Abstract
Background:
Passive-dynamic ankle–foot orthosis characteristics, including bending stiffness, should be customized for individuals. However, while conventions for customizing passive-dynamic ankle–foot orthosis characteristics are often described and implemented in clinical practice, there is little evidence to explain their biomechanical rationale.
Objectives:
To develop and combine a model of a customized passive-dynamic ankle–foot orthosis with a healthy musculoskeletal model and use simulation tools to explore the influence of passive-dynamic ankle–foot orthosis bending stiffness on plantar flexor function during gait.
Study design:
Dual case study.
Methods:
The customized passive-dynamic ankle–foot orthosis characteristics were integrated into a healthy musculoskeletal model available in OpenSim. Quasi-static forward dynamic simulations tracked experimental gait data under several passive-dynamic ankle–foot orthosis conditions. Predicted muscle activations were calculated through a computed muscle control optimization scheme.
Results:
Simulations predicted that the passive-dynamic ankle–foot orthoses substituted for soleus but not gastrocnemius function. Induced acceleration analyses revealed the passive-dynamic ankle–foot orthosis acts like a uniarticular plantar flexor by inducing knee extension accelerations, which are counterproductive to natural knee kinematics in early midstance.
Conclusion:
These passive-dynamic ankle–foot orthoses can provide plantar flexion moments during mid and late stance to supplement insufficient plantar flexor strength. However, the passive-dynamic ankle–foot orthoses negatively influenced knee kinematics in early midstance.
Clinical relevance
Identifying the role of passive-dynamic ankle–foot orthosis stiffness during gait provides biomechanical rationale for how to customize passive-dynamic ankle–foot orthoses for patients. Furthermore, these findings can be used in the future as the basis for developing objective prescription models to help drive the customization of passive-dynamic ankle–foot orthosis characteristics.
Background
In healthy gait, the plantar flexors contribute substantially to vertical support and forward progression.1–3 In particular, during midstance, the plantar flexors act eccentrically to provide dorsiflexion resistance to control the shank as it rotates over the foot. 4 Plantar flexor weakness, an impairment frequently seen in many neuromuscular diseases or pathologies such as stroke,4,5 leads to poor control of shank rotation, 4 which may result in crouch gait, 6 shorter step lengths, and decreased walking speeds.4,5,7
Ankle–foot orthoses (AFOs) are assistive devices regularly prescribed to aid individuals with plantar flexor weakness with the aim of improving gait function. Passive-dynamic ankle–foot orthoses (PD-AFOs) use orthosis shape, material properties, and design features to drive the orthosis bending stiffness and storage and return of mechanical energy. 8 PD-AFO bending stiffness, which can be calculated as the ratio of ankle moment to angle 9 and modeled as a torsional spring that acts about the ankle joint center, is an important functional characteristic that may enable orthoses to substitute for lost plantar flexor function. 10 It is believed that customizing AFO characteristics, including stiffness, to each individual’s needs can enhance patient function.11–14 Inadequate customization may explain why some patients find their orthoses effective while others experience limited or even inhibiting effects. 15 However, while conventions for customizing orthosis characteristics are often described and implemented in clinical practice, 16 there is little objective evidence to explain the biomechanical rationale behind these conventions.14,17 Such conventions include modifying orthosis trimlines to adjust the bending stiffness (level of dorsiflexion resistance) provided by the orthosis10,18 and setting the unloaded shank-to-vertical angle into dorsiflexion, either through the AFO or orthosis–footwear combination.6,19,20
The lack of biomechanical rationale guiding customization of PD-AFO characteristics is due in part to the limited understanding of the influence of these characteristics on muscle function. Muscles have multiple and sometimes counterintuitive functions during gait, and muscles classified in the same anatomical group can have different effects on joint and body accelerations. 21 In particular, studies have demonstrated distinct roles of the biarticular gastrocnemius and uniarticular soleus (SOL) during stance.1,22,23 Thus, the interactions between the musculoskeletal system and orthoses are not straightforward. However, musculoskeletal models and simulations provide a tool to explore the influences of perturbations, such as assistive devices or muscle deficits, on musculoskeletal function.24–27 For each perturbation, individual muscle function can be isolated and cause-and-effect relationships that cannot be readily determined via experimental studies explored.28,29
A few studies have probed the influence of orthosis bending stiffness on muscle or joint-level function during gait. Bregman et al. 30 used an experimental protocol to explore the interaction between PD-AFO bending stiffness and impaired gait. Alternatively, Crabtree and Higginson 26 used simulations to explore the interaction between AFO stiffness perturbations and muscle function; however, this study assumed normal gait could be achieved with simulated orthosis use. Furthermore, both studies lacked the ability to control the orthosis bending stiffness in an experimental environment and thus could not systematically explore the relationship between orthosis bending stiffness and gait function. Kobayashi et al. 31 designed an experimental AFO with bending stiffness that could be adjusted via an oil damper and experimentally studied the influence of the bending stiffness on the ankle joint kinematics of patients post-stroke. While this study provided a valuable tool to begin to explore the influence of AFO bending stiffness on gait function and demonstrated the feasibility of using AFO bending stiffness to influence ankle joint kinematics for individuals post-stroke, the heterogeneity of the impaired population used in this study made it difficult to link orthosis bending stiffness directly to resulting joint or muscle-level function during gait.
We previously developed a framework that enables the customization and manufacture of PD-AFOs with objectively-tuned fit and functional characteristics.32,33 This ability to tune and systematically vary PD-AFO characteristics enables controlled PD-AFO perturbations to be experimentally implemented. Musculoskeletal modeling and simulations can then be used to explore the influence of these perturbations on muscle function. The purpose of this study was to integrate a model of the customized PD-AFO into a healthy musculoskeletal model and use simulation tools to explore the influence of PD-AFO stiffness on plantar flexor function during gait. This understanding may provide insight into the biomechanical rationale for customizing PD-AFO characteristics for patients with plantar flexor weakness.
