Abstract
Background:
Charcot neuroarthropathy is one of the more devastating complications of diabetes. It is a progressive, destructive condition that is characterized by acute fracture, dislocation, and joint destruction in weight-bearing neuropathic foot. In its acute phase, it is often misdiagnosed and can lead to several deformities such as ulcerations and amputation. Early diagnosis and management is, therefore, imperative to avoid rapid progression.
Objectives:
Review current literature on the diagnosis and management of diabetic patients with Charcot neuroarthropathy.
Study design:
Narrative review.
Methods:
Particular attention is directed to the role of surgical management in achieving long term osseous stability and alignment so that appropriate footwear and bracing are possible.
Conclusion:
Charcot neuroarthropathy is a serious and potentially limb-threatening lower extremity late complication of diabetes. Correct timing, adequate fixation and a long post-operative weightbearing period are key to optimizing reconstructive surgery.
Clinical relevance
Primary care providers who treat diabetic patients should be cognizant of the possible complication among patients with diabetic neuropathy of which includes Charcot neuroarthropathy. In this paper, discussion is provided on a rational approach to functional limb salvage with various surgical techniques when non-operative management fails.
Introduction
The famous French neurologist, Jean-Martin Charcot, described a chronic and progressive neuroarthropathy resulting in joint destruction in a patient with tabes dorsalis. His work on diabetic neuroarthropathies was published in 1868 in Archives de physiologie normale et pathologique. 1 However, it was not until 1936 that William Riley Jordan 2 described the first case of neuropathic arthropathy in a patient with diabetes mellitus (DM). Today, diabetes is the most common cause of Charcot neuroarthropathy (CN) in developed countries.3 –7 Other known causes of neuroarthropathies include syphilis, HIV, leprosy, yaws, syringomyelia, meningomyelocele, spina bifida, amyloid neuropathy, neuropathies secondary to alcoholism and renal dialysis, and post-renal transplant arthropathy.8 –10 CN is characterized as a progressive process of deterioration of weight-bearing joints, most commonly affecting the foot and ankle (Figure 1). Pathologic fractures and dislocations are associated with CN, with collapse of the architecture of the foot and progression to plantar deformity, ulceration, infection, and the potential for amputation. Diabetic patients with CN have an increased risk of mortality and lower health-related quality of life when compared to diabetic patients without CN. 11
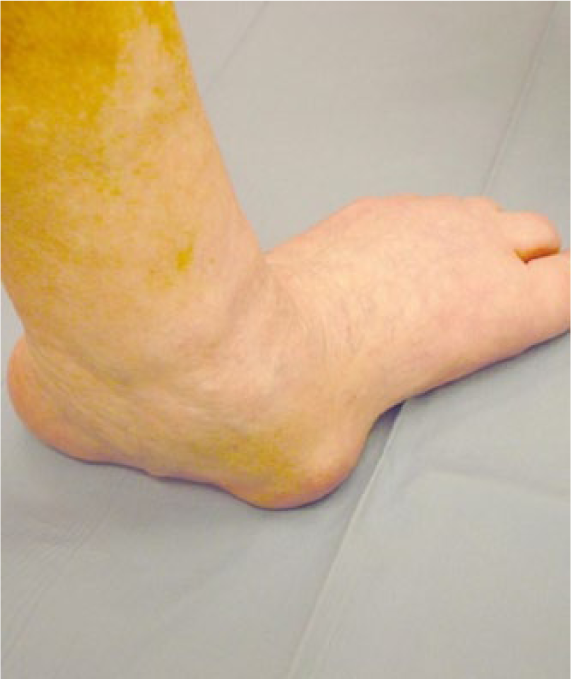
Clinical photograph of a 44-year woman with a Charcot fracture dislocation of the hindfoot.
Despite its relatively low incidence and prevalence rates, CN causes significant mortality and morbidity, with mortality rates estimated to be as high as 28%. In contrast, those patients with diabetes alone have 23% lower mortality risk. 11
A recent study by Sohn et al. shows that 34% of patients develop foot ulceration as a complication of CN. This work found no serious amputation risk in the presence of simple CN, but the amputation risk was noted to be much higher in patients with concomitant ulceration. 12 The study revealed a risk of amputation that was 7 times higher in patients with ulceration in the absence of CN, and 12 times higher in patients with both CN and ulceration when compared with patients with CN alone.
If the disease is diagnosed early in its course, appropriate management will lead to improved outcomes. With delayed diagnosis, subsequent pedal collapse may result in an inability to manage subsequent deformities conservatively, resulting in the need for surgical intervention to salvage the limb. The goal of early and accurate diagnosis includes prevention of amputation.13,14
The disorder is commonly misdiagnosed as sprain deep vein thrombosis, osteomyelitis, cellulitis, or rheumatoid arthritis. To prevent devastating outcomes associated with delayed diagnosis and treatment, such as gross foot deformity or amputation, it is critical for providers to be able to accurately diagnose and effectively manage CN.
