Abstract
Background:
Hospital-based studies have shown that mortality rates in individuals with diabetic foot ulcers are about twice those observed in individuals with diabetes without foot ulcers.
Objective:
To assess the etiology and management of chronic diabetic foot ulcers.
Study design:
Literature review.
Methods:
Systematic review of the literature discussing management of diabetic foot ulcers. Since there were only a few randomized controlled trials on this topic, articles were selected to attempt to be comprehensive rather than a formal assessment of study quality.
Results:
Chronic nonhealing foot ulcers occur in approximately 15% of patients with diabetes. Many factors contribute to impaired diabetic wound healing. Risk factors include peripheral neuropathy, peripheral arterial disease, limited joint mobility, foot deformities, abnormal foot pressures, minor trauma, a history of ulceration or amputation, and impaired visual acuity. With the current treatment for nonhealing diabetic foot ulcers, a significant number of patients require amputation.
Conclusion:
Diabetic foot ulcers are optimally managed by a multidisciplinary integrated team. Offloading and preventative management are important. Dressings play an adjunctive role. There is a critical need to develop novel treatments to improve healing of diabetic foot ulcers. The goal is to have wounds heal and remain healed.
Clinical relevance
Diabetic neuropathy and peripheral arterial disease are major factors involved in a diabetic foot ulcer. Despite current treatment modalities for nonhealing diabetic foot ulcers, there are a significant number of patients who require amputations. No known therapy will be effective without concomitant management of ischemia, infection, and adequate offloading.
Introduction
Chronic nonhealing neuropathic foot ulcers occur in approximately 15% of patients with diabetes. 1 In 2011, there were an estimated 366,000,000 adults with diabetes. Worldwide global projections indicate that this figure will increase to 552,000,000 by 2030. 2 In North America and Europe, the number of adults with diabetes is expected to increase by 42.4% and 20%, respectively.3,4 A major increase is also predicted in Africa, with the number of adults with diabetes expected to increase by 98.1% from 2010 to 2030.3,4 The main factors responsible for the increase in patients with diabetes are aging of the population and lifestyle changes.3,5 The dramatic increase in the worldwide prevalence of diabetes has resulted in a rise in diabetes-related complications.
Persons with diabetes mellitus have a 15%–25% chance of developing a diabetic foot ulcer during their lifetime and a 50%–70% recurrence rate over the ensuing 5 years.6–8 Foot lesions carry high morbidity and mortality and represent the most common cause of hospitalization in patients with diabetes. While diabetes affects more than 8% of the US population, it has been implicated in over 60% of all nontraumatic lower extremity amputations. 9
The risk of a person with diabetes undergoing a lower extremity amputation is estimated to be 23 times that of a person without diabetes. 10 Greater than 85% of major amputations in patients with diabetes are preceded by foot ulceration.8,11–13 Following amputation, 50% of patients either die or lose the contralateral limb within 5 years. 14 The early recognition of the high-risk foot and timely treatment may prevent foot ulcers, save limbs, potentially save lives, and improve patient quality of life. 15 A strategy which includes prevention, patient and staff education, multidisciplinary treatment of foot ulcers, and close monitoring can reduce amputation rates by 49%–85%. As a result, several countries and organizations (the World Health Organization and the International Diabetes Federation) have set goals to reduce the rate of amputation by up to 50%. 16 Unfortunately, despite evidence to suggest that targeted interventions resulting from multidisciplinary care can reduce limb loss, progress to date has been slow. 17
Diabetes and impaired healing
