Abstract
Background:
Diabetes mellitus causes a multitude of complications. Foot ulceration is one complication with serious consequences, amputation. Foot deformities contribute to ulcer development. It would be advantageous to ascertain whether foot deformities are preventable as their presence increases amputation risk.
Objectives:
The objectives were to understand the development of foot deformities in diabetes and explore their biomechanical effects.
Study design:
Literature review.
Methods:
In February 2014 CINAHL, Embase, Ovid and Medline were searched. Studies in English of adults with Diabetes that reported neuropathy, foot deformities or associated gait abnormalities were included for review. All study designs were considered. The articles’ quality was considered high overall, assessed using SIGN and CARS.
Results:
In total, 17 studies were reviewed. The main themes identified in relation to foot deformities were nerve function, intrinsic foot muscles, muscle weakness and limited joint mobility. Nerve function and intrinsic foot muscle atrophy did not display definitive associations with foot deformities. However, muscle weakness and limited joint mobility were associated with foot deformities, although the relationship is still unclear.
Conclusion:
The development of common foot deformities in diabetes is not well understood. The literature did not support the common belief that motor neuropathy, atrophy and muscle imbalance cause foot deformities.
Clinical relevance
An understanding of the aetiology of foot deformities in diabetes mellitus may allow for pro-active management of the foot in anticipation of the development of foot deformities and ulceration. If the aetiology of deformity was established preventative treatment may reduce the incidence of foot deformities and resultant ulcerations and amputations.
Background
Need for review
A full understanding of the common foot deformities within diabetes mellitus (DM) and the pathway that ultimately leads to them would allow for optimal treatment of diabetic patients within a multi-disciplinary team (MDT). It would be advantageous to ascertain whether foot deformities within DM are a preventable complication because amputation risk increases when foot deformities are present. The risk has been reported to rise from 1.7-fold once protective sensation is lost to 12-fold with both sensory loss and foot deformity within DM.1,2 DM affects approximately 60 million people in the European Region, about 10.3% of men and 9.6% of women aged 25 years and over according to the World Health Organization (WHO). 3 With such a significant portion of the population affected, it is essential that the effects and complications of this disease be fully understood so that they can be addressed in adequate time to prevent complications where possible. It is generally accepted that foot deformities within DM occur as a result of motor neuropathy causing atrophy and muscle imbalance. The structural foot deformities that are commonly reported are claw and hammer toes, 4 prominent metatarsal heads, pes cavus, 5 pes equinus and hallux valgus. 6 The functional foot deformities that are prevalent in DM are hallux limitus and hallux rigidus. 6
Overview of problem
The St Vincent Declaration established the target to reduce amputation rates by 50%. An expert opinion was obtained from David G Armstrong (DPM, MD, PhD, Professor of Surgery, and Director of Southern Arizona Limb Salvage Alliance) on the success of the St Vincent Declaration. He stated,
If one looks at the St. Vincent document, one can find successes and failures. Throughout the developing world, major amputation amongst people with T2DM is rising. This is clearly a failure. However, some nations in Northern Europe as well as in the USA can identify a drop (at least from the mid-1990s) in high-level amputation. The story is mixed. What is most mixed, however, is the so-called ‘postcode lottery’ that shows variability in amputations by up to 10-fold in the NHS and in the USA.7,8
This statement highlights the sporadic increase in amputations. This is a clear shortcoming that must be addressed.
Approximately 75%–85% of amputations are preceded by ulceration.9,10 Therefore, a pro-active approach to preventing ulceration would subsequently reduce the rate of amputations in accordance with the aims of The St Vincent Declaration. 11
Effect of problem on patients
Patients with diabetic foot ulcerations suffer from a reduced Quality of Life (QoL) 12 even compared to patients with DM without ulceration. 13 These observations included the following categories of assessment: physical functioning, role–physical, bodily pain, social functioning, and role–emotional. 13
Aside from a reduced QoL, diabetic foot ulcerations have also been associated with high morbidity and mortality rates. The mortality rate from onset of diabetic foot complications has been reported to be as low as 5 years. Alarmingly, this is worse than many common types of cancer. 14 However, neuropathic ulcerations are largely preventable.15,16 Therefore, this high mortality rate is unacceptable.
The whole problem is one of mechanics not of medicine. The biological response to these denervated limbs are qualitatively similar to those of normal limbs. It is the permitted pattern of mechanical stress that is different.
17
This highlights that diabetic foot disease is a condition that requires treatment within a MDT to fully address all aspects. The mechanical stress that the foot is placed under throughout ambulation must be considered. Therefore, an understanding of the biomechanical impact of foot deformities would be advantageous in the management of diabetic foot disease.
Clinical relevance of problem
Within a clinical environment when dealing with patients with DM, it is important to consider the main causal pathway to ulceration. Peripheral neuropathy, peripheral vascular disease (PVD), minor trauma and foot deformity are four of the major components in the main causal pathway.10,18,19 The best prevention of foot ulceration is the reduction of its risk factors. 20 Therefore, a focus on the influence of peripheral neuropathy, PVD, minor trauma and foot deformity would be appropriate. This literature review focused on the foot deformity component of the causal pathway and its effect on the biomechanics of gait.
