Abstract
The importance of preventing hypothermia in the perioperative period cannot be overemphasised. The use of active warming devices is advocated and practised widely. The forced-air warming (FAW) blanket is currently a popular modality. The BARRIER® EasyWarm® (Mölnlycke Healthcare, Belrose, Australia) self-heating blanket, which does not require a power unit, has been proposed as an easy-to-use alternative to the FAW blanket. We conducted a single-centre, randomized controlled trial to compare the efficacy of the EasyWarm® self-heating blanket to the Cocoon blanket (Care Essentials, North Geelong, Australia), a conventional FAW blanket, in preventing intraoperative hypothermia. Forty patients undergoing elective surgery under general anaesthesia lasting >1 h were randomized in a 1:1 ratio. Prewarming was not allowed; 39 patients were analysed (19 EasyWarm® group and 20 Cocoon blanket group). There was no statistically significant difference between the two groups with regard to the mean (±standard deviation) final nasopharyngeal temperature (35.89 ± 0.82 °C versus 35.74 ± 0.77 °C; P = 0.72 in the Cocoon and EasyWarm® blanket groups, respectively). Similarly, the difference between average temperatures at 1, 2 and 3 h was not statistically significant. We found the EasyWarm® blanket to be as effective as the Cocoon FAW blanket in preventing intraoperative hypothermia. Due to its ease of use, the EasyWarm® blanket may have an additional benefit in preoperative warming of patients.
Introduction
Hypothermia is common in the perioperative period with prevalence varying between 50% and 90%. 1 The incidence of intraoperative hypothermia depends on the duration and type of surgical procedures, anaesthetic techniques, ambient temperature and patient demographics. 2 The drop in core body temperature is highest in the first hour of surgery due to redistribution of heat from the core to the periphery.3,4
Maintenance of intraoperative normothermia is one of the principal goals of the anaesthetist. Intraoperative hypothermia causes shivering, and patient discomfort postoperatively and leads to multiple complications. It has been shown to increase the risk of morbid cardiac events4,5 and surgical site infection, 3 impair coagulation, 6 increase transfusion requirement, 7 and impair immune function, 8 all of which contribute to prolonged hospital admission and increased health costs.9,10
Several studies have shown that active warming is more effective than passive warming. 11 Forced-air warming (FAW) blankets are currently the most commonly used method of intraoperative active warming. 12 There is an ever-increasing number of FAW systems available in the market, 13 which include the Cocoon FAW system, an inflatable blanket (Care Essentials, North Geelong, Australia). A new BARRIER® EasyWarm® self-heating blanket (Mölnlycke Healthcare, Belrose, Australia) has been proposed as an alternative to FAW. FAW devices consist of an electrical heating unit which drives warm air into the blanket. 13 An EasyWarm® blanket does not require a power unit therefore reducing trip hazard from the cables, theatre clutter, and noise from the heating unit. Instead, it consists of 12 pockets, which contain iron, activated coal, clay, water, salt, and sodium polyacrylate. These chemicals, when exposed to air, lead to an exothermic reaction, resulting mainly from oxidation of iron. The blanket then warms up to a maximum temperature of 44 °C in about 30 min that lasts for 10 h. The device is comparable in price to the commonly used FAW blankets, and, to the best of the authors’ knowledge, no complications with regards to burns have been reported. Also, it can be disposed of like other hospital waste by incineration. Due to its ease of use, it has the potential to be used to preoperatively warm patients.
The efficacy of the BARRIER® EasyWarm® blanket (a self-heating system) in minimization of intraoperative hypothermia has not been compared with a FAW device such as the Cocoon system. We hypothesized that there is no difference between the Cocoon FAW blanket and EasyWarm® self-heating blanket in minimizing intraoperative hypothermia. We conducted this randomized controlled trial to compare the efficacy of the two devices in patients having relaxant general anaesthesia lasting >1 h.
Materials and methods
This study was a single-centre, prospective, randomized controlled trial (RCT), and was conducted after approval from the Austin Health research ethics committee (LNR/14/Austin/611) and registration with the Australian New Zealand Clinical Trials Registry (ACTRN12615000278538). Blinding was not performed due to the obvious use of two different warming blankets. The ethics committee granted waiver of consent as our study was categorized under low and negligible risk research. Patients were recruited at the Austin Hospital, Melbourne, between February and July 2015.
