Abstract
Background:
Direct skeletal attachment of limb prostheses is associated with high rate of transcutaneous infection and loosening of the fixture in the medullary canal prompting for careful assessment of various means for enhancing the skin-device and bone-device interface. The skin and bone integrated pylon system constitutes a technological platform for different modifications being evaluated previously.
Objectives:
The current study assessed the combination of nano-treatment skin and bone integrated pylon with its pre-seeding with dermal fibroblasts. We hypothesized that this combination will enhance cell interaction with skin and bone integrated pylon compared to nano-treatment and the fibroblast seeding when done separately.
Study design:
The feasibility and safety of in-bone implantation of the skin and bone integrated pylon with nanotubes was investigated in vitro and in vivo in the animal model.
Methods:
TiO2 nanotubes were fabricated on the skin and bone integrated pylon, and the fibroblasts taken from rabbit’s skin were cultured on the pylons before implantation.
Results:
The in vitro experiments demonstrated higher cellular density in the samples with a nanotubular surface than in the non-modified pylons used as control. There were no postoperative complications in any of the animals during the 6-month observation period. Subsequent scanning electron microscopy of the pylon extracted from the rabbit’s femur showed the stable contact between the pylon and soft tissues in comparison to control samples where the patchy fibrovascular ingrowth was detected.
Conclusion:
The promising results prompt further investigation of the integrative properties of the nanotextured skin and bone integrated pylon system seeded with dermal fibroblasts and its optimization for clinical application.
Clinical relevance
The study is devoted to the development of more safe and efficient technology of direct skeletal attachment of limb prostheses aimed in improving quality of life of people with amputations.
Background
The necessary condition for the success of direct skeletal attachment (DSA) of limb prostheses is the formation of a reliable interface between the implant and hosting tissues. Failure to create a suitable interface of the implant and skin leads to marsupialization and periprosthetic deep infections.1,2 It has been demonstrated that the roughness of the surface determines the degree of cell adhesion, as well as the tissue biocompatibility of the implants. 3 To date, several approaches have been introduced for modifying the implant surface with different bioactive coatings, including fibroblast growth factor-2-embedded apatite composites, 4 hydroxyapatite (HA) coatings,5,6 and with the electrochemical methods in various modifications.
Electrochemical anodization promotes the formation of a highly ordered nanostructure of nanotubes on the surface of the implant. Several studies demonstrated promising results relating to cellular interactions with the nanotubular implant’s surface, as compared to the smooth surface.7–9 Positive effect of nano-treatment on the bone-device interface was confirmed in previous studies.3,10–16 In the work of Ruckh et al., 17 it was demonstrated that nanotube arrays on the implant surface significantly enhance osteoblast cell adhesion, proliferation, and differentiation. Fewer reports while also with positive results are known for the effect of nano-treatment on the interface of a device with the skin.18,19
We previously developed a system for direct attachment of limb prostheses called the skin and bone integrated pylon (SBIP), in which in vitro and in vivo experiments were found to support an infection-free skin and bone interface.7,19–24 Recent study with the SBIP having nanotubular surface 7 demonstrated more consistent, diffuse fibrovascular ingrowth into the pores of the embedded portion of the device. Angio- and osteogenesis following the implantation of the SBIP seeded with autologous fibroblasts were evaluated in vivo. 25 Enhanced osseointegrative properties of the pylon were observed compared to the untreated porous titanium pylon. Greater cellular inhabitation within first 3 weeks of implantation compared to the unmodified implants was demonstrated.
As both independent studies suggested that seeding with fibroblasts and nano-treatment of the implants, if taken separately, were advantageous for the skin-device and skin–bone interface, the aim of the current pilot study was to evaluate the combined effect from the nano-treatment of the SBIP implants with their treatment with dermal fibroblasts. In this article, we present the results of in vitro tests and trials with rabbits.
Methods
Development of the SBIP with nanotubular surface
There are eight known implant systems for DSA, including the SBIP system, all of whose features are summarized in Pitkin. 26 The first DSA system was applied at the Rancho Los Amigos Hospital (RLAH) to three amputees in 1977. 27 Currently, three systems, Osseointegrated Prostheses for the Rehabilitation of Amputees (OPRA), 28 Endo-exo prosthetic system (EEFP), 29 and Intraosseous transcutaneous amputation prosthesis (ITAP), 30 have been used in human patients. The Percutaneous osseointegrated prostheses (POP) 31 and SBIP19,23 systems are being used in continuing pre-clinical and pilot animal studies, and the Alameda East Animal Hospital & BioMedtrix (AEAHBM) 32 and The University of Akron (UA) 33 systems are being used in pilot animal studies.
