Abstract
Background:
Plantar fasciitis often leads to disability. Optimal treatment for this clinical condition is still unknown.
Objective:
To compare the effectiveness of wearing a full-length silicone insole with ultrasound-guided corticosteroid injection in the management of plantar fasciitis.
Study design:
Randomized clinical trial.
Methods:
Forty-two patients with chronic unilateral plantar fasciitis were allocated randomly to have an ultrasound-guided corticosteroid injection or wear a full-length silicone insole. Data were collected before the procedure and 1 month after. The primary outcome measures included first-step heel pain via Visual Analogue Scale and Heel Tenderness Index. Other outcome measures were the Foot and Ankle Outcome Score and ultrasonographic thickness of the plantar fascia.
Results:
After 1 month, a significant improvement was shown in Visual Analogue Scale, Heel Tenderness Index, Foot and Ankle Outcome Score, and ultrasonographic thickness of plantar fascia in both groups. Visual Analogue Scale scores, Foot and Ankle Outcome Score pain, Foot and Ankle Outcome Score for activities of daily living, Foot and Ankle Outcome Score for sport and recreation function, and plantar fascia thickness were better in injection group than in insole group (p < 0.05).
Conclusions:
Although both ultrasound-guided corticosteroid injection and wearing a full-length silicone insole were effective in the conservative treatment of plantar fasciitis, we recommend the use of silicone insoles as a first line of treatment for persons with plantar fasciitis.
Clinical relevance
Silicone insole may be considered as a first-line treatment option in patients with plantar fasciitis.
Introduction
Plantar fasciitis (PF) is the most common cause of heel pain in adults. Approximately 10%–16% of the population suffers from PF. 1 The incidence peaks in people between the ages of 40 and 60 years with no bias toward either sex. 2 Although the exact cause is not known, some risk factors such as age (middle age), obesity, excessive foot pronation, pes cavus, excessive running, reduced ankle dorsiflexion, tight Achilles tendon, and prolonged standing have been identified.1,3 The underlying condition that causes PF is a degenerative tissue condition that occurs near the site of origin of the plantar fascia at the medial tuberosity of the calcaneus. The normal fascia tissue is replaced by an angiofibroblastic tissue that spreads itself throughout the surrounding tissue creating a self-perpetuating cycle of degeneration. 4
Patients feel the gradual onset of pain near the medial side of the calcaneal tuberosity. Patients may present with heel pain with their first steps in the morning or after prolonged sitting and sharp pain with palpation of the medial plantar calcaneal region. Diagnosis is primarily based on history and physical examination.5,6
Numerous methods have been employed for treating PF including stretching exercises, foot orthoses, cortisone injections, platelet-rich plasma injection, botulinum toxin A injection, physical therapy, night splints, and extracorporeal shock-wave therapy, but there is poor evidence on optimal management strategy and their long-term effects.7 –12 Local corticosteroid injections (CSIs) are a popular method in the treatment of PF but only seem to be useful in the short term and only to a small degree.13,14 In recent years, different shapes of insole have been commonly prescribed by physicians to treat PF. While common to clinical practice, studies of the effects of silicone insole are generally of poor quality, and to our knowledge, there are no randomized trials comparing the silicone insole with CSI. The aim of this prospective randomized clinical trial was to compare the short-term efficacy of ultrasound-guided CSI with wearing a full-length silicone insole in the conservative treatment of PF.
Methods
This study is designed as a randomized controlled trial with a follow-up of 1 month. A total of 67 consecutive patients with chronic unilateral plantar heel pain from outpatient clinic of Physical Medicine and Rehabilitation Department, Necmettin Erbakan University Medical Faculty, Konya, Turkey were screened for admission into this study. The diagnosis of PF was based on tenderness localized to the medial tubercle of the calcaneus and pain, which started with the first step in the morning, receded thereafter, and worsened with weight-bearing activity.
Inclusion criteria required patients to be between the ages of 18 and 65 years, with a primary report of unilateral plantar heel pain for more than 3 months, and have pain score in the morning by first steps more than 4, by using 0–10-cm Visual Analogue Scale (VAS). The exclusion criteria were previous surgical intervention, CSI or shock-wave treatment, presence of foot deformity, acute heel trauma, PF due to systemic rheumatic disease, radicular or neuropathic pain, local infections, coagulation disorders, and pregnancy.