With the increasing prevalence of diabetes around the world, understanding the mechanisms responsible for poor diabetic wound healing is an important public health issue. The increased risk of chronic ulcer formation stems from disruption of the complex process of wound healing by the pathophysiological abnormalities associated with diabetes.18–20 Wound healing is a dynamic interactive process involving coagulation, inflammation, granulation tissue formation, and tissue remodeling.18,21 Analysis of human diabetic ulcers has revealed the differential expression of growth factors, chemokines, cytokines, and their receptors, which are critical to several phases of the normal wound healing process.22,23 Matrix-metalloproteinases involved in tissue remodeling are also differentially expressed in chronic wounds, causing the dysfunctional breakdown of the extracellular matrix.24,25 Macrophages isolated from the wounds of diabetic mice have exhibited a decreased ability to remove dead cells, resulting in a prolonged inflammatory response.26,27
Tumor necrosis factor-α (TNF-α), a potent proinflammatory cytokine, contributes to the stimulation of fibroblasts and keratinocytes, the expression of growth factors, and upregulation of antimicrobial defenses. 28 In normal wound healing, the highest levels of TNF-α are seen from 12 to 24 h after wounding. 29 After completing the proliferation phase of wound healing, TNF-α returns to basal levels. During the early phase of wound repair, it is predominantly expressed in polymorphonuclear leukocytes and later by macrophages. It is also expressed in the hyperprolific epithelium at the wound edge. In diabetic wound healing, impaired fibroblast proliferation has been linked to increased levels of TNF-α. 30 The high levels of TNF-α inhibit angiogenesis, proliferation, differentiation, migration, and increase apoptosis. TNF-α inhibition attenuates the impact of diabetes-enhanced TNF-α which offers potentially new therapeutic avenues for treatment of chronic diabetic foot ulcers. 31
Diabetes is characterized by significantly increased cross-linking and nonenzymatic glycation of collagen and elevated levels of advanced glycation end products (AGEs).32,33 Blocking the receptor for advanced glycation end products (RAGEs) restores the wound healing properties of diabetic (Db/Db) mice. 34 Hyperglycemic animals have been shown to have significantly higher concentrations of glycated collagen and higher levels of collagenase activity. 35 Furthermore, diabetic (Db/Db) mice have also been shown to have a prolonged inflammatory phase with sustained expression of the inflammatory cytokines. 36 Diabetic skin (both murine and human cadaveric) is biomechanically inferior to nondiabetic skin. 37 These results suggest the defect in tissue integrity may be inherent to diabetic skin at baseline.
Treatments that enhance diabetic wound healing are often associated with reducing inflammatory cytokines in the diabetic wound environment. 38 An important yet still unanswered question during diabetic wound healing is how a high-glucose environment affects chemotactic activity.
Diabetic foot ulcers
Diabetic neuropathy and peripheral arterial disease (PAD) are usually the major factors involved in diabetic foot ulcers. These two factors may act alone, together, or in combination with other conditions, such as microvascular disease, biomechanical abnormalities, limited joint mobility, and increase susceptibility to infection.39,40
For every 1% increase in hemoglobin A1c, there is a corresponding 26% risk of PAD. 41
Neuropathy is a common complication of diabetes. This is characterized by a progressive loss of peripheral nerve fibers caused by decreased blood flow and high glycemic levels.42,43 The duration and intensity of hyperglycemia strongly influences the severity of the neuropathy. 42 Neuropathy leads to an insensate and sometimes deformed foot, often with an abnormal walking pattern. In people with neuropathy, minor trauma (ill-fitting shoes, walking barefoot, or an acute injury) can precipitate a chronic ulcer.
Diabetes may cause stiffening of capsular structures and ligaments. Decreased ankle, subtalar, and first metatarsal phalangeal (MTP) joint mobility results in higher focal plantar pressures with increased risk of ulceration. Loss of sensation, foot deformities (Figure 1(a) and (b)), and limited joint mobility can result in abnormal biomechanical loading of the foot. 16 Whatever the primary cause, whenever patients continue walking on the insensate foot, subsequent healing is impaired (Figure 2).
