Abstract
Major bleeding in noncardiac surgery is common and associated with serious complications. The antifibrinolytic agent tranexamic acid (TXA) reduces bleeding and may reduce the risk of these complications. TXA also may have immunomodulatory effects that could reduce surgical site infection. Clinical trials of TXA in noncardiac surgery have been insufficiently powered to evaluate its efficacy and safety. Therefore, large randomised controlled trials of its use in noncardiac surgery are required. To ensure that future clinical trials are feasible and acceptable, we undertook a survey of Fellows of the Australian and New Zealand College of Anaesthetists (ANZCA). Our aims were to ascertain current patterns of TXA administration and to assess the acceptability of randomising patients to intravenous TXA or placebo. A 12-item survey was electronically mailed to 1001 ANZCA Fellows. Two hundred and eighty nine responses were received and analysed (response rate 29%). Ninety-eight percent of respondents had used intravenous TXA in noncardiac surgery; 67% give TXA routinely for lower limb arthroplasty, with smaller proportions giving TXA for spinal surgery (40%) and other major orthopaedic surgery (28%). Almost half (49%) give TXA routinely for major trauma surgery. Thirty-six percent indicated that they did not give TXA for major vascular, abdominal, pelvic or thoracic surgery. The majority administered TXA as a single, fixed dose. Fifty-seven percent agreed that there is uncertainty about the relative risks and benefits of perioperative TXA in noncardiac surgery and 87% agreed that large definitive trials determining the safety and efficacy of perioperative TXA in noncardiac surgery are required. These results indicate that for ANZCA Fellows the use of TXA in noncardiac surgery is highly variable, that there is uncertainty about the safety and efficacy of TXA, and that a large trial would be acceptable.
Introduction
Major bleeding in noncardiac surgery is common and associated with serious complications. In the second Vascular Events In Noncardiac Surgery Patients Cohort Evaluation (VISION) Study (a prospective cohort study of 21,000 noncardiac surgery patients), major bleeding (defined by haemoglobin (Hb) ≤70 g/L, transfusion of ≥1 unit of red blood cells (RBC), or death) occurred in 14.2% of patients and was independently associated with mortality at 30 days (adjusted hazard ratio (aHR), 2.77; 95% confidence interval (CI), 2.11 to 3.62). 1 Perioperative bleeding is also associated with vascular complications. Secondary analysis of the Perioperative Ischemic Evaluation (POISE) trial 2 demonstrated that perioperative bleeding (defined by bleeding that was disabling or required ≥2 units of RBC) was independently associated with myocardial infarction (MI) (adjusted odds ratio, 3.62; 95% CI, 2.01 to 6.36). 3 Analyses of POISE-2 trial data confirmed that perioperative major bleeding (defined by Hb ≤70 g/L and transfusion of ≥2 units of RBC; Hb drop ≥50 g/L and transfusion of ≥2 units of RBC; transfusion ≥4 units of RBC in 24 hours, or intervention; pressor requirement; surgery; death) was independently associated with MI (hazard ratio, 1.82; 95% CI, 1.40 to 2.36) 4 and acute kidney injury (aHR 2.2; 95% CI, 1.72 to 2.83). 5 Bleeding-related hypotension and anaemia leading to a mismatch in end-organ oxygen supply and demand is a plausible explanation for the consistently observed association between perioperative bleeding and these complications.
Given that most perioperative bleeding occurs prior to postoperative day two, 4 administration of an antifibrinolytic agent such as tranexamic acid (TXA) at the time of surgery may have substantial impact on subsequent bleeding risk. Several large randomised controlled trials (RCTs) of TXA have been reported recently in cardiac surgery (Aspirin and Tranexamic Acid for Coronary Artery Surgery, ATACAS 6 ), post-partum haemorrhage (World Maternal Antifibrinolytic Trial, WOMAN 7 ) and trauma (Clinical Randomisation of an Antifibrinolytic in Significant Haemorrhage 2, CRASH-2 8 ). These trials demonstrated that TXA is associated with reductions in bleeding,6–8 blood transfusion (ATACAS 6 ), death due to bleeding (CRASH-2 8 and WOMAN 7 ) and all-cause mortality (CRASH-2 8 ). However, results of these trials may not be generalisable to noncardiac surgical patients, who tend to be older and have more co morbidities than trauma and obstetric patients.