Epidemiology
CN is an uncommon complication among diabetic patients. The incidence of the disease among diabetic patients is approximately 3–11.7/1000 patients per year.7,9 The prevalence ranges from 0.08% to 0.40% among diabetic patients but may be as high as 13% in high-risk patients. 5 The wide variation is partly attributed to underreporting of the disease as a result of misdiagnosis or delay in diagnosis particularly in its early stages. 15
The age of onset of CN varies. In patients with type 1 DM, the most frequent presentation of the disease occurs during the fifth decade with an average duration of diabetes for 20–24 years. In patients with type 2 DM, CN frequently presents during the sixth decade with duration of diabetes for 5–9 years. 16 There are risk factors common to both type 1 and type 2 DM that significantly increase the likelihood of development of CN. These risk factors include peripheral neuropathy, obesity, diabetes for duration of 6 years or more, hemoglobin A1c of 7% or more, and renal failure (p < 0.001). 17
Pathoanatomy
The process of Charcot arthropathy is characterized by a phase of hypertrophic repair after initial bone resorption in the neuropathic joint. 5 There is a reduction of bone density and increased osteoporosis, accompanied by abnormal collagen formation. Rupture of the plantar fascia due to glycosylation of collagen causes failure of the bony support as a result of undue stress on the joint arch. 5 Additionally, shortening of the Achilles tendon may render joints prone to abnormal pressure distribution, resulting in deformity and Charcot foot. 18 These factors contribute to impaired healing of a foot that has lost its biomechanical support, resulting in an unstable fracture. 19
Pathogenesis
Although the pathophysiology of the disease remains unknown, two principal theories have been proposed. The neurovascular, or French theory, postulates that vasomotor change related to autonomic dysfunction accompanying the neuropathy leads to vasodilation with increased blood flow to the extremity, resulting in an imbalance of bone destruction and synthesis.20,21 There is increased sympathetic activity, hyperthermia, and facilitation of bone resorption with periarticular fractures and subluxation of the joints.4,22 The neurotraumatic theory postulates that repetitive micro-trauma in the insensate foot results in unrecognized subchondral fractures that, with continued activity, lead to joint fragmentation and subluxation of the joints.4,6 An exaggerated inflammatory response to a local trauma may be a primary pathogenic factor. Because the foot is insensate, with loss of proprioception, the patient is not aware of the bony destruction occurring with repeated stress to the joint. 20
CN is probably a combination of autonomic neuropathy and subsequent abnormal bone formation, with a sensory neuropathy leading to susceptibility to trauma. It is most likely that the abnormal bone development results in an inability to protect the joint, with gradual fracture and subluxation. An emerging hypothesis in the pathogenesis of Charcot involves the receptor activation of the nuclear factor kappa B ligand (RANKL)/RANK signaling pathway.23 –25 Jeffcoate et al. theorized that an exaggerated inflammatory response to injury of the neuropathic foot is responsible for the cycle that results in the destructive bony and joint changes seen in CN. They postulate that proinflammatory cytokines, primarily tumor necrosis factor (TNF) alpha and interleukin 1-beta, trigger increased expression of RANKL, leading to activation of RANK, inducing osteoclast differentiation and maturation. 23 This theory is supported by evidence of significantly lower bone mineral density in the affected foot of patients with CN when compared to the healthy foot (p < 0.01). 26
Classification systems
Numerous systems have been proposed to classify CN. None of the systems adequately categorized the disease in early stages or provide prognostic information or direct the treating physician to specific treatment options. Eichenholtz 27 proposed a systematic classification in 1966 to stage the progression of CN and recommend treatment based on corresponding clinical and radiographic patterns. The newer modified Eichenholtz classification now includes magnetic resonance imaging (MRI) to differentiate the qualitative findings at each of the four stages of the disease.28,29
Stage I, the fragmentation or developmental phase, is characterized by increased edema, erythema, warmth, and minor joint deformity and instability; radiographic changes include osteopenia, subchondral erosions, periarticular fragmentation, and joint subluxation or frank dislocation of joints. Stage II, the coalescence phase, subacute CN (reparative stage) is characterized by decreased edema, erythema, and warmth, as well as marked joint deformity and instability; radiographic changes include remineralization of regional bone, new bone formation, sclerosis, absorption of osseous debris, and callous formation.During stage III, the consolidation or remodeling phase, edema, erythema, and warmth are no longer present; radiographic changes include osteoarthritis, joint space collapse, subluxation, and deformity. Stage III, con-solidation stage, reconstructive or bone healing stage is characterized by the absence of inflammation and restoration of joint stability; however, joint deformity is permanent. Stage 0, the clinical phase, is characterized by edema, erythema, and warmth without any radiographic changes. Stages 0 and I constitute the acute phase of the disease and last 2–6 months. Stages II and III constitute the chronic phase of the disease.
There are several classifications utilized in the characterization of the anatomy of Charcot foot. A commonly used anatomic system was defined by Saunders and Mrdjenovich and is based upon the location pattern of the arthropathy:
Forefoot, 10%–30%;
Lisfranc’s joint, the most common presentation;
Involvement of the midtarsal joint, frequently including the naviculocuneiform joint;
Involvement of the ankle and subtalar joints, seen in 8%–10% of cases;
Approximately 2% of cases involve “posterior pillar” fractures of the calcaneus. 30 The forefoot seems to have a better prognosis than hindfoot arthropathy since lesions of the calcaneum may have detrimental effects on weight distribution during walking. 31
Brodsky’s 32 anatomic classification identifies four patterns of collapse in patients with CN (Figure 2(a) to (c)). Type 1 affects the tarsometatarsal and naviculocuneiform joints—the most commonly affected joints in CN. Collapse of this joint leads to the classic fixed “rocker bottom” foot with valgus angulation. This deformity commonly leads to the development of recurrent plantar foot ulcerations. Type 2 collapse involves the subtalar and midtarsal joints and frequently leads to significant joint instability. Type 3A involves the ankle joint and leads to recurrent ulceration and osteomyelitis over the malleoli. Type 3B involves fracture of the posterior calcaneal tuberosity. The distribution of CN fracture and dislocation pattern includes 50% midfoot (type 1), 28% hindfoot (type 2), 19% ankle (type 3A), and 3% forefoot (type 3B). 32
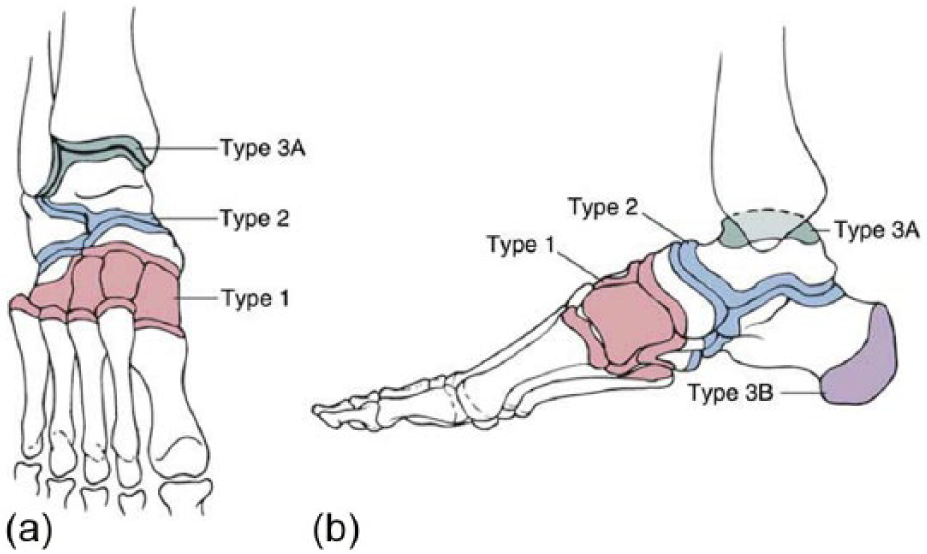
Brodsky’s anatomic classification: (a) anterior–posterior and (b) lateral drawings demonstrating Brodsky’s anatomic classification of the Charcot foot. Type 1 involves the tarsometatarsal and naviculocuneiform joints. Type 2 involves the subtalar joint. Type 3A involves the ankle joint. Type 3B involves fracture of the posterior calcaneal tuberosity.