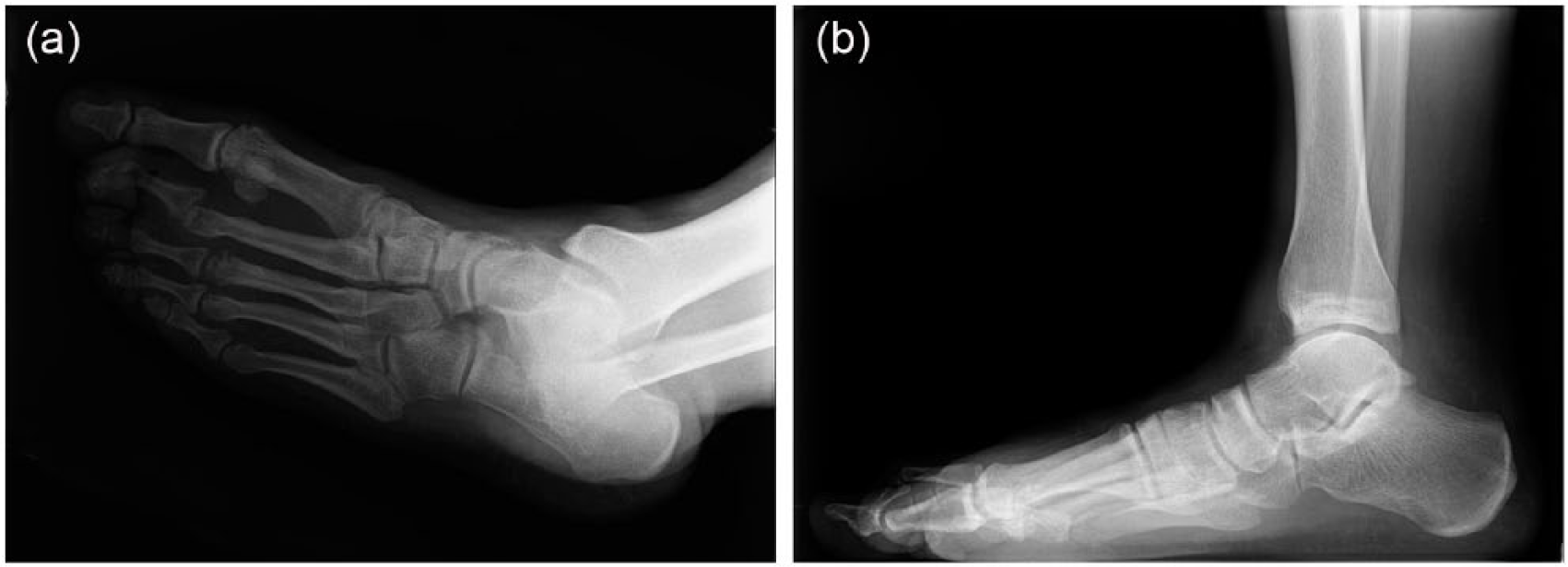
(a) Dislocation of the second toe at the MTP joint and (b) standing view showing prominence of the dislocated second MTP joint.
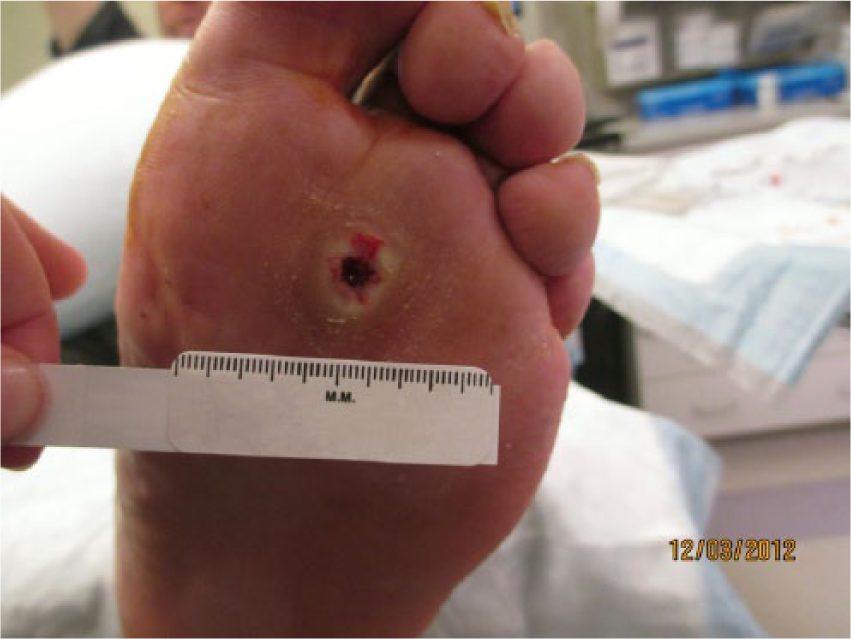
Chronic nonhealing wound overlying prominent second metatarsal head following debridement (patient’s X-rays shown in Figure 1(a) and (b)).
Imaging
Plain radiographs help identify foreign bodies, bone deformities, gas in the tissues, or evidence of osteomyelitis. Plain films lack sensitivity for detecting early osteomyelitis, especially in the presence of osteopenia associated with neuropathy, since 30%–50% of bone loss is required to produce detectable changes. 44 It is helpful if radiographic findings are positive, but if these are negative, it does not exclude the diagnosis of osteomyelitis. Despite the poor predictive value of a normal radiograph, it is recommended that a plain radiograph be taken in all patients presenting with foot ulcers. 45 Plain films should be repeated in 2–3 weeks as radiographic changes of osteomyelitis may be delayed.
