Abstract
Background:
Three-dimensional printer technology can produce the personalized orthosis in various forms.
Objective:
To develop a personalized wrist orthosis using a three-dimensional scanner and three-dimensional printer for patients with wrist pain.
Study design:
A preliminary, prospective, randomized, open-label study.
Methods:
A total of 22 patients with wrist pain were randomly assigned to the control and experimental groups. The control group wore a cock-up orthosis and the experimental group wore a three-dimensional-printed wrist orthosis for 1 week. The Patient-Rated Wrist Evaluation, Jebsen Hand Function Test, and Orthotics and Prosthetics Users’ Survey were checked before and 1 week after the application.
Results:
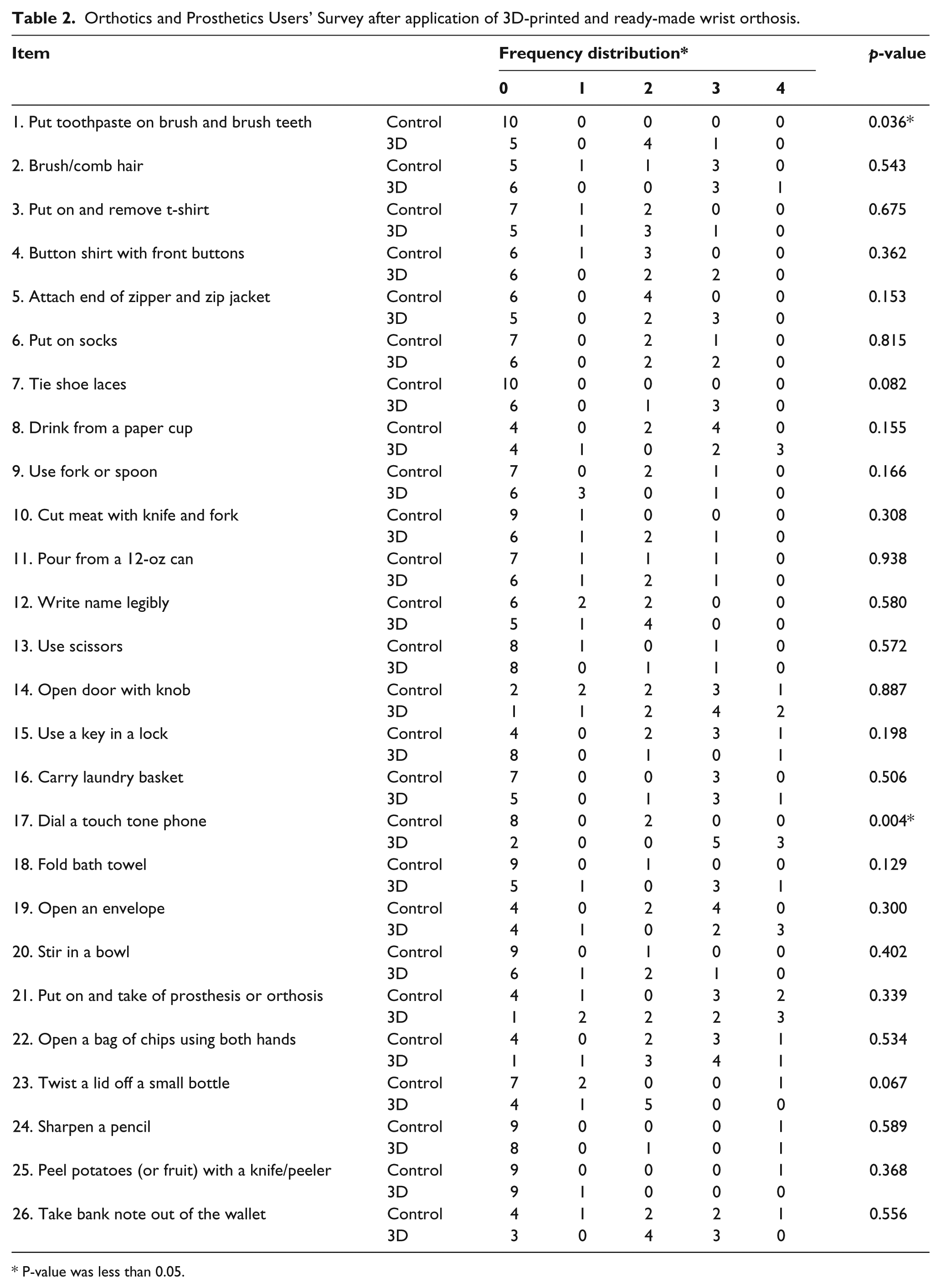
The Patient-Rated Wrist Evaluation showed significant pain relief in both groups. Two items of the 28 Orthotics and Prosthetics Users’ Survey questions, “Put toothpaste on brush and brush teeth” and “Dial a touch tone phone,” showed high satisfaction scores, with statistically significant difference in the experimental group (p = 0.036 and 0.004).
