Abstract

We present our recent experience affirming the benefit of early echocardiography in the management of presumed amniotic fluid embolism (AFE). Written patient consent has been obtained prior to submission for publication.
Case description
A healthy 34-year-old, gravida 5 para 2 woman presented to the antenatal clinic for routine assessment at 29 weeks gestation. She reported a two-day history of lower abdominal cramping pain with associated minor vaginal bleeding, and was found to be in active labour. Ultrasound demonstrated a singleton fetus in transverse lie, and internal examination revealed a fully dilated cervix with bulging membranes. A decision was made to perform an emergency lower segment caesarean section (LSCS).
The patient underwent a routine spinal anaesthetic achieving a satisfactory neuraxial block prior to incision. Upon opening of the membranes, the patient became confused, had seizure-like activity, lost consciousness and became profoundly cyanosed. Bradycardia was rapidly followed by complete loss of cardiac output. Advanced life support was commenced, with a live male infant delivered immediately prior to commencement of chest compressions. The uterus was exteriorised and compressed during cardiopulmonary resuscitation. Tracheal intubation was performed, and femoral venous and arterial sheaths were placed in preparation for extracorporeal life support (ECLS).
Return of spontaneous circulation (ROSC) was obtained after 17 min of advanced life support and blood pressure was supported by an epinephrine (adrenaline) infusion. The uterus was internally repositioned. Uterine atony and bleeding were treated with intravenous oxytocin and intra-uterine balloon tamponade. Haemostasis was difficult to achieve, with clinically apparent coagulopathy.
Pathology results demonstrated disseminated intravascular coagulopathy (DIC); International Normalized Ratio 7.8, prothrombin time 65.4 s, activated partial thromboplastin time >200 s, platelets 32 × 109/L, fibrinogen 0.14 g/L; as well as a lactic acidosis: pH 7.1, lactate 6.3 mmol/L. The patient received 2 units of packed red blood cells, 2 units of pooled platelets, 2 units of fresh frozen plasma, 26 units of whole blood cryoprecipitate, tranexamic acid 1 g, and calcium chloride 10 mmol.
A presumptive diagnosis of AFE was made. Transoesophageal echocardiography (TOE) performed 18 min post ROSC revealed extensive echodense masses within the right heart (Figures 1 and 2) and inferior vena cava. The masses in the right atrium and the right ventricle were large, highly mobile and heterogeneous, with a gelatinous appearance. No patent foramen ovale was seen.
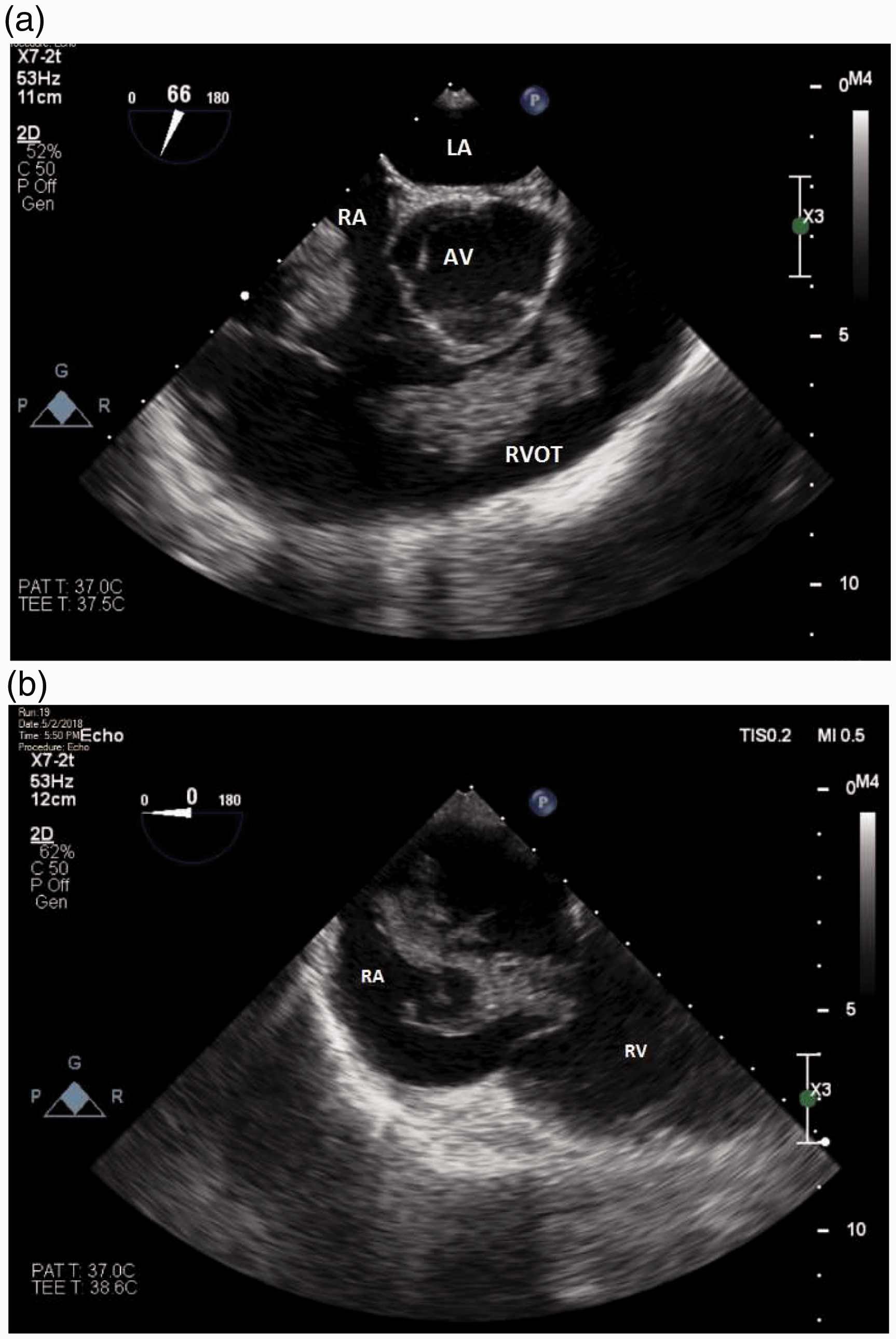
(a) and (b) Transoesophageal echocardiographic mid-oesophageal views showing large heterogeneous right heart masses. LA: left atrium; AV: aortic valve; RVOT: right ventricular outflow tract; RA: right atrium; RV: right ventricle.