Indium plus bone scan and magnetic resonance imaging (MRI) are used when the diagnosis is equivocal to help gauge the extent of bone and soft tissue infection. An MRI might be needed to better define the presence of bone or deep soft tissue infection.46,47
MRI scans have the advantage over radionucleotide bone scans of being able to accurately define the anatomic location and extent of inflammatory changes or any soft tissue infection in the foot, including sinus tracts, deep tissue necrosis, or abscesses. A meta-analysis comparing the performance of MRI scans to bone pathology for the diagnosis of osteomyelitis showed a sensitivity of 90% and specificity of 79%. 48 The lower specificity in diabetes is usually attributable to the difficulty of distinguishing osteomyelitis from other causes of bone edema, in particular Charcot-neuroarthropathy. 49
Antimicrobial therapy
All open wounds are colonized with microorganisms. The generally accepted clinical definition of infection is the presence of purulent secretions or at least two signs or symptoms of inflammation (erythema, warmth, tenderness, pain, and induration). 50 Patients with chronic ulcers or those who have recently received antibiotic treatment often have a polymicrobial infection with aerobic gram-negative bacilli and gram-positive cocci. 51
The aim of antimicrobial therapy is to cure the infection, not to heal the wound. Extended antibiotic treatment increases the risk of antibiotic resistance and drug-related toxic effects. Antibiotic treatment without offloading a plantar wound is unlikely to result in ulcer healing. There is no role for routine suppressive antibiotics. Antibiotics should not be used unless a wound infection is present.
Osteomyelitis
Approaches to the management of osteomyelitis in diabetic foot ulcers vary widely from center to center and country to country. At present, there are a few studies with robust data to guide clinicians in the choice, duration, or route of antimicrobial therapy.52,53 Osteomyelitis is probably present if the bone is visible or palpable by probing. Other clinical clues to the presence of osteomyelitis include general malaise, local pain, worsening diabetic control, abnormal friable exuberant granulation tissue in the wound, and laboratory findings of an elevated sedimentation rate or C-reactive protein.
Although there is no clear consensus as to whether management should be primarily medical (antibiotics) or surgical, 52 when possible, bone infection is best treated by surgical resection of the infected and necrotic bone.54,55 A potential complication of surgical management is altered foot architecture. Careful offloading is important in the perioperative period to avoid skin breakdown at a new high-pressure site (Figure 3(a)–(c)). The highest risk of recurrent ulceration occurs after surgery to the first metatarsal head (28%), and the lowest risk occurs to the fifth metatarsal head (8%). 56 Surgical management allows shorter duration of antibiotic treatment and reduces the likelihood of bacterial resistance to antibiotics.
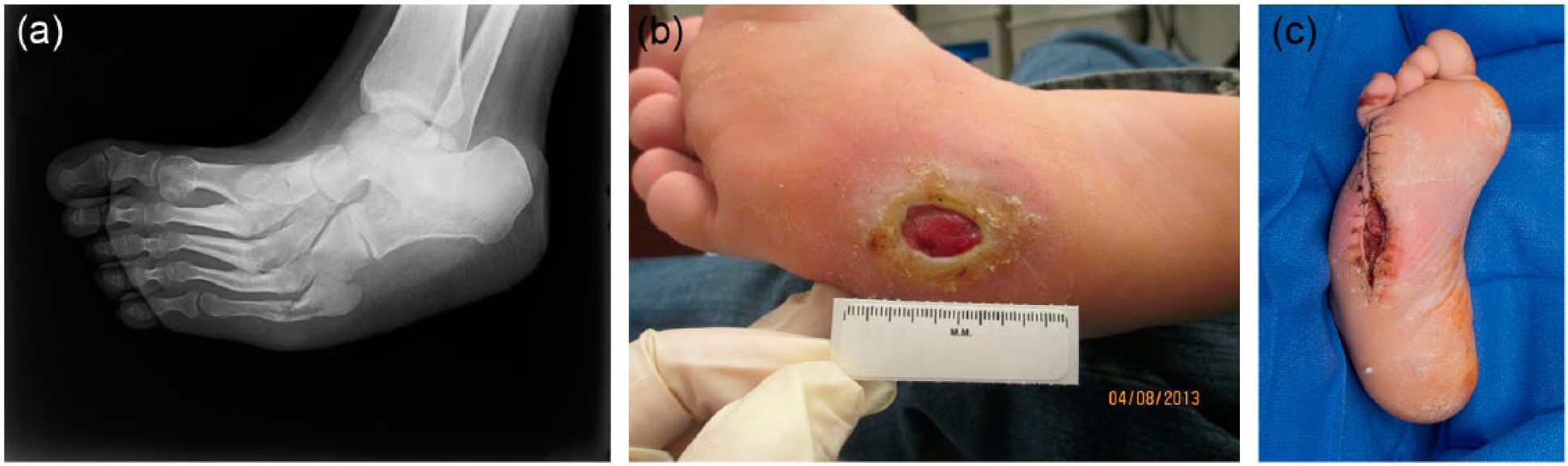
(a) Chronic fracture deformities of the proximal right second through fourth metatarsals. Ununited fracture of the proximal right fifth metatarsal with overlying wound. (b) Neuropathic wound right lateral midfoot overlying ununited fracture of the proximal right fifth metatarsal (Figure 3(a)). Note: Periulcer callus; undermining; pale, flat tissue in the wound base; slough around the edge of the wound. (c) Right foot 6 weeks following fifth ray amputation despite casting and instructions to offload the foot. The patient ultimately required a transtibial amputation 7 months later.