A two-stage system that classifies CN as active or inactive (quiescent) according to the status of the inflammation appears to be more clinically relevant.31,33
Clinical presentation and diagnosis
Accurate diagnosis may be elusive and can be a considerable clinical challenge for the treating physician. The diagnosis of acute CN is primarily clinical requiring a high index of suspicion. It should be suspected in a diabetic patient with an inflamed foot, profound neuropathy, and bone and joint abnormalities in the absence of fever and elevated erythrocyte sedimentation rate (ESR).4,7,31
This presentation often is confused with cellulitis and can make a definitive diagnosis difficult. A diagnosis of CN should be considered in all diabetic patients with peripheral neuropathy who present with leg edema, erythema, and increased skin temperature with or without foot ulceration.
In the acute phase, patients present with unilateral erythematous and edematous lower extremity, which may or may not be painful. Patients may complain of warmth or change in foot shape. Deformity may or may not be present in the foot, and true acute stage radiographic abnormalities may be absent.
In the acute active stage, the patient typically presents with unilateral-dependent erythema, edema, increased skin temperature, and joint effusion in an insensate foot.34,35 The skin temperature is greater than 2°C warmer than the contralateral foot and ankle. 4 In the acute active stage, the disease is often mistaken for cellulitis.
If the skin is intact, these findings are pathognomonic of CN. In some patients, the diagnosis may be complicated with a concomitant ulceration that raises the possibility of osteomyelitis. Signs of systemic infection also require further investigation. Ulceration of the foot may be a consequence of the joint deformity.
Pain may be absent due to peripheral neuropathy. Pain occurs in more than 75% of patients, but normally it is less severe than expected in the presence of the significant clinical and radiological findings.16,35 Repetitive micro-trauma on an insensate foot may be responsible for CN.36,37
Patients may present with a history of a minor trauma or may have recurrent episodes of swelling, warmth, and edema over a longer period of time.3,8 Recent surgical procedures on the foot may be a precipitating factor for development of acute CN.38,39 Local inflammation after pedal amputation may lead to alterations in weight bearing, resulting in micro-trauma.40,41
If the Charcot process is allowed to progress to bone destruction, the foot collapses and results in a “rocker bottom deformity” with difficult exostoses predisposing to ulcerations.31,33 Chronic late stage disease presents without erythema or warmth, and edema may or may not be present. Both acute and chronic stages can be complicated by plantar ulcerations on the bony prominences of the foot. The ulcerations may be refractory to care 3–6 months after the acute stage.
Investigation and differential diagnosis
Accurate diagnoses may be elusive and can be a considerable clinical challenge for the health-care provider. A correct diagnosis of CN within 3 months of initial signs and symptoms results in better functional outcome and walking distance; conversely, a delay of diagnosis of more than 3 months adversely affects the quality of life and functional outcome of diabetic patients. 42
Furthermore, early diagnosis and treatment is linked with a significantly decreased incidence of gross foot deformities and complete foot fractures (p < 0.001). 42 For these reasons, a prompt and correct diagnosis of CN is vitally important.
The acute stage is commonly misdiagnosed as cellulitis, septic arthritis, deep vein thrombosis, gout, abscess, sprain, and osteomyelitis. Gout can be excluded by measuring the serum uric acid levels; the deep vein thrombosis can be ruled out by a duplex venous scan.
To differentiate CN from osteomyelitis, a careful patient history, physical examination, specific diagnostic tests, and imaging must be performed. 43 Osteomyelitis can be ruled out when there is no presence or history of foot ulceration.31,44 Reduction of the erythema and swelling with elevation of the affected limb may help differentiate acute CN from an infectious process. 3
A useful clinical examination is the “probe to bone” test; if bone can be palpated through an open foot ulcer, osteomyelitis is likely. Systemic signs of inflammation such as an elevated ESR), C-reactive protein, fever, or leukocytosis are sensitive tests but lack specificity and are of limited usefulness in discriminating between CN and osteomyelitis.
Imaging
The initial workup of CN should involve weight-bearing radiographs to determine whether bone injuries are present. Radiographic analysis aids in the staging process and also assists in determining the presence of active disease or joint stability. Early disease findings on plain films are frequently nonspecific or minimal, with swelling of soft tissue, osteopenia, and loss of joint space. 21 Findings are also dependent upon the location of the arthropathy. When involvement of the forefoot occurs, there is often a predomination of bone resorption with osteolysis of the phalanges.13,45 The metatarsal heads may disappear, or the shafts of the metatarsals and phalanges may acquire a deformity known as “pencil-pointing.” 5 When involvement of the midfoot or hindfoot occurs, the most common radiologic findings are osseous fragmentation, subluxation, dislocation, sclerosis, and formation of new bone.5,43
MRI
A contrast-enhanced magnetic resonance imaging (MRI) may help to differentiate noninfected from infected bone pathology in CN. The presence of a sinus tract, replacement of soft tissue fat, and fluid collection on a contrast-enhanced MRI favor osteomyelitis over CN. 46 Diffuse marrow involvement on MRI is frequently observed in the setting of CN and rarely without infection.46,47 Ring positron emission tomography (PET) has advantages over MRI because it can be used with patient with hardware and is more effective in differentiating bone infection from CN. 48
Radionuclide bone scanning and labeled white blood cell (WBC) scans or the combination of these two tests are useful for discriminating between CN and osteomyelitis.49,50 Bone scans show areas of increased bone turnover (a finding present in both disorders), but labeled WBC scans highlight areas of infection.49,51 This combination of imaging studies allows the detection and localization of osteomyelitis versus adjacent soft tissue infection.