An informed consent was obtained from each patient, and this study was approved by the local Medical Ethical Committee of Selcuk University. Following the baseline examination, patients were randomly assigned to injection or insole group. Concealed allocation was performed by using a computer-generated randomized table of numbers created prior to the beginning of the study.
Outcome measures
All outcome measures were collected by a researcher blinded to the patient’s group assignment at the beginning of the study and at the follow-up of 1 month. A patient-assessed 10-cm VAS (0 cm = no pain, 10 cm = incredibly severe pain) and a physician-assessed Heel Tenderness Index (HTI) on palpation (0 = no pain; 1 = painful; 2 = painful and winces; 3 = painful, winces, and withdraws) were used to evaluate pain. The function and quality of life were measured using the Foot and Ankle Outcome Score (FAOS).15,16 FAOS is a 42-item questionnaire divided into five subscales: “pain,” “other symptoms,” “activities of daily living,” “sport and recreation function,” and “foot- and ankle-related quality of life.” The subscale “pain” contains 9 items, the subscale “other symptoms” contains 7 items, the subscale “activities of daily living” contains 17 items, the subscale “sport and recreation function” contains 5 items, and the subscale “foot- and ankle-related quality of life” contains 4 items. Each question can be scored on a 5-point Likert scale (from 0 to 4), and each of the five subscale scores is calculated as the sum of the items included. Raw scores are then transformed to a 0 to 100, worst to best score.
Measurement of plantar fascia thickness
Real-time ultrasonography (USG) with a linear 13-–5 MHz array transducer (Siemens G40, Germany) was used for the evaluation of symptomatic heels and for steroid injections. The USG evaluations were performed by the same radiologist. Patients were in the prone position during examination, and ankle motion was free, so as to allow easy examination of plantar fascia. The thickness of the plantar fascia was measured in a longitudinal view of the heel from the nearest point to the calcaneal insertion of the plantar fascia (Figure 1).
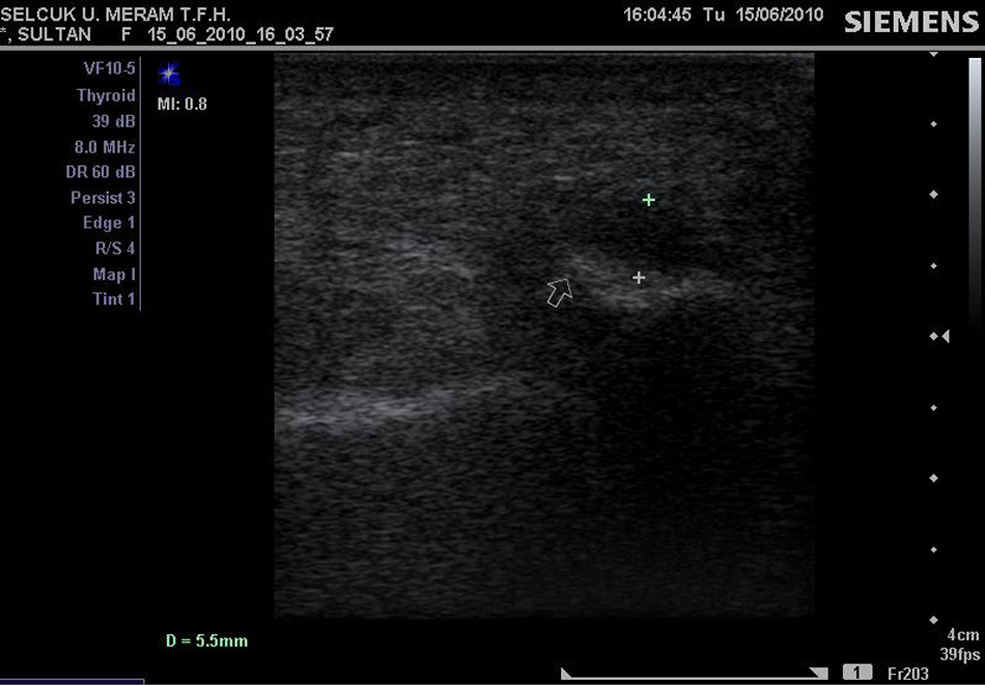
Longitudinal heel sonogram showing thickened proximal plantar fascia (calipers). The arrow indicates plantar fascial attachment to the calcaneus.