Methods
Experimental movement analysis data were collected via an Institutional Review Board-approved study on two healthy subjects (Subject A: male, age: 25 years, height: 1.75 m, mass: 79.8 kg; Subject B: female, age: 24 years, height: 1.65 m, mass: 61.2 kg). The subjects walked under three randomly presented conditions: a no PD-AFO condition and two unilateral PD-AFO condition (AFO Flexible, AFO Stiff), with the orthoses tuned to different percentages of each subject’s natural ankle pseudo-stiffness. 34 Subjects were given 10 min of walking time to accommodate to each condition. Subjects were permitted more accommodation time if needed, but both subjects stated they felt comfortable after the 10-min accommodation period for all conditions. The subjects walked overground at a controlled speed of 0.80 ± 0.02 statures/s for all conditions, which is an average normal walking velocity. Speed was measured during the data collections via two photocell beams located approximately 3.0 m apart. After each trial, subjects were given verbal cues to guide them to the targeted speed (e.g. “please walk a little faster/slower for the next trial”). Data were collected until three useful trials (speed 0.80 ± 0.02 statures/s and feet entirely on the force plates) were obtained. Bilateral kinematics were collected at 240 Hz using a six-camera optical motion analysis system (Motion Analysis Corporation, Santa Rosa, CA, USA) and a 6-degree of freedom (DOF) marker set. 35 Kinematics of the PD-AFO strut-cuff were tracked separately from the subject’s shank using three additional tracking targets placed directly on the PD-AFO cuff. Kinetic data were collected at 1200 Hz from four force plates (AMTI, Watertown, MA, USA). Kinematic and kinetic data were filtered at 6 and 25 Hz, respectively, using a zero-lag low-pass Butterworth filter. Additionally, bilateral muscle activity was collected from the medial gastrocnemius (MGAS), SOL, and tibialis anterior (TA) using bipolar silver/silver chloride electromyography (EMG) surface electrodes (Myotronics Inc., Kent, WA, USA) with a MA-300 EMG System (Motion Lab Systems, Baton Rouge, LA, USA).
Two PD-AFOs were customized and stiffness tuned for each subject via the established framework.32,33 PD-AFO stiffness levels were targeted at 40% (AFO Flexible) and 80% (AFO Stiff) of each subject’s natural ankle pseudo-stiffness in order to span the natural ankle pseudo-stiffness curve. Once customized and stiffness tuned, the PD-AFOs were manufactured via fused deposition modeling. 33 In this PD-AFO design, ankle and knee joint centers as well as foot and shank coordinate systems were established. 32 The PD-AFO ankle joint alignment, defined as the orientation of the shank coordinate system relative to the foot coordinate system when the PD-AFO was unloaded, was set to a neutral alignment such that the long axis of the shank (knee joint center to ankle joint center) was perpendicular to the bottom, flat surface of the footplate (Figure 1). PD-AFOs were worn on the dominant (right) leg without shoes to minimize variability and instead the footplate, with controlled characteristics, served as the foot–ground interface. The foot was secured in the footplate via straps across the top of the foot. The footplate was rigid so that bending stiffness was isolated to the strut and could therefore more readily be modeled. Thus, to promote more natural roll over shape36,37 with a rigid footplate, the footplate had heel and toe rockers separated by a flat stability region.
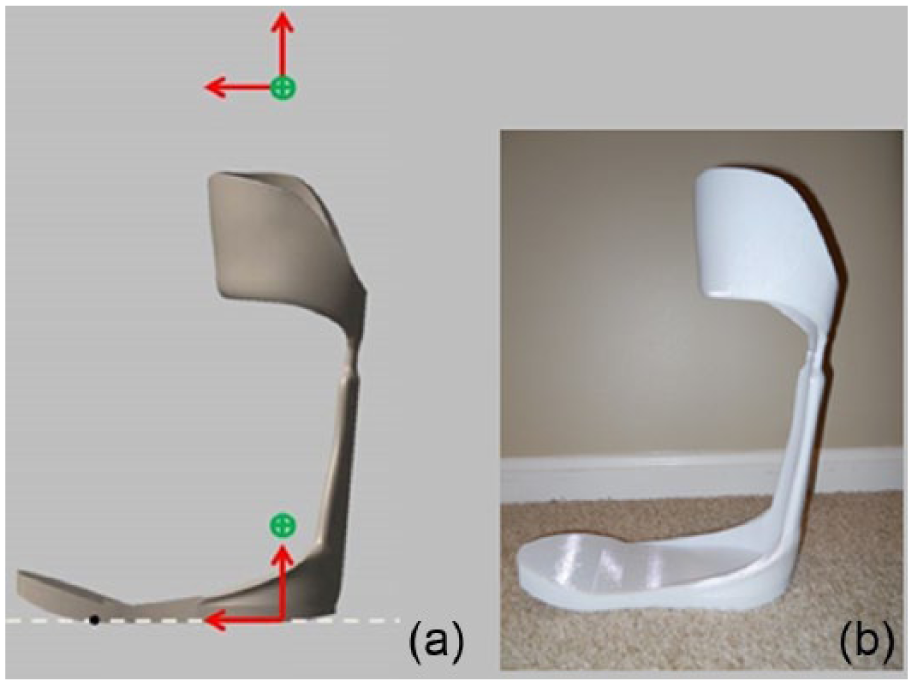
Sagittal view of PD-AFO: (a) neutral ankle joint alignment of PD-AFO. Ankle and knee joint centers (green circled plus) and shank and foot coordinate systems shown and (b) experimental PD-AFO before padding and straps were added.
In OpenSim, a lower extremity musculoskeletal model with 23 DOFs and 92 muscle actuators was used. 29 The characteristics of the customized PD-AFOs were integrated into this healthy musculoskeletal model and used in simulating the PD-AFO conditions. This PD-AFO-integrated model added two bodies, a footplate and a strut-cuff, to the right lower leg of the musculoskeletal model (Figure 2). The footplate body was welded to the calcaneus. The strut-cuff body was connected to the tibia via a custom joint that only allowed translation of this body along the tibia’s longitudinal axis, which allowed for cuff pistoning. Since the strut-cuff body could move separately from the shank, the experimental kinematics of the PD-AFO strut-cuff were needed in the simulation in addition to the shank kinematics to track movement of both segments. The two PD-AFO bodies were connected at the ankle joint center by a bushing force, which simulated a torsional spring with user-prescribed stiffness value(s) for its DOF(s). In this model, the ankle joint had one DOF (rotation in dorsi/plantar flexion), thus, for each PD-AFO condition the stiffness of the bushing force’s dorsi/plantar flexion DOF was set to match the stiffness of the PD-AFO used in the experimental condition. A torsional spring was used to model the PD-AFO bending stiffness because deformation of the PD-AFO was constrained by the anatomy, so the PD-AFO primarily rotated congruently with the anatomical ankle joint in the sagittal plane. This deformation pattern was confirmed using the motion capture data comparing the kinematics of the PD-AFO cuff to the kinematics of the shank. For both subjects across all conditions, the largest translation difference between the PD-AFO cuff and shank was 8.8 mm. Relative to the PD-AFO struts, which were 380–400 mm in length, these data showed that there was minimal pistoning of the PD-AFO cuff along the longitudinal axis of the shank, and therefore, the majority of the PD-AFO deformation was confined to the anatomical ankle joint (distal end of the strut) and the PD-AFO bending stiffness acted like a torsional spring.