TXA has been widely reported to reduce bleeding and the risk of blood transfusion in patients undergoing lower limb arthroplasty, and TXA has been incorporated into patient blood management guidelines to reduce perioperative bleeding.9,10 A meta-analysis of 824 patients undergoing knee joint replacement and randomised to intravenous (IV) TXA or placebo demonstrated significant reductions in total blood loss as well as the proportion of patients requiring blood transfusion in those who received TXA. 11 A meta-analysis of 350 patients undergoing hip joint replacement showed significant, but clinically modest, reductions in total blood loss as well as a reduction in the proportion of patients requiring blood transfusion among those who received IV TXA. 12
TXA is an antifibrinolytic drug for which in vivo data shows an association with thrombus formation, 13 and there are numerous case reports of clinically significant thrombotic events associated with TXA use.14–16 However, the majority of RCTs of TXA in the arthroplasty setting have been small, moderate-quality trials primarily designed to examine bleeding endpoints. None have had sufficient power to demonstrate a difference in clinically important adverse events. Nevertheless, existing meta-analyses have failed to demonstrate any increased risk of clinically significant adverse events, including pulmonary embolus, with TXA.11,12,17
There is laboratory evidence to suggest that TXA may have a role in reducing surgical site infection. Laboratory data indicates that plasmin has both pro-inflammatory and immuno-modulatory effects, including induction of a tolerogenic phenotype in dendritic cells, which could promote the development of perioperative infection.18–21 TXA blocks the activation of plasminogen to plasmin thus providing a biologically plausible mechanism by which it may have an impact on perioperative infection risk. 22 Although the clinical significance of this potentially important effect of TXA remains unknown, large clinical trials to address this question are currently being planned.
With the potential indications for perioperative TXA expanding there is a need for large RCTs to determine the safety and effectiveness of TXA in noncardiac surgery. 23 However, to gauge feasibility and optimise the design and value of large, pragmatic trials it is essential to first ascertain current practice and beliefs regarding perioperative TXA administration. We therefore undertook a survey of Fellows of the Australian and New Zealand College of Anaesthetists (ANZCA). Our primary aim was to ascertain current patterns of TXA administration. Secondary aims included assessing anaesthetists’ understanding of the evidence and their views on the acceptability of randomising patients from different surgical specialties to IV TXA or placebo.
Materials and methods
This survey was prospectively approved by the Central Adelaide Local Health Network Human Research Ethics Committee (CALHN HREC Reference number: HREC/18/CALHN/190; date of approval: 11 April 2018). The survey was generated using SurveyMonkey® (SurveyMonkey Inc., San Mateo, CA, USA). The survey complied with the ANZCA Survey Research Policy. 24 After trialling the survey items with the investigators to ascertain ease of use, the survey was approved and administered by the ANZCA Clinical Trials Network. The survey items are provided in Appendix 1. A cover letter and link to the survey was sent by electronic mail to a randomly selected sample of 1001 fellows of ANZCA on 22 May 2018. With an expected response rate of 30% we anticipated 300 responses, which in a recent similar survey provided a broad representation of demographic characteristics and practice type of ANZCA Fellows. 25 The investigators were not involved in the selection process and were blinded from the identity of those who received the survey. Accordingly, information about non-responders could not be obtained. All responses were anonymous, and no internet protocol addresses were collected. A reminder was sent two weeks after the initial invitation and the survey closed two weeks after the reminder was sent (19 June 2018).
Questions 1 to 3 asked for basic demographic information (sex, location of practice and age) to enable comparison of respondent demographics to those of the entire ANZCA Fellowship. Questions 4 to 6 were designed to ensure that respondents currently practise adult anaesthesia and have previously administered IV TXA in a noncardiac surgical setting. Respondents not currently practising adult anaesthesia were taken to the final two questions, 11 and 12. Respondents indicating that they had never administered IV TXA were asked to provide a reason for this response (Question 6). Questions 7 to 10 sought to characterise the pattern and administration of IV TXA use in terms of use of TXA as prophylaxis or rescue therapy, dose adjustments, dosing pattern (i.e. single dose/repeated doses) and criteria for use in specific surgical groups in which the respondent administers TXA. Question 11 examined respondent beliefs about absolute contraindications to TXA administration and Question 12 assessed attitudes to the current level of evidence and the perceived need for one or more large RCTs to provide level-one evidence to guide the use of IV TXA in noncardiac surgery.
Eight of the items offered multiple choice options requiring a single response, of which three had additional free-text options. Two of the items offered checkboxes that allowed multiple responses, of which one provided an additional free-text option. Two of the items were a matrix of dropdown menus, one of which had an additional free-text option. There was an additional free-text box for comments at the end of the survey.
Data were downloaded from SurveyMonkey® as a .csv file and imported into Stata 12 (StataCorp, College Station, TX, USA) for analysis. Frequency distributions and summary statistics were generated as counts and proportions, with 95% confidence intervals (CIs) to characterise current TXA usage patterns. Due to the nature and purpose of the survey there were no pre-specified hypotheses for testing.