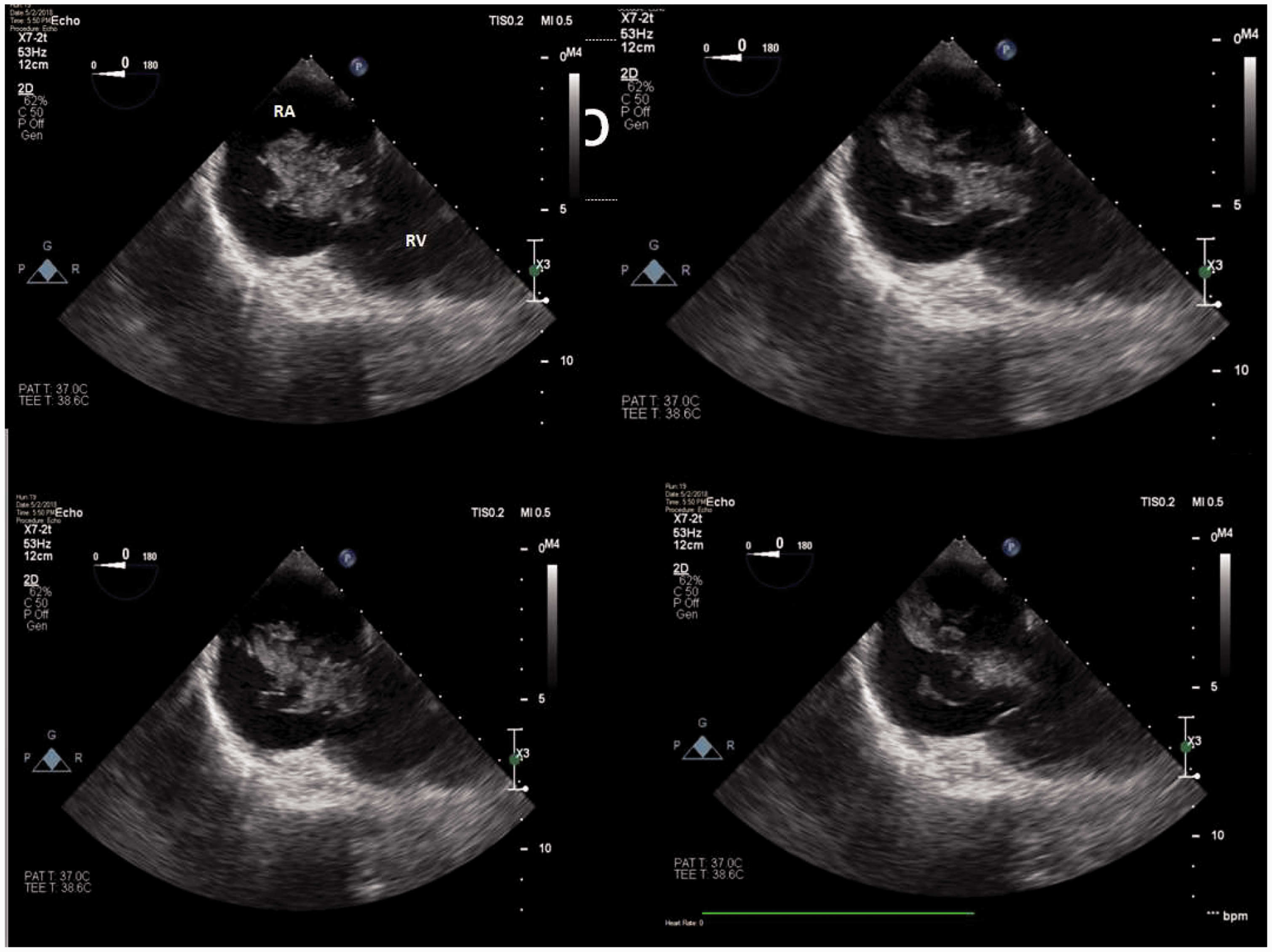
Transoesophageal echocardiographic mid-oesophageal views showing large heterogeneous right heart masses. RA: right atrium; RV: right ventricle.
The patient was reviewed intraoperatively by specialists from cardiothoracic surgery, interventional radiology, and intensive care. Bedside bilateral lower limb ultrasonography did not demonstrate deep vein thrombosis. Once sustained haemodynamic improvement was obtained, the patient was transferred to the intensive care unit (ICU) for ongoing management.
After 3.5 h, there was only moderate output from the surgical drains and biochemical and coagulation parameters were improving. Formal Doppler sonography confirmed the absence of lower limb thrombi.
Twelve hours later, the patient no longer required inotropic or vasopressor support. Transthoracic echocardiography demonstrated complete resolution of the intracardiac masses, with normal biventricular size and systolic function. Coagulopathy had resolved. On clinical assessment, generalised bulbar dysfunction and a dense right-sided hemiparesis were identified. Computed tomography of the brain demonstrated focal hypodensities within the left midbrain and right inferior cerebellum, confirmed as infarcts on subsequent magnetic resonance imaging. The patient made a complete cardiac recovery, and after six weeks was transferred to a rehabilitation centre with ongoing neurological improvement.