It is important to review the pros and cons of each approach with patients on an individual basis. 52 Retrospective studies suggest that long-term treatment (at least 4–6 weeks) with drugs that penetrate well into bone (fluoroquinolones) can produce a remission of infection. 54
Risk factor modification
Risk factor identification is fundamental for effective preventative management of the diabetic foot. Hospital-based studies have shown that mortality rates in individuals with diabetic foot ulcers are about twice those observed in individuals with diabetes without foot ulcers. 57 Nearly all patients with lower limb ulcers can benefit from evidence-based therapy aimed at reducing the risk of atherosclerotic vascular disease. Consistently using evidence-based wound risk assessment coupled with standardized wound interventions may result in fewer wounds and improved therapeutic outcomes. Unfortunately, the quality of the evidence is low. The Cochrane reviews have attempted to answer many treatment questions with meta-analysis and systematic evaluation of the literature. These reviews indicate that even when combining these small studies, the results do not provide solid conclusions regarding the efficacy of current wound treatment options. Clinicians rely on consensus views and personal experience to guide wound treatment options. Most randomized control trials (RCTs) evaluate one specific intervention (often topical), resulting in many common practices being untested or assumed. In many wound care RCTs, the authors report results from a small number of total subjects (often less than 100) with limited duration of subsequent follow-up.58,59
Adequate nutrition supports wound healing. Decreased serum albumin is associated with poor wound healing and poor clinical outcomes. 60 Serum pre-albumin provides a more sensitive indicator of protein status in acute stages of malnutrition and helps evaluate the adequacy of nutritional therapy. 61 Clinically, significant malnutrition is defined as a pre-albumin of less than 15 mg/dL or a decrease in one’s total body weight by more than 5% experienced within 1 month. If a patient develops malnutrition, they should eat protein in the amount of 1.2–1.5 gm/kg of body weight every day. 62
All individuals with diabetes should have a thorough foot examination at least once per year to identify high-risk foot conditions. This examination should assess protective sensation, foot structure and biomechanics, vascular status, and skin integrity. Clinicians should perform a comprehensive physical examination on all patients with ulcers, giving particular attention to the wound characteristics, vascular status, and overall health of the patient. The wound site should be evaluated on every visit, noting wound size and characteristics. 63 Assessing the wound margins (callous, undermining) provides clues about offloading.
Patients at risk should understand the implications of loss of protective sensation, the importance of foot monitoring on a daily basis, the importance of proper care of the foot (nail and skin care), and the importance of always wearing protective footwear. Patients with evidence of increased plantar pressure (erythema, warmth, callus, or measured pressure) should use footwear that provides soft tissue supplementation and redistributes pressure. Patients with neuropathy should be advised to break in shoes gradually to minimize the formation of blisters and ulcers.
Thermography
The use of temperature is a quantifiable, reproducible measurement of inflammation. One study evaluated the effectiveness of at-home infrared temperature monitoring as a preventative tool in patients with diabetes at high risk of lower extremity ulceration or amputation. The study group patients had temperatures measured in the morning and evening, were instructed to reduce their activity, and contact the study nurse when temperatures increased. This group had significantly fewer foot complications owing to their early warning of inflammation and tissue injury. 64
Offloading
The treatment of diabetic foot ulcers includes pressure relief (offloading) by limiting walking, wearing special footwear, or both.