Results
Of the 1001 surveys sent, 986 were successfully delivered. From this there were 289 responses, a response rate of 29.3%. The characteristics of the respondents were similar to those for the entire ANZCA Fellowship (Table 1).
Characteristics of the respondents.
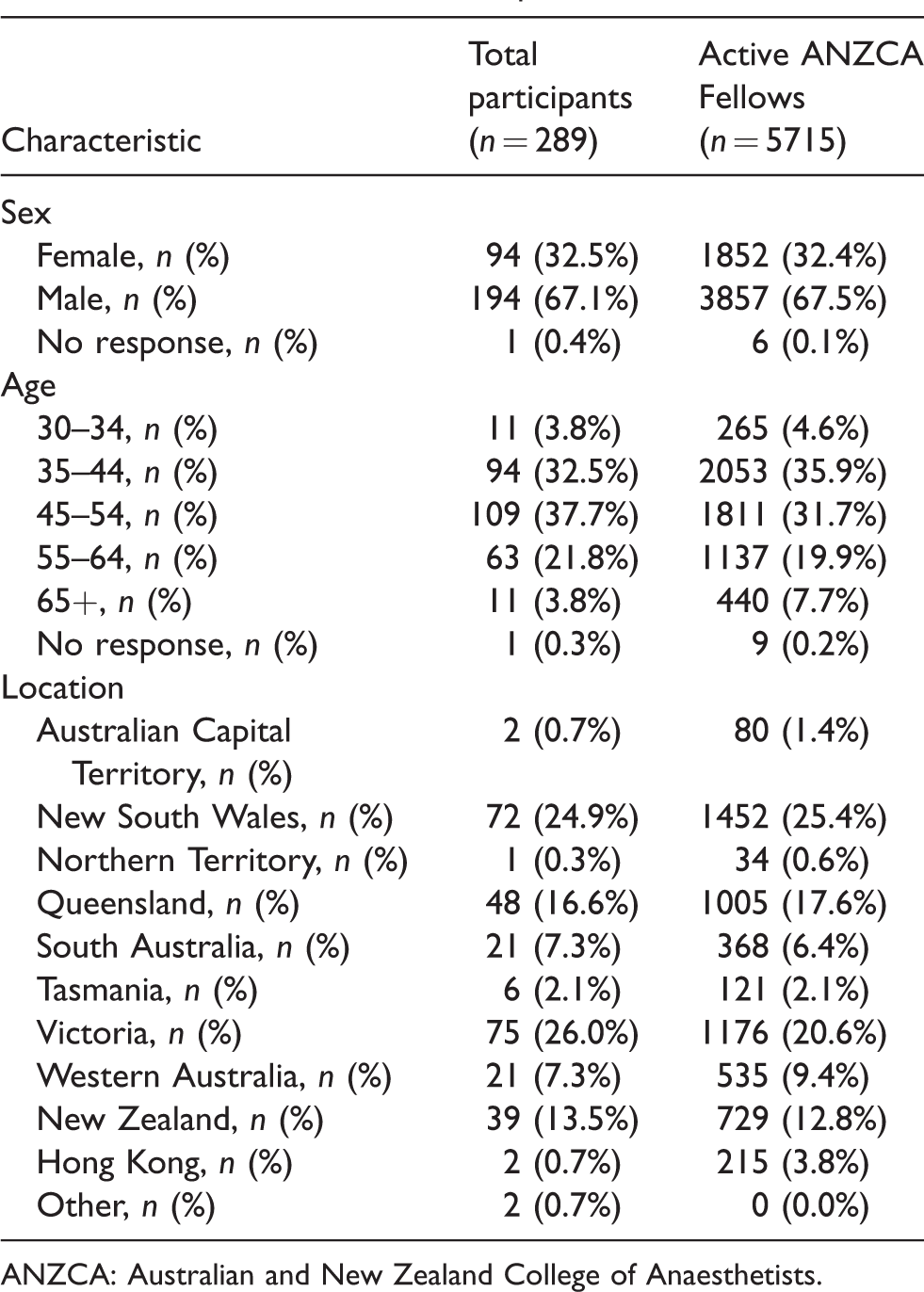
ANZCA: Australian and New Zealand College of Anaesthetists.