The principal innovation of the SBIP system and its distinction from existing systems for DSA is the total permeability of the pylon. This provides for deep ingrowth of the hosting tissues, both of the bone and of the skin. The permeability is achieved in a composite structure consisting of porous cladding and perforated inserts. 20 Specification for all modifications of the SBIP pylons, defining the four critical parameters of particle size, pore size, porosity, and volume fraction, is uniquely selected and protected by US Patent # 8,257,435. Durability, resistance to fatigue, safety, and efficiency of the SBIP specification have been confirmed in mechanical and animal studies.7,20,21,26,34–40
For the current study, porous titanium rods (0.3 cm diameter × 2 cm length) were sintered by ADMA Products Group, Hudson, OH, with Ti6–AL–4V ELI powders and using 0.3-cm-diameter cylindrical boron nitride molds (Payne Engineering & Fab. Co., Canton, MA, USA). The high-temperature sintering was conducted in vacuum using titanium powders sieved to (−80 to +200) mesh. Samples were sintered at 1090°C for 4 h which was above the beta transus temperature of 996°C. Before the sintering cycle (Vacuum Industries Super VII furnace (Centorr Vacuum Industries Inc., Nashua, NH, USA) under vacuum 3.333 × 10–3 Pa), three thin (0.05 cm diameter) titanium wires Ti6–AL–4V ELI (Small-Parts, Seattle, WA, USA) were inserted inside the molds. The rods had pore sizes 40–100 µm and porosity 45% ± 5% (Figure 1(a) to (c)).

SBIP sample images with magnification (a) 100×, (b) 500×, and (c) 1000× of SBIP pylon surface before nano-treatment (anodization). Images after nano-treatment (d) 1000×, (e) 10,000×, and (f) 65,000×.
Titanium nanotubular surface treatment was performed at the Colorado State University, Fort Collins, CO, using a simple anodization process 17 as being described elsewhere. 7 Prior to anodization, the rods were cleaned with soap, acetone, and isopropanol. A titanium foil as the anode and platinum foil as the cathode formed a two-electrode cell. The cell was introduced into an electrolyte solution, prepared with diethylene glycol (DEG, 99.7%), 2% hydrofluoric acid (HF, 48% solution), and 3% de-ionized water. The treatments were carried out at room temperature for 24 h at 60 V. After anodization, the samples were rinsed with isopropanol and dried with nitrogen gas. Crystallized substrates were obtained by annealing the anodized titania nanotube arrays at 530°C in an oxygen ambient environment for 3 h. The nanotube architecture was examined with field emission scanning electron microscope (SEM) JSM-6300 by JEOL (Paebody, MA). Prior to imaging at 15 kV, the substrates were coated with a 10-nm layer of gold. The fabricated nanotubular surfaces had pore diameters ranging from 50 to 250 nm (Figure 1(d) to (f)).
In vitro assessment of fibroblasts’ interaction with the SBIP system
In a series of in vitro experiments, samples of porous titanium pylons with an electrochemically modified surface were put in a nonpolymerized collagenous gel. The gel covered the surface of the pylon (there were nine samples in total). Pylons with a non-modified surface were used as controls (there were nine samples in total). Following incubation with the gel, the pylons were incubated in 6% CO2 and 37°C for full polymerization of the collagenous gel and were then transferred to Petri dishes seeded with dermal fibroblasts (Figure 2). The fibroblasts were obtained from rabbit derma, as described elsewhere.