Conclusion:
The three-dimensional-printed wrist orthosis was superior to the cock-up orthosis for two items of the Orthotics and Prosthetics Users’ Survey. Wrist pain was reduced in the group wearing the three-dimensional-printed wrist orthosis as well as the group wearing the cock-up orthosis, so the three-dimensional-printed wrist orthosis could possibly play the same role as the cock-up orthosis.
Clinical relevance
A three-dimensional-printed wrist orthosis can be a substitute for a conventional ready-made wrist orthosis for patients with wrist pain with more satisfaction.
Background
Computers and smart phones are often used for prolonged periods each day, and occupations that involve video display terminals (VDTs) are increasing every year. 1 VDT use has been linked with increasing likelihood of work-related musculoskeletal disorders (WMSDs) caused by accumulated damage to extremely fine muscles or tissue by repetitive work-related movements. WMSDs mainly involve parts of the back, neck, shoulders, arms, and wrists. Upper-extremity musculoskeletal disease can occur in the wrists2–5 and is referred to as “overuse syndrome.” The persistent pain caused by overuse syndrome can affect the function of the entire hand.6,7
Common treatments for overuse syndrome include refraining from the aggravating activity, injection of pain relief medication, or wearing of a wrist orthosis.8,9
Wearing a wrist orthosis allows unhindered continuation of the existing work and maintains the durability of work. Mass produced, ready-made orthoses are of lower quality and are bulky, and uncomfortable to wear compared to custom-made wrist orthoses. However, custom-made wrist orthoses are more expensive than ready-made and take a long time to produce.
Three-dimensional (3D) printer technology can overcome these problems by producing personalized medical products with low cost and reduced time.10,11 3D printing data files are also advantageous for the production of individualized products at no extra cost, even when different designs are used in consecutive production runs. In addition, because the 3D printer is controlled by a computer, it can produce objects in various forms and is easy to use compared to other manufacturing techniques. 12
The purpose of this study was to develop a personalized wrist orthosis using a 3D scanner and 3D printer for patients with wrist pain caused by overuse syndrome and to evaluate the improvement of pain relief, work performance, and daily life satisfaction after wearing the orthosis.
Subjects and methods
The study was conducted with the approval of the Institutional Review Board of our hospital (IRB 2015-10-097), and the clinical trial registration number is PRE20170118-001 in the clinical research information service (http://cris.nih.go.kr).
Subjects
Subjects were recruited from an advertisement posted publicly in the outpatient clinic of the university hospital. Informed written consent was obtained from each participant. The preliminary study involved 22 subjects (20 women, 2 men) with overuse syndrome in the upper wrist area. They were randomly assigned to the control group (n = 11) and the experimental group (n = 11). The mean age was 31.8 ± 7.8 years for the control group and 33.8 ± 8.5 years for the experimental group (p = 0.591). The inclusion criteria were an age of 19–65 years, wrist pain that had persisted for more than 1 month, and relief of wrist pain upon resting for more than 30 min. The exclusion criteria were disease related to the central or peripheral nervous system, musculoskeletal disorder of the upper limb except wrist pain, history of upper extremity surgery or trauma, deformation or arthritis of the wrist bone on X-ray, and cognitive impairment with inability to express feelings of pain, itching, or skin rash.
Randomization
Patients who met both the inclusion criteria and the exclusion criteria were randomly assigned to the control or experimental group using a random number table with a random permuted block design and a block size of 4. Those in the control group wore a ready-made device (Spectra Wrist Brace; Ossur, Reykjavik, Iceland). Those in the experimental group wore a custom-made 3D-printed device that was made using an Eva 3D scanner (Artec Group, Luxembourg City, Luxembourg).
Methods
For a 3D-printed wrist orthosis, scanning was performed from the center of the forearm to the fingertip while keeping the wrist joint at a slightly dorsiflexed position and the fingers loose (Figure 1). A 3D scanner (Artec™ Eva, Artec Group) was used in this study to scan the wrist and hand. The scanner captured up to 16 frames per second and these frames were aligned automatically in real time, which made scanning easy and fast. With automatic alignment and pairing of the two-dimensional (2D) captured images, 3D surface images were acquired (Figure 1).