Almost all respondents (284, 98.3%) had administered IV TXA in noncardiac surgery with nearly half (127, 46%) administering TXA prophylactically only and half (137, 49.6%) administering TXA as both a prophylactic and a rescue intervention. Only 12 (4.3%) reported using TXA only as a rescue intervention. A single intraoperative dose (174, 63.5%) was the most common pattern of dosing for IV TXA. Approximately 10% of respondents either administer a second dose at the end of surgery or follow the initial bolus with an infusion. Only 27 respondents (9.9%) gave three boluses over 16–24 hours, which is consistent with the regimen recommended in the TXA product information for lower limb arthroplasty 26 (Table 2). A majority of respondents (179, 65%) administered IV TXA as a fixed dose, while 72 (26%) adjusted TXA dose for weight. Only 18 respondents (6.5%) reported adjusting dose according to renal function (Table 3).
Typical dosing pattern for prophylactic intravenous tranexamic acid.
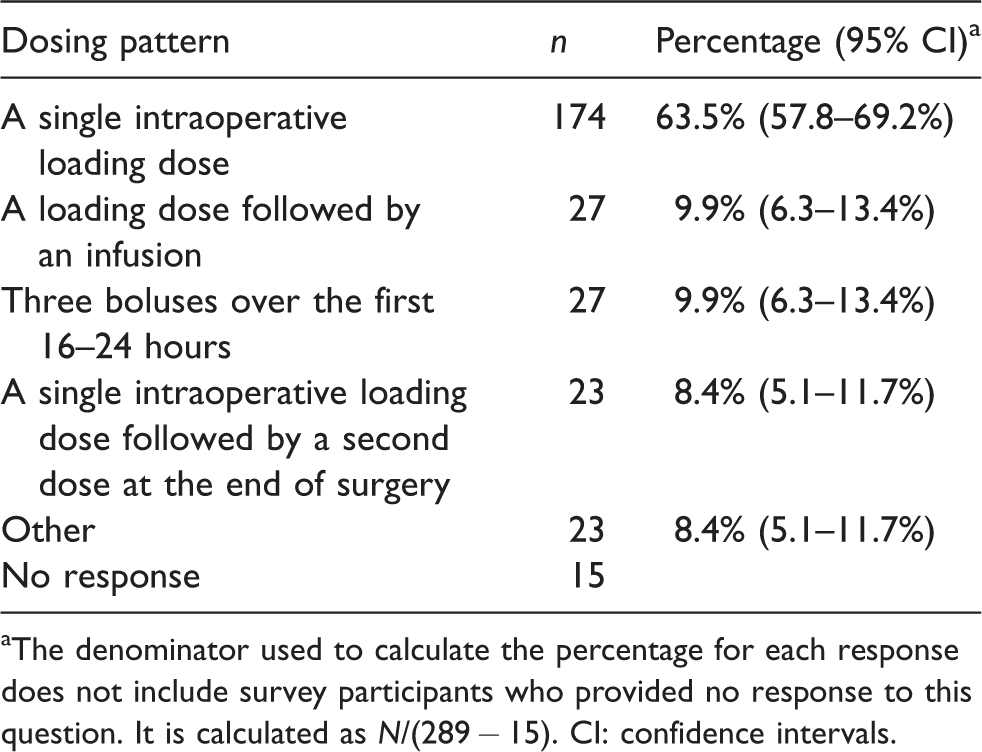
aThe denominator used to calculate the percentage for each response does not include survey participants who provided no response to this question. It is calculated as N/(289 − 15). CI: confidence intervals.
Typical dosing adjustments for prophylactic intravenous tranexamic acid.
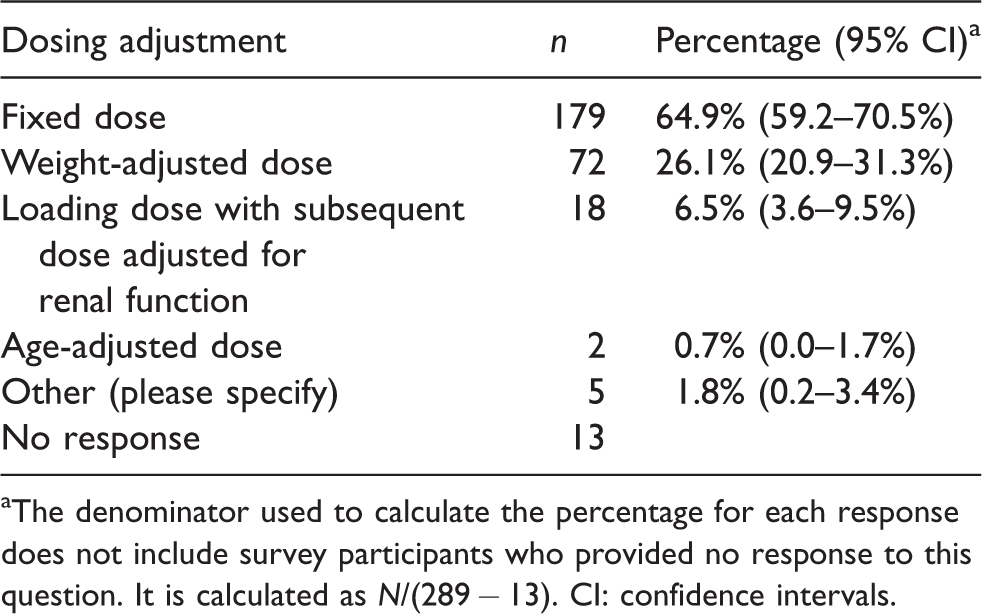
aThe denominator used to calculate the percentage for each response does not include survey participants who provided no response to this question. It is calculated as N/(289 − 13). CI: confidence intervals.