Injection technique
To injection group, USG-guided injection was performed via a medial approach. A 4-cm 21-gauge needle was positioned in a caudocranial oblique manner into the area of maximal ultrasound abnormality (thickness plus hypoechoic changes) with real-time imaging guidance. A quantity of 1 mL of betamethasone dipropionate (6.43 mg/mL) and betamethasone sodium phosphate (2.63 mg/mL) combination (Kenakort-A Retard®; Bristol-Myers Squibb) plus 1 mL of lidocaine HCl (Jetokain Simplex® 20 mg/2 mL; Adeka) were used for injection. The dispersal of the steroid was confirmed by USG.
Insole
The patients in insole group were asked to wear a prefabricated full-length silicone insole (Figure 2) in their daily lives for 1 month both indoors and outdoors as possible. Patients are allowed to proceed with normal sporting or recreational activities as tolerated but not to change their usual diet, daily activities, and sporting habits for the duration of this study. Usage of simple analgesics (such as acetaminophen) was allowed if necessary, except last 24 h before evaluations.

Silicone insoles.
Statistical analysis
Statistical analysis was performed with the SPSS for Windows 15.0 software. The Mann–Whitney U test was used for comparisons of the mean values for plantar fascia thickness, fat pad thickness, and VAS between the groups, and the Wilcoxon signed-rank test was used for comparisons of the values within the group. The chi-square test was used for comparisons of demographic data between the groups. A p value of <0.05 was considered to be statistically significant.
Results