Identification of the biomechanical needs of patients with DM would allow the MDT, in particular Orthotists, to provide the most appropriate care to reduce peak plantar pressures that occur as a result of biomechanical abnormalities. It would be advantageous to establish whether the weakness caused by motor neuropathy leads to damage and deformity of the lower extremity joints in DM. If the aetiology of deformity could be established, it would allow for preventative treatment that could potentially reduce the incidence of foot deformities and the resultant ulcerations.
Objectives
The primary objective of this literature review was to understand the development of the most common foot deformities that result from DM. The secondary objective was to explore the biomechanical effects of foot deformities within DM.
Methods
Search strategy
To locate the literature for this review, four electronic databases were searched in February 2014, these included CINAHL, Embase, Ovid and Medline. The search was for studies that reported on foot deformities within DM or the biomechanical impact of these deformities. The participant intervention comparison outcome (PICO) method was utilised to identify keywords and phrases, listed below:
Diabet* OR Diabetes Mellitus;
Sensorimotor Neuropathy OR Motor Neuropathy OR Diabetic Neuropathy;
Foot OR Lower Limb;
Weakness OR Atrophy;
Type 1 OR Type 2;
Deform*;
Gait;
Biomechanic*.
Study selection
Duplicates were removed from the search results. The titles and abstracts that the databases returned from the search were screened using the inclusion and exclusion criteria. For those papers that remained valid after the initial screening process, the full texts were retrieved and the articles were then closely examined. All articles that met any of the exclusion criteria were disregarded.
The inclusion criteria were as follows:
Studies in the English language;
Studies of adult populations with DM;
Studies reporting on diabetic peripheral neuropathy (DPN);
Studies examining foot deformity within DM;
Studies that assessed gait abnormalities as a result of foot deformities.
The exclusion criteria were as follows:
Studies that discussed treatment of active ulcers;
Studies that discussed surgical intervention;
Studies that focused on Charcot neuropathic arthropathy;
Studies that focused on interventions for treatment;
Studies that focused on painful DPN;
Studies that assessed gait abnormalities as a result of DPN.
Figure 1 displays the results from each stage of the literature search. At the end of the process, 17 articles remained eligible for inclusion.

Literature review; flow diagram of article inclusion process.
Quality assessment of studies
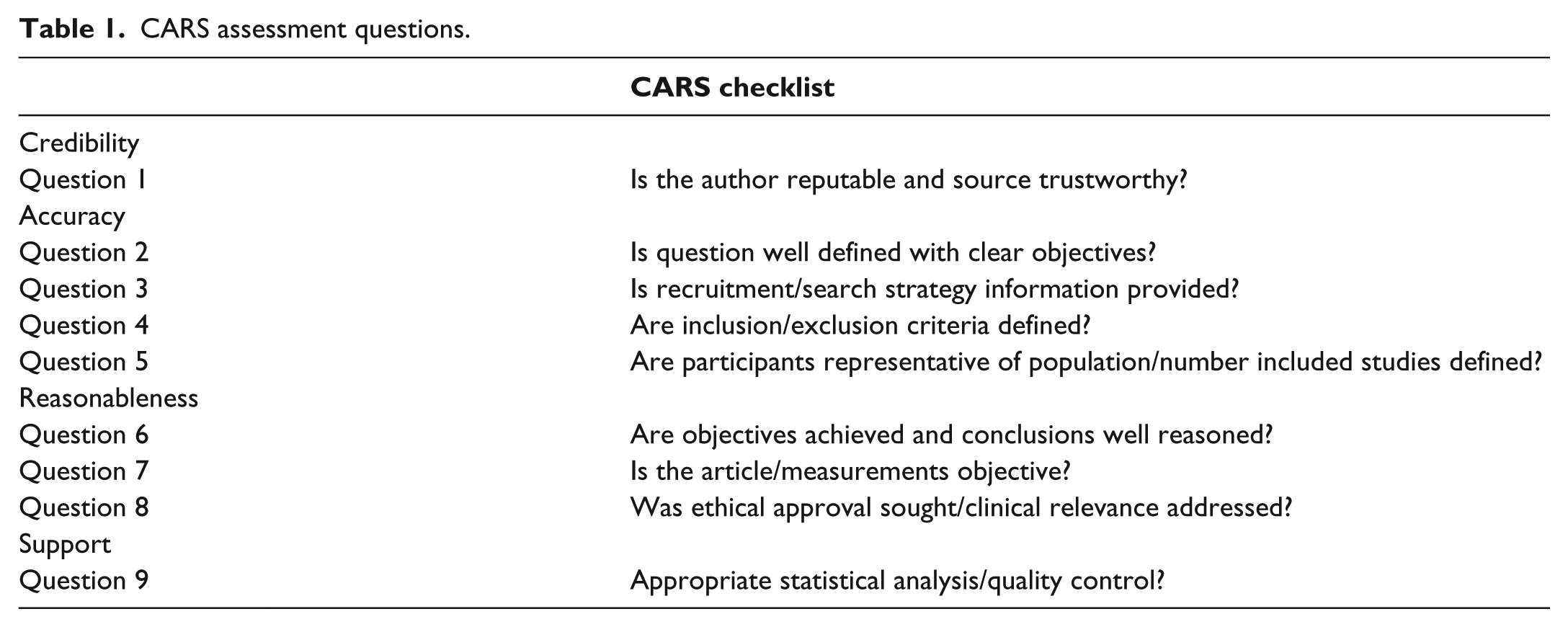
There were two assessment tools utilised to evaluate the quality of the included studies. Scottish Intercollegiate Guidelines Network (SIGN) Critical appraisal: Notes and checklists 21 were utilised to assess the retrospective cohort study and the systematic review and meta-analyses. However, this assessment tool was inappropriate for assessing cross-sectional studies and narrative reviews. Therefore, the CARS checklist (credibility, accuracy, reasonableness, support) 22 was utilised to help determine whether the article was of high, acceptable or low quality. The CARS assessment tool scored each article out of 9 using a series of questions (Table 1). A score >6 was considered high quality, a score 4–6 acceptable quality and a score <3 as low quality.