Known allergy to TXA was identified by 271 respondents (94%) as an absolute contraindication to its use. Up to 30% of respondents identified a history of arterial thromboembolism, spontaneous venous thromboembolism or a prothrombotic disorder (such as Factor V Leiden deficiency) as absolute contraindications to IV TXA. A small minority of respondents identified a history of ischaemic stroke (10%) or provoked venous thromboembolism (12%) as absolute contraindications to IV TXA (Table 4).
Perceived absolute contraindications to tranexamic acid administration.
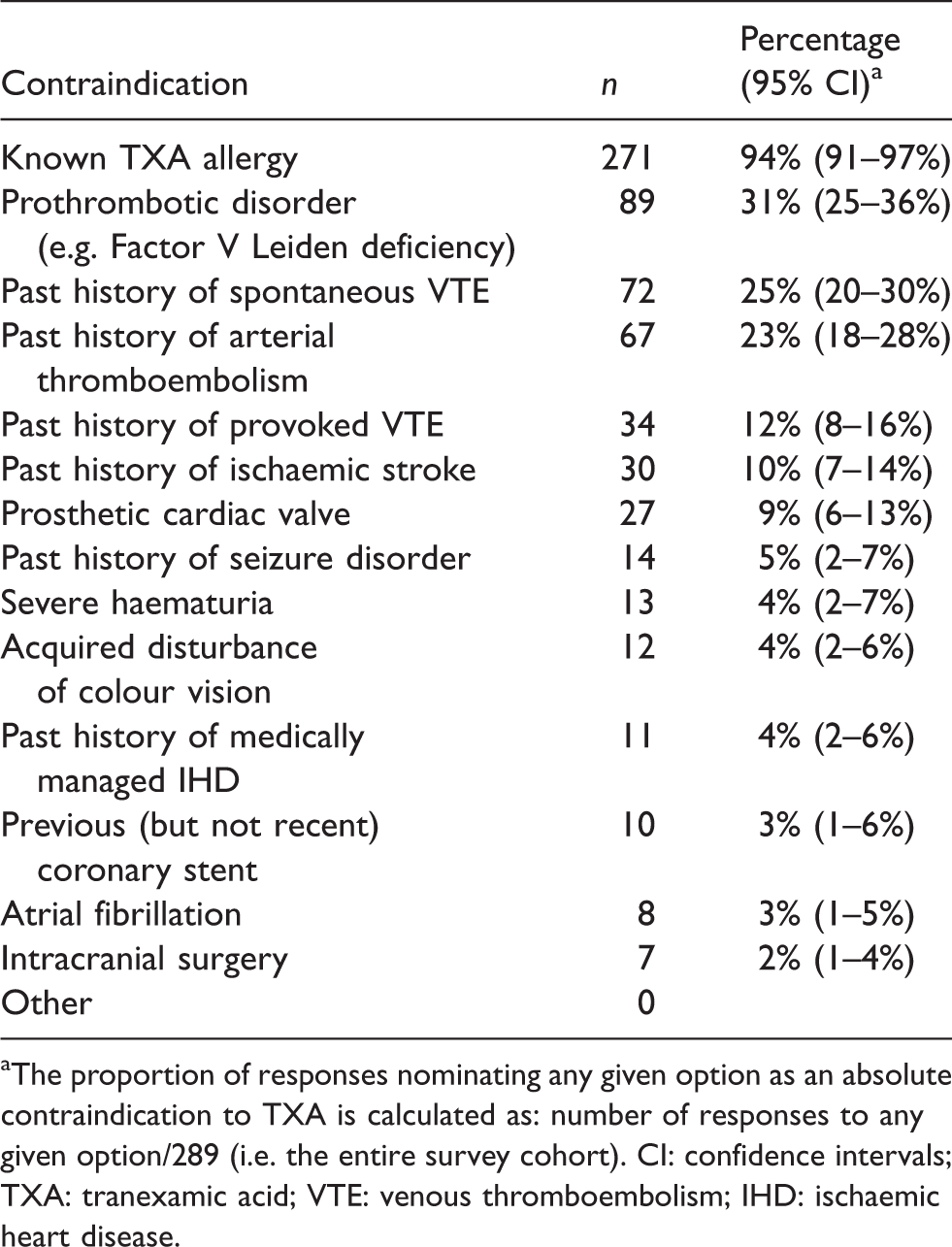
aThe proportion of responses nominating any given option as an absolute contraindication to TXA is calculated as: number of responses to any given option/289 (i.e. the entire survey cohort). CI: confidence intervals; TXA: tranexamic acid; VTE: venous thromboembolism; IHD: ischaemic heart disease.