Sixty-seven consecutive patients were screened for possible eligibility criteria. Forty-four patients satisfied the eligibility criteria, and 40 (age (mean ± standard deviation (SD)) = 46.4 ± 8.7 years; 80% female) completed the 1-month follow-up (Figure 3). Baseline demographics and clinical characteristics of groups are shown in Table 1. The mean values for age, gender, body mass index (BMI), job, duration of symptoms and sporting habits were similar in each group (p > 0.05). At the study onset, there were no statistically significant differences between the groups with respect to VAS, FAOS scores, HTI scores or plantar fascia thickness.
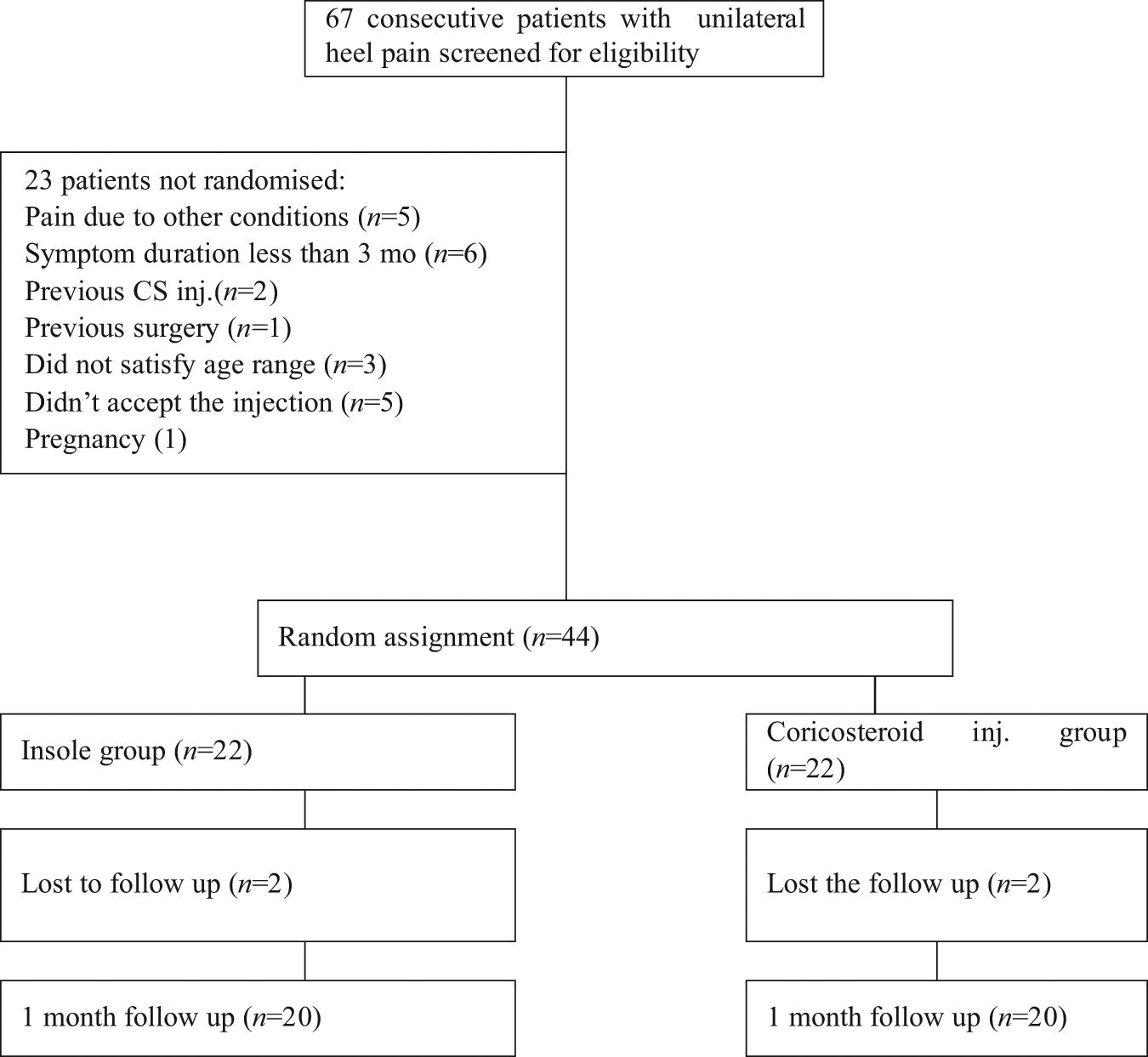
Flow diagram of patients recruited for this study.
Baseline demographic and clinical characteristics of participants.
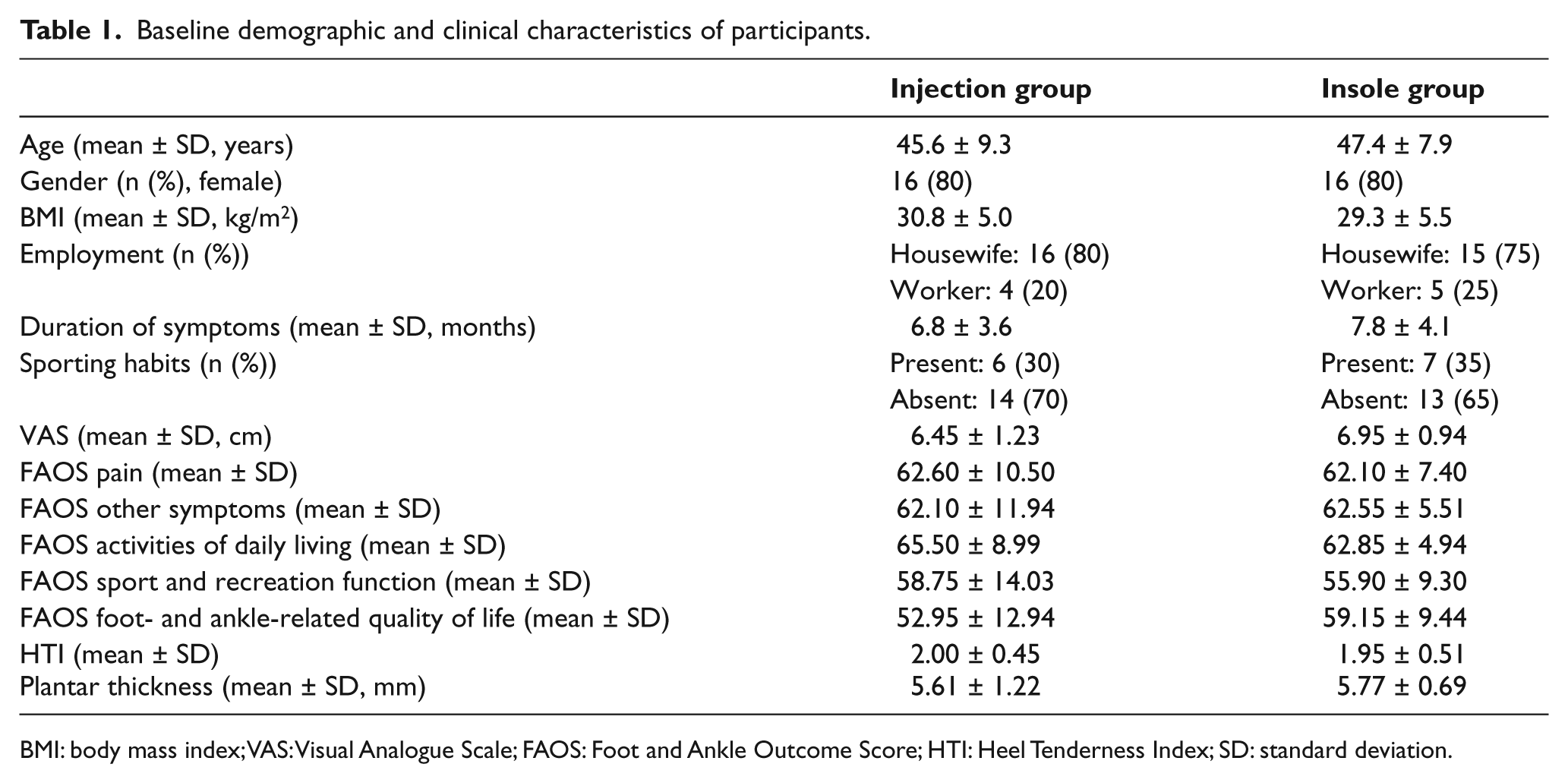
BMI: body mass index; VAS: Visual Analogue Scale; FAOS: Foot and Ankle Outcome Score; HTI: Heel Tenderness Index; SD: standard deviation.
One month later, both groups showed significant improvement for all parameters except subscore of FAOS sport and recreation function in insole group (p > 0.05). When the groups were compared after treatment, VAS scores, FAOS pain, FAOS activities of daily living, FAOS sport and recreation function, and plantar fascia thickness were better in injection group than in insole group, whereas significant differences were not observed between groups with respect to FAOS other symptoms subscale, FAOS foot- and ankle-related quality of life, and HTI (Table 2). No adverse effects were observed due to CSI, and noncompliance with silicone orthoses was not observed in any of the patients.
VAS, FAOS, and fat pad thickness before and after treatment in the groups.
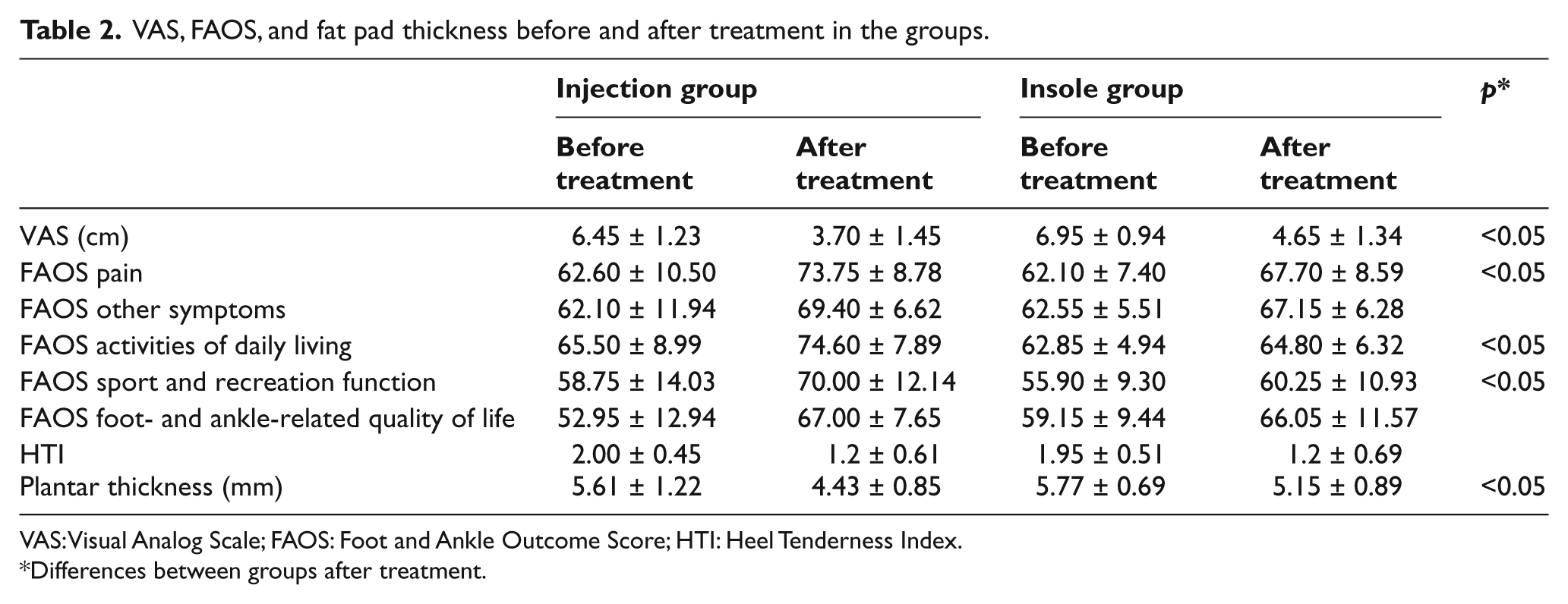
VAS: Visual Analog Scale; FAOS: Foot and Ankle Outcome Score; HTI: Heel Tenderness Index.
Differences between groups after treatment.
Discussion