Patients should be counseled never to walk in shoes that contributed to a foot ulcer. 65 The most compelling evidence that offloading accelerates ulcer healing comes from studies using total contact casting for healing of noninfected neuropathic ulcers. Neuropathic ulcers that resisted healing for many months or years typically heal in about 6 weeks in a total contact cast. 66
Debridement and biofilm disruption
Wound debridement helps to correct cellular and molecular abnormalities. The fundamental goal of basic wound care involves keeping the wound base free of nonviable tissue to facilitate wound healing. More recently, the treatment of diabetic foot ulcerations has focused on biofilm reduction. 67 Biofilm is defined as bacterial and/or fungal colonies populating the surface of wounds that are highly resistant to antibiotic treatment.68,69 Biofilm formation is a multistage process in which microbial cells adhere to the surface (initially reversible attachment). The subsequent production of an extracellular matrix containing polysaccharides, proteins, and DNA results in a firmer attachment. 70 Biofilm has been identified in up to 60% of chronic wounds (greater than 30 days) versus 6% of acute wounds. 71 Biofilm reduction, eradication, and inhibition are essential considerations in the treatment of chronic diabetic foot wounds. 67
Debridement is not the only method of biofilm disruption. The use of low-frequency ultrasound has been beneficial. Ultrasound therapy is typically painless. It requires multiple treatments (up to five times per week) for 4–6 weeks. Although direct measure of biofilm disruption has not been fully elucidated utilizing ultrasound technology, biofilm disruption may contribute to the reported increase in healing of chronic wounds. 72
Topical therapies
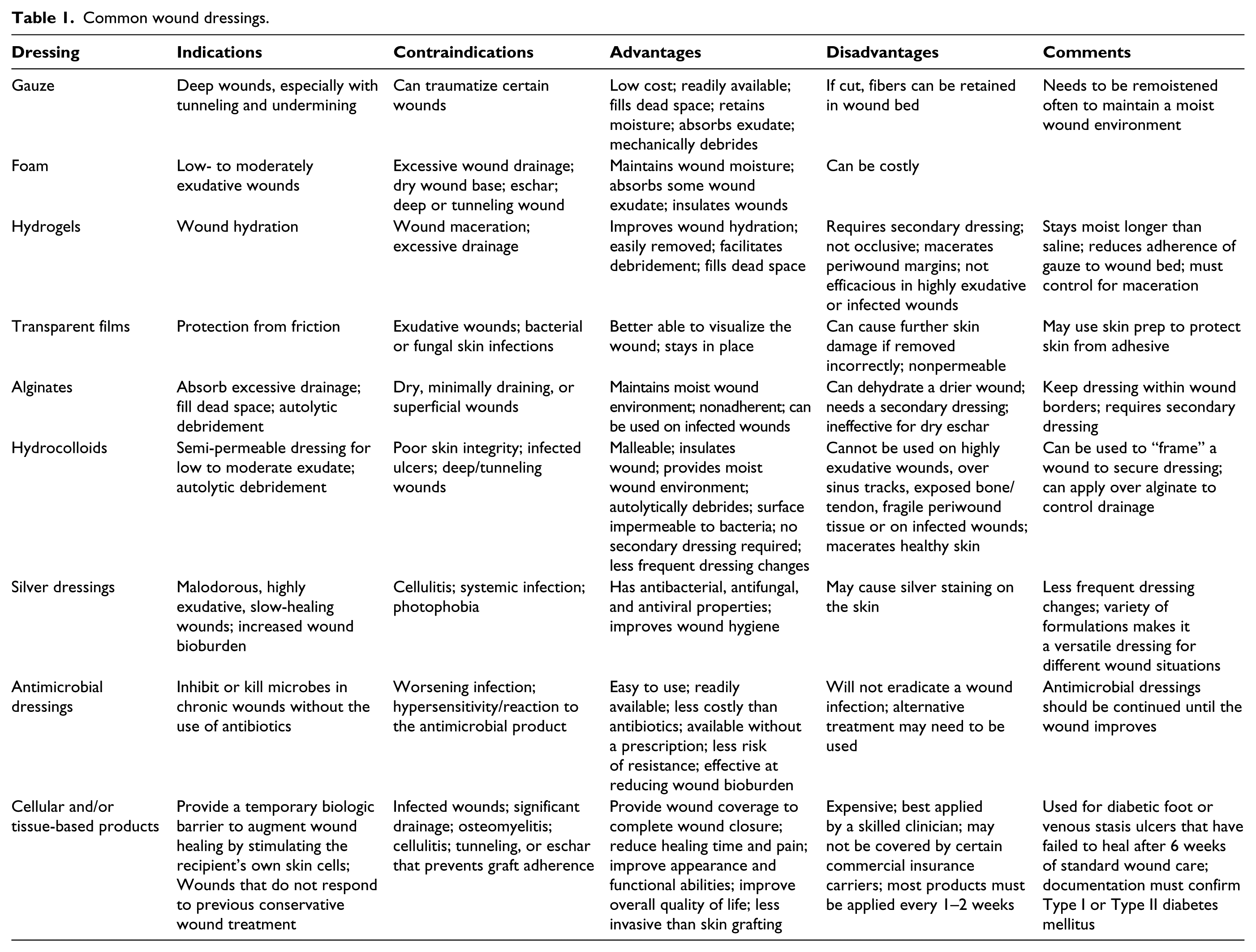
Dressings play an adjunctive role in the treatment of diabetic wounds; however, this role can be limited, and dressings should never be considered the sole treatment modality. Clinicians typically use three types of dressings: hydrating; debriding; and antimicrobial (Table 1). Clinicians should always consider cost when choosing wound care products. Wound characteristics such as location, size, depth, and presence of drainage may influence the topical agent chosen.
Common wound dressings.
Topical antimicrobials (ointments, creams, and gels) have long been utilized for the treatment of wounds. Despite their widespread use, there is a paucity of evidence to support the use of topical therapies for diabetic foot ulcers. 73 Topical antiseptics can impair wound healing. Dressings containing silver or iodine appear to be safe and possibly useful. 74
Cellular and/or tissue-based products
Cellular and/or tissue-based products (CTP) include a number of products derived from human, animal, and synthetic tissues that have been manufactured, cleaned, or otherwise altered. CTP products are typically indicated when split thickness skin graft, local or free tissue flaps, or primary closures are not feasible options. These products work to awaken and activate senescent cells in the chronic diabetic wound. Some CTP products contain viable cells including fibroblast and keratinocytes which are delivered to the nonhealing wound site (Apligraf® and Dermagraft® (Organogenesis, Canton, MA, USA)). In contrast, other CTP products provide an organized scaffold to facilitate cell migration (Integra™ (Integra Life Sciences, Plainsboro, NJ, USA) and MatriStem® (ACell, Columbia, MD, USA)). These products provide the durable dermal layer necessary for granulation tissue formation.
