Abstract
Clonidine has been used successfully to prolong the duration of action of local anaesthetics in peripheral nerve blocks, but its mechanism of action in this setting remains unclear. Some studies suggest that clonidine exerts a vasoconstrictor effect, limiting the washout of local anaesthetic from its site of deposition. We investigated this potential vasoconstrictor effect, using plasma ropivacaine concentrations as a surrogate measure of vasoconstriction, in patients who received transversus abdominis plane (TAP) blocks with and without clonidine. Eighty women undergoing laparoscopic gynaecological surgery were randomly assigned to receive one of four TAP block solutions: 0.2% ropivacaine (control), ropivacaine with clonidine 2 μg/kg (clonidine), ropivacaine with 1:400,000 adrenaline (adrenaline) or ropivacaine and a subcutaneous injection of clonidine 2 μg/kg (SC clonidine). The primary outcome was total venous plasma ropivacaine concentrations up to 6 h after the block. There were no significant differences in plasma ropivacaine concentrations between the control group and the clonidine group at any timepoint in the study, nor were there differences in either the mean maximum ropivacaine concentration (Cmax) (1.99 μg/mL versus 2.05 μg/mL, P = 0.712) or the time to maximum concentration (Tmax) (51.0 min versus 56.0 min, P = 0.537). The SC clonidine group also did not differ significantly from the controls (Cmax 2.13 μg/mL versus 1.99 μg/mL, P = 0.424; Tmax 43.5 min versus 51.0 min, P = 0.201). Plasma ropivacaine concentrations in the adrenaline group were significantly lower than the controls from 10 to 90 min (P < 0.003 for each comparison), and the Cmax was less than that of the control group (1.36 μg/mL versus 1.99 μg/mL, P < 0.001) with a longer Tmax (103.5 min versus 51.0 min, P = 0.001). These findings indicate that clonidine at a concentration of 1.35 μg/mL added to ropivacaine for TAP blocks did not produce a reduction in plasma ropivacaine concentrations. This suggests a lack of vasoconstrictor effect during TAP blocks. Further studies should evaluate whether vasoconstriction occurs when clonidine is used at higher concentrations or for other blocks.
Introduction
Several studies have demonstrated that the addition of clonidine to local anaesthetic in peripheral nerve blocks reduces postoperative pain and analgesic requirements and increases the duration of sensory nerve block.1–4 The mechanism by which it prolongs local anaesthetic action remains unclear and several potential receptor and channel-mediated mechanisms have been postulated.5–9 The site of action of clonidine in this setting is also unclear, with some previous studies supporting a central mechanism of action,10,11 but others indicating that the likely site of action is peripheral at the nerve itself.12–14
Some previous studies have shown that when clonidine was added to local anaesthetic it resulted in increased tissue and decreased plasma local anaesthetic concentrations over time in peripheral nerve blocks and epidural blocks,15,16 respectively. From these results, the authors of these studies concluded that clonidine had a local vasoconstrictor effect. By causing vasoconstriction, the rate of washout of the local anaesthetic from its site of deposition might be reduced, allowing the local anaesthetic to remain at its site of action longer, thereby prolonging the duration of action.
Several possible mechanisms of action of clonidine have been postulated and may operate to varying degrees in different anatomical locations and at different concentrations. We designed a randomized controlled study in an effort to help clarify whether clonidine has a vasoconstrictor effect when added to ropivacaine for tranversus abdominis plane (TAP) blocks, either centrally or peripherally, using plasma ropivacaine concentrations as a surrogate measure of vasoconstriction. We expected that if clonidine produced local vasoconstriction then the rate of washout of ropivacaine from the TAP block would be reduced and therefore the plasma ropivacaine concentrations would also be reduced. A secondary objective of this study was to assess the effect of adding clonidine to ropivacaine in TAP blocks on abdominal wall sensation.