Patients undergoing elective open or laparoscopic surgery requiring relaxant general anaesthesia in the supine position lasting >1 h were included. Patients with sepsis, untreated thyroid dysfunction, fever, age <14 years, body mass index (BMI) >40 kg/m2 or having operations in the lithotomy position were excluded.
A temperature difference of 0.5 °C between the two groups was considered clinically significant, as previously described. 3 A prospective sample size calculation stipulating a clinically important difference of 0.5 °C in final temperature (primary outcome variable) suggested a sample size of 31 patients (α = 0.05; β = 0.8; standard deviation (SD) = 0.5 °C). To account for possible dropouts, a total of 40 patients were recruited. Randomization was performed by random selection of shuffled opaque envelopes containing group allocation into Cocoon FAW blanket or EasyWarm blanket in a 1:1 ratio.
Preoperative warming was not performed. After obtaining intravenous (IV) access in an anaesthetic bay, 1 L of compound sodium lactate at room temperature was connected to the patient. Subsequent fluids used were from a warming cabinet (temperature 42 °C). Choice of anaesthetic agent, monitoring, and fluid management were left to the discretion of the attending anaesthetist. The room temperature, then baseline temperature, of the patient were recorded using a nasopharyngeal probe, after induction of anaesthesia before warming blankets were applied.
For patients in the Cocoon FAW group, prior to surgical draping, a single rectangular Cocoon blanket was placed over the lower limbs and as much of the abdomen as was accessible outside the surgical field, and set to 42 °C on the power unit once draping was completed, unless the patient temperature rose above 37.5 °C, when the unit was turned off. The upper body and outstretched arms were covered with the patient’s standard issue unheated blanket, consistent with hospital standard practice. If patient temperature fell below 34 °C, a second Cocoon FAW device and upper body blanket were obtained and deployed.
For patients in the EasyWarm® group, blankets were opened to air and activated 30 min prior to commencement of surgery. The EasyWarm® blanket consists of upper and lower halves joined by Velcro® (VELCRO, London, UK) lining, which were divided and placed over the lower limbs/abdomen and the upper body/arms prior to draping.
Data were collected at the point of care by the attending anaesthetist. Demographic data collected included age, sex, weight and height. Other intra-operative data included room temperature, baseline nasopharyngeal temperature of the patient, then patient temperature every hour for up to 6 h and final temperature at completion of wound closure. Type and duration of surgery, whether or not regional anaesthesia was performed in addition to general anaesthesia, volume of fluid used, use of sequential compression devices (SCDs), use of different drugs including paracetamol, non-steroidal anti-inflammatory drugs (NSAIDs) and dexamethasone were all recorded.
Data analysis was performed using Stata12.0 (StataCorp College Station, TX, USA). Continuous data was tested for normality using the Shapiro–Wilk test. Normally distributed data were described by the mean and standard deviation (SD) and skewed data by median and inter-quartile range. Normally distributed data was compared using the unpaired t-test and the Wilcoxon rank-sum test was used to compare skewed data.
Results
Forty patients were recruited; however, positioning of one patient was changed to the lithotomy position during the surgery and this patient’s data were removed from the analysis. Data for 39 patients were analysed (20 Cocoon FAW blanket and 19 EasyWarm® blanket) (Figure 1). Both groups were comparable with regard to age, gender, BMI and baseline patient temperature (Table 1).