24
The fibroblasts were taken from cells that had migrated from fragments of the rabbit’s skin that was obtained during surgery. The skin was cut into small pieces and placed in a Petri dish with a Dulbecco’s modified Eagle’s medium (DMEM) medium, supplemented with 10% fetal bovine serum (FBS), 2 mm
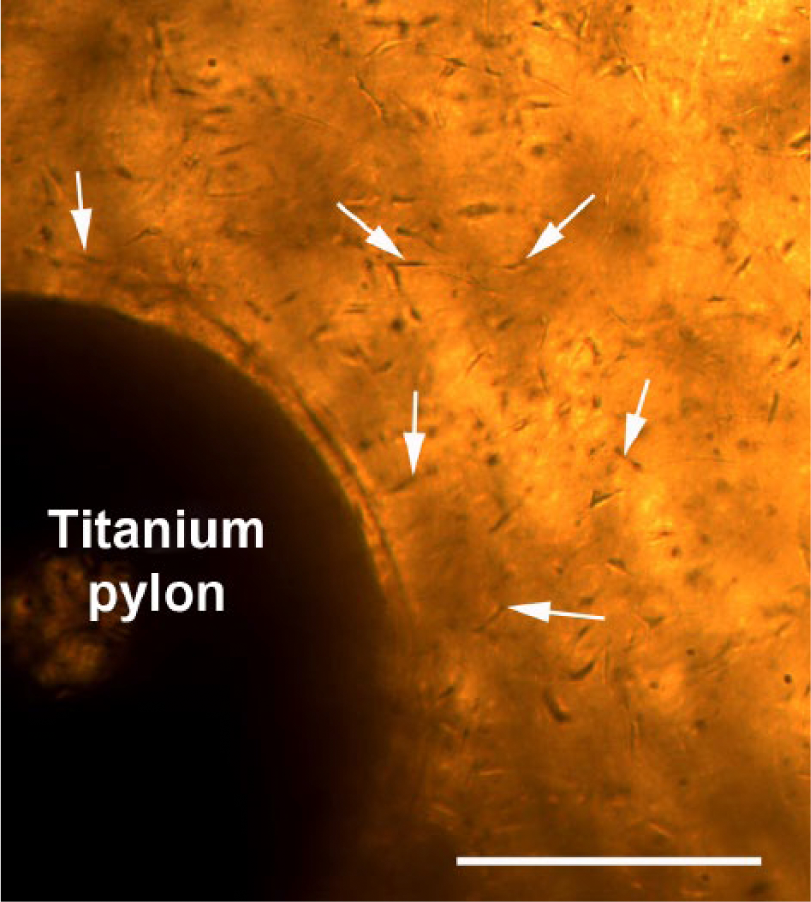
Light microscopy photograph of the SBIP in the collagen gel with autologous fibroblasts (shown with white solid arrows) on the 10th day of cultivation. Scale bar: 1 cm.
Titanium pylons were cultured with dermal fibroblasts for various time intervals (5, 10, and 15 days) and were then removed from the gel, fixed in 4% formaldehyde (Sigma, St. Louis, MO, USA). To assess the fibroblasts’ interaction with the pylon, three samples from each time interval were used (nine samples in total). Cell morphology on nanotube arrays was analyzed with SEM: JSM-6300 by JEOL (Paebody, MA).
In vivo animal experiments
The protocol of the study followed the institutional guidelines of the I.P. Pavlov State Medical University of St. Petersburg (St. Petersburg, Russia) in compliance with the US Department of Health and Human Services Guide for the Care and Use of Laboratory Animals. The protocol was submitted for approval to the local ethics committee and the ethical approval was obtained.
The feasibility of the in-bone implantation of the electrochemically modified SBIP system was assessed in New Zealand male rabbits. The animals (3.5–4 kg) were purchased from the animal nursery “Rappolovo” of the Russian Academy of Medical Sciences (RAMN) (St. Petersburg, Russia). Before the insertion of the implants into the rabbit’s femur, they were pretreated with fibroblasts in collagenous gel for 7 days. For anesthesia, we used intravenous injection of ketamine (10–50 mg/kg) and xylazine (1–3 mg/kg) mixture.
We performed unilateral above-knee amputation in six rabbits that were randomly divided into two groups (three animals each): (1) in experimental group, we implanted intramedullarily the modified titanium pylon with nanotubular surface and (2) in control group, we inserted pylon with non-modified surface.
In all animals, we performed above-knee amputation described elsewhere. 21 The anterior and posterior skin flaps at the level of bony resection were marked 4 cm above the knee with a marking pen. The skin and subcutaneous tissues were incised down to the fascia, provided that the anterior flap was 2.5 cm longer than the posterior flap. The greater saphenous vein was ligated and divided on the medial aspect of the thigh. Subcutaneous tissue and fascia were divided in line with the skin and reflected proximally. Femoral vessels and nerves were identified deep to the sartorius muscle. Posteriorly, the sciatic nerve was identified deep to the hamstrings on the adductor magnus. Nerves were gently pulled down from their muscular bed approximately 2 cm, ligated with nonabsorbable monofilament sutures, transected with a surgical knife and allowed to retract back to the muscle mass. Deep femoral vessels were divided; the artery and vein were sutured and ligated individually. Then, we cut the bone with a circular saw. Muscle flaps were approximated (myoplasty). The quadriceps and hamstrings muscles were myodesed to each other in covering the bony end of the femur. During the surgery, we implanted modified SBIP system with nanotubular surface immediately after the above-knee amputation. The position of the pylon inside the femoral canal was monitored using fluoroscopy. The pylon was press fit intramedullarily into the residual femoral bone, and the soft tissues and skin were closed over the residuum.