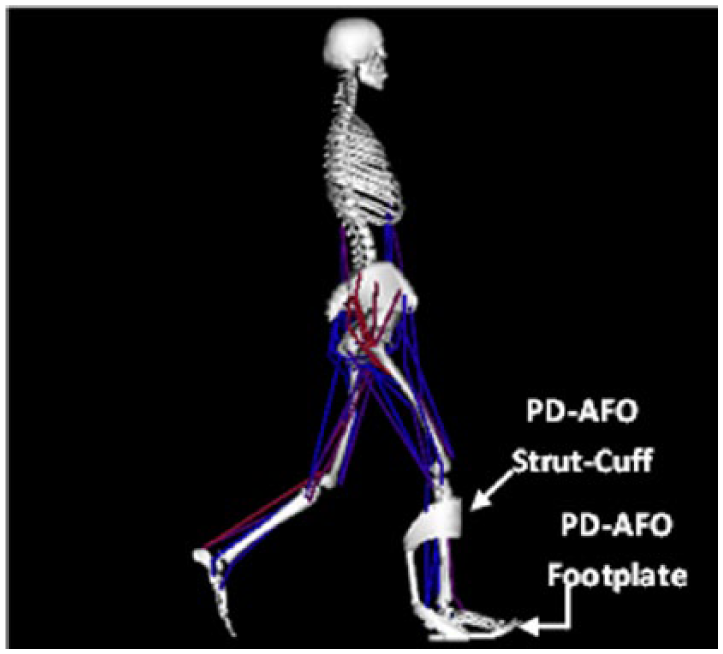
Schematic of PD-AFO-integrated musculoskeletal model.
Using OpenSim v2.4, models were customized and simulations were run for one representative trial from each experimental condition for both subjects. After scaling the appropriate musculoskeletal model to the subject’s anthropometric measurements, a reduced-residual algorithm (RRA) was used to account for dynamic inconsistencies between the experimental kinematics and force plate data. Predicted muscle activities that drove the quasi-static forward dynamic simulations were calculated through a computed muscle control (CMC) optimization scheme that minimized the difference between the experimental and simulated gait data. 38 The experimental EMG data were used to constrain the simulated muscle excitation patterns if they did not mimic the experimental muscle activity. Finally, an induced acceleration analysis39–41 was run for the primary plantar flexors and PD-AFO for each condition.
To identify the influence of the PD-AFO on ankle muscle function, sagittal plane net ankle joint moments, knee joint angles and knee joint moments were determined from the experimental data using Visual 3D (C-Motion Inc., Germantown, MD, USA). From the simulation results, right MGAS and SOL activations and moments as well as PD-AFO moments about the right ankle during stance were examined. Additionally, accelerations of the right knee induced by the MGAS, SOL, and PD-AFO were determined during stance to help identify the functional role of these components across all conditions as the differences in anatomy between these structures (uniarticular vs biarticular) might influence their functional role on the knee. Finally, in the Visual 3D pipeline, EMG data were full wave rectified and low-pass filtered using a fourth order and phase corrected Butterworth digital filter set at a cut-off frequency of 6 Hz. EMG data were visually compared to muscle activity predicted by the simulation.
Results
For Subject A, AFO Flexible and AFO Stiff had stiffness values of 3.51 and 8.17 N m/°, corresponding to 37% and 86% of the subject’s average natural ankle pseudo-stiffness, respectively. For Subject B, AFO Flexible and AFO Stiff had stiffness values of 1.90 and 3.90 N m/°, corresponding to 35% and 72% of the subject’s average natural ankle pseudo-stiffness, respectively.
Results reported and discussed in this study focus on the period of ankle dorsiflexion, approximately 20%–100% stance, as the bending stiffness of these PD-AFOs was designed to provide plantar flexor assistance via dorsiflexion resistance. From the experimental data, the net joint plantar flexion moment began around 20% stance for all conditions (Figure 3). Net plantar flexion moments were very similar between 70% and 100% stance (Region III in Figure 3) for all conditions, with peak plantar flexion moments occurring around 80% stance. However, a premature increase in the net plantar flexion moment occurred with PD-AFO use between 20% and 70% stance (Region II in Figure 3).
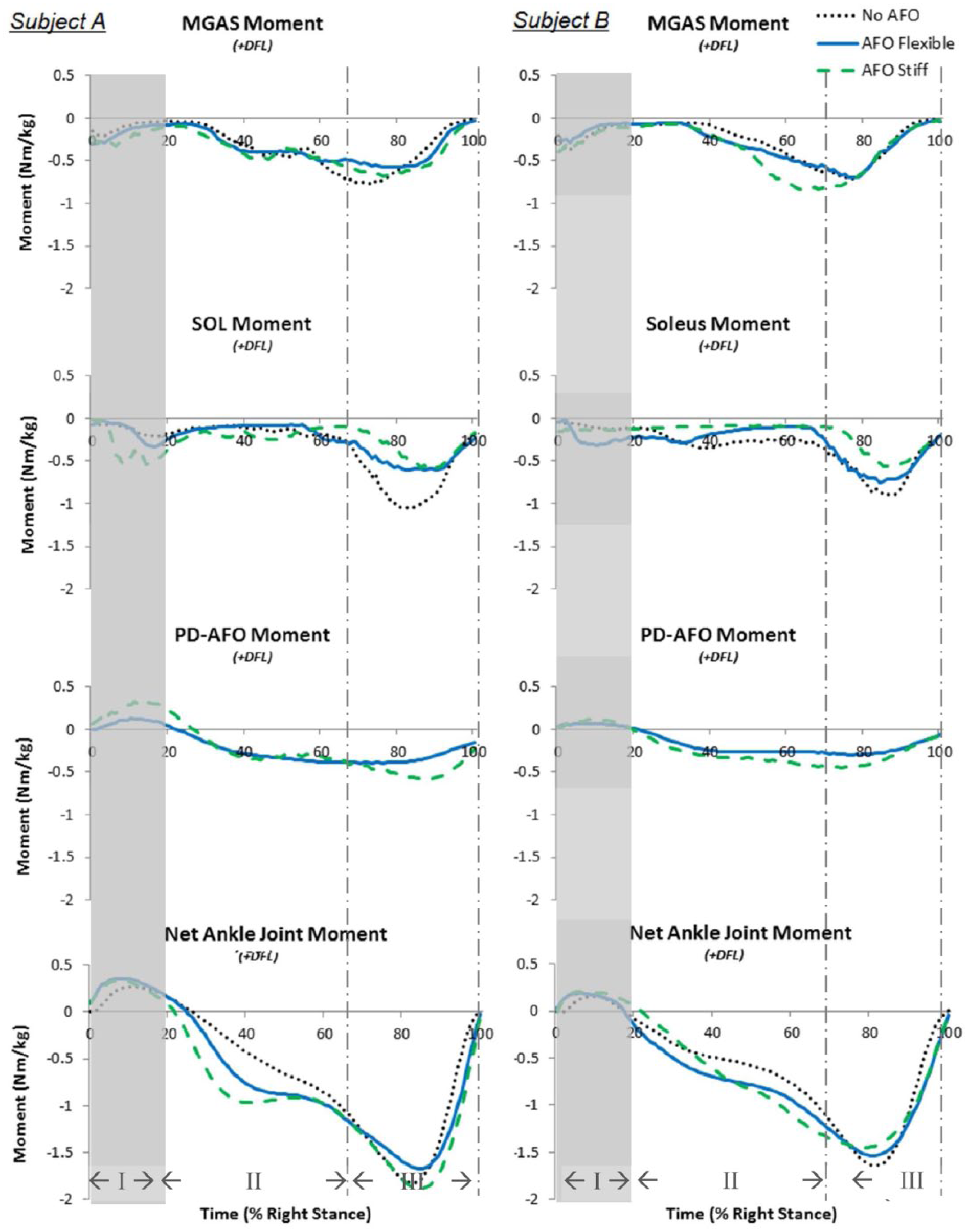