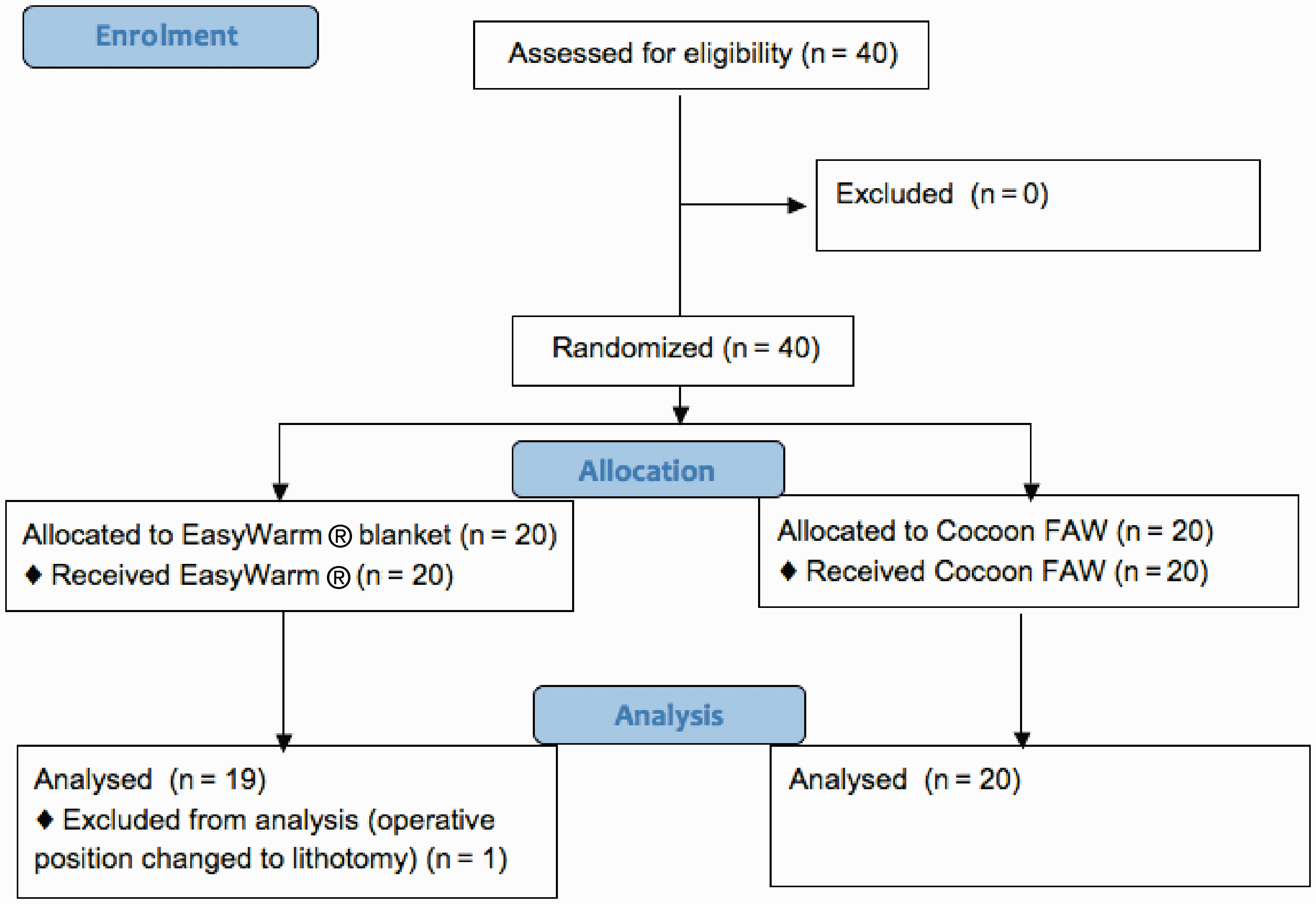
Participants’ progress through the phases of the trial.FAW: forced-air warming. Cocoon system (Care Essentials, North Geelong, Australia); EasyWarm® ((Mölnlycke Healthcare, Belrose, Australia).
Demographics of the patients.