Three-dimensional scanning was performed from the center of the forearm to the fingertip while keeping the wrist joint at neutral position and the fingers loosened.
After acquiring 3D surface images from the scanning technique, the 3D-printed wrist orthosis was designed using scanned 3D images through Geomagic Touch (3D SYSTEMS Corp., Rock Hill, SC, USA) and Geomagic Freeform software (3D SYSTEMS Corp.; Figure 2). The thickness of the orthosis was set to be 1.8 mm and the proximal border of the orthosis was the midpoint of the forearm. The inner surface of the orthosis was separated 1 mm from the 3D surface images, which made the orthosis well ventilated and well drained. Based on the prepared design, the wrist orthosis was printed using a fused filament fabrication (FFF) FINEBOT Z420 3D printer (TPC Mechatronics Inc., Incheon, Korea). An occupational therapist who is an expert in manipulating 3D printers was involved in printing the orthosis. Thermoplastic polyurethane (TPU) filament was used as the output material, and the output surface directly touching the skin was smoothed through a post-printing process. The post-printing process was the removal of by-products used as supporting structures during the FFF-type printing process. This post-printing process was conducted manually using pincers and a grinding machine. Pincers were used to remove the support materials from the printed orthosis and grinding machine was used to smooth the surface of the orthosis. There was no manual adjustment of the orthosis after the post-printing process. Those in the control group wore a ready-made, cock-up orthosis, which is most commonly used for wrist pain in clinics. The cock-up orthosis was made from a fabric of similar shape to the 3D-printed wrist orthosis and a steel bar in the midline area to limit wrist motion (Figure 3). The size was selected among “xlarge,” “large,” “medium,” and “small” according to patient wrist size. Both groups wore the orthosis for 1 week.

The 3D-printed wrist orthosis was designed using scanned 3D images through geomagic touch and geomagic freeform software.

The wrist orthosis was printed out using fused filament fabrication (FFF) type 3D printer. Thermoplastic polyurethane (TPU) filament was used as the output material, and the inner surface directly touching the skin was smoothed through post-printing process. A ready-made wrist orthosis was used for control group.
Evaluation
The evaluation was performed before and after wearing the wrist orthosis for 1 week. The Patient-Rated Wrist Evaluation (PRWE) method was used to evaluate pain and function. PRWE was developed to evaluate hand function after distal radius fracture 13 and has been widely used to evaluate wrist pain and function. 14 It consists of 15 questions divided into pain and function subscales. The Jebsen Hand Function Test (JHFT) was used to assess hand functionality during use of the wrist orthosis. The JHFT was designed to assess broad hand functions that are commonly used in activities of daily living. 15 The test consists of the seven tasks of writing, turning over cards, picking up small objects, simulated feeding, stacking checkers, picking up large light cans, and picking up large heavy cans. The Orthotics and Prosthetics Users’ Survey (OPUS) was used to assess overall satisfaction with the wrist orthosis. 16 The OPUS has been validated in many conditions.17–19 The Consolidated Standards of Reporting Trials (CONSORT) flow diagram for randomized controlled study to improve the reporting of a randomized controlled study 20 is presented in Figure 4.