There was marked variation in IV TXA use across specific surgical subgroups. After excluding those who either did not respond or indicated ‘Not applicable’ to each of these questions, 67% of respondents indicated that they routinely administer IV TXA in elective lower limb arthroplasty. The next most common surgical group receiving routine IV TXA was major spinal surgery (40%) followed by other major orthopaedic surgery (28%). No other elective surgical group received IV TXA routinely from more than 11% of respondents. On average, IV TXA was given on surgical request in approximately 30% of elective surgical settings listed in the survey. Approximately half (49%) indicated that they give IV TXA routinely for major trauma surgery. Thirty-six percent of respondents indicated that they did not give IV TXA for major vascular, abdominal or thoracic surgery (Table 5).
Tranexamic acid use in specific surgical groups.
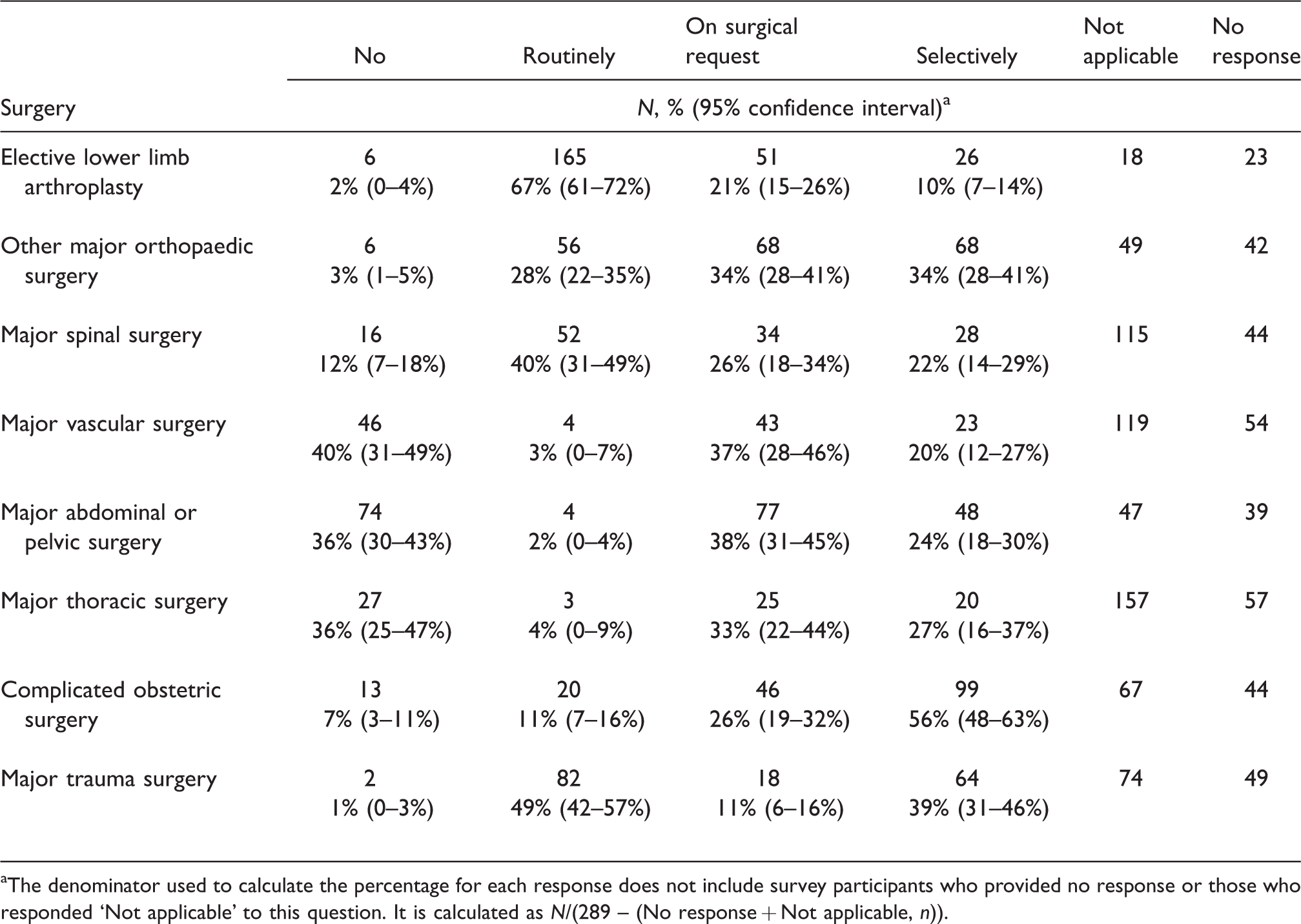
aThe denominator used to calculate the percentage for each response does not include survey participants who provided no response or those who responded ‘Not applicable’ to this question. It is calculated as N/(289 – (No response + Not applicable, n)).