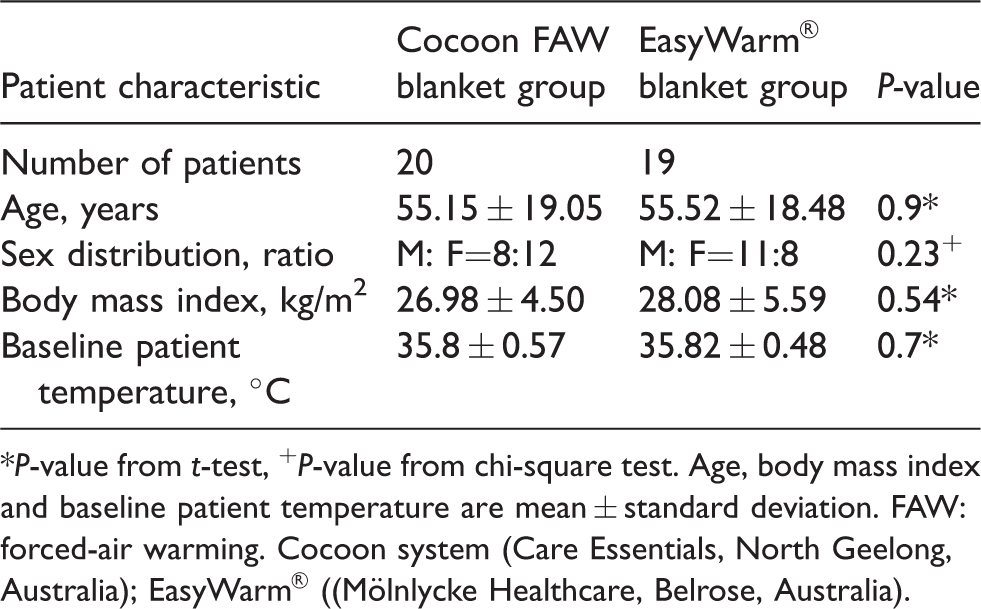
*P-value from t-test, +P-value from chi-square test. Age, body mass index and baseline patient temperature are mean ± standard deviation. FAW: forced-air warming. Cocoon system (Care Essentials, North Geelong, Australia); EasyWarm® ((Mölnlycke Healthcare, Belrose, Australia).
The temperature of one patient from the Cocoon Blanket group dropped to below 34 °C intraoperatively and an additional Cocoon power unit and upper body FAW blanket were deployed in accordance with the protocol. Active warming continued throughout surgery for both groups as no patient reached a temperature above 37.5 °C. Room temperature, anaesthetic techniques, volume of infused intravenous fluid, use of medications and SCDs in both groups were similar (Table 2). There were no statistically significant differences between types and duration of surgery (Table 2).
Intraoperative data.
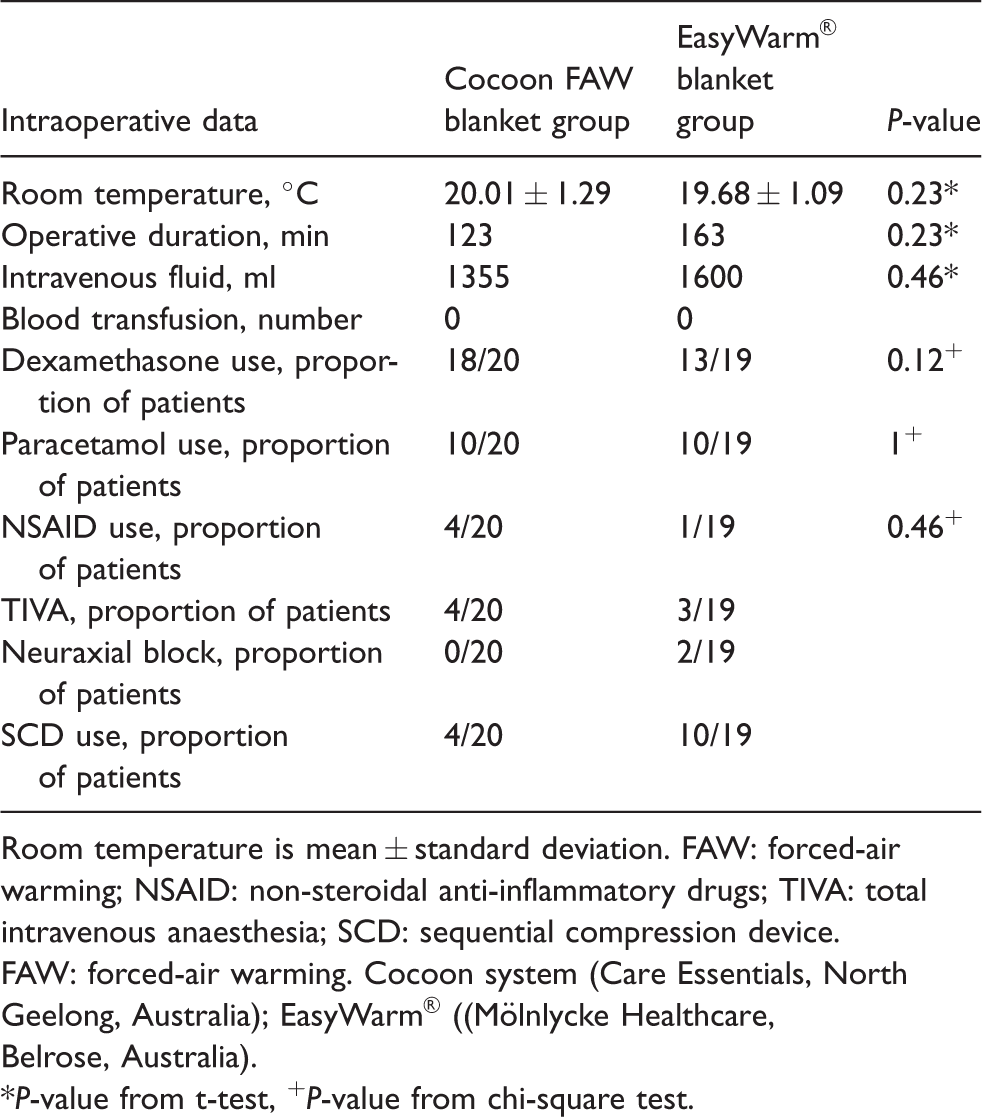
Room temperature is mean ± standard deviation. FAW: forced-air warming; NSAID: non-steroidal anti-inflammatory drugs; TIVA: total intravenous anaesthesia; SCD: sequential compression device.FAW: forced-air warming. Cocoon system (Care Essentials, North Geelong, Australia); EasyWarm® ((Mölnlycke Healthcare, Belrose, Australia).