Radiographs (46 kV, 200 mA, 32 ms, Trophy N800 HF, Fujifilm 24 × 30 cm2 IP cassette type C, 1 m film-focus distance) were taken prior to surgery, 1 month after the implantation of the intraosseous component, and then monthly for up to 6 months. For radiography, rabbits were sedated by an intramuscular injection of xylazine (1–3 mg/kg) and ketamine (10–50 mg/kg) mixture.
Results
Attachment of fibroblasts to the nanotubular surface of the pylon in vitro
The fibroblasts’ morphology, cellular adhesion, migration, and proliferation on the SBIP with electrochemically modified surface were assessed with SEM. After 5 days of in vitro cultivation, the dermal fibroblasts on the pylons could be observed on the surface of the rods; they were adjacent to the pores covered with nanotubes by cellular bundles (Figure 3). Observed with SEM, the pattern of cellular growth on the surface of the pylons resembled the one when non-modified control titanium pylons were used (Figure 4). 24 As in that series of in vitro experiments, 24 cells were seen not only on the surface of the rods but also in the interior of the pylon. On the transverse sections of the titanium pylons, we could observe the fibroblast bundles throughout the section, thus proving that cells can migrate through the pores of the SBIP. Cellular density increased significantly after the 15th day of incubation (Figure 3). Well-spread fibroblasts formed large bundles that overlapped pores in the pylon and multilayer coatings (Figure 3). The cellular density on the control titanium samples with non-modified surface did not demonstrate any increase on the 15th day of cultivation.
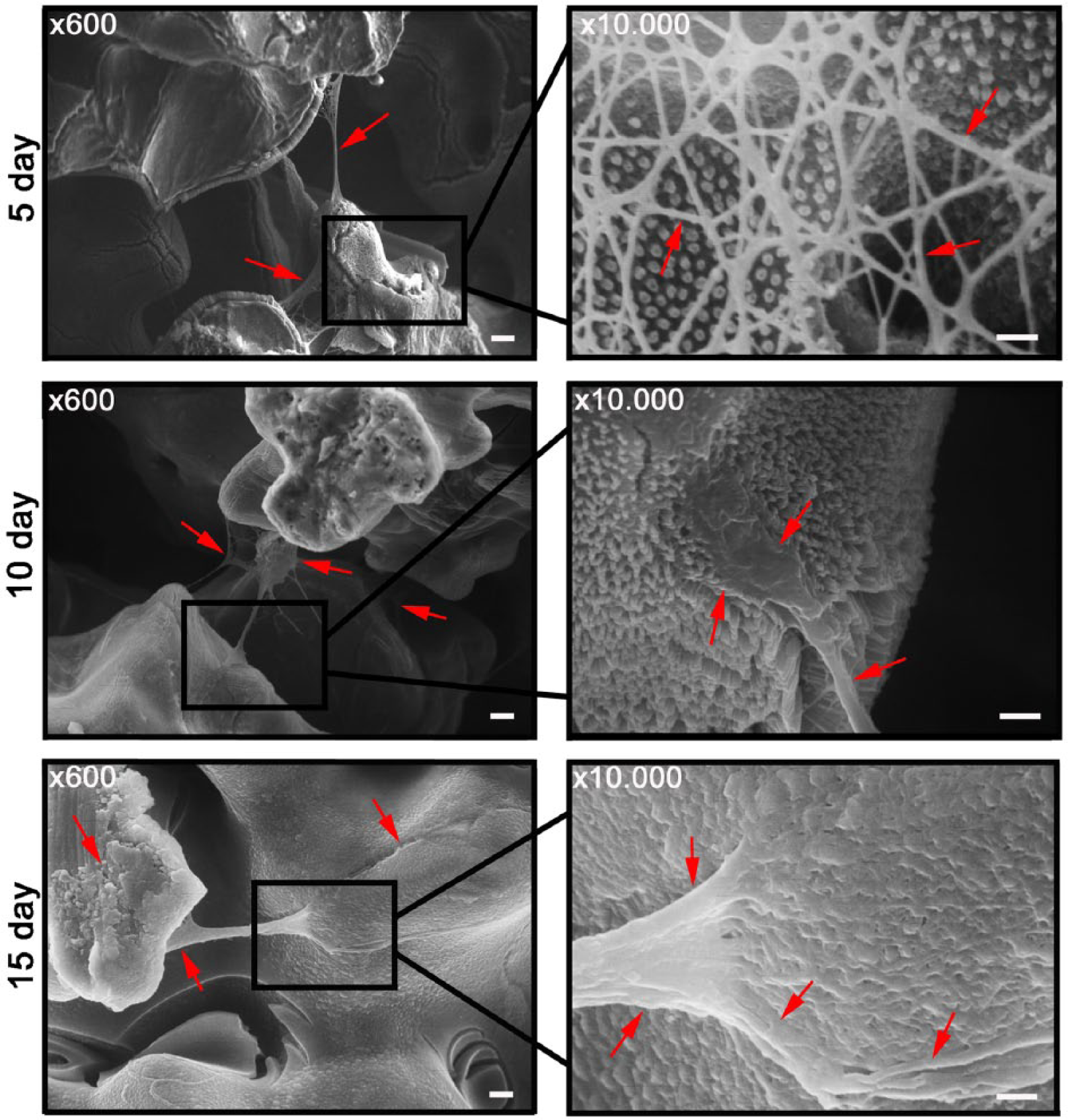