Prior to applying a CTP product, it is important that the wound bed and local perfusion are optimized. Although CTP products can be applied repeatedly until complete epithelialization occurs, complete epithelialization is not always the goal of this therapy. The relatively high cost of these products may prohibit this approach. Close monitoring of wound quality and size is essential.
Regenerative medicine
With an estimated annual cost of over US$20 billion, there is a critical need to develop novel treatments to improve healing of chronic nonhealing cutaneous wound, in particular diabetic foot ulcers. 75 There have been encouraging results in preclinical models of diabetic wound healing. The plasticity of bone marrow-derived mesenchymal stem cells (MSCs) is well established, differentiating various cells including skin cells, specifically keratinocytes.76–78 The addition and incorporation of stem cells into wounds, particularly if they are capable of differentiating into a number of cell types, is promising. Similarly, MSCs have been shown to suppress the local immune response, reduce inflammation, and stimulate the differentiation and proliferation of local progenitor cells through the secretion of growth factors and their ability to modulate the immune system of the local soft-tissue.75,79–81 Stem cells are thought to modulate all the phases of wound healing, allowing for a wound to progress from the initial inflammatory state, and preventing them from becoming a chronic wound. MSCs have been shown to reduce inflammation by decreasing the amount of TNF-α and interferon-γ, both proinflammatory cytokines, helping to regulate the body’s response to injury. 82 Along with reducing inflammation, MSCs have also been shown to have an antimicrobial effect.83,84 They are able to do this through the secretion of antimicrobial factors and by upregulating the phagocytosis and bacteriocidal effect of immune cells.83,84
It is thought that one of the greatest actions of MSCs in ameliorating chronic wounds is through their paracrine effects. Although studies have shown MSCs are capable of differentiating into keratinocytes, the secretion of various growth factors, including vascular endothelial growth factor (VEGF), stromal cell-derived factors (SDF), platelet-derived growth factor (PDGF), fibroblast growth factor (FGF), and keratinocyte growth factor (KGF), allows MSCs to modulate the local tissue environment to increase the survival and proliferation of local repair cells.85–87 The addition of MSCs to fibroblasts has been shown to accelerate wound closure and increase the proliferation of keratinocytes, endothelial cells, and dermal fibroblasts.85,87,88
Preclinical models of diabetic wound healing MSCs have resulted in new granulation tissue formation, increased blood vessel formation and cellularity,89–92 and increased wound closure. In the treatment of chronic wounds, including diabetic foot ulcers, MSCs have been shown to be beneficial. 93 Animal studies have shown that when MSCs are injected into the wounds of chronic wound model mice, there is an increased rate of wound closure, reepithelialization, and angiogenesis. 94 Small clinical studies have also shown the effectiveness of using MSCs in the treatment of chronic wounds.95–97 In one of the largest clinical studies examining the use of MSCs for the treatment of chronic wounds, MSCs were added to a dermal replacement in 20 patients and the authors noted a 90% healing rate, with regeneration of the native tissue. 97