When other diagnostic modalities fail, synovial biopsy may reveal small fragments of bone and cartilage debris embedded in the synovium due to destruction of the joint, a finding that is highly suggestive of CN, if not pathognomonic of the disorder. 52 Bone biopsy and culture may be useful to establish a diagnosis. Prompt distinction between these disorders is crucial since treatments differ and timely intervention leads to improved patient outcomes.
Management
Immediate referral to a specialist clinic or a multidisciplinary foot clinic for management of acute Charcot neuropathy is indicated.42,53 This will allow the patient all treatment options from trained specialists who can provide wound care, management of infection, casting, orthoses, and surgery.
The primary goals of treatment are structural stabilization of the foot and ankle, with maintenance of a plantigrade, stable foot that is able to fit into a shoe and to also prevent a recurrent ulceration. Management of CN varies upon the stage of the disease process, location of pathology, and the presence or absence of ulceration and infections.
Nonsurgical treatment
Immediate immobilization and offloading of the foot and ankle remains the cornerstone therapy during the active acute stage of Charcot arthropathy.3,9,44 The goal of conservative treatment is to interrupt the destruction process and to maintain adequate foot and ankle alignment.
Historically, total contact cast (TCC) is considered the gold standard immobilization therapy for acute CN. It reduces mechanical stresses and edema and redistributes the plantar pressure while maintaining a plantigrade foot for weight-bearing ambulation. Nonweight bearing on the affected joint should be initiated until the resolution of the destructive phase in order to break the cycle of repetitive trauma and minimize fracture and debilitating deformities.54,55
The first cast change is typically done after 1 week. The frequent cast changes allow for assessment of the skin and adjustment of cast size as edema subsides. In the presence of ulceration, debridement and evaluation of the ulcer are required every week. Cast immobilization should continue until erythema warmth and edema of the foot and ankle have resolved, with no further radiographic evidence of bone destruction. The mean time required in a cast varies from 12.5 to 18.5 weeks, according to two studies.5,55 The length of healing will depend upon the location of the pathology. Forefoot pathology heals in two-thirds the time of other patterns. When the acute inflammation has subsided and coalescence safely achieved, assisted weight bearing can be gradually started.
During the chronic late stages (stages II and III), edema, erythema, and warmth have been resolved, and radiographic studies demonstrate fracture healing, sclerosis, and bone remodeling. The patient should progress into accommodative bracing devices such as Charcot Restraint Orthotic Walker (CROW) or ankle–foot orthosis (AFO) followed by a return to depth-inlay shoes and custom foot orthoses. Management of the patient after removal of a CROW or TCC should incorporate lifelong protection of the extremity.56,57 This should include both patient education and foot care by a professional. Custom footwear may include extra-depth shoes with a plastic, carbon fiber, or metal shank. Protection of the contralateral extremity, although practically difficult, should not be neglected because contralateral fracture may occur in almost 80% of cases.3,4
Pharmacologic agent for CN
There is as yet no pharmacological agent licensed for use in acute Charcot foot. Bisphosphonates can inhibit osteoclastic bone resorption, so they are usually used in treatment of conditions characterized by abnormal turnover, especially acute active phase of the disease and sometimes in chronic phases.58 –61 Pamidronate is most commonly used, and it acts on hydroxyapatite crystals in newly synthesized bone matrix, blocking access of osteoclast precursors to the matrix.
Several studies have shown improvement in active Charcot arthropathy after an infusion of Pamidronate. 62 A study of patients randomized to treatment with alendronate while treated with a TCC boots progressing to pneumatic walkers revealed significantly decreased pain in the group treated with alendronate. 60 Bisphosphonate treatment is not approved by the Food and Drug Administration (FDA) for use for diabetic neuroarthropathy and should be used as an adjunct to standard offloading treatment.
Surgical management
When all nonoperative measures have been exhausted, surgical reconstruction of the deformed foot and ankle may be indicated. The main indications for surgical treatment of Charcot include gross instability, chronically recurrent ulcers, progressive joint destruction despite rest and immobilization, associated osteomyelitis, and selected acute fractures.
The goals of surgical treatment are to preserve functional activity, to restore stability and alignment so that appropriate footwear or bracing is possible, and to prevent amputation. The reported surgical management of CN includes exostectomy of bony prominence, debridement, resection–realignment, arthrodesis, open reduction and internal fixation, Achilles tendon lengthening, autologous bone grafting, external fixation, and amputation
Surgical treatment varies depending upon the surgeon’s preference and experience in combination with the location of the disease. There are controversies in the literature with respect to timing of surgical intervention for foot and ankle deformities. Historically, surgical reconstruction has been reserved for stage III deformities because of the high rate of nonunion, infection, and wound complication. However, other authors report successful arthrodesis and open reduction and internal or external fixation during the developmental stage.63,64 In the report by Simon et al., 64 the authors studied the use of arthrodesis of the tarsometatarsal and advocated early arthrodesis for Eichenholtz stage I Charcot arthropathy of the foot noting likelihood anatomical reduction, clinical union, and stability with or without increased risk of complications.
Preoperative evaluation and workup
A complete workup and optimization is paramount to a successful surgical outcomes and stratifying risk of complication. The preoperative assessment should include knowledge of patient’s ambulatory status, quality of the soft tissue, and the presence or absence of infection. An elevated hemoglobin A1c indicates poor hyperglycemic control in diabetes. Complete blood count and differential, ESR and C-reactive protein, and blood cultures to rule out underlying osteomyelitis. In the presence of an ulceration or chronic open wound, staged reconstruction is necessary to convert a contaminated (i.e. bacterial count > 105) or infected wound to a clean wound. 65
The preoperative assessment should include a thorough vascular evaluation with transcutaneous oxygen pressure (TcPO2), ankle brachial index, and toe pressures to document evidence of sufficient arterial perfusion to the limb. It requires a comprehensive team approach to optimize patient diabetes.