CARS assessment questions.
Results
SIGN assessment
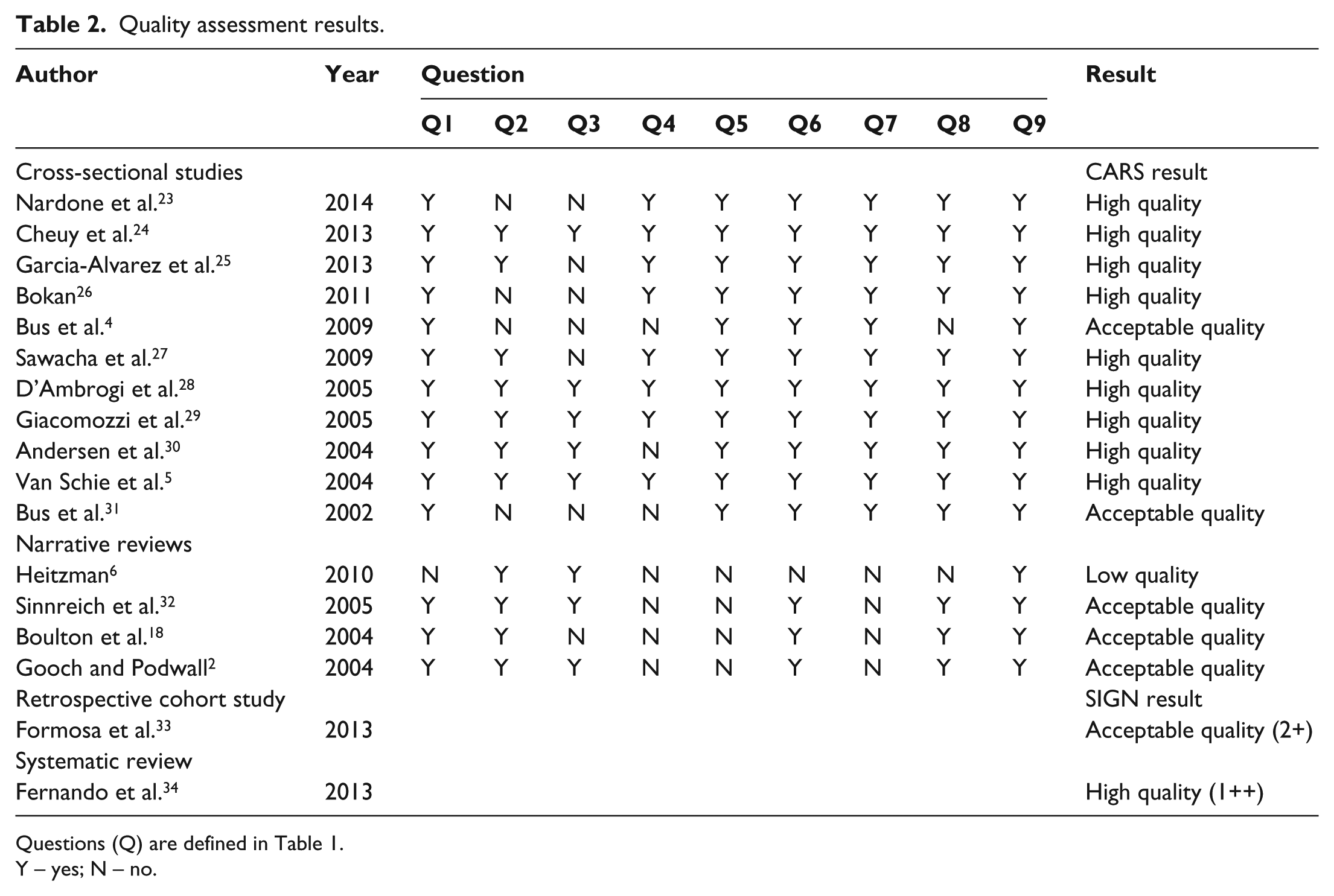
Of the 17 articles included, the majority were of cross-sectional design or narrative reviews with only one retrospective cohort study and one systematic review and meta-analyses. The Retrospective Cohort Study and the Systematic Review and Meta-Analyses were assessed using SIGN Critical appraisal: notes and checklists. 21 The SIGN Guidelines were based on a systematic review that was executed by members of the guideline development group. It was created for the development of Clinical Guidelines and does not include checklists for all types of study designs. This was a reliable tool that provided consistent quality assessment results with low risk of bias. There was an algorithm provided to assist in the classification of study design to ensure that the correct checklist was utilised. 21 The retrospective cohort study was graded as acceptable quality (2+) and the systematic review and meta-analyses was graded as high quality (1++) (Table 2).