*P-value from t-test, +P-value from chi-square test.
The baseline patient temperatures were 35.8 ± 0.57 °C and 35.8 ± 0.48 °C, in the Cocoon and EasyWarm® blanket groups, respectively (P = 0.7). Ambient room temperatures were comparable between groups.
There was no statistical difference in the primary outcome (nasopharyngeal temperature at the end of wound closure). The mean final temperature in the Cocoon blanket group was 35.89 ± 0.82 °C and in the EasyWarm® blanket group was 35.74 ± 0.77 °C, (P = 0.76) (Table 3), a difference of 0.14 °C, which is clinically insignificant. The average temperatures during the case, which was our secondary outcome, were similar between the two groups at 1, 2 and 3 h (Figure 2). The temperature drop was highest in the first hour of surgery in both groups as anticipated.
Primary and secondary outcomes.
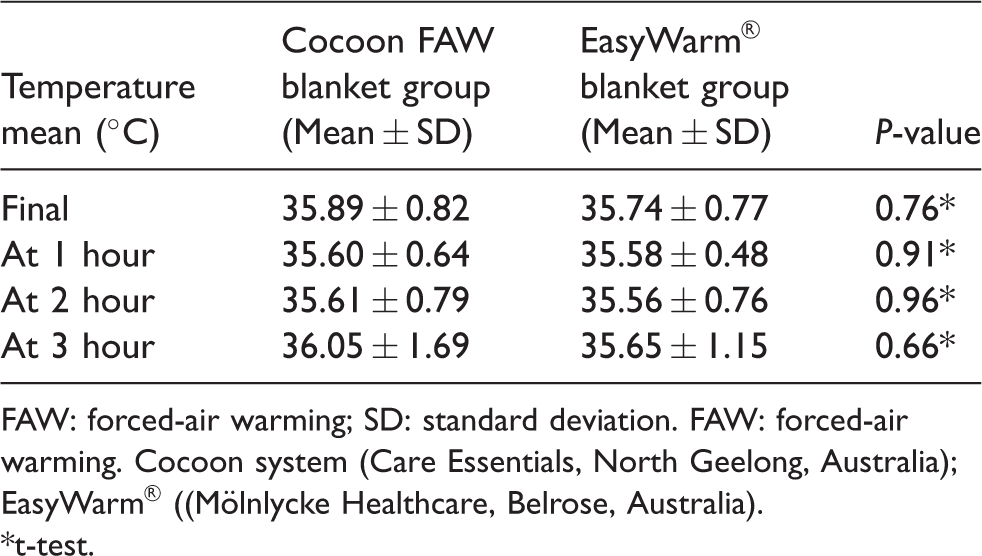
FAW: forced-air warming; SD: standard deviation. FAW: forced-air warming. Cocoon system (Care Essentials, North Geelong, Australia); EasyWarm® ((Mölnlycke Healthcare, Belrose, Australia).