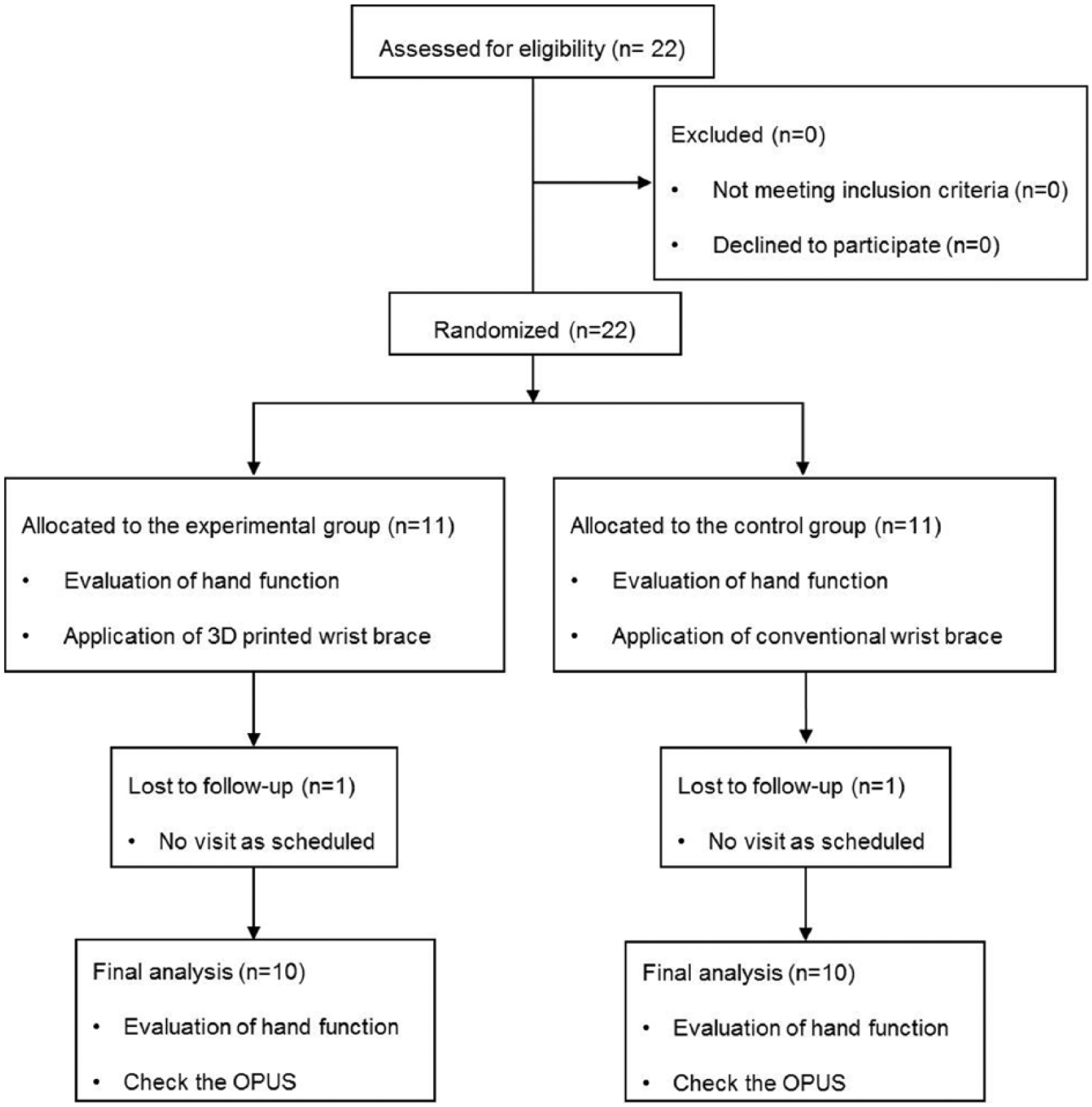
The Consolidated Standards of Reporting Trials (CONSORT) flow diagram was presented. Data from total 20 patients were analyzed in the final analysis.
Patients were educated to report any possible side effects related to the orthosis and occurrence of skin rash or ulcer were investigated after 1 week of use of the orthosis.
Statistical analysis
SPSS version 18.0 program (IBM Corp., Armonk, NY, USA) was used for statistical analysis of the data. A Mann–Whitney U test was performed to analyze the difference in the continuous variables between the control and experimental groups and a chi-square test for the categorical and ordinal scale variables. The statistical significance level was α = 0.05.
Results
One patient in the experiment group and one patient in the control group were lost to follow-up and were excluded from our analysis. Thus, data from 20 patients were analyzed for this study.