Quality assessment results.
Questions (Q) are defined in Table 1.
Y – yes; N – no.
CARS assessment
Of the included studies, 15 were assessed using CARS. 22 The assessment questions were developed before the papers were assessed to ensure correct protocol and minimise bias. However, due to the self-creating nature of CARS, 22 this method is extremely subjective and the results cannot be treated as definitive. From this analysis, a total of 9 were of high quality, 5 of acceptable quality and 1 of low quality (Table 2).
Participant characteristics
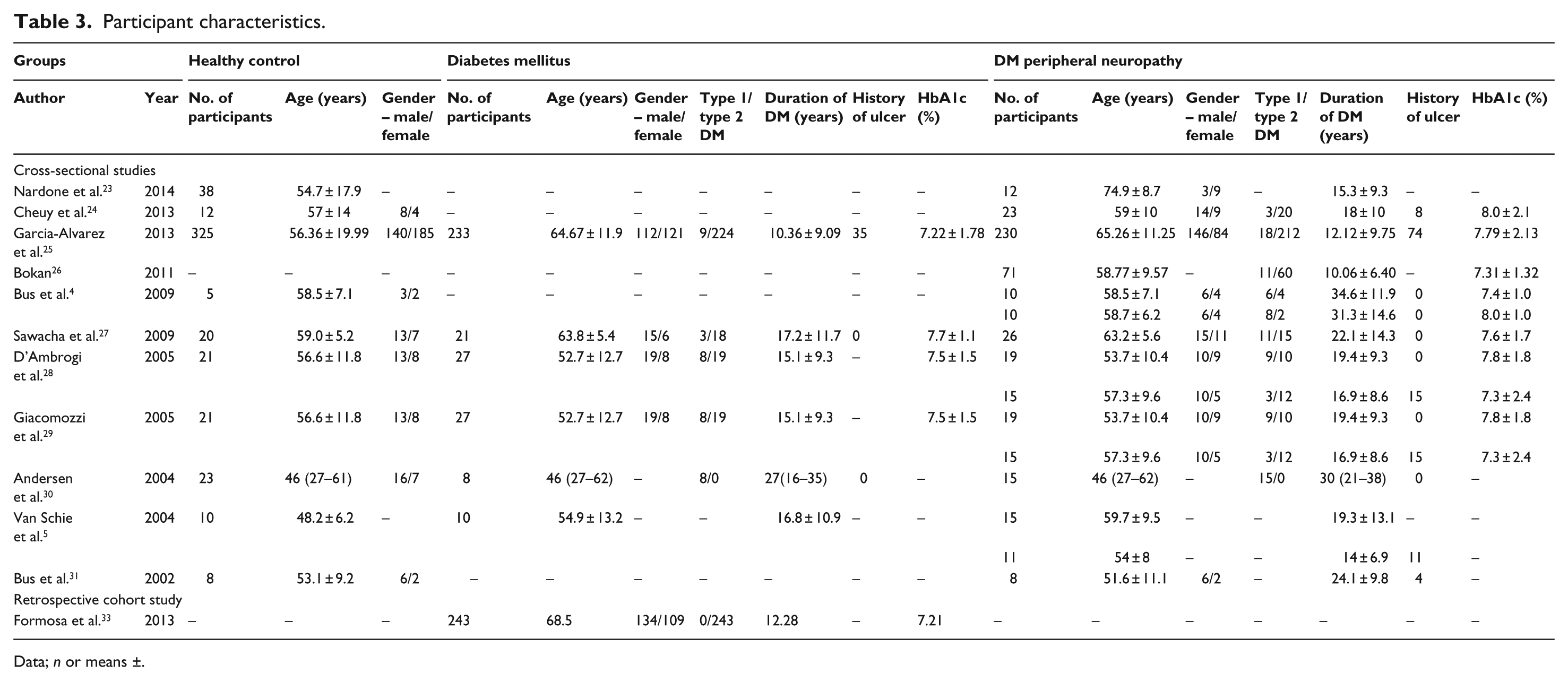
The participant characteristics from the included studies are displayed in Table 3. There were 1551 participants with a large range in group size, ranging from 5 to 325 participants. It was inappropriate to take an average of the group size, as the large variability would skew the results. Therefore, a median was calculated for each of the groups. All of the studies except 1 had a healthy control (HC) group. The median group size was 20.5 participants. From the included studies, there were a total of 1068 patients with DM, of which 499 had DPN. The median group size was 27 and 15, respectively.