Muscle, PD-AFO and net joint moments about the ankle. MGAS (row 1), SOL (row 2), and PD-AFO (row 3) moments about each subject’s dominant (right) ankle shown. The experimentally determined net ankle joint moment (row 4) shown for comparison. Subject A (left column) and Subject B (right column). Region I is early stance, Region II is midstance, and Region III is late stance.
The characteristics of the customized PD-AFO were successfully modeled and integrated into OpenSim’s healthy musculoskeletal model. All simulations successfully tracked the experimental kinematics. Average residuals resulting from RRA were under ±15 N, although the vertical force residual often spiked at heel strikes to around ±60 N. The reserve actuators required for CMC were minimal with the actuators occasionally turning on but actuator torques never exceeding 10% of the peak joint moment. For both subjects, TA excitations were constrained to better mimic the pattern of the experimental EMG. From the experimental data, TA EMG activity dropped off from its peak activity in early stance to minimal activity in midstance, which is typical TA activity for gait. 4 However, simulated TA activity remained near full activation (1.0) during midstance when left unconstrained, possibly in order to offset biarticular MGAS activity. Therefore, TA excitations were constrained to a maximum of 0.2 between 15% and 86% stance for all simulations for all conditions. Due to uncertainty in the actual activation magnitudes since the EMG data were not normalized, a conservative threshold of 0.2 was chosen, which enabled simulated TA activity to mimic the pattern of the TA EMG but allowed the simulation some flexibility in solving the optimization. Constrained TA excitations made subtle but not substantial changes in MGAS and SOL function. All other muscle excitations were left unconstrained as the simulated muscle activities sufficiently mimicked the experimental EMG for the muscles collected.
MGAS generated substantial activity (Figures 4 and 5) and ankle moments (Figure 3) beginning around 30% stance and persisting through late stance for all conditions. MGAS activity and moments showed no distinct differences with and without PD-AFO use, except for the AFO Stiff condition for Subject B where the MGAS activity and moment increased to near its peak level faster and remained at a peak level longer than the No AFO and AFO Flexible conditions. In contrast, SOL function generally showed notable differences with and without PD-AFO use. SOL activity was substantially lower for both PD-AFO conditions compared to the no orthosis condition for both subjects (Figure 4). A similar trend was seen with the SOL moments; however, the SOL moment was similar for the No AFO and AFO flexible conditions for Subject B except for a slightly lower peak SOL moment for the AFO flexible condition (Figure 3). Across all conditions, SOL moments (Figure 3) were minimal until a burst from 70% to 100% stance (Region III). Peak SOL moments occurred between 82% and 88% stance across all conditions for both subjects; however, peak SOL moments were lower in the PD-AFO conditions compared to the no PD-AFO conditions.
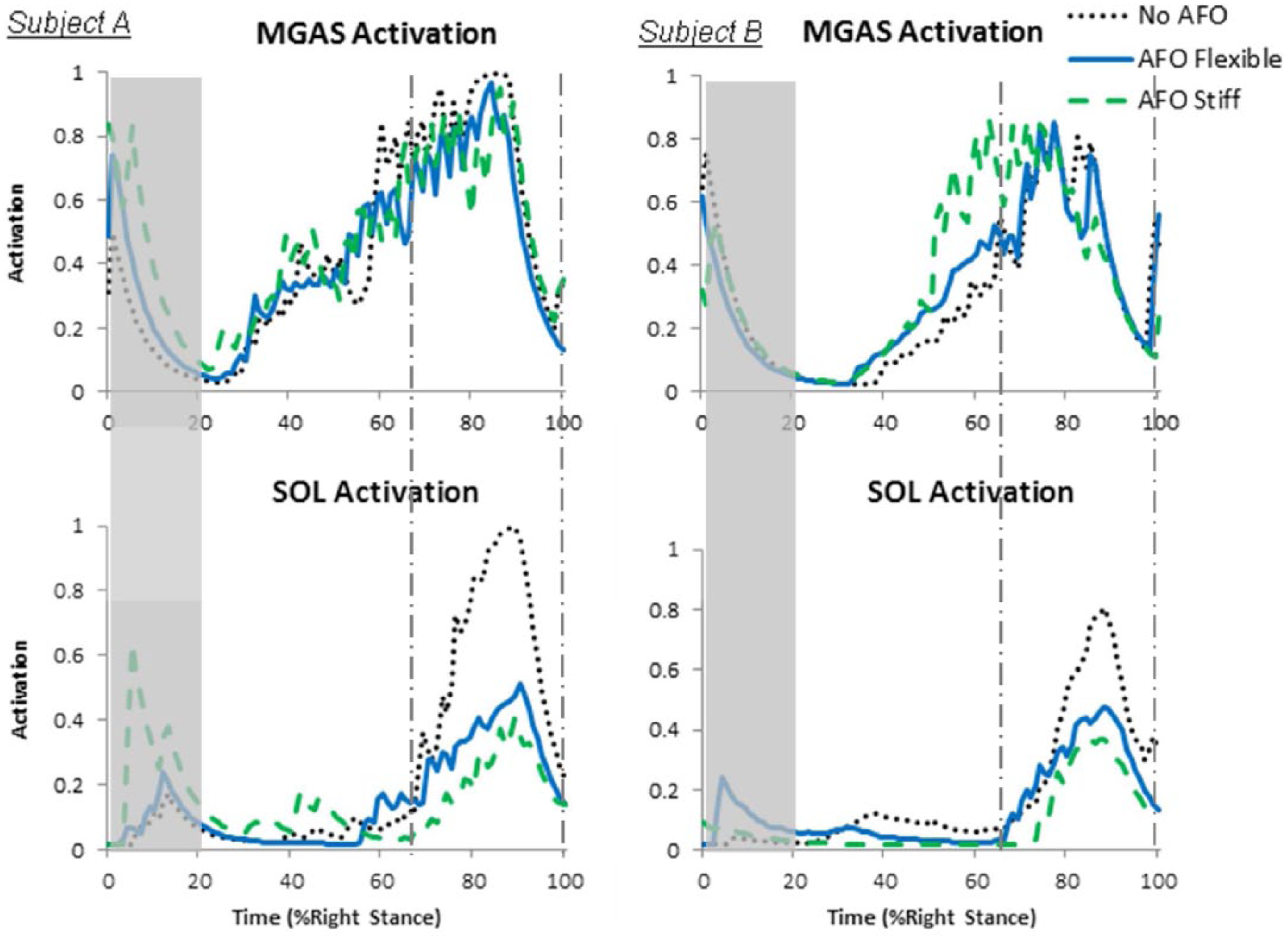
Medial gastrocnemius (MGAS) and soleus (SOL) activations for Subject A (left column) and Subject B (right column). Muscle activations predicted by the simulation, with maximum activation of 1.0 for each muscle.