In terms of perceptions about the safety and efficacy of IV TXA in noncardiac surgery, 57% agreed that there is uncertainty about the relative risks and benefits of perioperative IV TXA (although 34% disagreed with this statement). The majority (87%) of respondents agreed that RCTs to determine the safety and efficacy of IV TXA in noncardiac surgery are required (Table 6).
Perceived uncertainty about risks and benefits and need for large trials of tranexamic acid.
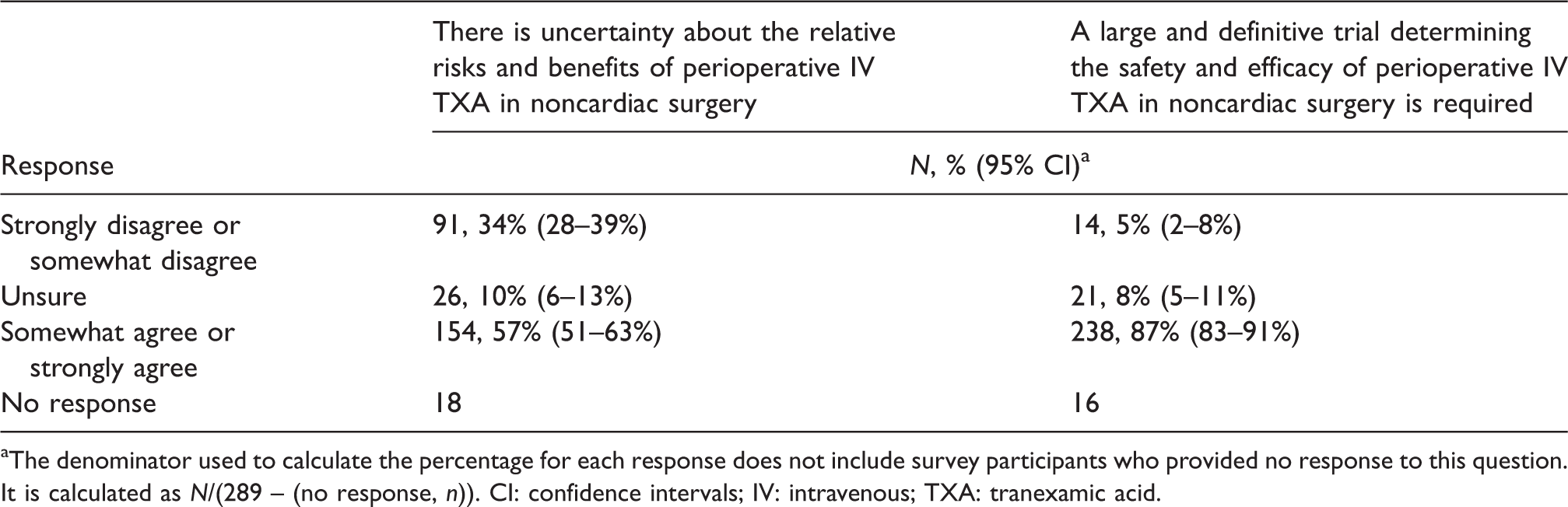
aThe denominator used to calculate the percentage for each response does not include survey participants who provided no response to this question. It is calculated as N/(289 – (no response, n)). CI: confidence intervals; IV: intravenous; TXA: tranexamic acid.