Participant characteristics.
Data; n or means ±.
The weighted means of the age of the participants within the DM group were calculated to be 64.70 years while the Diabetes Mellitus Peripheral Neuropathy (DMPN) groups were calculated to be 61.33 years. This indicates that in general, those with DMPN were slightly younger than those solely with DM. However, when considering a complex disease process such as that of DM, it is important to consider either the age of onset with respect to the age at the time of the study or the duration of the disease. For the included studies, the weighted average of the duration of DM was 12.23 years for the DM group and 15.36 years for the DMPN group. This is the more relevant figure when comparing the study participants, as it is a major factor in the progression of DMPN.35,36 The weighted mean of the HC group was calculated to be significantly younger at 55.68 years.
Summary of literature quality
The majority of studies included within this literature review were considered to be of high quality after assessment using SIGN 21 and CARS. 22 Due to the overall high quality of the included studies, the results were considered to be reliable. There were no major limitations with the methods in any of the 17 studies. The main limitation of the included studies was their cross-sectional design that cannot be used to determine causality. 37 All of the studies except 1 had a HC group, although the weighted average of the ages of participants showed the HC group to be significantly younger than both the DM and DMPN groups. This variation slightly reduces the credibility of any comparisons drawn between the HC and the other groups. The group size across the studies displayed large variability with a range of 5–325 participants. This affected the quality of the studies with the smaller sample sizes graded at acceptable quality.4,31
Main themes
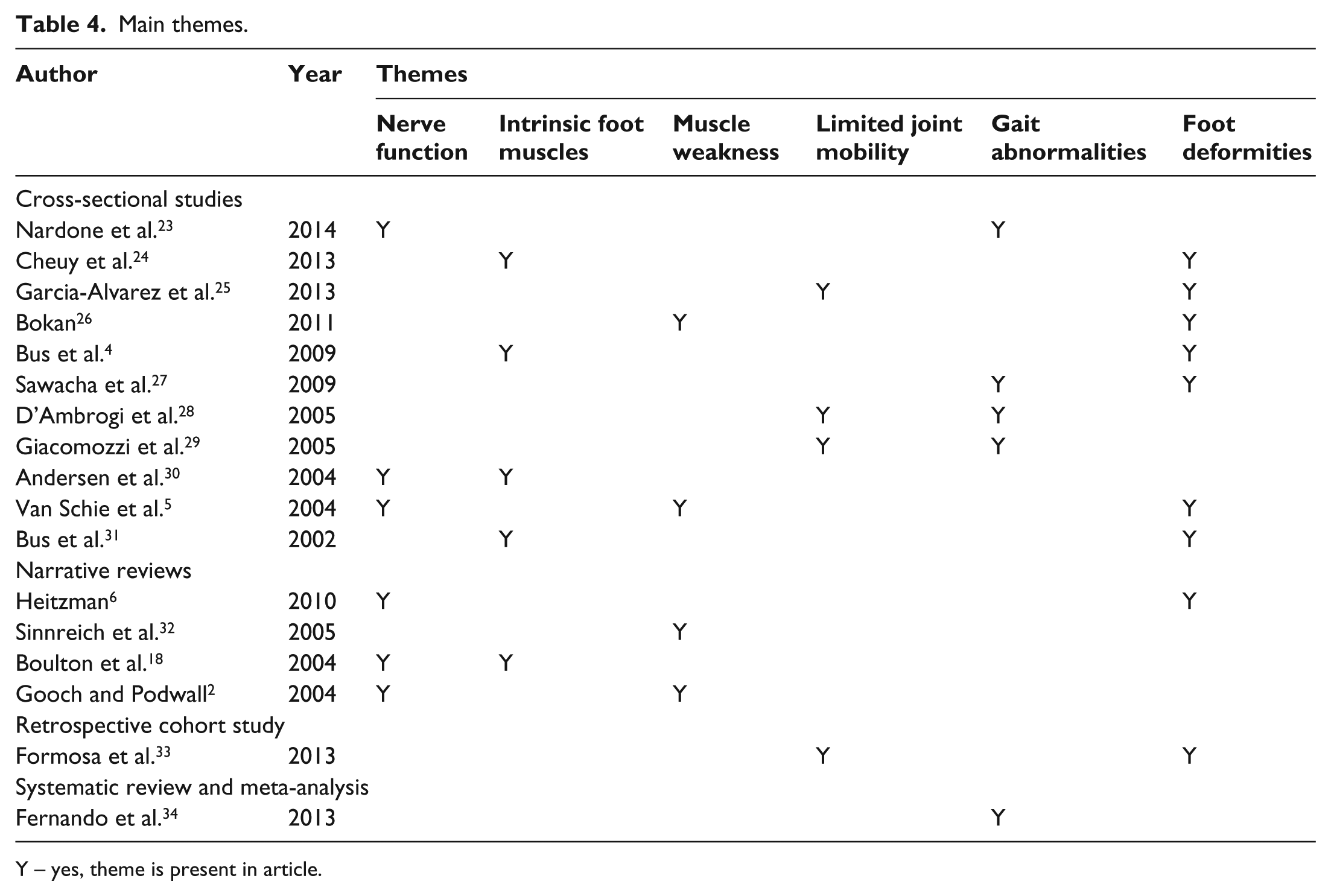
A thematic analysis was employed to identify and report on the main themes within the literature (Table 4). The six main themes identified in this literature were nerve function, intrinsic foot muscles (IFMs), muscle weakness, limited joint mobility (LJM), gait abnormalities and foot deformities. These continuously appeared throughout the literature reviewed and so it was decided to focus on these within the discussion.