Scanning electron microscopy (SEM) of the SBIP with titanium oxide (TiO2) nanotubes on the 5th, 10th, and 15th day of incubation in vitro with dermal fibroblasts in collagen gel. On the fifth day of cultivation, fibroblasts formed bundles over the pores of the pylon (red arrows). On magnification, cells were seen attached to nanotubes by forming filopodia (red solid arrows). On the 10th day, fibroblasts formed thick cellular bundles with firm attachment to the nanotubes (red arrows). On the 15th day, cells overlapped the pores in multilayer coatings (red arrows) and nanotextured surface of the pylon could barely be detected. Scale bar for the left column: 10 µm and for the right column: 1 µm.
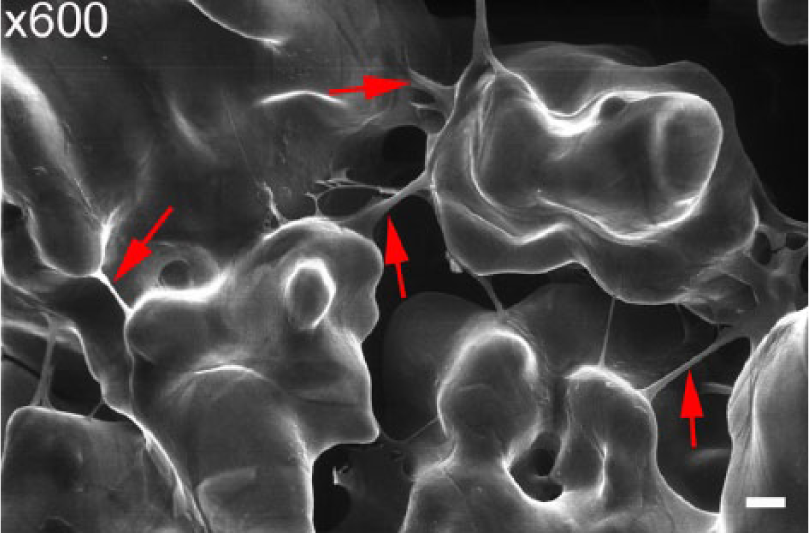
Scanning electronic microscopy of the non-modified SBIP with dermal fibroblasts on the fifth day of cultivation in vitro. Cells formed bundles over the pores of the pylon (shown by red solid arrows). Scale bar: 10 µm.
Feasibility of in-bone implantation of the SBIP system with a nanotubular surface
The intraoperational photograph of the press fit pylon with nanotextured surface is shown in Figure 5. There were no postoperative complications in any of the animals after surgery, nor during the follow-up period of 6 months. There were no clinical signs of infection during the observation period, and all animals behaved normally. For a deep infection control, we performed anterio-posterior (AP) and lateral radiographs before the operation and every month after surgery for 6 months (seven radiographs in total). To evaluate the bone infection, we applied parameters of the scoring system of Schmidmaier et al., 41 which includes the following parameters: (1) periosteal reaction, (2) osteolysis, (3) soft-tissue swelling, (4) deformity, (5) general impression or destruction, (6) loosening of the prosthesis, and (7) sequestrum formation.