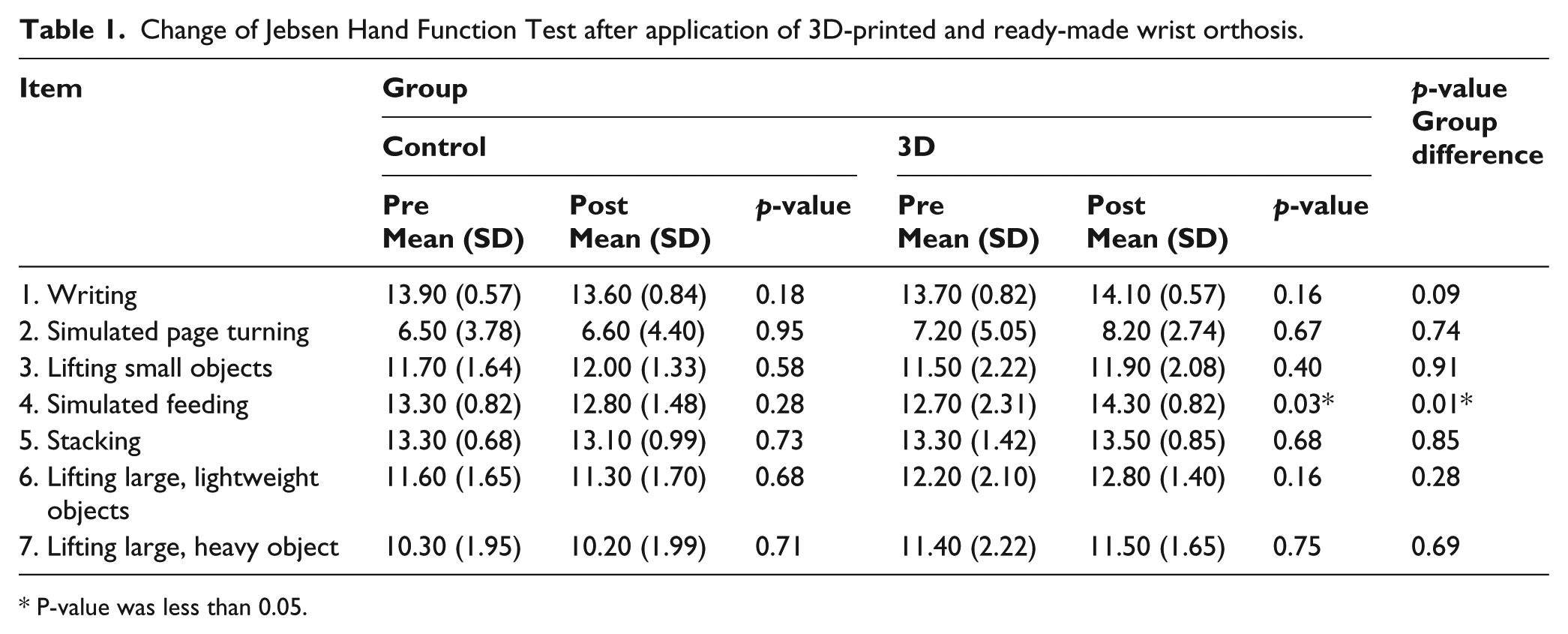
In the PRWE, the mean pain score before wearing the wrist orthosis was 28.8 ± 4.8 in the control group and 29.3 ± 8.6 in the experimental group, without a significant difference (P = 0.874). The mean pain score after 1 week of wearing the wrist orthosis was 23.4 ± 6.9 in the control group and 19.2 ± 6.2 in the experimental group. Both groups showed pain relief, but pain tended to be relieved more in the experimental group although the difference was not significant (p = 0.109). The change of JHFT after 1 week was not significantly different between the control and experimental groups (1.0 ± 6.8 and 4.3 ± 6.9, p = 0.101). The sub-items of the JHFT are presented in Table 1. The experimental group spent significantly more time wearing the device (10.0 ± 6.3 h per day) than the control group (3.7 ± 1.3 h per day, p = 0.006). Two items of the 28 OPUS questions—“Put toothpaste on brush and brush teeth” and “Dial a touch tone phone”—showed high satisfaction scores, with statistically significant difference in the experimental group wearing the 3D-printed wrist orthosis (p = 0.036 and 0.004). The sub-items of the OPUS are presented in Table 2.
Change of Jebsen Hand Function Test after application of 3D-printed and ready-made wrist orthosis.
P-value was less than 0.05.
Orthotics and Prosthetics Users’ Survey after application of 3D-printed and ready-made wrist orthosis.
P-value was less than 0.05.
Discussion
The 3D-printed wrist orthosis showed better scores for two items of the OPUS than the ready-made wrist orthosis. Wrist pain was reduced in the group wearing the 3D-printed wrist orthosis as well as the group wearing the ready-made wrist orthosis. There was no difference in PRWE between the two groups, possibly due to wearing the wrist orthosis for just 1 week, which is a very short period to observe differences in pain. Because of the similar pain reduction effect in the two groups, the 3D-printed wrist orthosis could possibly play the same role as the ready-made device.
JHFT revealed no significant group-wise difference in work performance immediately after and 1 week after wearing a device, indicating that the 3D-printed wrist orthosis and the ready-made wrist orthosis have the same work performance. The 3D-printed wrist orthosis was thought to be superior in performance because of its lightweight and simple design. However, the two groups showed no difference since the JHFT test was developed for patients with functional disorders like stroke, so there might be a ceiling effect in this study.
