greatest potential to impact global warming with a GWP20 (GWP over 20 year timeframe) of 6810. 5 Sevoflurane, in comparison, has a GWP20 of 440. 5 In Christchurch Hospital, sevoflurane is used during many more cases than desflurane. To illustrate, for the 12-month period from 1 July 2017 until 30 June 2018, 1198 (250 mL) bottles of sevoflurane were used at a cost of NZ$167,720. In contrast, 102 (240 mL) bottles of desflurane were used at a cost of NZ$22,815.51. A focus to reduce sevoflurane consumption will have both an environmental and economic benefit.
In this audit of practice at Christchurch Hospital we found that cases performed with a gas induction consumed a median of 11 mL more sevoflurane than those with an IV induction. There was little difference between the groups during the maintenance phase. The induction phase is thus critical to determine the amount of sevoflurane consumed during a case and future interventions to reduce sevoflurane consumption should focus on this period. Furthermore, adding nitrous oxide to sevoflurane increases greenhouse gas emissions significantly. 9 Most sevoflurane inductions in this cohort were performed with the addition of nitrous oxide.
High flows are commonly used during inhalational inductions to increase the speed at which the sevoflurane concentration in the breathing system approaches that on the vaporiser dial. The default setting on our machines is 6 L/min. Mathematically, the concentration in the circuit approaches the concentration delivered via FGF following an exponential time course described by a time constant (τ) which equals the volume of the breathing system divided by the FGF. 7 The volume of the system includes the volume of the circuit (1.6 L), the anaesthetic bag (typically 2 L) and the volume inside the anaesthetic machine and CO2 canister (2.7 L in the GE Aisys Carestation). For simplicity, if we estimate this volume at 6 L and the FGF is set at 6 L/min, after 1 min (τ) the concentration of sevoflurane in the circuit will be 63.2% of the concentration of the FGF. Therefore, if the vaporiser dial is set at 8%, the concentration after 1 min will be approaching 5% (excluding the uptake of sevoflurane by the patient). An FGF of 6 L/min with 8% sevoflurane will consume 480 mL of vaporised sevoflurane which represents approximately 2.6 mL of liquid sevoflurane. 8 Maintaining an FGF of 6 L/min at 8% sevoflurane beyond the first minute will continue to consume 2.6 mL/min of liquid sevoflurane, with most if not all of this being scavenged and lost into the atmosphere.
The most effective way to reduce the contribution of paediatric anaesthesia to greenhouse gas emissions is to consider methods to reduce volatile waste, particularly during the induction of anaesthesia. For anaesthetic agents, propofol probably has the least impact on greenhouse gas emissions, 9 and for this cohort, propofol induction halved the sevoflurane expenditure and almost eliminated the requirement for nitrous oxide.
However, inhalational inductions are, and will remain, a practical method for inducing anaesthesia especially for paediatric patients. Consideration can be given to what FGF is used during inhalation induction and to reduce the flow rate as quickly as practical. Further study to determine the optimum FGF for gas inductions will be helpful to guide practice.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