*t-test.
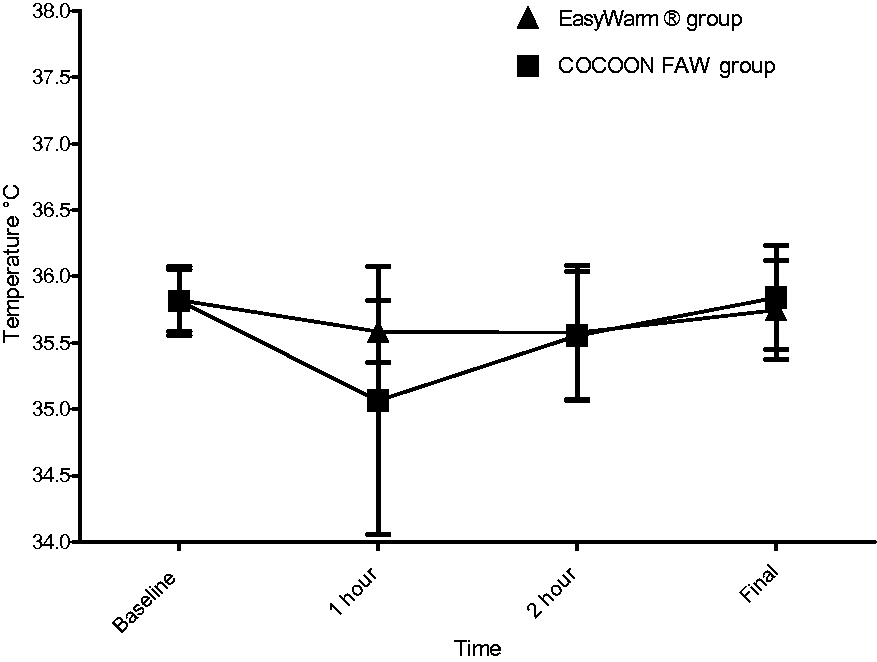
Line diagram showing the mean body temperatures (error bars represent standard errors) in the two groups at different timepoints during the surgeries. There was no significant difference between the groups.FAW: forced-air warming. Cocoon system (Care Essentials, North Geelong, Australia); EasyWarm® ((Mölnlycke Healthcare, Belrose, Australia).
Discussion
This single-centre RCT aimed to compare the efficacy of the two intraoperative warming devices under standard operating conditions. Our data demonstrated that there was no statistically significant difference in the final temperature between the Cocoon FAW and EasyWarm® blanket groups.
While there is no previous study that has compared the efficacy of the EasyWarm® blanket to FAW blankets in the literature, there is a multicentre RCT by Torossian et al. that compared passive thermal insulation using hospital cotton blankets to active warming using the BARRIER® EasyWarm® in the perioperative period. 11 This larger trial found that active warming with the EasyWarm® self-heating blanket marginally increased perioperative core body temperature compared with hospital cotton blankets (36.5 ± 0.4 °C versus 36.3 ± 0.3 °C; P < 0.001), measured using a tympanic infrared thermometer. Higher thermal comfort scores were recorded in the EasyWarm® group. However, our study did not find any significant difference in final temperature between the two groups. A potential explanation is that our study compared the efficacy of two active warming devices, whereas the above-mentioned study compared a passive warming to an active warming device. Another reason for a positive finding in the study by Torossian et al may be that they stipulated a significant mean temperature difference of 0.2 °C. Notably, the average temperature of the patients in both groups in the study by Torossian et al was higher compared to ours, with a quite narrow range. In contrast, the mean final temperature of both groups of patients in our study, measured using a nasopharyngeal probe, was under 36 °C, with a wider range in our sample, despite meticulous covering of patients, using warm IV fluids after induction of anaesthesia and maintaining ambient temperature. This could be explained by the benefit of preoperative warming, which was performed in their study and not in ours. It has been shown that preoperative warming is one of the most efficient interventions to reduce the incidence of perioperative hypothermia.1,14 Preoperative warming has been strongly advocated by the National Institute of Health and Care Excellence (NICE) in the guideline for prevention of hypothermia in patients having surgery. 15 Due to the requirement of a power unit for FAW systems it might not always be feasible to routinely preoperatively warm patients awaiting surgery. EasyWarm® blankets do not require any power source and could easily be used to pre-warm patients in the day surgery area, during surgery and also after surgery in the post-anaesthesia care unit.