Discussion
This survey of the perioperative use of IV TXA in noncardiac surgery by ANZCA Fellows reveals a number of key messages. Firstly, TXA administration in this context is common: almost all respondents have administered IV TXA to patients undergoing noncardiac surgery. Secondly, while most respondents administer IV TXA routinely for lower limb arthroplasty (the only approved surgical indication for IV TXA in noncardiac surgery), TXA is also frequently administered in non-approved (off-label) surgical settings either on surgical request or selectively on a case-by-case basis. Thirdly, there is marked variation in dosing regimen, with only 10% of respondents administering TXA as recommended in the product information. 26 Finally, although there was variation in the level of uncertainty expressed about the relative risks and benefits of TXA in noncardiac surgery, there appears to be strong support for large RCTs to examine the efficacy and safety of TXA within this context.
Most respondents indicated that they administer TXA as a single fixed intraoperative dose. With an elimination half-life of two hours for a 1g bolus of IV TXA, such a dosing regimen is unlikely to provide adequate serum concentrations of TXA for more than eight hours. 27 The dosing regimen recommended in the TXA product information requires three doses over a 24-hour period, adjusted for weight and tapered for declining renal function. 26 While there is minimal literature comparing the effectiveness of different dosing regimens in relation to perioperative bleeding, a meta-analysis of RCTs of IV TXA in lower limb arthroplasty suggests that a bolus dose followed by either a second bolus or an infusion is more effective in reducing the need for blood transfusion than a single bolus alone. 28 Moreover, existing data from noncardiac surgery confirms that the risk of significant bleeding extends well beyond the conclusion of surgery. 4 The observed discrepancy between the current typical practice of single dose TXA and existing pharmacokinetic and clinical evidence highlights the need for careful consideration of the dosing regimen for future RCTs, needing to provide both optimal antifibrinolytic activity of TXA and maximal feasibility of continued administration of TXA beyond the completion of surgery, a pattern that is currently different to the majority of respondents. Finally, the frequent identification of ‘surgical request’ as a reason to use TXA suggests that surgeons continue to be closely involved in decisions about the perioperative administration of TXA. As a result, their acceptance of clinical equipoise in this debate and their engagement in trial development will be essential for trial success.
Respondents’ beliefs regarding absolute contraindications for TXA were variable. For example, fewer than 100% of respondents appeared to recognise known TXA allergy as a contraindication to its administration. This may reflect the fact that TXA allergy is comparatively rare. The product information states that TXA should not be given to patients with a history or risk of thrombosis unless it is possible to co-administer an anticoagulant. 26 Nevertheless, only 25–30% of respondents answered that a history of thromboembolic disease or high risk of arterial thrombosis was a contraindication to TXA. This most likely reflects the fact that no trial of TXA to date has shown an increased risk of thrombosis and underscores the fact that there is indeed uncertainty about the risk of giving TXA in noncardiac surgery.
This survey has several limitations. The response rate of 29.3%, while consistent with other ANZCA surveys, is still low and may have led to unrecognised bias in the results. Respondents may have been those who tend to use TXA, creating inadvertent selection bias and limiting the generalisability of the results to the entire cohort of current ANZCA Fellows. However, respondent demographics appear broadly representative of ANZCA Fellowship. In order to keep the survey short and minimise the burden on respondents, we used a limited number of items that spanned a broad range of noncardiac surgical settings. This restricted the range of possible responses meaning our survey may not have captured the full breadth of clinical practice patterns. Although the wording of survey questions and available responses were carefully considered by the investigators prior to conducting the survey, it was notable that up to 20% of respondents recorded ‘No response’ to Question 10 that pertained to TXA use in different surgical settings (Table 5). It is not possible to determine whether the item was unclear, or whether these respondents intended to reply ‘Not applicable’ (i.e. indicating that they do not provide anaesthesia for a particular surgical group). Lastly, because of our limited item set, we collected minimal demographic information from respondents about practice type, experience and case mix.
In conclusion, assuming that the responses are representative of the broader ANZCA Fellowship, TXA is currently administered in a wide variety of noncardiac surgical settings by Fellows of ANZCA. The majority give TXA routinely in elective lower limb arthroplasty surgery, and on surgical request or selectively for other elective noncardiac surgery, including many where TXA is not approved. 26 There is marked variation in dosing regimen although the majority give a single, fixed dose of TXA. Fifty-seven percent agree that there is uncertainty about the risks and benefits of TXA in noncardiac surgery, and 87% agree that large and definitive RCTs determining the safety and efficacy of perioperative IV TXA in noncardiac surgery are required. The results of this survey support the premise that large RCTs of IV TXA in noncardiac surgery in our region are acceptable and of interest to Fellows of ANZCA.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