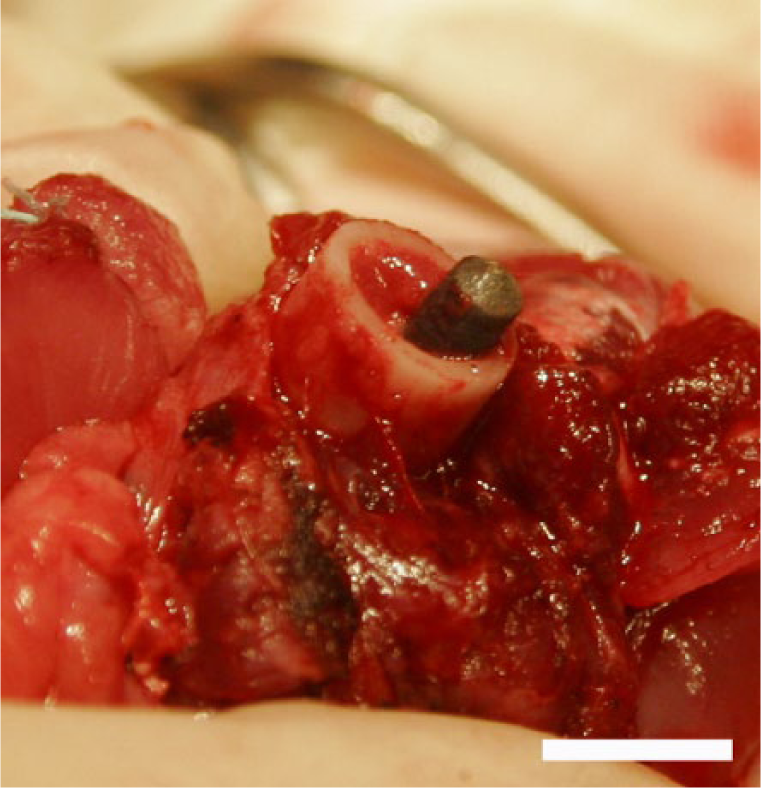
Intraoperative photograph of the SBIP with TiO2 nanotubes implanted into the femur after the above-knee amputation (animal 2). Prior to insertion into the femur, the pylon was pretreated with autologous rabbit’s fibroblasts in collagenic gel for 7 days. Scale bar: 1 cm.
On the postoperational radiographs, we did not observe any signs of the osteomyelitis. We did not observe osseous changes on the radiographs during the entire period of observation in the rabbits. One such radiograph, presented as an example from animal 2 is shown in Figure 6(a). At the end of the follow-up period, we extracted the pylon from the femur and analyzed with SEM the following surfaces of the SBIP system (Figure 6): (a) contact between the pylon and soft tissues (Figure 6(b)), (b) cross section of the pylon (Figure 6(c)), and (c) contact between the pylon and the inner surface of the femur canal (Figure 6(d)). After 6 months, we could observe reliable contact between the pylon and connective tissue (Figure 6(b)).

Animal 2 with intramedullary inserted SBIP system with TiO2 nanotubes 6 months after surgery. (a) Radiograph of the in-bone implanted SBIP. Scale bar: 1 cm. (b) Scanning electron microscopy (SEM) of the extracted pylon. Presented is the surface of the pylon in contact with soft tissues of the stump. Scale bar: 10 µm. (c) SEM of the pylon cross section. The dramatic ingrowth of the soft tissues inside the pores of the pylon is observed. Scale bar: 10 µm. (d) SEM of the surface of pylon in contact with bone tissues. All of the area is covered with tissues. Scale bar: 10 µm.
On the cross section of the titanium rod, we observed ingrowth of the soft tissues into the pores of the pylon throughout the cross section (Figure 6(c)). No zones could be found where the pylon was not in contact with soft tissues. Also, when we analyzed the surface on the pylon that was in contact with the inner surface of the femur, we observed reliable contact of the nanotubular surface with tissues (Figure 6(d)). Subsequent analysis of the radiographs demonstrated that the pylon did not migrate within the femur canal in any of the animals, thus proving the integration of the SBIP system with surrounding tissues.