Methods
This study was approved by the local ethics committee (Sydney Local Health District Human Research Ethics Committee, Protocol number X14-0093) and registered with the Australian New Zealand Clinical Trials Registry (ACTRN12614000587606). Written informed consent was obtained from all patients. Female patients aged 18–65 years who were undergoing TAP blocks and general anaesthesia for laparoscopic gynaecological surgery were recruited. Exclusion criteria included allergy to any of the study drugs, body mass index (BMI) > 40 kg/m2, evidence of infection at the site of block placement, pre-existing neurological or muscular disease, coagulopathy, significant renal or liver disease, and pregnancy. Eighty patients were included and randomized using a web-based randomization tool to one of the control group (TAP block with 3 mg/kg of 0.2% ropivacaine), the clonidine group (TAP block with 3 mg/kg of ropivacaine and 2 μg/kg of clonidine), the adrenaline group (TAP block with 3 mg/kg of ropivacaine and 1:400,000 adrenaline) or the SC clonidine group (TAP block with 3 mg/kg of ropivacaine and a subcutaneous injection of 2 μg/kg of clonidine).
The study was conducted between June 2014 and November 2015. Data collected included patient characteristics such as age, height, weight, American Society of Anesthesiologists (ASA) physical status classification and comorbidities, as well as surgical procedures performed, duration of surgery, estimated blood loss, total venous plasma ropivacaine concentrations over time, abdominal wall sensation over time, intraoperative and postoperative changes in haemodynamic status, incidence of postoperative nausea and vomiting (PONV), and sedation scores.
Standard monitoring (pulse oximetry, electrocardiography and automated non-invasive blood pressure) and intravenous access were established prior to induction of general anaesthesia with propofol 2 mg/kg and fentanyl 1.5 μg/kg. Endotracheal intubation was facilitated with rocuronium 0.6 mg/kg. Anaesthesia was maintained with sevoflurane. After induction of anaesthesia, an 18 gauge intravenous cannula was placed in the external jugular vein of the patients for blood sampling and the TAP blocks were performed prior to the start of surgery. Patients received a further 1.5 μg/kg of fentanyl during the procedure as well as paracetamol 1 g, parecoxib 40 mg, and ondansetron 4 mg.
Ultrasound-guided bilateral posterior TAP blocks were performed by a single blinded investigator. A Sonosite M-Turbo ultrasound machine with a linear probe (SonoSite Australasia, Brookvale, New South Wales) was used to locate the TAP. The overlying skin was disinfected with an alcohol swab prior to using an in-plane approach to guide a SonoPlex® cannula (Pajunk, Geisingen, Germany) into the plane using a hydrodissection technique. The block solution was then injected and the procedure repeated on the contralateral side. The patients, the investigator collecting data, measuring plasma ropivacaine concentrations and performing sensory assessments, and the nurses in the recovery area were all blind to the group allocation of the patients. The anaesthetists caring for the patients mixed the block solutions and therefore were not blinded.
Postoperatively patients received intravenous boluses of fentanyl 20 μg as needed up to 200 μg until oral analgesia was tolerated at which point they were provided with regular oral paracetamol 1 g every 6 h and oral oxycodone 5–10 mg every 2 h as required. Anti-emetics were administered as required according to a PONV protocol that included ondansetron 4–8 mg, prochlorperazine 6.25–12.5 mg and droperidol 0.5 mg.
Total venous plasma ropivacaine concentrations were measured at 10, 20, 30, 40, 50, 60, 90, 120, 150, 180 and 360 min after TAP blocks were performed. Gas chromatograph mass spectrometry (QP2010SE, Shimadzu Scientific Instruments, Rydalmere, New South Wales) was used. The internal standard was bupivacaine hydrochloride 0.5% (AstraZeneca, Sydney, New South Wales). The maximum total plasma ropivacaine concentration (Cmax) and time to maximum total plasma ropivacaine concentration (Tmax) were calculated for each study group from the pharmacokinetic profiles of the individual patients to enable comparisons between study groups.
Abdominal wall sensation to mechanical stimuli, warm, and cold was tested from T8 to L1 dermatomes prior to induction of anaesthesia and at 3 and 6 h after the TAP blocks. The mechanical detection threshold was established in each dermatome bilaterally using Von-Frey Hairs (DanMic Aesthesio®, Precise Tactile Sensory Evaluator 20 piece Kit, Campbell, CA, USA). Then a method described in a previous similar study was employed to compare groups, 17 which involved using the 180 g filament as the cut-off point between normal/minimally impaired sensation and abnormal sensation. Warm and cold sensation were assessed using heated pouches (Pocket Hand Warmer, Kathmandu®, Melbourne, Victoria) and alcohol swabs (Kendall Webcol® Alcohol Prep —saturated with 70% isopropyl alcohol, Minneapolis, MN, USA), respectively.