Main themes.
Y – yes, theme is present in article.
Discussion
The primary aim of this literature review was to understand the development of the most common foot deformities that result from DM and explore the commonly accepted association with motor neuropathy. The secondary aim of this review was to explore how the common foot deformities within DM influenced the biomechanics of gait. Motor neuropathy is commonly believed to be the cause of foot deformities as a result of IFM atrophy and muscle imbalance. This review has attempted to identify the common foot deformities that occur as a direct result of DM. However, upon examination of the literature, the deformities were not easily identified. The literature presented four main themes in relation to foot deformities within DM (nerve function, IFMs, muscle weakness and LJM). Each of these main themes will be discussed in relation to foot deformities within DM.
Nerve function and foot deformities
Of the 17 articles included in the review, 6 of them discussed the effect of DM on nerve function. The aim of these articles was to ascertain whether nerve function was related to foot deformities within DM. It was identified that there are a variety of methods through which the nerves can become damaged in patients with DM.2,18 Understanding of the exact pathogenesis of DPN in human remains unclear. 18 A multitude of factors including chronic hyperglycaemia, insulin deficiency and ischaemia contribute to the development of DPN. 2 The interaction between these components is complex and at present, not completely understood. A few of the suggested methods of damage are the polyol pathway and ischaemia. In animal models, the polyol pathway has been significantly associated with a reduction in nerve-conduction velocity. 18 The results have not been as definitive in human. Ischaemia in patients with DM has been shown to correlate with the extent of neuropathy. 38 However, ischaemia does not present independently in patients with DM and it is unclear how it interacts with other factors that may influence DPN. The lack of clarity of the pathogenesis of DPN is extremely problematic when attempting to find treatments as it is unclear exactly what requires treatment. It would be beneficial to fully understand the pathogenesis of damage so that effective treatment methods could be developed. An effective treatment for DPN would allow for pro-active management as opposed to reactive treatment of this serious diabetic complication. Ideally, effective treatment would prevent the onset or at least slow the progression of DPN. The severity of nerve-conduction abnormalities was correlated with the severity of hyperglycaemia. 32 This supports the evidence that the only current successful treatment of DPN is good glycaemic control.2,18
Although the pathogenesis of damage to the nerves is unclear, it is accepted within the literature that DM does cause a reduction in motor nerve-conduction velocity (MNCV).5,30 The mean MNCV has been reported to be 10%–30% below normal and the reduction to be more pronounced the longer the duration of DM. 32 The measurement of the reduction in MNCV is potentially a useful clinical tool as it can be utilised to trace the onset and progression of DPN. It has also been noted to be of use as an indication of the severity of DPN and the QoL of the patient, although it is not indicative of the development of foot deformities within DM. 15 However, MNCV tests are not always readily available within a clinical environment and often require a Neurologist to carry out and interpret the results of the test.
The primary aim of this literature review was to understand the development of the most common foot deformities that result from DM. A strong inverse relationship was identified between peroneal and tibial MNCV and the foot deformity score. 5 This would imply that a reduction in MNCV is not responsible for foot deformities within the diabetic foot. Therefore, assessing MNCV would not be advantageous in assessing which patients may be at risk for developing foot deformities.