Discussion
Despite the rare occurrence of AFE, identified in an estimated 1:40,000 deliveries, it contributes to 5% to 15% of all maternal deaths and has a maternal mortality rate of 13% to 44%.1–3 AFE resulting in cardiac arrest has a mortality approaching 50% and is one of the more common causes of cardiac arrest during labour. 4 In addition, historically a high percentage of survivors may exhibit residual neurological deficits. 5
Our case fulfils the proposed uniform diagnostic criteria for AFE. 6 AFE typically occurs during labour or immediately thereafter. Risk factors include advanced maternal age, caesarean section, placenta praevia or abruption, multiparity, male fetus and Rhesus isoimmunisation.2,5 The diagnosis is clinical, with hypotension, respiratory failure, and disseminated intravascular coagulopathy the hallmark features. 7 Acute dyspnoea or sudden agitation and anxiety are common premonitory symptoms, and seizures occur in approximately 10% to 50% of patients 2 .
The pathogenesis of AFE remains poorly understood. The historical concept of amniotic fluid causing physical obstruction of the pulmonary vasculature has not been supported by radiological evidence, post mortem findings or animal models. 8 Current theories suggest an immunological pathogenesis, with entry of amniotic fluid into the maternal circulation triggering release of vasoactive and procoagulant substances. 8 This has led to the label ‘anaphylactoid reaction of pregnancy’. Increased levels of pulmonary vasoconstrictors result in acute right ventricular failure and haemodynamic collapse. 9 The associated coagulopathy may be primarily a consumptive process or due to massive fibrinolysis.2,8 Amniotic fluid itself has procoagulant activity, with thrombus forming around amniotic fluid. and it has been proposed that massive intravascular thrombosis during the initial phase of AFE may be the missing link in the pathogenesis of this syndrome.7,10 We postulate that the masses we saw on TOE were the result of this process.
TOE is able to rapidly identify the potential causes of haemodynamic collapse during labour and delivery, and to differentiate between AFE and other conditions such as myocardial ischaemia and peripartum cardiomyopathy. It may aid management, including placement of ECLS cannulae, placement and assessment of effect of intra-aortic balloon counterpulsation, and assessment of effects of inotropic agents. 11 It has been proposed as a standard of care in the evaluation of AFE. 10
Early TOE findings in AFE include right heart dilation with deviation of the interventricular septum into the left ventricle, and acute pulmonary hypertension.9,12 Transient thrombi or emboli may occur7,13; however, the presence of large cardiac masses, as seen in our patient, has only been rarely described previously. 14 In those who survive the initial insult, subsequent left ventricular failure may ensue. 9 Rapid and complete recovery of right ventricular function occurs, and differentiates AFE from other diagnoses such as acute pulmonary thromboembolism. 11
The early resolution of intracardiac masses without specific treatment is a notable feature of our case, and is in keeping with other reports.13,14 This transience may explain the lack of thrombus noted in other studies, with clot resolution or distal embolisation occurring prior to echocardiography. In our case, the absence of clinical or echocardiographic features of ongoing right heart strain favours clot dissolution over distal embolisation.
There is no established specific treatment for AFE. Heparin treatment during the consumptive phase of DIC is not recommended. ECLS, cardiopulmonary bypass, pulmonary thromboembolectomy and intra-aortic balloon pump usage carry significant risk in the presence of profound coagulopathy and are not currently recommended. 9 Significantly in this case, ECLS was not pursued as a result of the ultrasound findings, given the potential for the large intracardiac masses to occlude the ECLS cannulae or entrain into the ECLS circuit with resultant circuit failure. Sudden circuit failure in this case may have been catastrophic. Removal of masses via open sternotomy, suction thrombectomy and directed thrombolysis were all considered; however, these interventions were not required as the patient stabilised rapidly.
Our case provides further support for the role of intravascular thrombosis in the pathogenesis of AFE. It highlights the benefits of TOE in the early diagnosis of AFE and also in guiding ECLS decision-making. The rapidity of haemodynamic improvement and resolution of intracardiac masses without specific treatment, along with the presence of DIC, favour a conservative approach to management. Focus should be on cardiovascular support, delivery of the neonate and rapid correction of coagulopathy. We suggest echocardiography be performed in cases of maternal arrest during delivery, with further benefit in cases where ECLS is considered.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