Exostectomy
Simple exostectomy is indicated for stable midfoot CN in order to prevent primary or recurrent ulcerations and to relieve shoe fitting problems63 –68 (Figure 3(a) to (c)). It involves surgical removal of the osseous prominence from the apex of the rocker bottom deformity of the foot. Many recommend management of the overlying plantar foot ulcer first before proceeding with bony resection. A concomitant percutaneous lengthening of the tendon or gastrocnemius recession is often required to achieve a plantigrade foot and to decrease the chances of ulcer recurrences.66,67 One potential complication from exostectomy is an iatrogenic midfoot instability from an aggressive resection or failure to recognize potential preoperative instability.
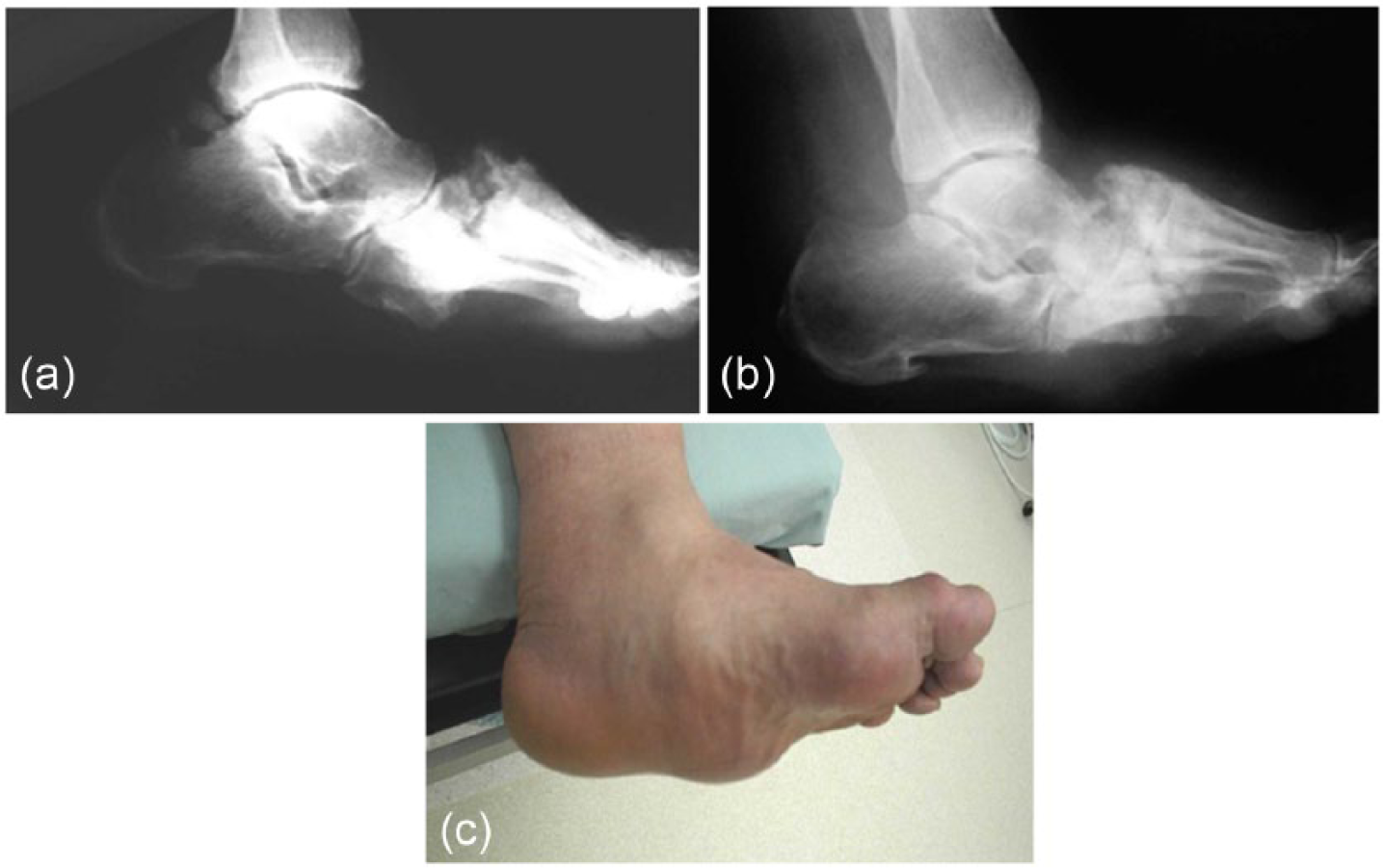
A 50-year-old woman with a Charcot neuroarthropathy with a “rocker bottom deformity.” (a and c) Lateral weight-bearing radiographs and Preoperative photographs. (b) Lateral radiographs following plantar exostectomy and a tendo Achilles lengthening.
Surgical realignment arthrodesis
The major goal of reconstructive arthrodesis is to restore the stability and alignment of the foot and ankle, so that prescription footwear can be worn. It involves resection of nonviable bone with reduction of the deformity followed by stabilization and/or arthrodesis of multiple tarsal and/or ankle joint.
This procedure is indicated for failed exostectomy, gross instability, severe fixed deformity, and acute dislocation. The surgical approach depends on the severity and location of the deformity, level of instability, and the presence or absence of infection. Reported method of stabilization for midfoot CN includes multiple screw fixation, staples, intramedullary screws, and compression plating and screw constructs.69 –72 The use of a plate on the plantar aspect of the medial column of the midfoot has been advocated to enhance the rigidity of an arthrodesis of the midfoot 73 (Figure 4(a) to (d)).