We also assessed the SEM images of the pylon’s non-modified surface interaction with soft tissues (Figure 7). After 6 months of surgery, we extracted the pylon from the femur, and as in experimental group of animals, we analyzed the region of the pylon in contact with soft tissues (distal end), with bone (proximal end), and the cross section. There was no difference in the soft-tissue ingrowth in the distal end between modified and non-modified pylons. However, when we compared the surface being in contact with bone, we observed patchy fibrovascular ingrowth in control titanium samples (Figure 7(c)). On the cross sections of the control rods, we also observed the regions of the pylon that were not covered by the soft tissues.
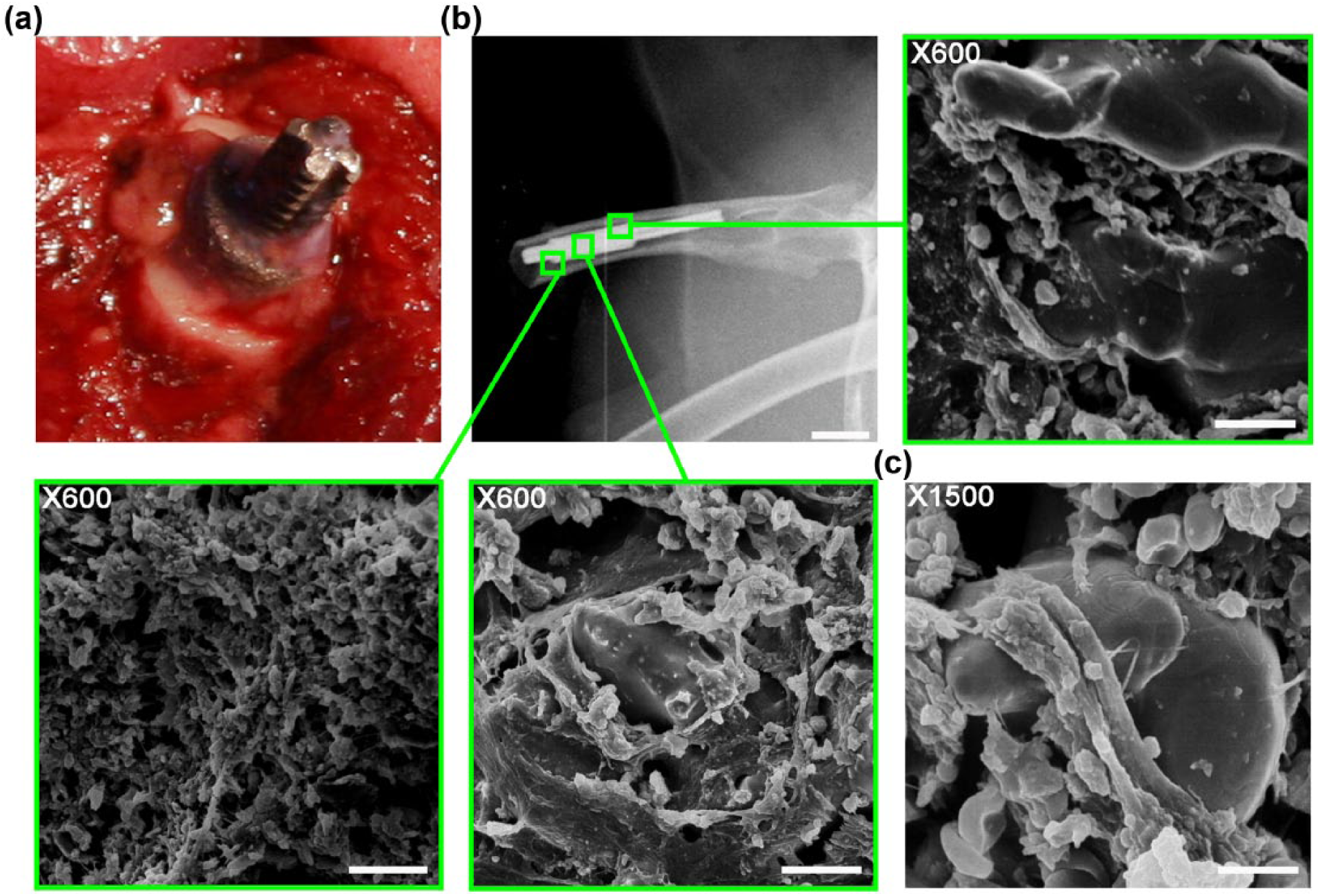
Animal 3 with intramedullary inserted SBIP system without nano-surface 6 months after surgery. (a) Intraoperative photograph of in-bone insertion of the pylon. (b) Radiograph of the in-bone implanted SBIP (scale bar: 1 cm) with subsequent scanning electron microscopy (SEM) of the extracted pylon. Presented is the surface of the pylon in contact with soft tissues (distal end), cross section of the pylon, and surface in contact with bone (proximal end). Scale bar: 10 µm. (c) Magnified SEM image of the pylon’s cross section with patchy fibrovascular ingrowth. Scale bar: 10 µm.