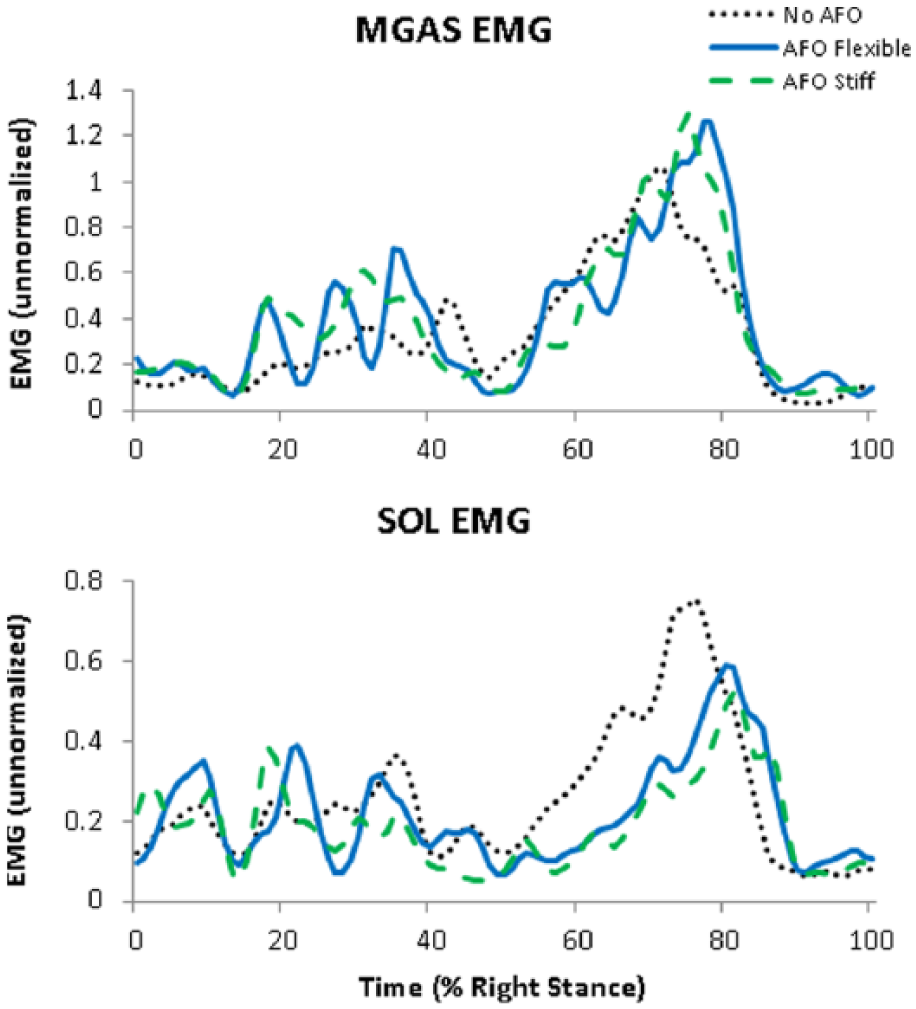
MGAS and SOL EMG excitations. Experimental EMG activity shown for Subject A.
All PD-AFOs began generating a plantar flexor moment around 20% stance (Figure 3). The PD-AFOs generated larger peak moments as stiffness increased for both subjects. Timing of the peak PD-AFO moments remained consistent across conditions for both subjects, occurring between 74% and 79% stance except for the stiffest orthosis for Subject A, which had a peak moment at 88%.
Induced acceleration analysis revealed that MGAS induced minimal or slight knee flexion accelerations throughout stance in the No PD-AFO condition (Figure 6). In contrast, for the No PD-AFO condition, SOL induced substantial knee extension accelerations when active, from 70% to 100% stance (Region III), with peak knee extension accelerations occurring during 80%–90% stance, corresponding to peak SOL moments (Figure 6). These induced acceleration findings for both the MGAS and SOL agree with previous literature. 1 Similar to the SOL, the PD-AFOs induced knee extensions accelerations when dorsiflexed. However, the PD-AFOs began inducing the knee extension accelerations much earlier than SOL. The PD-AFO induced knee extension accelerations from around 20% stance through the remainder of stance (Figure 6). Furthermore, net knee joint extensor moments decreased from 20% to 70% stance (Region II) in the PD-AFO conditions compared to the no PD-AFO conditions for both subjects (Figure 7). Despite these kinetic changes, the knee flexion–extension kinematics remained consistent across conditions for Subject A (Figure 8). For Subject B, the general knee flexion–extension pattern remained consistent across conditions, but there were noted variations in the amplitudes of the knee angle with and without PD-AFO use (Figure 8).