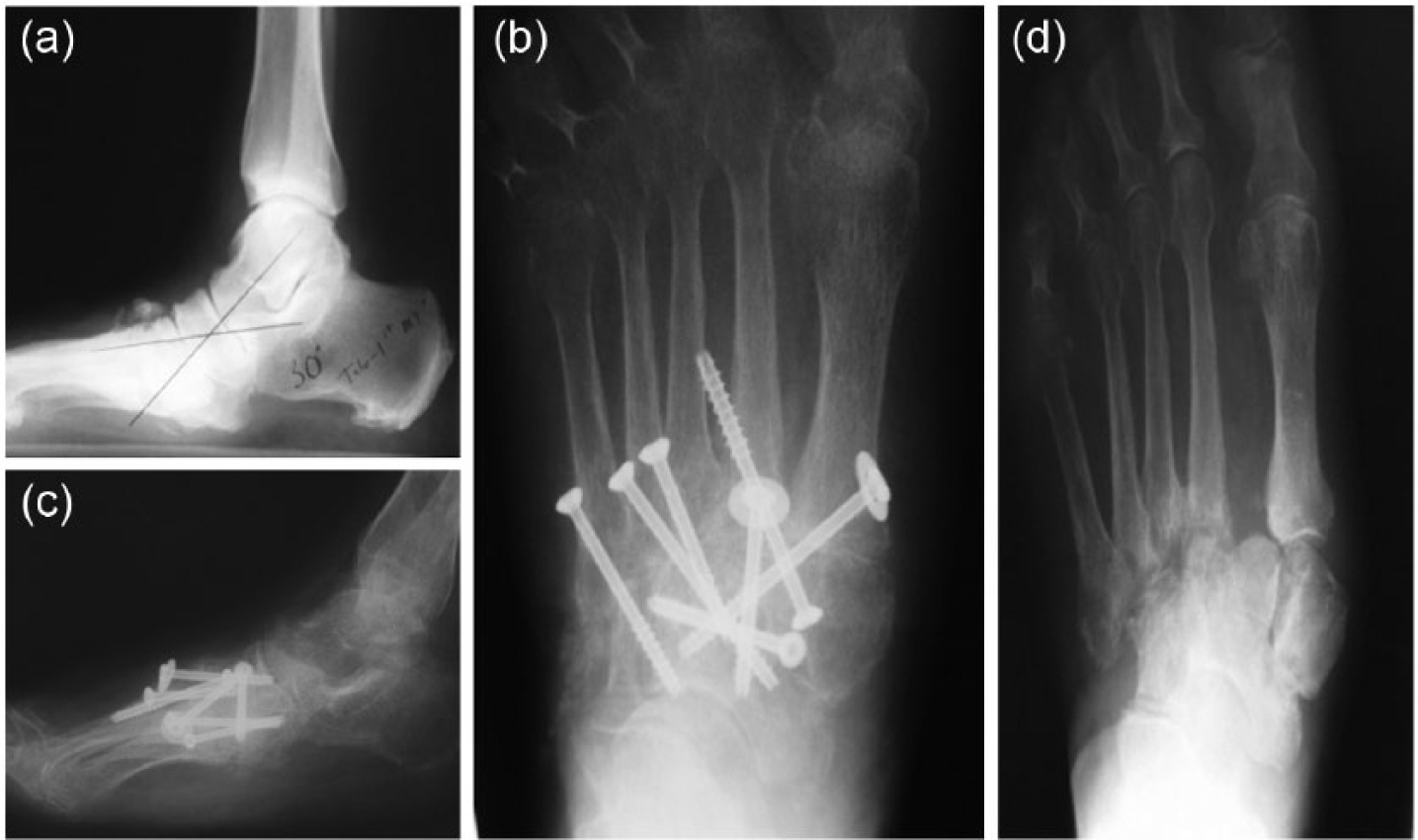
A 50-year-old woman with an Eichenholtz stage III nonplantigrade foot deformity. (a and d) Preoperative anterioposterior and lateral weight-bearing radiographs. (b and c) Postoperative radiographs following a midfoot resection–realignment arthrodesis with multiple screw fixation.
Open reduction and arthrodesis with use of multiple axially placed intramedullary screws provided reliable construct to achieve and maintain correction of deformity 69 (Figure 5(a) to (k)).
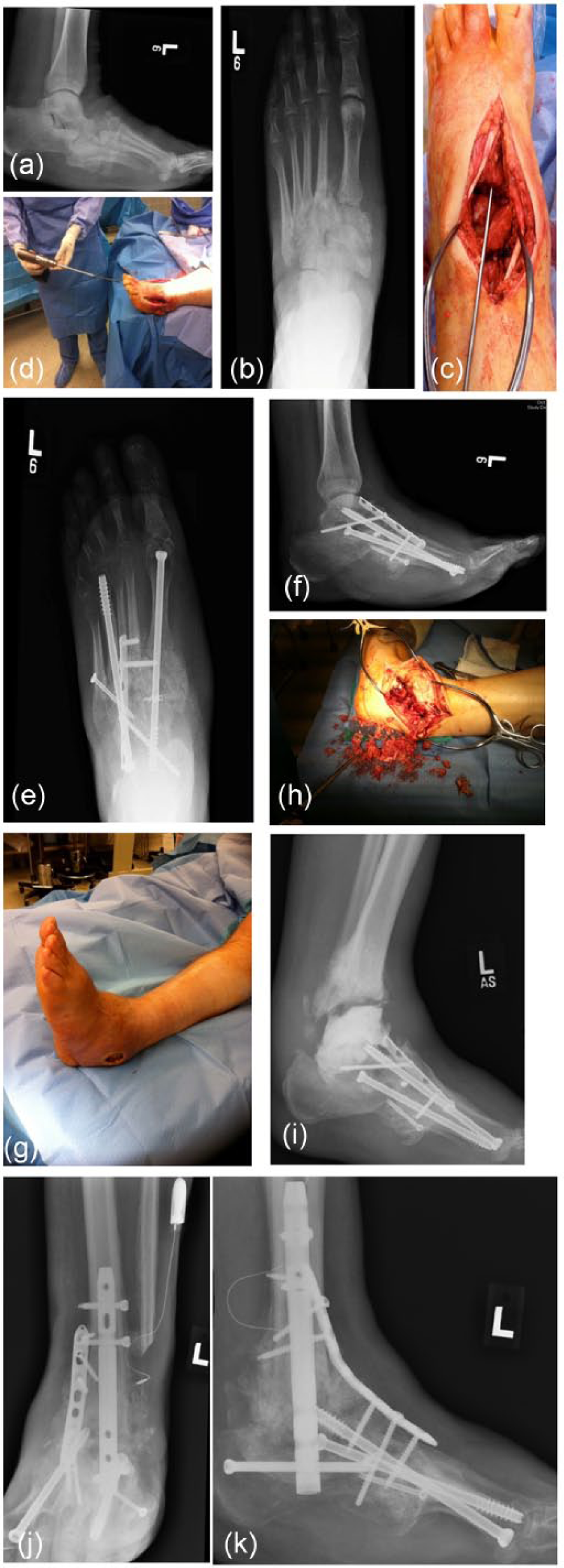
A 61-year-old man with a Brodsky type 1 midfoot Charcot neuroarthropathy. (a and b) Preoperative radiographs. (c and d) Intraoperative photographs. (e and f) 7 months after midfoot arthrodesis. (g and h) presented 2 years following midfoot arthrodesis with an infected ankle instability. (i) Infection managed with debridement followed by antibiotic spacer. (j and k) Postoperative radiographs showing the arthrodesis with internal fixation.