Significant intraoperative and postoperative haemodynamic instability (decrease in systolic blood pressure >30% from baseline systolic pressure and decrease in heart rate to less than 45 per minute), and medications administered to treat haemodynamic changes were recorded. Level of sedation was recorded at 30 and 60 min after extubation using the Ramsay sedation score.
An adrenaline group was included because, as a known vasoconstrictor, if reduced plasma ropivacaine levels were noted in this group, it would support the notion that the reduction was caused by local vasoconstriction. The SC clonidine group was included to determine, if indeed a vasoconstrictor effect was observed with clonidine, whether this might be mediated by a central rather than a peripheral mechanism.
Statistical analysis
To estimate the required sample size, we used the effect size from a previous study (0.62 μg/mL), 18 which investigated plasma local anaesthetic concentrations with the addition of adrenaline to ropivacaine in paravertebral blocks, specifically the Cmax data (control group 2.47 μg/mL, adrenaline 1.85 μg/mL, standard deviation, SD 0.6 μg/mL). No similar studies using clonidine had been performed so we assumed the minimum clinically important effect of clonidine, compared with control, would need to be the same as adrenaline. With α = 0.05 and power 0.80 a minimum sample size of 15 was required, which we increased to 20 subjects per group to account for potential dropouts.
Results were reported as number and frequency (percentage) or mean and SD, as appropriate. Parametric data including demographic, surgical, and plasma ropivacaine concentration data were analysed using Student’s t-test (two-sided, equal variances, unpaired). Non-parametric data including sensation data were compared using chi-square analysis and Fisher’s exact test. Statistical analyses were performed using IBM® SPSS® Statistics v20 (IBM Corporation, Armonk, NY, USA) and GraphPad Prism Software version 6.0 (GraphPad Software, La Jolla, CA, USA).
Results
All 80 patients completed the study. The study groups were comparable with respect to age, height and ASA physical status; however, the weight of the patients in the clonidine and SC clonidine groups was greater than that of the control group (Table 1). Estimated blood loss and duration of surgery were comparable between study groups. In two patients (one in the control group and one in the clonidine group) the surgical access was inadequate and the procedures were converted to open procedures via Pfannenstiel incisions.
Patient characteristics and surgical data.
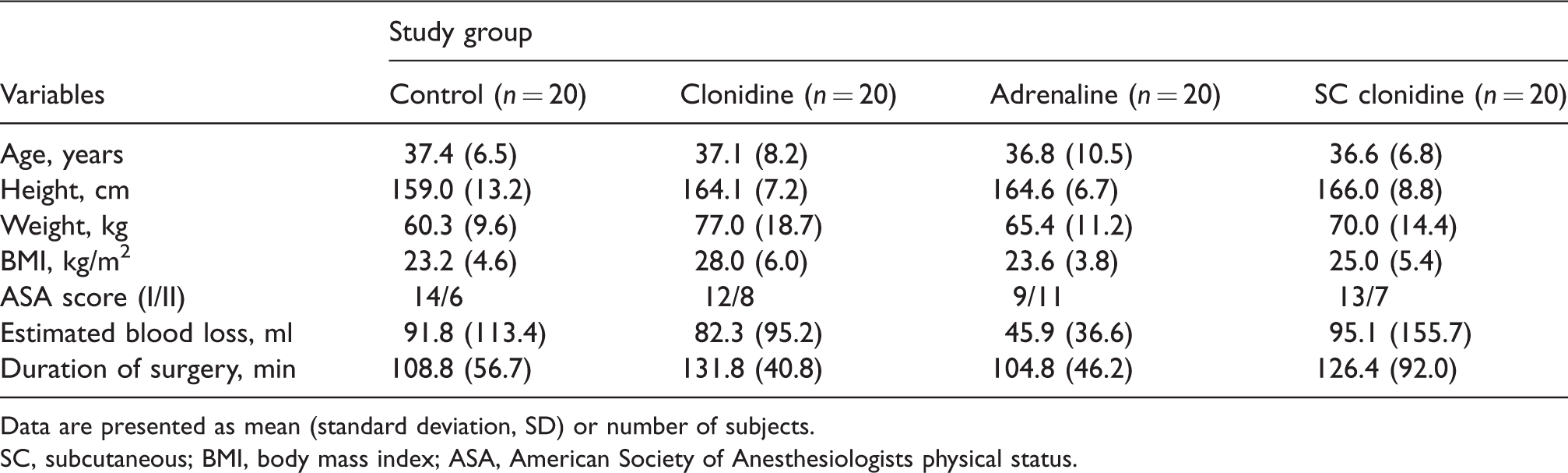
Data are presented as mean (standard deviation, SD) or number of subjects.