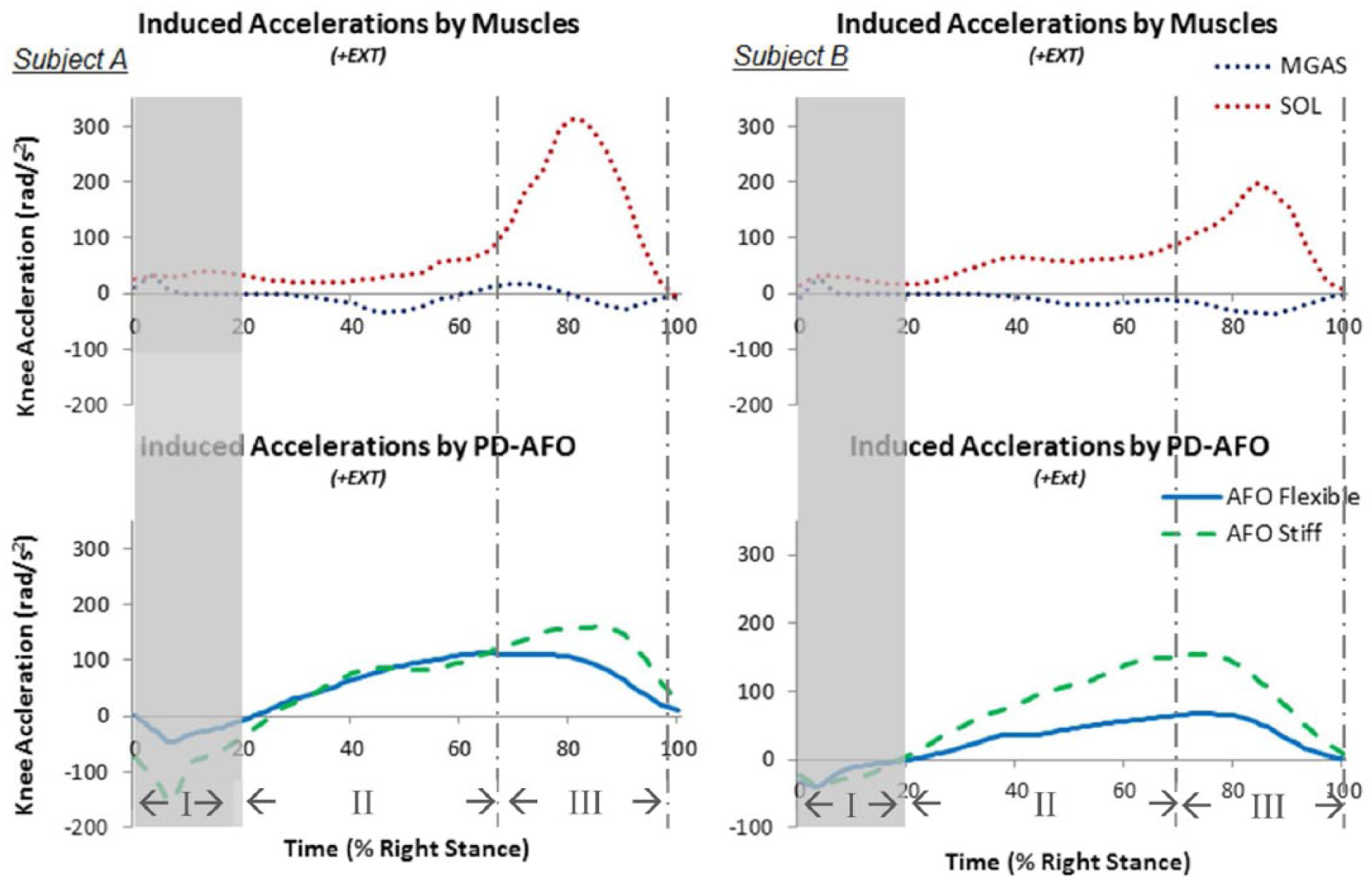
Flexion–extension accelerations of the knee joint induced by the MGAS and SOL (top) and PD-AFO (bottom) for Subject A (left column) and Subject B (right column). MGAS and SOL induced accelerations shown for the no PD-AFO condition.
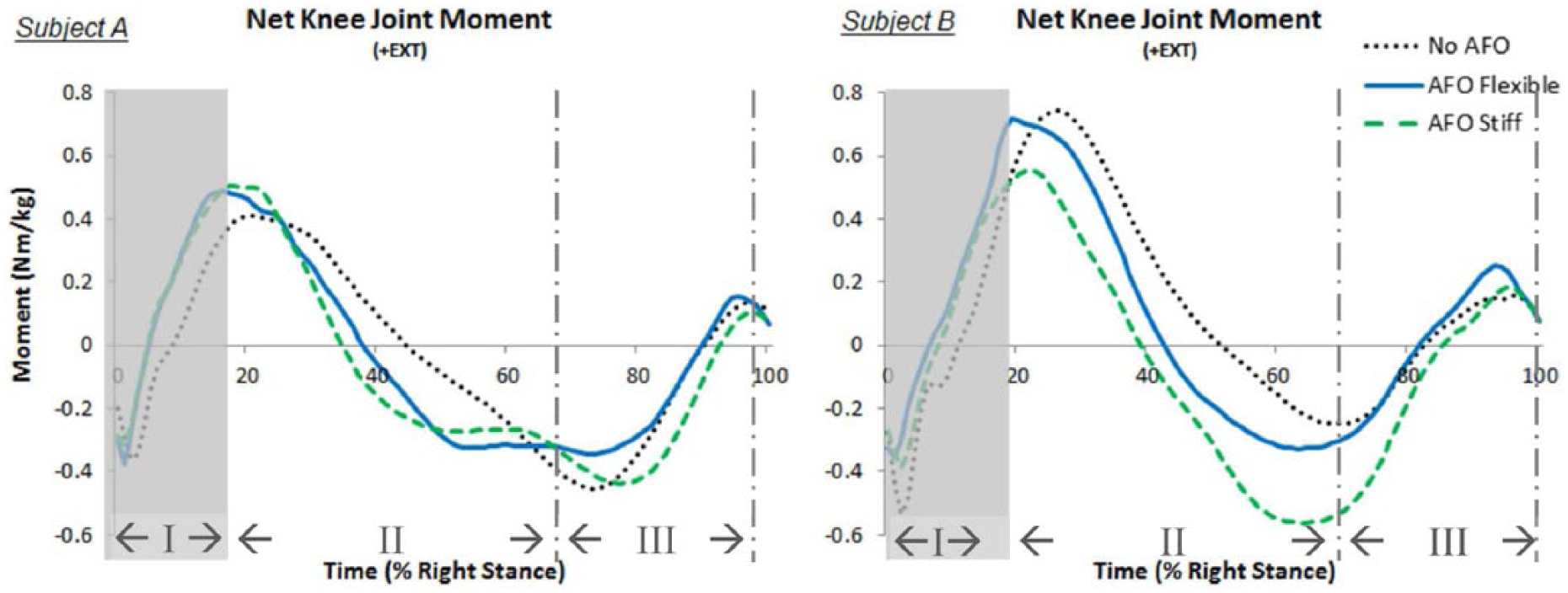
Net knee joint flexion–extension moments. Subject A (left) and Subject B (right). These joint moments were determined from the experimental data.