Ankle and hindfoot CN can be successfully managed with a retrograde intramedullary revision ankle nail (Figure 6(a) to (e)).
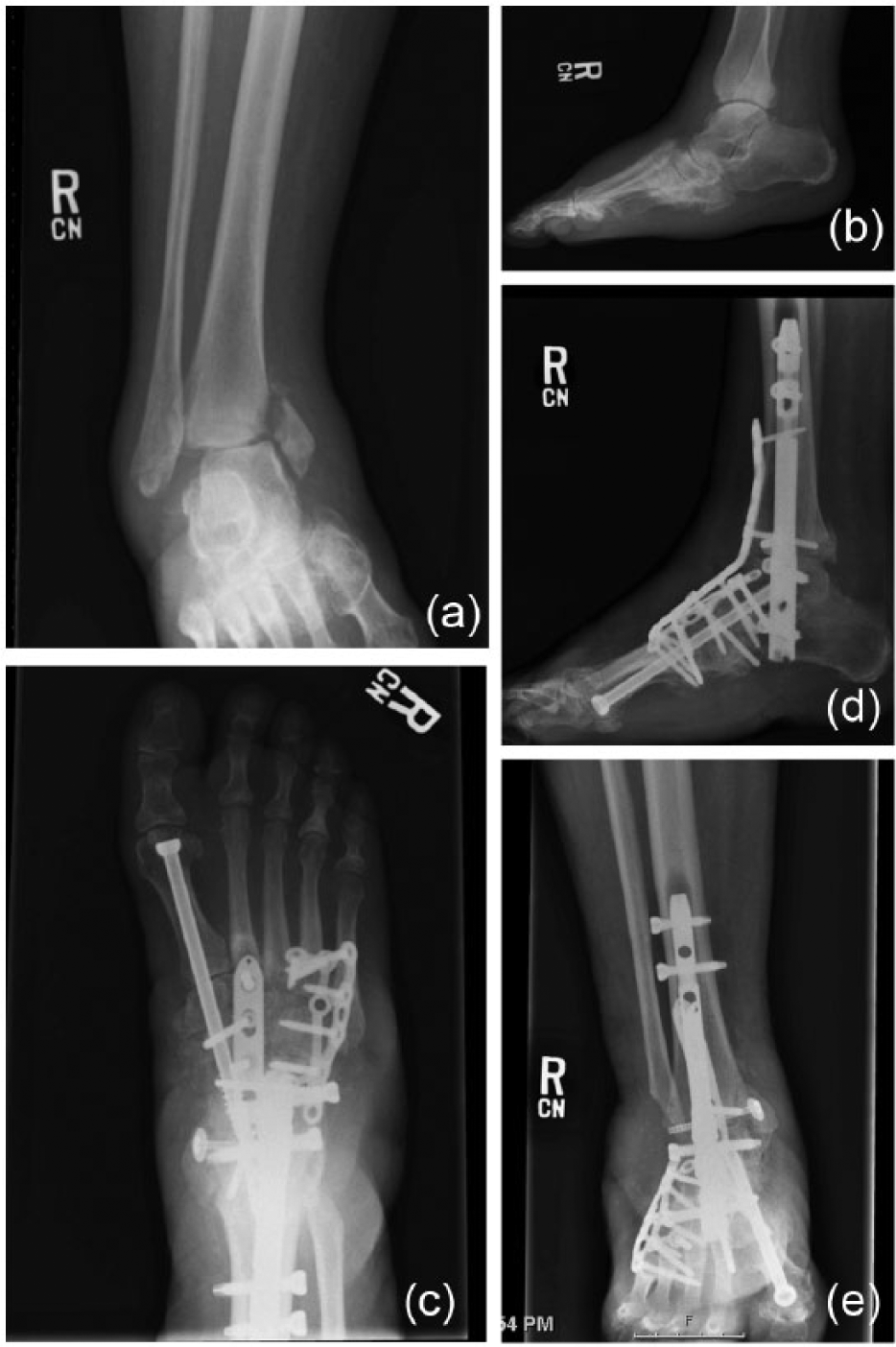
A 36-year-old man with insulin-dependent diabetes mellitus with a midfoot Charcot neuroarthropathy presenting with an acute ankle fracture. (a and b) Preoperative radiographs and (c, d, and e) postoperative radiographs showing 5 months after foot and ankle reconstruction.
A displaced unstable ankle fracture is an indication for immediate open reduction and internal fixation (Figure 7(a) to (d)).