SC, subcutaneous; BMI, body mass index; ASA, American Society of Anesthesiologists physical status.
The individual patient pharmacokinetic profiles for ropivacaine according to study group are presented in Figure 1. There were no significant differences between the total venous plasma ropivacaine concentrations of the patients in the control group and the clonidine group at any of the study timepoints (Figure 2). The mean Cmax and Tmax of the clonidine group were 2.05 (95% confidence interval (CI) 1.81–2.29) μg/mL and 56.0 (95% CI 42.9–69.1) min, respectively, which was not different from that of the control group (1.99 (95% CI 1.73–2.25) μg/mL; P = 0.712 and 51.0 (95% CI 42.2–59.8) min; P = 0.537, respectively) (Table 2). The plasma ropivacaine concentrations of the patients in the adrenaline group were significantly lower than in the control group at 10, 20, 30, 40, 50, 60 and 90 min timepoints (P < 0.003 for every comparison). The Cmax of the adrenaline group, 1.36 (95% CI 1.20–1.52) μg/mL, was less than that of the control group (1.99 (95% CI 1.73–2.25) μg/mL; P < 0.001) and the Tmax was delayed (103.5 (95% CI 79.4–128.0) min versus 51.0 (95% CI 42.2–59.8) min; P < 0.001). There were no significant differences between the plasma ropivacaine concentrations of the SC clonidine group and the control group, nor did the Cmax or Tmax differ significantly from the control group.
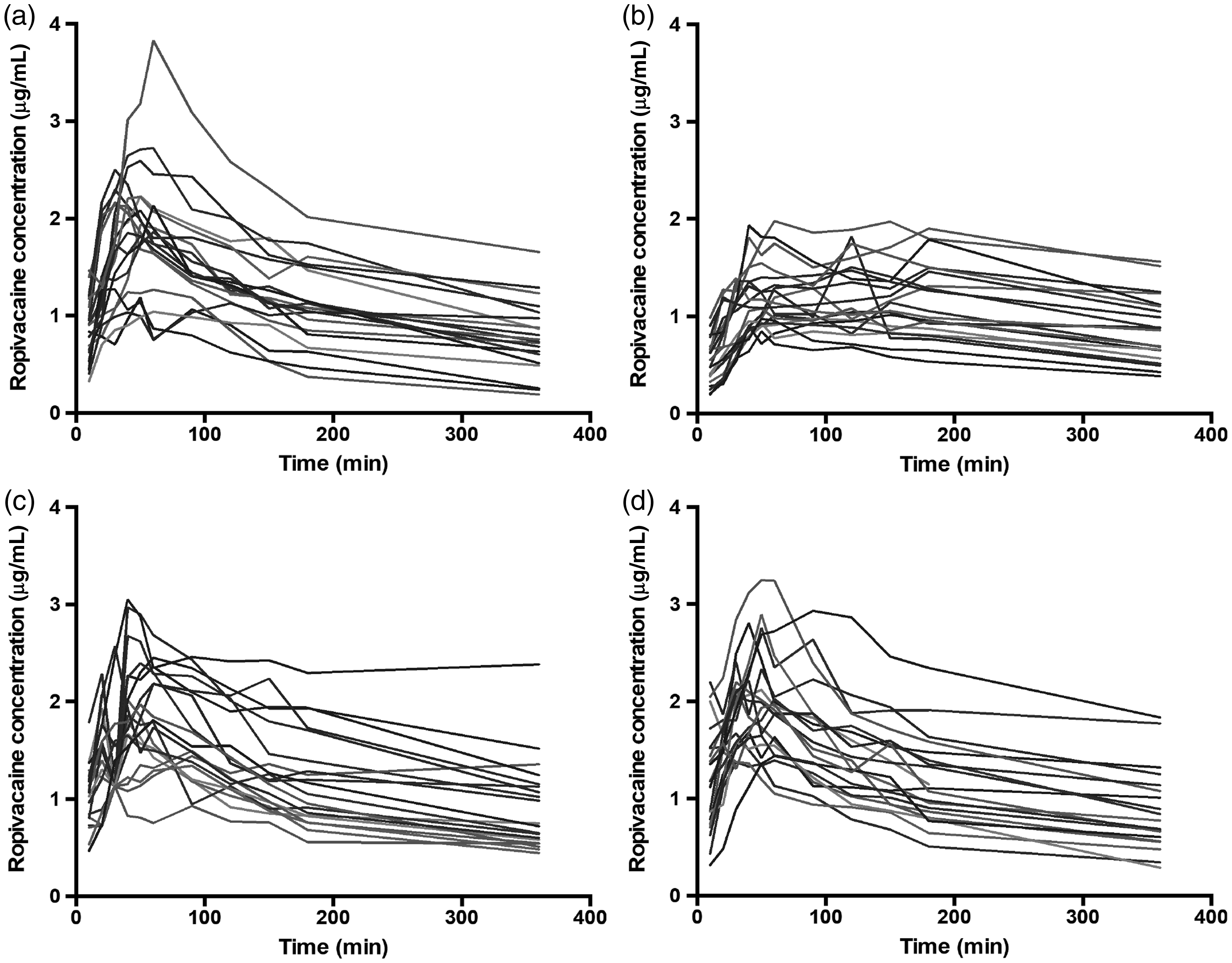
Individual data of total venous plasma ropivacaine concentrations over time of patients in the (a) plain ropivacaine group, (b) ropivacaine with adrenaline group, (c) ropivacaine with clonidine group and (d) ropivacaine and subcutaneous clonidine group.
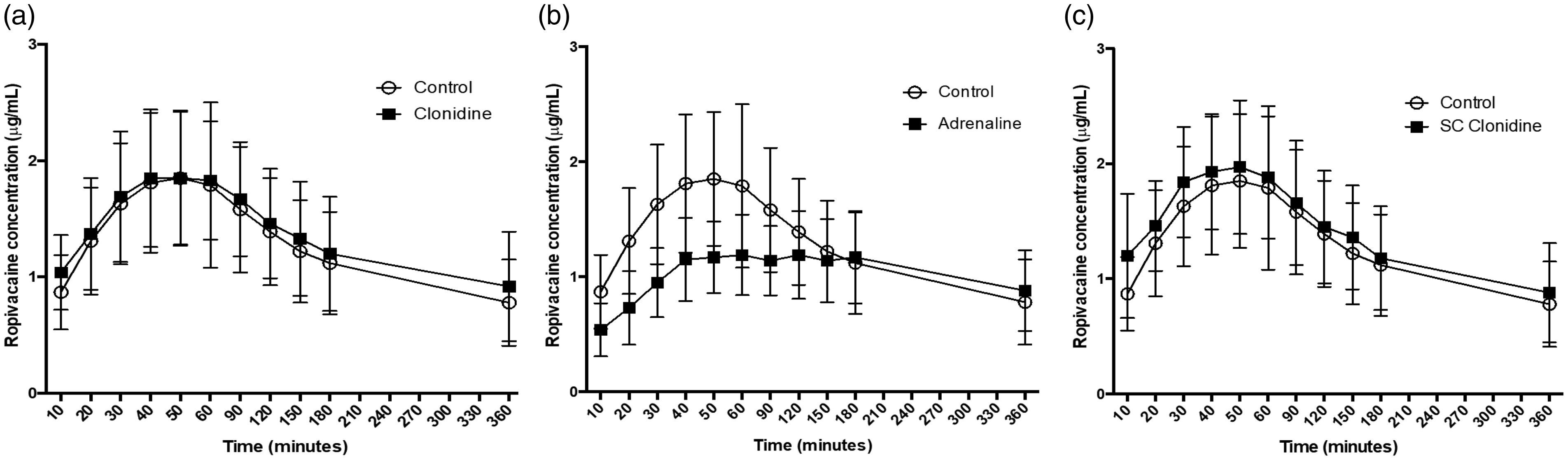
Mean total venous plasma ropivacaine concentrations over time in the (a) control and clonidine groups, (b) control and adrenaline groups and (c) control and subcutaneous clonidine groups.