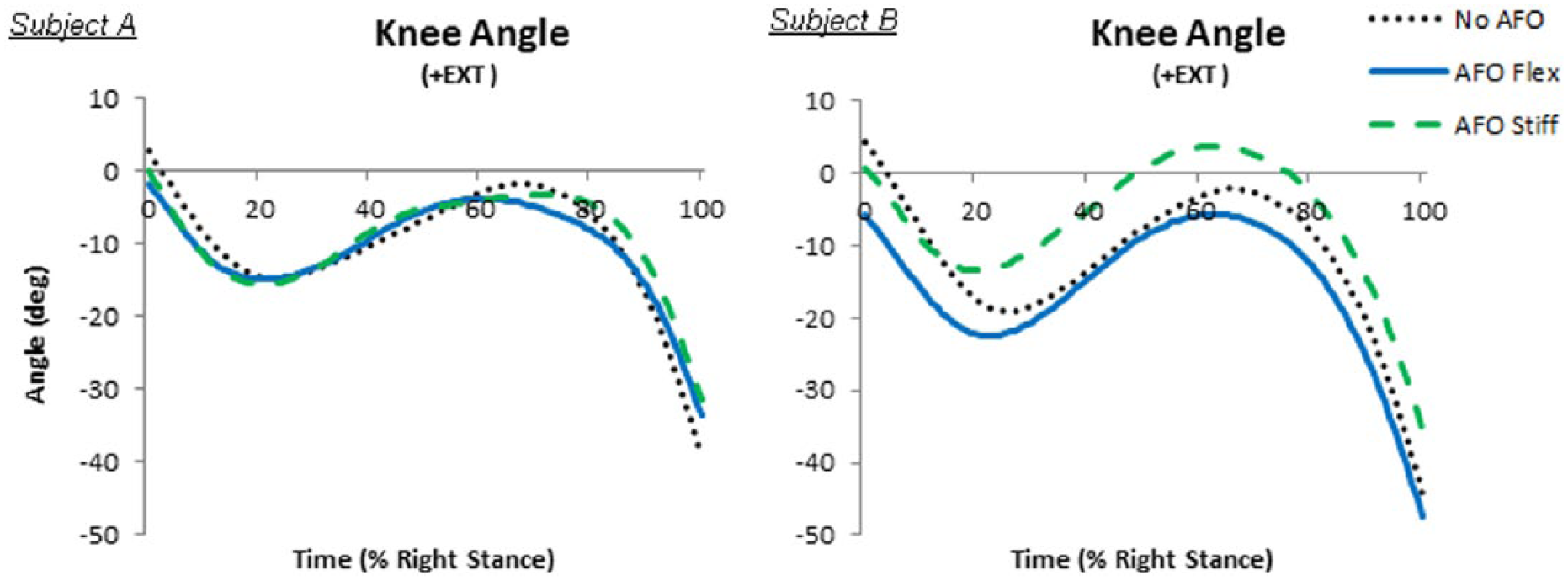
Net knee joint flexion–extension angles. Subject A (left) and Subject B (right). These joint moments were determined from the experimental data.
Discussion
The purpose of this study was to use musculoskeletal modeling and simulations to investigate the influence of the PD-AFO on plantar flexor function during the stance phase of gait. The results showed that the PD-AFO could substitute for some SOL function but not MGAS function. Furthermore, the PD-AFOs induced premature, unnatural knee extension accelerations, and this finding may explain why PD-AFO use down regulated SOL but not MGAS function. Finally, this study explored the influence of different levels of PD-AFO bending stiffness on healthy plantar flexor function. Understanding how the plantar flexors adapt to different levels of bending stiffness at the ankle provides some insight into the adaptive capacity of the musculoskeletal system across a range of stiffness levels, which is essential to being able to effectively customize PD-AFO bending stiffness for patients.
During stance, the PD-AFOs always resisted deformation and tried to return the ankle to the neutral position, producing moments as a function of ankle angle and orthosis stiffness. Consequently, with the PD-AFO ankle joint set to a neutral alignment, the PD-AFOs produced plantar flexion moments throughout the time when the ankle was dorsiflexed. Despite these added PD-AFO moments, the peak net plantar flexion moments remained consistent with and without orthosis use. While peak MGAS moments also remained virtually constant across all conditions, peak SOL moments decreased with PD-AFO use. Since the PD-AFOs acted like uniarticular plantar flexors, PD-AFO stiffness substituted for some SOL function, primarily during its peak activity (70%–100% stance). This downregulation of SOL function by the PD-AFO likely enabled peak net ankle joint moments to remain the same without and with PD-AFO use (Region III).
However, MGAS displayed a different adaptation with PD-AFO use than SOL. In particular, MGAS maintained normal, or sometimes increased, moments beginning around 30% stance and persisting through the remainder of stance, even though the PD-AFOs provided some contribution to the net plantar flexion moment. Thus, since MGAS activity was not down regulated with PD-AFO use, a premature increase in the net ankle joint plantar flexion moment occurred (Region II) because the moment contributed by the PD-AFO was added to the moment contributed by MGAS.
The downregulation of SOL function but not MGAS function concurs with other findings reported in the literature. One research group designed a pneumatically powered ankle exoskeleton that used proportional myoelectric control to provide assistance to the plantar flexors and used it to study how human adapt to this assitance.42,43 While this power assistance is not the same assistance provided by the PD-AFO bending stiffness in this study, the findings regarding the adaptations of the SOL and MGAS can be compared. In concurrence with this study, the research by Ferris and colleagues42,43 found that healthy individuals decreased SOL activity when assistance was provided by the powered ankle exoskeleton, but gastrocnemius activity remained constant or decreased minimally. These findings were consistent regardless of whether the ankle exoskeleton was controlled by the SOL or gastrocnemius. 43 Furthermore, some prosthetic feet have been modeled in a similar manner to the PD-AFO, and these models have been used to study the role of prosthetic feet as it relates to typical muscle function in gait. Studies have shown that energy storing and returning prosthetic feet can provide the primary functions of the SOL, but not the gastrocnemius, with respect to accelerations and energetics of the trunk and leg.44,45 Thus, results from this study agree with these previous findings in that they show that these devices can replicate some SOL, but not gastrocnemius, function during stance in gait. However, uniquely, this study demonstrated that the PD-AFO stiffness can downregulate healthy SOL function with different bending stiffness levels, where the prosthetic feet simply replaced the non-existent SOL function. Furthermore, this study examined the influence of different PD-AFO bending stiffness levels on the ankle and knee joints instead of trunk and whole-limb accelerations.