Discussion
Positive effect on osseointegration following nano-treatment of the surface of the solid implants has been reported in many studies. Lesser data are available on osseointegration with the nano-treated porous scaffolds. In a previous study, 42 such scaffolds were fabricated using both nano-HA powder (20 nm average particle size) and micro HA powder. The osteoblast attachment and proliferation was greater on the nano-HA scaffolds compared to similarly processed micro HA scaffolds 5 days after seeding.
Current in vivo study demonstrated the feasibility and safety of in-bone implantation of the titanium pylon with nanotubular surface pretreated with fibroblasts in collagenous gel. The rabbits did not experience any postoperative complications (i.e. implant loosening, deep infection, bone fractions, etc.) during the entire 6 months observation period. Moreover, we observed that the SBIP reliably integrated with the surrounding tissues, thereby providing stability to the pylon in the medullary canal (Figure 6). The cross section of the pylon clearly demonstrated the ingrowth of the soft tissues into the pores of the titanium, which was similar in nature to our previous experiments with non-modified pylons.21,43,44 With the non-modified pylons (control), following 6 months after surgery, we observed the patchy fibrovascular ingrowth (Figure 7). These data are in line with recent results of Farrell et al., 7 when the authors assessed the skin ingrowth in pylons with nanotubular surface in rats. The authors reported that the implants with nanotubular surface had more consistent and diffuse soft tissues attachment in comparison with the pylons without nano-surface, although the period of postoperative observation was limited to 6 weeks. 7 In our study, we extended the period of long-term implantation to 6 months and confirmed the better tissue ingrowth in experimental group of the nanotextured pylons.
The in vitro analysis of cells’ interaction with titanium rods demonstrated that electrochemical modification of the pylon surface with nanotubes significantly increased the cellular density when compared to the non-modified rods. On the 15th day of co-incubation of fibroblasts with titanium rods, we observed multilayer coatings on the pylon (Figure 3). The fibroblasts’ cellular coatings on the nanotubular surface were denser in comparison to the non-modified pylons from our previous observations. 24
In our study, we observed a formation of a confluent fibroblast monolayer on nanotubes, 130–140 nm in diameter. The presented data are in accord with the results by Oh et al., 45 when it was demonstrated that osteogenic differentiation of human mesenchymal stem cells (MSCs) was highest on the 100-nm nanotubes. At the same time, the rates of adhesion were maximal at 30 nm. Wang et al. 46 investigated the effects of TiO2 nanotubes with different diameters (30, 70, and 100 nm) on the biological attachment mechanism of implants to bone in minipigs by studying the gene expression and bone formation around the implants. The authors reported a significant increase in bone-implant contact (BIC) and gene expression (i.e. alkaline phosphatase (ALP), osterix (Osx), collagen-I (Col-I), and tartrate-resistant acid phosphatase (TRAP)) in bone attached to implants with TiO2 nanotubes, especially with 70 nm diameter. Probably, the processes of cell adhesion, migration, proliferation, and differentiation depend not only on the size of the pores of the nanotubes but also on the particular cell biology and the chemical modifications of nanotubes. Thus, Sul 3 demonstrated the importance of the roughness of the pylon surface for the proper cell adhesion over smoother surfaces. The author proved the superior bone response from fluorinated TiO2 nanotube implants, despite their low roughness values. In experiments in vivo on rabbits, the new bone formation in case of nanotubes was increased from 57.5% to 65.5%, whereas the osseointegration strength was also elevated (from 29 to 41 N cm). 3 In the studies by various groups, the importance was emphasized on the chemical modifications of nanotubes, especially by including fluoride (F) and strontium (Sr), and their influence on bone formation.3,14,47
Conclusion
In summary, in the presented work, we demonstrated the feasibility of DSA of the SBIP system with nanotubular surface in vitro and in animals. The promising results beg further investigation of the integrative properties of the nanotextured SBIP system and its optimization for clinical applications.
Limitations of the current study include a small number of animals, and the fact that the implantation was performed subcutaneously. The authors will address these limitations in their further research.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This study was funded in part by Eunice Kennedy Shriver National Institute of Child Health and Human Development, NIH (grant number: R44HD057492).