IFMs and foot deformities
In total, 5 of the 17 articles explored the effect of DM on IFMs. The articles’ primary objective was to ascertain whether IFMs played a role in the development of foot deformities within DM. Following on from the previous section, the amplitude of the compound muscle action potential of the peroneal nerve was closely related to the volume of IFMs. 30 The volume of IFM in patients with DPN was shown to be half that of HC subjects and also half that of diabetic controls without DPN. Patients with DPN were also shown to have a considerable loss of striated muscle in the feet. In a separate study, patients with DPN were shown to not only have a reduction of lean muscle volume but also to have an increase in the adipose tissue within their IFM. 4 A close relationship was shown between the volume of IFM and the degree of neuropathy. Despite these clear relationships and associations, it may not be worthwhile assessing IFM within a clinical environment. The magnetic resonance imaging (MRI) technique that was utilised to obtain accurate measurements of the volume of IFM is an expensive and time-consuming procedure. Similar depth of information could be obtained through other less expensive means, such as ultrasound, that has been used successfully to evaluate the thickness of soft tissue structures in the foot.28,29
The atrophy that occurs within diabetic foot disease has been discussed within numerous studies.4,24,30,31 MRI was utilised in all of the studies to evaluate the extent of atrophy. A reduction or absence of definable muscle cross-sections in the majority of slices was noted in DPN patients in the forefoot, 31 the muscle bundles had been replaced by disorganised tissue with fatty infiltration. This is in agreement with the findings of volume of IFM where it was noted that there was an increase in the adipose tissue within the IFM. In relation to foot deformities within DM, the articles presented a variety of views as a result of their own key findings. All agreed that there was IFM atrophy, although it was not considered to be a primary causative factor of foot deformities within DM. However, Cheuy et al. 24 identified a possible threshold effect. As the IFM deterioration progressed, a more linear relationship was noted in comparison to metatarsal phalangeal joint (MTPJ) angle. If this relationship was to be confirmed, it has the potential to be the first step in understanding the development of foot deformities in DM, specifically in relation to more advanced DPN. Atrophy of the IFM was also recognised to be closely reflective of the extent of motor dysfunction.30,31 Despite the fact that the extent of IFM atrophy does not appear to be closely correlated with the incidence of toe deformities within DM, it may still be of value. However, the method of obtaining the information on extent of atrophy may have to be reconsidered to make it a viable clinical tool as MRI procedures bring their own problems as previously discussed. Due to the lack of associations between IFM and foot deformities within DM, assessing IFM does not appear to be beneficial when attempting to identify those patients who are at risk of developing foot deformities.
Muscle weakness and foot deformities
Just four of the included articles addressed muscle weakness in relation to DM. Muscle weakness within DM was associated with duration of DM >10 years and a higher score for DPN. 26 The articles explored the commonly believed theory that muscle weakness results in foot deformities within DM. The two narrative reviews that discussed muscle weakness simply identified that distal weakness in the legs and feet may be an issue in severe cases of DPN.2,32 Both of the articles agreed that the muscle weakness may result in foot drop but neither associated the muscle weakness with the development of foot deformities. This lack of consideration results in an inconsistency between the narrative reviews and the cross-sectional studies. Both the cross-sectional studies that examined muscle weakness identified that there was a relationship between muscle weakness and foot deformities within DM.5,26 The intrinsic muscles were found to be more severely affected and significantly weaker than the extrinsic muscles that would be supported by the MRI studies that examined muscle atrophy.4,24,30,31 This is supportive of the length-dependent nature of DPN.
The MNCV of the peroneal and tibial nerves displayed a strong inverse relationship with the muscle weakness present in the muscles that they innervated. The muscles that were innervated by the tibial nerve had a greater proportional decrease in weakness than the muscles innervated by the peroneal nerve. 5 This is also indicative of a length-dependent process as the peroneal nerve originates more proximally than the tibial nerve. However, the weakness associated with DPN and its association with foot deformities is not well documented within the literature. The two articles that were found to discuss this had contradictory views. Van Schie et al. 5 intimated a causal relationship between muscle weakness and foot deformities within DM. However, due to the cross-sectional design of the study, it was unclear if it was muscle weakness that resulted in foot deformities or foot deformities that resulted in muscle weakness. Conflictingly, Bokan 26 found that muscle weakness increased the frequency of foot deformities, in particular the presence of a high arch, which was found to be present in 80% of participants with muscle weakness. Further work is required to determine the precise relationship that exists between muscle weakness and foot deformities. Based on the work by Bokan, 26 the association between muscle weakness and foot deformities may be a useful clinical tool in detecting the risk of developing foot deformities. Although, this should be treated with care and not used definitively due to uncertainty over what is present first, the muscle weakness or the foot deformities. 5
It is commonly accepted that muscle imbalance plays an influential role on the development of toe deformities within DM. However, the literature does not support this theory. Only one article explored muscle imbalance in relation to foot deformities. 4 It found that muscle imbalance did not allow for a distinction between patients with and without foot deformities and therefore is not a primary causative factor. However, this article was of acceptable quality and had a small participant population. Therefore, the role of muscle imbalance in the development of foot deformities should not be completely disregarded.
LJM and foot deformities
Only 4 of the 17 included articles considered LJM within DM. These articles explored LJM and the presence of foot deformities within DM with conflicting results. All of the articles identified that LJM was an issue within the diabetic population25,29,33,39 with it being reported to be around 30%–40% of the diabetic population affected. The primary focus of the articles was the first MTPJ. A progressive reduction of motion was recognised as the degree of DPN became more advanced, this was also correlated with the duration of DM. 25 This association could have use within a clinical environment as degree of DPN and duration of DM are easily assessed and monitored, clinical signs of DPN or significant duration of DM should draw attention to the need for a biomechanical assessment to members of the MDT who do not specialise in this area.