In the OPUS, the two items “Put toothpaste on brush and brush teeth” and “Dial a touch tone phone” showed higher satisfaction scores in the experimental group than in the control group. For “Put toothpaste on brush and brush teeth,” this high satisfaction was due to the design that enabled water drainage. This design improved the satisfaction and wearing sensation of the patient. For “Dial a touch phone,” the high satisfaction was due to the relatively small size and snug fit of the 3D-printed wrist orthosis.
In addition, the group with the 3D-printed wrist orthosis wore the orthosis for 7 h longer per 24-h day than those wearing the ready-made orthosis, suggesting increased comfort of the 3D-printed wrist orthosis. The 3D-printed wrist orthosis is thinner than a ready-made wrist orthosis (1.8 vs 4 mm). Compared to a ready-made wrist orthosis made of synthetic fibers, the 3D-printed wrist orthosis has the advantage of being made of TPU filament, material that is similar to urethane and adheres well to the skin. However, there was a limitation in evaluating the satisfaction of both devices in the same patient because it was a parallel study design, not a cross-over study design. We could not adopt a cross-over study design because wrist pain is not a chronic stable disease.
There was no significant difference in pain relief effect and task-performing ability between those wearing a 3D-printed wrist orthosis and those wearing the ready-made wrist orthosis. In addition, the two devices have the same function. The possible explanation is the small sample size because it was a preliminary study. Further study with a large sample size must be conducted to clarify the difference in pain relief and task-performing ability between the 3D-printed and ready-made wrist orthoses.
Higher user satisfaction was observed in the group with the 3D-printed wrist orthosis. This is not surprising since the 3D-printed wrist orthosis is a personalized product. If the 3D-printed wrist orthosis has the same functionality as the ready-made wrist orthosis, the clinical preference of the 3D-printed wrist orthosis is expected to be greater. In addition, there are other additional advantages in the production of 3D-printed wrist orthoses. The design data is stored, so the orthosis can be easily re-printed when the product is lost or worn out. Furthermore, since the 3D data can be easily stored for a long time and can be transferred via e-mail or cloud system, the device can be produced remotely. However, there is one problem hindering the widespread popularity of the 3D-printed orthosis. The FFF-type 3D printer using TPU filament must use supporting structures to produce the 3D-printed orthosis. Post-printing processing to remove these structures is mandatory but is time-consuming and onerous.
The total amount of time to make the 3D-printed wrist orthosis was approximately 6 h. This was faster than the several days to a week needed to make an individualized wrist orthosis produced with a cast molding technique.
The total cost for scanning, image fusion, modeling, and post-printing was approximately US$50 and required two people, at a salary of approximately US$4000/month, with the capacity to make 80 wrist orthoses in 1 month. The cost of materials used for manufacturing one wrist brace was approximately US$20, so the total cost for one wrist brace was approximately US$70. This was similar to the cost of the ready-made cock-up orthosis, but had better satisfaction than the ready-made cock-up orthosis. Although we did not compare the function and satisfaction between the 3D-printed orthosis and a commercialized personalized orthosis with similar function, the 3D-printed orthosis is cheaper than a personalized orthosis which is made by direct forming of low-temperature thermoplastics (about US$300). Considering that 3D printing techniques are being developed and introduced quickly, the post-processing is expected to be eliminated, and the cost of 3D-printed materials will decrease, suggesting the development and the use of personalized 3D-printed wrist orthoses.
In this study, we show the possibility of 3D-printed wrist orthosis as a substitute for conventional ready-made wrist orthoses for patients with wrist pain. This 3D printing technique will become more popular with further refinement including addressing any post-printing process hurdles.
Conclusion
The 3D-printed wrist orthosis was designed using scanned 3D images and printed out using thermoplastic polyurethane filament by 3D printer.
The 3D-printed wrist orthosis was superior to the cock-up orthosis in some items of the OPUS.
Wrist pain was reduced in the 3D-printed wrist orthosis as well as the cock-up orthosis, so the 3D-printed wrist orthosis could possibly play the same role as the off-the-shelf cock-up orthosis.
Footnotes
Author contribution
Sang Jun Kim and Sung Jae Kim are co-first authors. All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the ICT R&D program of MSIP/IIPT (Grant No. B0101-17-1081) and by the Korea Institute for Advancement of Technology (Grant No. R0004587).