Maximum ropivacaine concentration (Cmax) and time to peak concentration (Tmax) for each study group.
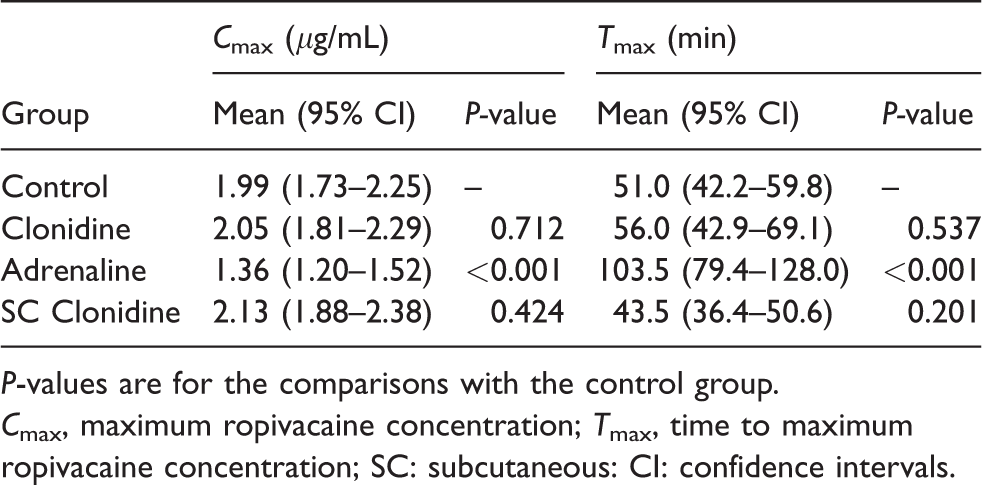
P-values are for the comparisons with the control group.
Cmax, maximum ropivacaine concentration; Tmax, time to maximum ropivacaine concentration; SC: subcutaneous: CI: confidence intervals.
No significant differences in mechanical sensation were found between groups using Von Frey filaments. Few differences in cold and warm sensation were found between groups at individual dermatomal levels, and these were not consistent across adjacent dermatomes, with the other sensory modalities, or across timepoints.
The groups were comparable in terms of the incidence of haemodynamic instability (defined as a decrease in systolic blood pressure >30% from baseline, decrease in heart rate to <45 per minute, or the administration of metaraminol or atropine by the treating anaesthetist). The level of sedation and incidence of PONV were also comparable between groups.
Discussion
Our results demonstrate that the addition of 2 μg/kg of clonidine to ropivacaine in TAP blocks does not result in a significant reduction in plasma ropivacaine concentrations. The total venous plasma ropivacaine concentrations were not reduced in the clonidine group compared to the control group and, furthermore, the addition of clonidine did not reduce the Cmax, nor increase the Tmax, of the clonidine group. SC clonidine administration also had no observed effect on the pharmacokinetic profile of ropivacaine in our study. In contrast, adrenaline, when administered with ropivacaine, resulted in significantly lower total venous plasma ropivacaine concentrations from 10 to 90 min following the TAP blocks, and a lower Cmax and prolonged Tmax compared to the control group. The finding that the administration of clonidine when added to ropivacaine in TAP blocks or as a separate subcutaneous injection did not result in reduced plasma ropivacaine concentrations suggests that clonidine does not cause vasoconstriction, whether administered locally at the site of the block, or when administered systemically by an SC injection (i.e. by either by a peripheral or central mechanism). However, a limitation of our study was that the sample size was calculated to detect a difference between the control and clonidine groups only and was not adjusted for multiple simultaneous comparisons. Therefore, only limited conclusions can be drawn regarding the SC clonidine and adrenaline groups. Additionally, we cannot exclude differences of <0.62 μg/mL (the effect size used in our sample size calculation) between groups, although our results do not suggest that there are such real differences.