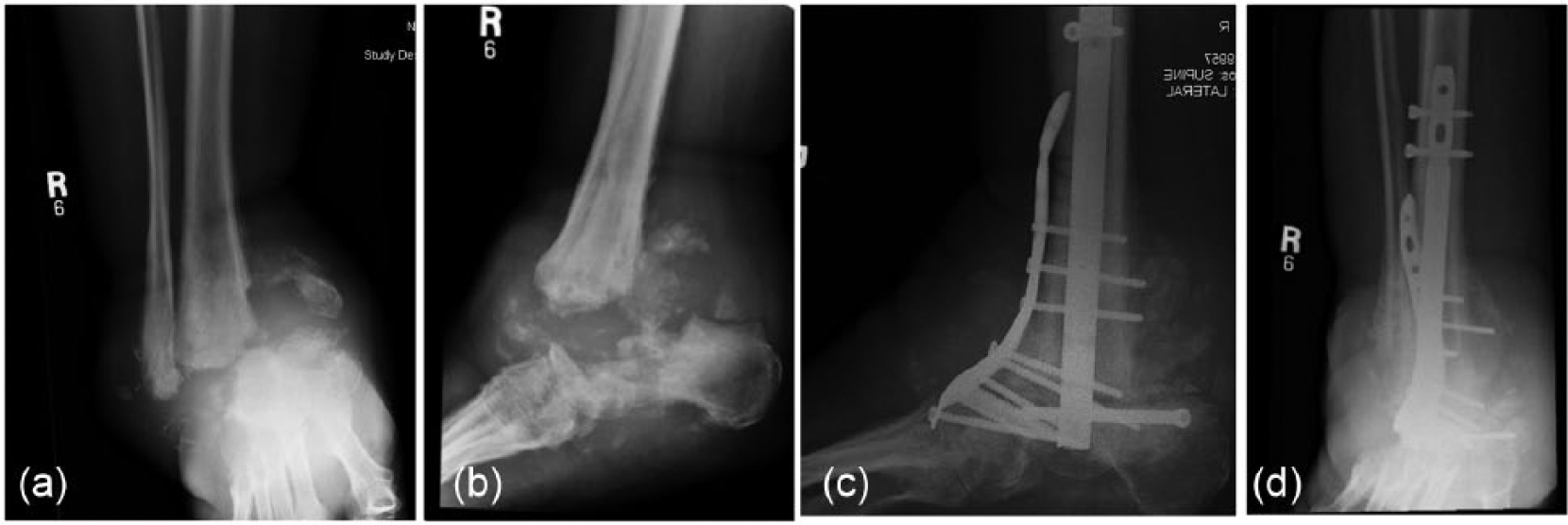
A 55-year-old man with insulin-dependent diabetes mellitus with an infected Charcot neuroarthropathy of the ankle referred for possible amputation. (a and b) Preoperative radiographs and (c and d) postoperative radiographs showing 6 months after arthrodesis following eradication of infection with debridement and antibiotic spacer.
Long-term immobilization is crucial for achieving union. In general, the duration of immobilization after arthrodesis for patients with CN is twice as long compared to nondiabetic patients. Postoperative treatment consists in casting and total nonweight bearing for at least 12–18 weeks and further partial weight bearing for 3–6 months until consolidation has been achieved. The patient is then managed with a bivalved AFO with rocker sole added to the foot plate until the use of custom footwear is possible.
External fixation
External fixation recently has gained popularity as a less invasive treatment of CN.74 –80 Potential advantages include single use in patients with ongoing ulcers and osteomyelitis and those with poor soft tissue envelope. Other indications include patients with severe bone loss, morbid obesity, osteopenia, and multiple comorbidities. Several authors have reported good to excellent results with external fixation techniques in patients who were not suited for internal fixation and otherwise may require amputation.74 –77
Small wire circular fixators may be used when there is an open wound or when bone loss preludes satisfactory open reduction. A study by Dalla Paola et al. revealed that in select patients, external fixation was an alternative to amputation. The study followed up 45 patients with CN; 39 of whom were healed after emergent surgery to drain acute infection with subsequent fixation for an average period of 25.7 weeks. Two patients in the cohort were treated with intramedullary nails, and in the remaining four patients, amputation resulted from uncontrolled infection. 80
Complications of surgery
Complications after Charcot foot surgeries include hardware failure, deep and superficial wound infection, wound dehiscence, pseudoarthrosis, instability, and amputation. Recurrent CN may occur in as many as 15% of patients treated surgically. Reported problems with external fixation include pin or wire tract infections, failure of hardware, stress fractures, osteomyelitis, and psychological difficulty.4,74,75,81 –83
Amputation surgery in the management of CN
The role of major amputation for the management of Charcot arthropathy has been decreased significantly in recent years as a result of improved surgical techniques and wound care management. In a retrospective review of CN deformity patients, Saltzman et al. 84 reported that amputation rate was 2.7%.
Amputation may be indicated in those with uncontrollable and overwhelming sepsis, severe peripheral arterial disease, failed reconstruction, and poor ambulatory potential.71,85 Limb salvage and surgical reconstruction although more technically difficult offer severe advantage over major amputation. These include better overall cost–benefit, lesser energy expenditure, and decreased chance of a contralateral amputation.86 –88 Prudent multidisciplinary evaluation should be made on a case-by-case basis to determine the risk–benefit ratio of reconstruction and salvage of the lower limb versus amputation.
Surgical correction versus accommodative bracing
High-level evidence is lacking to support the rationale of surgical correction over accommodative bracing as it relates health-related quality of life or functional impact of the disease in the management of patients with CN.
Accommodative bracing technique devices such as AFO or the custom-fabricated CROW avoid the surgical complications and define favorable outcomes based on limb preservation and avoidance of new ulcers. Advocates for surgical correction perceive these devices as cumbersome and not well tolerated by their patients. Additionally, earlier surgical correction may prevent late deformity which can be very challenging to accommodate with footwear and difficult to surgically reconstruct.
Prevention
While medical and surgical intervention is extremely important in the management of CN, patient education is the cornerstone of both treatment and preventive care. As with all diseases, well-informed patients have a greater understanding of how to best manage their disease. The importance of strict glucose control should be stressed in diabetic patients. Patients with diabetes should be referred to a comprehensive multidisciplinary program to improve outcomes.
Conclusion
CN is a serious and potentially limb-threatening lower extremity late complication of DM. A high index of suspicion for this disorder will often lead to early diagnosis. Early diagnosis and prompt treatment can prevent progressive deformity which may result in morbidity and amputation. The prerequisites of successful reconstructive surgery are correct timing, adequate fixation, and a longer postoperative nonweight-bearing period.
Footnotes
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