The source of the LJM at the first MTPJ within DM was identified by two of the articles. They found an association between the thicknesses of the Achilles tendon (TA) and plantar fascia (PF) and the reduction of motion at the first MTPJ.28,29 An association was also made between the stiffness of the PF and pronation of the foot. The foot position was assessed in relation to six criteria; both the hind foot and forefoot position were examined for signs of pronation. Pronation of the foot was significantly linked to DPN and was more prevalent in patients who had a longer duration of DM. 33 Those patients who had pronation of the foot also had an increase in LJM of the first MTPJ. This would be indicative that assessing the thickness of the TA and PF would be advantageous as it may be the first indicator that the patient is at risk of developing pronation of the foot. However, assessing the thickness of these tendon and ligamentous structures with MRI is costly, time consuming and not readily available within a clinical environment. Therefore, assessing joint mobility would be more beneficial as it is not costly, is relatively quick and easily assessed within a clinical environment. A reduction in motion available at joints within diabetic foot disease may be indicative that the patient is at risk of developing pronation of the foot which may, in turn, lead to other foot deformities. Therefore, utilising the assessment of LJM as a predictor for the development of foot deformities within DM may be clinically relevant.
Gait abnormalities and foot deformities
There were many studies that examined the effect of DPN on the biomechanics of gait. However, very few that considered the biomechanical impact of foot deformities in DM, five have been included in this review. As previously discussed, a thickening of the TA and PF resulted in LJM affecting the first MTPJ.23,33 The thickening of the PF was also shown to trigger the Windlass mechanism. 28 The Windlass mechanism ensures that the foot is a rigid lever arm through which propulsion forces can be generated at terminal stance. However, in patients with DPN where the PF was thickened, the Windlass mechanism was triggered prematurely and was sustained for a longer period of time. This was associated with a longer stance duration that all five articles recognised as a prevalent factor in patients with DPN.23,27–29,34 Therefore, it would be advantageous to identify whether there was LJM at the first MTPJ as this may be indicative that there will be a prolonged period of stance. This may also be an important factor even for those patients who do not have clinical indications of DPN as functional changes were noted in some patients even before they were symptomatic.33,34
The thickening of the TA and PF were also associated with a more rigid foot that was less adaptable to walking surfaces.28,29 This is possibly a factor in the unsteadiness that was reported,23,34 although the exact association is still unclear. Muscle weakness that resulted in a reduction of the single support phase may also be a contributory factor to unsteadiness. 27 Therefore, assessment of both TA and PF thickness along with muscle strengths would be advantageous when determining whether the patient is at risk of unsteadiness when standing and ambulating.
The LJM at the first MTPJ was also significantly associated with alterations in loading of the foot. The changes in foot loading became more severe as the LJM at the first MTPJ advanced. The primary change that was noted was the increase of loading times at the metatarsal heads. 34 This was an extremely important finding that would be beneficial within a clinical environment. Knowing that patients with LJM at the first MTPJ will experience significantly increased loading times at the metatarsal heads may influence treatment plans. This is an important consideration that could potentially influence insole design or material selection to help reduce the forces that the metatarsal heads will undergo.
Muscle weakness was associated with foot deformities. 26 Muscle weakness of the lower limbs in patients with DPN was also associated with a reduction in velocity and step length. 34 Due to these associations, it is essential that the muscle strengths of patients with DM be assessed regularly as muscle weakness may lead to further complications.
Despite the little research that has been carried out in this particular area, the results so far have been promising. It is extremely important for biomechanical abnormalities to be identified and treated quickly to prevent further complications.
Conclusion
The primary purpose of this literature review was to understand the development of the common foot deformities that result from DM. It became apparent that there is a lack of understanding about the aetiology of foot deformities. The common belief that motor neuropathy causes atrophy and muscle imbalance that results in foot deformities is not supported within the literature. IFM atrophy, muscle imbalance and a reduction in nerve function did not display definitive associations with foot deformities. However, muscle weakness and LJM were associated with foot deformities within DM, although these associations should still be treated with care.
The secondary aim of this review was to explore the biomechanical effects of the common foot deformities within DM. There is limited literature on this. This may be due to the uncertainty of the common foot deformities within DM. A prolonged stance phase was linked to thickening of the TA and PF due to decelerated braking forces at loading response and reduced propulsion forces at terminal stance. The only other gait abnormality identified in relation to foot deformity was unsteadiness. This was associated with LJM and muscle weakness.
Future directions
Studies of prospective design would be beneficial and have potential to further the understanding of the common foot deformities within DM. To fully understand what the common foot deformities are it is essential to fully understand their aetiology. Further work should be done to establish whether muscle weakness results in deformity or whether deformity results in muscle weakness. Also, it would be advantageous to investigate the possible threshold effect that was identified between IFM deterioration and MTPJ angle. The final piece of work that the author would recommend is further investigation into the association of LJM with pronation of the foot and other foot deformities. This work is important, as it would establish whether the assessment of muscle weakness, IFM deterioration or LJM could be utilised within a clinical environment to predict the development of foot deformities. Once the common foot deformities have been identified, it will be possible to evaluate the impact they have on biomechanics.
Footnotes
Acknowledgements
The authors would like to thank Professor David G Armstrong for his expert opinion on the success of the St Vincent Declaration. Also, the authors thank Sally Bell and Suzanne Faulkner for their contribution to this review of the literature.
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