The dose of clonidine used in our study was based on other previous studies that used a dose of 150 μg or 2 μg/kg.2,19,20 However, because 0.2% ropivacaine and high volume TAP blocks were used in our study, the final concentration of clonidine in the injectate was relatively low at 1.35 μg/mL. Previous studies,15,16 which have demonstrated a vasoconstrictor effect of clonidine, used higher clonidine concentrations in the injectate. Kopacz and Bernards used microdialysis catheters to assess tissue lidocaine concentrations after peroneal nerve blocks using lidocaine with and without clonidine 10 μg/mL and found that clonidine caused vasoconstriction as indicated by a reduced area under the lidocaine concentration versus time curve. 15 Mazoit et al. demonstrated that adding clonidine at a concentration of 13.64 μg/mL to lidocaine in the epidural space resulted in a reduced Cmax compared to epidural lidocaine alone. 16 On the contrary, a previous study using a low concentration of clonidine (3.75 μg/mL) similar to our study demonstrated that compared to adrenaline, clonidine caused minimal or no vasoconstriction, and therefore it may be that clonidine only results in vasoconstriction at higher concentrations. However, the lack of an observed vasoconstrictor effect in our study may instead indicate that clonidine exerts its effect predominantly via a mechanism other than vasoconstriction.
The finding that addition of adrenaline caused a reduction in plasma ropivacaine concentrations from 10–90 min and a reduced Cmax and increased Tmax, likely mediated by vasoconstriction, is in agreement with the findings of other studies.21,22 A direct comparison of the results is difficult due to differing drug doses, but one study showed a similar 65% reduction in the Cmax and a twofold increase in the Tmax, 21 as was found in our study.
Despite randomization, patients in the clonidine and SC clonidine groups had a greater mean body weight than did the control group. This represented a possible confounding factor as the doses in our study were weight-based.
Several previous studies have found that clonidine prolongs the duration of action of local anaesthetics,2,23,24 however that was not demonstrated in our study. This is in agreement with other studies that showed that the addition of clonidine did not result in a prolonged duration of anaesthesia or analgesia.19,25,26 Despite the fact that our sample size was calculated to detect a difference in plasma ropivacaine concentrations rather than abdominal wall sensation, the sample size was similar to other previous studies powered to detect differences in duration of analgesia and anaesthesia.26–28 It is therefore likely that our sample size was adequate to detect a difference in abdominal wall sensation should one have existed. The lack of significant prolongation of the TAP blocks with clonidine in our study may suggest that clonidine, when added to ropivacaine, is not effective in this type of block and therefore, TAP blocks may be an unsuitable type of block with which to assess the vasoconstrictor effects of clonidine.
A meta-analysis of the addition of clonidine to peripheral nerve blocks found an increase in haemodynamic adverse effects (arterial hypotension, orthostatic hypotension or fainting and bradycardia) and sedation. 29 However, other previous studies,19,24,27 which have used a similar dose of clonidine to our study (150 μg), have found it to be a safe dose without increasing the incidence of haemodynamic instability or sedation. In our study, the study groups were comparable in terms of the incidence of haemodynamic instability and sedation, although it was not powered to detect a difference in these endpoints as this was not the primary focus of the study.
In summary, the addition of low concentrations of clonidine to ropivacaine in high volume TAP blocks did not result in a reduction in plasma ropivacaine concentrations, suggesting that clonidine does not exert a vasoconstrictive effect under these conditions. However, our results pertain only to TAP blocks, ropivacaine, and low concentrations of clonidine, and further studies would be required to assess the effect of clonidine in other circumstances. Studies evaluating the effect of the addition of clonidine to low-volume blocks on plasma local anaesthetic concentrations may be useful to better evaluate the existence of a vasoconstrictor mechanism.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