In this randomized study, we compared the full-length silicone insoles with ultrasound-guided CSI in patients with PF. To our knowledge, this is the first study to compare silicone insoles with CSI. We showed significant improvement in VAS, HTI, FAOS, and USG thickness of plantar fascia in both groups except subscore of FAOS sport and recreation function in insole group when compared with the beginning evaluation. VAS scores, FAOS pain, FAOS activities of daily living, FAOS sport and recreation function, and plantar fascia thickness were better in injection group than in insole group. These findings indicate that commonly prescribed silicone insoles are at least as effective as CSI in reducing pain and improving functions in short term.
The efficacy of foot orthoses in patients with PF has been shown in previous studies. A recent meta-analysis 17 found that prefabricated and custom foot orthotics can decrease rearfoot pain and improve foot function in PF. A Cochrane review 18 found that custom foot orthotics may not reduce foot pain any more than prefabricated foot orthotics.
Recent studies reported that foot orthoses can reduce heel pain by relieving strain on the plantar fascia19,20 and reduce pronation of the foot and collapse of the foot arch.21,22 Total contact insoles can redistribute plantar pressure 23 and transfer pressure from the rearfoot to the midfoot region with a view to moderating plantar heel pain.24,25 Scherer 26 stated that the most important part of orthotic success was to mechanically control the midtarsal joint. A recent clinical practice guideline 5 recommends that both CSI and over-the-counter (OTC) arch support/heel cup should be considered as part of the “initial tier 1 treatment options.” CSIs are a popular method of treating the condition but only seem to be useful in the short term and only to a small degree. 