There is accumulating evidence to confirm the efficacy of stem cells in the treatment of critical limb ischemia. 98 Stimulation of angiogenesis and arteriogenesis represents attractive approaches to the diabetic foot ulcer. Angiogenesis is essential for wound healing, and multiple studies have demonstrated the proangiogenic nature of MSCs and their ability to secrete PDGF, VEGF, and EGF.90,99,100 A large level I RCT using bone marrow-derived MSCs in wounds of the limbs was published by Dash et al. 101 This trial showed that MSC therapy significantly reduced wound size and increased several clinical parameters when compared with controls. A study in China demonstrated improved ulcer healing rates with bone marrow mesenchymal stem cells (BMMSC). 102
An interdisciplinary approach involving scientists, biologists, and wound care providers will be important to develop novel therapies for tissue regeneration and treatment of complex wounds. Hopefully cell-based therapy will augment angiogenesis, optimize healing, and avoid amputation.
Hyperbaric oxygen
Hyperbaric oxygenation (HBO) has been proposed as an adjunctive treatment for diabetic foot ulcers.103–105 A recent review provides evidence that HBO therapy in patients with diabetic ulcers decreases the overall risk of amputation, especially major amputation, when compared to therapy without HBO (13.63% vs 30.07%). 103
Reported benefits of hyperbaric oxygen therapy (HBOT) include detrimental effect on bacteria via the production of oxygen free radicals and enhanced leukocyte activity. 106 Chen et al. 107 reported increased limb salvage rates (78.3%) for infected diabetic wounds with greater than 10 HBOT treatments. A meta-analysis found no statistically significant difference in major amputation rate with adjunctive HBOT (pooled data of five trials with 312 patients). 108
Prevention of ulcer recurrence
An adequate care plan must ensure offloading and sufficient arterial blood flow as prerequisites for healing. After the ulcer heals, the patient and their caregivers must incorporate preventative measures in care plans to reduce the risk of wound reoccurrence. Prevention of ulcer recurrence is a major clinical challenge, with recurrence rates ranging from 28% at 12 months to 100% at 40 months. 109
A newly healed ulcer is covered with fragile skin; after complete healing, there is an area of higher density tissue (scar), and sheering between the different tissue densities often contributes to new ulcers. The highest incident of reulceration is in the site of a previous ulceration.
A major goal of preventative treatment is the removal and prevention of calluses. Removal of plantar calluses can reduce peak plantar pressure by 26%. 110 In addition, removal of callus can help decrease forces at the site of the wound.
Conclusion
Advances in treating chronic diabetic wounds are promising; however, the intrinsic pathophysiologic abnormalities that lead to ulcers in the first place cannot be ignored. No known therapy will be effective without concomitant management of ischemia, infection, and adequate offloading.
Not all diabetic foot complications can be prevented, but it is possible to dramatically reduce their incidence through appropriate management and prevention programs. The multidisciplinary team approach to diabetic foot disorders has been demonstrated as the optimal method to achieve favorable rates of limb salvage in the high-risk diabetic patient.
Footnotes
Author contribution
All authors contributed equally in the preparation of this manuscript.
Conflict of interest
No disclosures of funding were received for this work from NIH, Wellcome Trust, or HHMI. No conflicts of interest are declared by any author on this study.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