Induced acceleration analysis helped explain this complex role of MGAS during orthosis use. As a result of the stiffness, the PD-AFOs, acting like uniarticular plantar flexors, induced knee extension when the ankle was dorsiflexed, which began around 20% stance. However, results showed that SOL did not start inducing knee extension accelerations until around 70% stance, which is typical of healthy gait. 4 Thus, the PD-AFOs induced considerable knee extension accelerations not normally present from 20% to 70% stance. In contrast to SOL, the biarticular MGAS induced no or minimal knee accelerations throughout stance. Thus, this study’s findings suggested that while the PD-AFO could substitute for some SOL function, MGAS function could not be down regulated in these neurologically healthy individuals because it had a crucial and complex role, working to eccentrically control ankle dorsiflexion while not interfering with the natural knee flexion–extension mechanism, a function that the PD-AFOs could not replicate. These induced acceleration findings were further supported by the net knee joint extensor moments. From 20% to 70% stance, the net knee joint extensor moments decreased with PD-AFO use, indicating that the muscles crossing the knee had to produce less knee extension moment likely because of the knee extension accelerations induced by the PD-AFO. Even with these kinetic changes at the knee, Subject A was able to maintain natural knee joint kinematics and Subject B was able to maintain the general flexion–extension pattern of the knee joint but had changes in knee angle amplitudes.
Previous studies have investigated the influence of AFOs with different bending stiffness levels on gait function. Results from a recent study by Russell Esposito et al. 46 examined the influence of different orthosis bending stiffness levels on gait biomechanics of individuals post-limb salvage concur with the net ankle joint moment results from this study, as both show that AFO stiffness can substitute for ankle moments. However, unlike this study, the study by Russell Esposito did not include any muscle-level analysis to shed light onto the muscle function that enabled that substitution. However, with the same population, Harper et al. 47 studied the influence of different orthosis bending stiffness levels on EMG-based muscle function and found altered gastrocnemius activity with different AFO stiffness levels. This finding differs from the results of our study, which found that SOL, not gastrocnemius, adapted its function in response to different PD-AFO bending stiffness levels. The study by Harper was conducted on individuals with lower-limb impairments, and as a result of these impairments, the subjects may have not been able to adapt their uniarticular SOL function and instead relied on adaptations from their biarticular gastrocnemius. Finally, a study by Haight et al. 48 evaluated the influence of PD-AFOs with three different bending stiffness levels on the biomechanics of uphill walking and showed a similar substitution pattern for the net ankle joint moment across stiffness levels. Uniquely, this study also provided induced acceleration analysis to help explain the muscle function that resulted from use of the PD-AFOs with different bending stiffness levels.
Some study limitations should be noted. Simulation results are dependent on many factors including the definition of the musculoskeletal model and the optimization cost function used, so the results and conclusions may vary if simulation parameters are modified. However, the experimental EMG generally supported the muscle function conclusions drawn from the simulations, showing a decrease in SOL EMG, but not MGAS EMG, with PD-AFO use for Subject A (Figure 5). EMG of the SOL was not analyzed for Subject B because a clean EMG of the SOL was not obtained. Additionally, only one stiffness value for each rotational DOF could be specified in the bushing force used to model the PD-AFO, so the dorsiflexion and plantar flexion rotations had the same stiffness value. In reality, the stiffness when deforming the PD-AFOs into plantar flexion was much higher than when deforming into dorsiflexion. However, the ankle was in dorsiflexion during the majority of stance and certainly during the regions of interest for this study, so this simplification of the PD-AFO model likely did not affect the findings. Moreover, while the manufactured PD-AFO stiffness levels were slightly different than targeted, the bending stiffness levels were within previously reported accuracy levels 33 and the differences between the two PD-AFO for each subject were greater than 30%, which should be large enough to investigate the influence of different bending stiffness levels on plantar flexor function during gait. Finally, data from only two healthy subjects were used in this study. However, as this was primarily a simulation study that used a healthy musculoskeletal model scaled to each subject’s anthropometrics, the simulation results would only change if the pose of the system (kinematics) changed substantially. Thus, while kinematics and kinetics varied slightly between subjects, the same conclusions were drawn from the simulation results and it is unlikely that additional subjects would change the study conclusions.
This study’s findings have important implications for providing biomechanical rationale for customizing PD-AFO characteristics for individuals with plantar flexor weakness. This study revealed that the PD-AFO can substitute for some SOL function, supporting the belief that a PD-AFO can supplement a patient’s weakened plantar flexors. However, the PD-AFO negatively influenced the knee earlier in stance. While the healthy subjects in this study were mostly able to maintain the typical knee flexion–extension pattern, patients may not have the capacity to maintain this knee kinematic pattern. Therefore, future research should harness this study’s findings to investigate whether the PD-AFO design can be modified to minimize the negative influence of the PD-AFO on the knee. For example, setting the PD-AFO ankle joint into dorsiflexion in its unloaded position may allow for gravity to flex the knee to counteract the knee extension accelerations induced by the PD-AFO, and future research should explore this hypothesis. Future research should also examine the influence of these PD-AFOs on the activity of other muscle groups. Finally, methods and findings from this study may be able to be used in the future to develop objective prescription models to optimally customize PD-AFOs for individuals.
Conclusion
The findings in this study demonstrated that the PD-AFO can substitute for some SOL function and thus can potentially supplement a patient’s weakened plantar flexors in mid and late stance. However, the PD-AFO acted liked SOL, not MGAS, by inducing knee extension accelerations. With the PD-AFO ankle joint aligned in a neutral position as was done in this study, the PD-AFO induced knee extension accelerations much earlier in stance than the SOL typical does.
Footnotes
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs. Steven Stanhope and Elisa Arch are the co-inventors of the patented technology used to customize and manufacture the passive-dynamic ankle–foot orthoses used in this study (US Patent No. 8,538,570).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This material is based upon work supported under a National Science Foundation Graduate Research fellowship.