13 Results of a Cochrane review 14 showed that CSIs improved PF symptoms at 1 month but not at six months when compared with control groups. Treatment with corticosteroids has a high frequency of relapse and recurrence, probably because intrafascial injection may lead to permanent adverse changes within the structure of the fascia and because patients tend to overuse the foot after injection as a result of direct pain relief.27,28
In addition, it is worthwhile to note that CSIs may have side effects such as tendon rupture, post-injection pain, local skin atrophy, facial flushing, post-injection flare, hyperglycemia, sepsis, and hypersensitivity reactions,28 –32 which may limit its acceptability. CSIs are also associated with high recurrence rates and delayed recovery. 14 Therefore, insole is continued to be used, which reduces the chance of recurrence.
The strengths of this study include that it is a prospective follow-up of a randomized clinical trial and it used validated outcome measures assessed by a blinded researcher. Furthermore, dropout rate was kept to a minimum. A limitation of our study was that responses to treatment were assessed only at a 1-month follow-up. So, we had no data regarding the long-term benefits of treatment. Additionally, the small sample size, single center, and selected population in this study limit the generalizability of our findings to the entire population of patients with PF.
Conclusion
We recommend the use of silicone insoles as a first line of treatment for persons with PF. Silicone insoles are also affordable and readily available. The promising results of our study need replication in future researches that would focus on the long-term effectiveness.
Footnotes
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