Our secondary outcome, average temperature during surgery, was not significantly different between the two groups (Figure 2). Both groups had the highest decrease in temperature in the first hour. The fall in temperature slowed in the second hour and plateaued in the third hour (Table 3 and Figure 2). This coincides well with the known physiology of thermoregulation under general anaesthesia. 10
Other potential advantages of the EasyWarm® blanket are that it remains warm for 10 h, hence unsoiled blankets can be sent to the wards with the patient post-surgery. Given its flexibility to be used anywhere and its long heating duration, it can potentially cut down the costs of processing multiple hospital cotton blankets. It may also have the benefit of reducing clutter in the operating theatre and trip hazards due to the absence of electrical or other cords. No burns due to the use of EasyWarm® or overheating were observed in our study, as was also the case in the multicentre trial by Torossian et al. 11
Nosocomial infection, especially at the surgical site due to disturbance of laminar air flow caused by FAW devices, has been a concern, particularly in orthopaedic surgery, and studies have conflicting results.16–19 Use of EasyWarm® blankets might be a preferable option for such cases. It may also be valuable in intra- and inter-hospital transfers.
The drawback of using the EasyWarm® blanket over the FAW blanket is that it is cumbersome to remove intraoperatively if the patient becomes hyperthermic. It is also difficult to use for specific positions such as the lithotomy position. Also, it has to be removed during patient X-rays or magnetic resonance imaging. The vacuum in the packet needs to be broken and the blanket needs to be exposed to air for 30 min before it warms up fully and is ready for patient use. This makes its use less favourable in urgent surgeries.
There are some limitations to our study. While we calculated our sample size based on an SD of 0.5 °C, an SD of 0.8 °C in the final temperature was observed in our study. With this SD, 80 patients would have been required to achieve the planned precision of our final comparison. We did not look at thermal comfort scores postoperatively, which would have given us valuable information about patient comfort, although the different designs of the devices would have somewhat confounded their comparison. The EasyWarm® blanket was used for both the upper and lower body, but the Cocoon FAW was used only for the lower half of the body, which means a larger surface area was covered and warmed in the EasyWarm® blanket group. In pragmatic fashion, we tested their use in a manner consistent with standard clinical practice at our institution, which is to use only one FAW blanket unless we consider the patient to be at particularly high risk of, or to be developing, severe intraoperative hypothermia (defined by us as a temperature of 34 °C or less for the purposes of the study).
There are myriad active warming systems available for perioperative warming. 20 FAW is a common clinical standard against which other systems are often compared. Multiple studies have compared FAW systems with radiant warmers and have found FAW to be superior.12,21 Other studies have compared resistive polymer devices to FAW warming systems in healthy volunteers and found that the heating efficacy and rewarming rates were similar with the two systems.3,22
In conclusion, we found the EasyWarm® blanket to be as effective as the Cocoon FAW blanket in preventing intraoperative hypothermia. Its ease of use and its greater flexibility may be additional advantages over Cocoon FAW blankets.
Declaration of conflicting interests
There is no conflict of interest to declare. Both companies of the active warming devices are unaware of this trial.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We would like to thank the Anaesthesia Research Fund, administered by the Department of Anaesthesia, Austin Hospital for funding the EasyWarm® blankets for the research.
